Abstract
Objective:
We present a case with prenatal diagnosis of submucous cleft palate (SMCP) which was described using 2- and 3-dimensional (3D) ultrasonography in utero.
Case Report:
A 25-year-old pregnant woman was referred to our department for fetal ultrasound screening. After the detection of cardiac and spinal malformations of fetal, further detailed examination detected SMCP, which showed a gap within the hard palate on axial transversal view with the soft palate visible on sagittal view. The imaging of a defective hard palate in prenatal 3D ultrasonography is similar to that in postmortem 3D computed tomography reconstruction.
Conclusion:
A gap within the hard palate and verification of the visibility of the soft palate should be key points in the prenatal diagnosis of SMCP. Three-dimensional ultrasonic imaging is helpful for displaying the shape and extent of the bony defect in SMCP.
Introduction
Submucous cleft palate (SMCP) is a rare congenital anomaly, with a prevalence of 1:1250 to 1:5000. 1 Calnan 2 presented the classic triad describing it, which includes a bifid uvula, a bony defect in the posterior end of the hard palate, and a translucent zone in the midline of the soft palate. Submucous cleft palate can be encountered as an isolated finding or as part of syndromic diseases. 3 The OMIM database lists SMCP as a clinical finding in about 40 distinct syndromes, but in about 70% of cases, SMCP is an isolated finding. 4 The diagnosis of SMCP is often delayed, even if an individual presents with velopharyngeal insufficiency, open-nasal speech, and conductive hearing loss. 5 The purpose of this article is to present a case of SMCP that was diagnosed prenatally by ultrasonography.
Case Report
A 25-year-old woman, gravida 2 para 1, was referred to our department for ultrasound screening of a fetal anomaly. She and her husband were both healthy and had no remarkable family history. A prenatal ultrasound scan was performed at 22 weeks of gestation. Tricuspid stenosis, right ventricular dysplasia, ventricular septal defect, horseshoe kidney, and cleft vertebra were detected in the examination. We also detected a gap within the hard palate on the axial transversal view of the palate, without cleft lip and alveolus (Figure 1A). On the sagittal view of the palate, we could recognize the hyperechoic hard palate, but the backward soft palate was invisible (Figure 1B). While the tongue was moving, the arc-shaped band composed of hyperechoic hard palate and isoechoic soft palate could be identified clearly (Figure 1C). On ultrasonic 3-dimensional (3D) imaging of the palate (GE voluson E10, using Omniview and VCI technology), the shape and extent of the cleft in the hard palate could be shown (Figure 1D). Ultimately, the woman chose to terminate the pregnancy and the mouth of the abortus was examined. No gap was observed on the lip, the alveolus, or the surface of the palate. The uvula looked abnormal. Only a tiny nodule was located on the trailing edge of the soft palate (Figure 2A). The cleft hard palate was shown well on the computed tomography (CT) 3D reconstruction (Figure 2B).
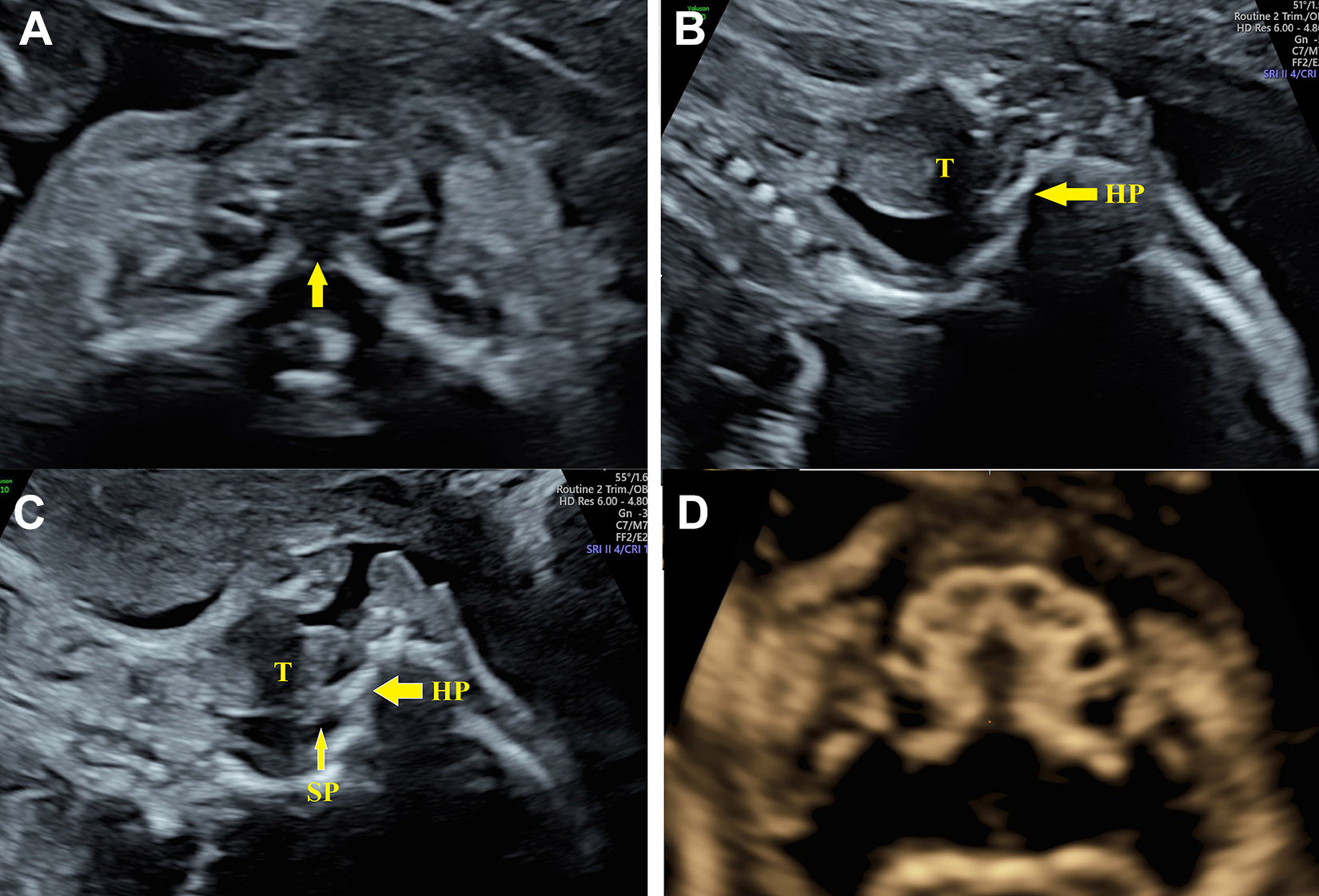
A, A gap (arrow) in the hard palate on the axial transversal view of the palate was shown. B, The soft palate was invisible on the sagittal view of the palate. C, The arc-shaped band of the palate was shown on the sagittal view of palate. Both the hyperechoic hard palate (thick arrow) and the isoechoic soft palate (thin arrow) were visible. D, The defect in the hard palate was visualized on the 3D ultrasonic image of the palate. HP indicates hard palate; SP, soft palate; T, tongue; 3D, 3-dimensional.
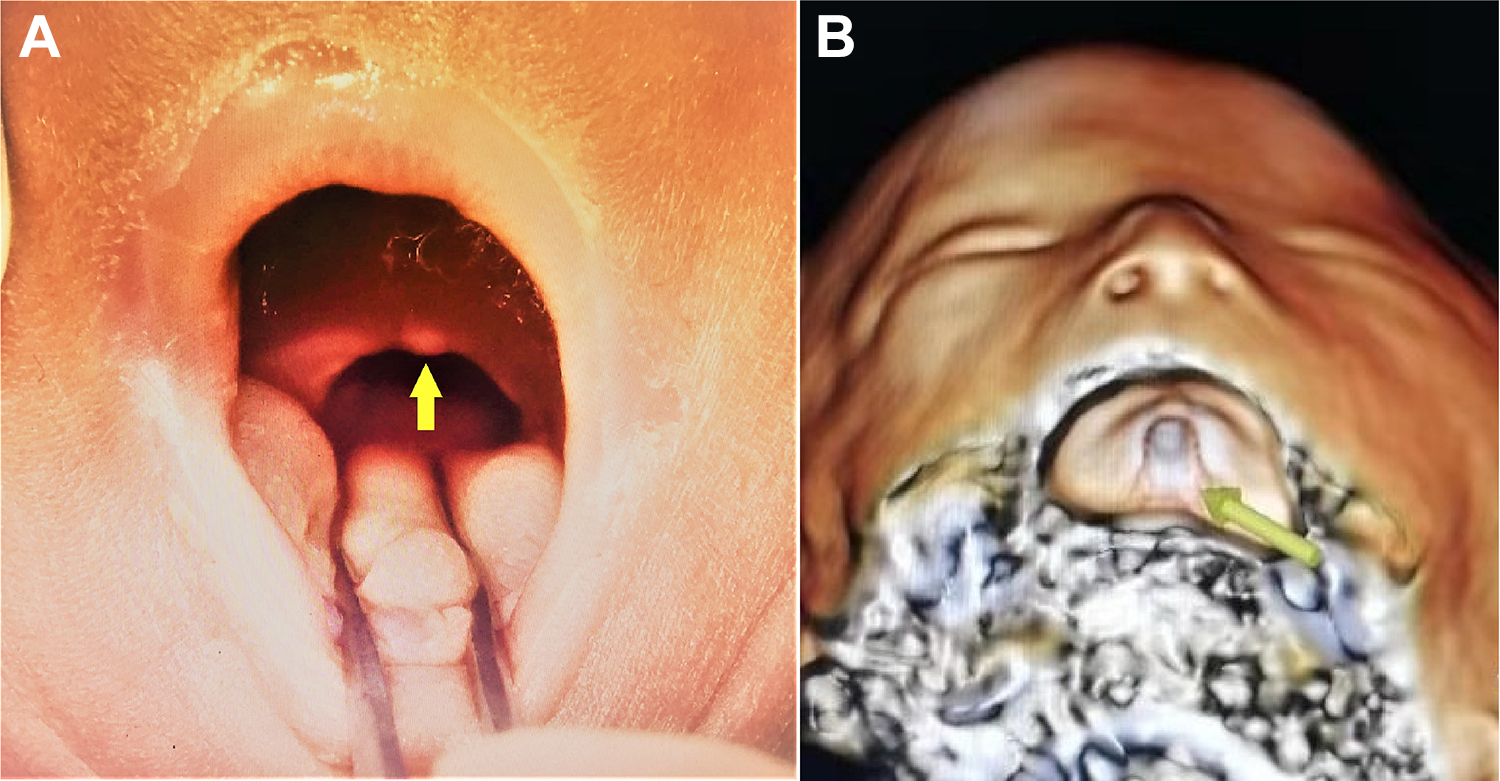
A, The postmortem examination of the palate. No gap was observed on the surface of the palate, and only a tiny nodule (arrow) was located on the trailing edge of the soft palate. B, The CT 3D reconstruction of the palate, showing the cleft (arrow) in the hard palate. CT indicates computed tomography; 3D, 3-dimensional.
Discussion
Orofacial clefts can be classified as cleft lip with or without cleft palate (CL/P) and CP only. Submucous cleft palate is a special type of cleft palate, with insufficient median fusion of the muscles of the soft palate hidden under the mucosa. 1
Postnatally, radiological imaging and ultrasonography are both used to identify SMCP. The 3D CT reconstruction divides the bone defect of SMCP into 4 types: type I, involving a posterior nasal spine and no V-shaped hard-palate cleft; type II, involving part of the hard bone; type III, involving the complete palate and extending to the incisive foramen; and type IV, involving the complete palate and the alveolus. 6 Jeremy and his colleagues used transoral ultrasonography to evaluate the defects of the muscle of the velum in SMCP. 7
It is difficult to detect the secondary palate by ultrasound prenatally because of the acoustic shadowing caused by surrounding bony structures. With the correct probe maneuverer and insonation angle in 2-dimensional ultrasonography, an axial transverse view of the hard palate can be obtained to insonate the horizontal plate of the palate bone 8 or the soft to hard palate interface (SHPI). 9 Both the hard and soft palate can be viewed in the sagittal plane by angled insonation. 10 Noncontinuous or nonvisual SHPI 9 can be a strong indication of a cleft secondary bone palate.
Magnetic resonance imaging (MRI) can increase the confidence in the ultrasound diagnosis of cleft palate.11,12 However, in studies of the prenatal diagnosis of orofacial clefts by ultrasound or MRI,8-12 SMCP was not included, and there has been no key point for the prenatal diagnosis of SMCP reported. In our case, the gap within the horizontal plate in the axial transverse view reminded us of a cleft in the hard palate. However, we could still identify the soft palate in the sagittal plane. We think this may be an important identification point between SMCP and the other forms of cleft hard palate. Nevertheless, sometimes the soft palate could not be visualized in the sagittal plane during our examination (Figure 1D). This was probably due to the enfoldment of the soft palate because of defects in the muscle and squeezing by the tongue. This may become an impediment to diagnosis. Therefore, it was necessary to observe patiently until there was some amniotic fluid in the fetus’s mouth, especially during the tongue’s activity.
The bony notch in the posterior end of the hard palate associated with SMCP is hidden under the mucosa, and the soft palate looks normal or has only a bifid uvula or a congenital fistula. 13 Therefore, both postnatal and postmortem, the oral difference between SMCP and normal anatomy is challenging to observe. Even for professional stomatologists, it is difficult to assess the extent of the bone involvement. However, 3D ultrasonography can visualize the cleft of the hard palate by many imaging methods, such as “reverse face,” “flipped face,” “oblique face,” and “surface-rendered oropalatal view.”14,15 In our case, the bony defect of SMCP was visualized in the axial view of the palate, rendered by 3D ultrasound. Significantly, the shape and extent of this defect of the hard palate presented in 3D ultrasonic imaging were similar to the postmortem 3D CT reconstruction of the palate.
In conclusion, a gap within the hard palate in the axial transversal view of the palate is a key point for identifying the bone defect of the secondary palate in SMCP prenatally. While the verification of the visibility of the soft palate in the sagittal view of the palate could be an identification point between SMCP and the other types of cleft hard palate. Three-dimensional ultrasonic imaging is helpful for displaying the shape and extent of the bony defect of SMCP.
Footnotes
Authors’ Note
The authors declared that materials described in the manuscript, including all relevant raw data, will be freely available to any scientist wishing to use them for non-commercial purposes, without breaching participant confidentiality. Patient guardian signed a document of informed consent.
Acknowledgments
The authors would like to acknowledge the hard and dedicated work of all the staff that implemented the intervention and evaluation components of the study. I confirm that I have read the Editorial Policy pages. This study was conducted with approval from the Ethics Committee of Renmin Hospital of Wuhan University. This study was conducted in accordance with the declaration of Helsinki. Written informed consent was obtained from patient guardian.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
