Abstract
Background:
Olfactory dysfunction evaluated with time-consuming tests was more common in patients with multiple sclerosis (MS) than in controls and correlated with neurological deficit. The aim of the present study was to compare olfactory function between patients with relapsing-remitting MS (RRMS) and controls with short and simple screening tool—the Sniffin’ Sticks Identification Test (SSIT)—and search for its association with clinical and radiological features of the disease.
Methods:
The study included 30 controls and 30 patients with RRMS treated with disease-modifying therapies—injectables (interferon β or glatiramer acetate, N = 18) and oral drugs (dimethyl fumarate or fingolimod, N = 12). Hyposmia was defined as a score of 6 points or fewer in the SSIT olfactory test. The data concerning number of previous relapses, disability in Expanded Disability Status Scale (EDSS), and recent brain magnetic resonance imaging (MRI) scan were collected. Moreover, thalamic volume and third ventricle width were recorded in every patient. Additionally, cognition and fatigue in patients were evaluated 24 months after olfactory assessment with the Symbol Digit Modalities Test (SDMT) and Fatigue Scale for Motor and Cognitive Functions (FSMC), respectively.
Results:
Patients with RRMS had a higher risk of hyposmia than controls (66.7% vs 36.7%, OR = 1.82, 95% CI, 1.10-3.67, P = .02). Neither inflammatory (number of previous relapses or new brain MRI lesions) nor neurodegenerative (EDSS, SDMT, and FSMC scores; thalamic volume; third ventricle width) MS features did not correlate with SSIT score (P > .05). In patients treated with oral drugs, olfactory dysfunction correlated with FSMC cognitive subscale (r = −0.90, P = .006).
Conclusions:
Olfactory dysfunction is nearly twice as common in RRMS as in controls and correlates with fatigue level in patients treated with dimethyl fumarate or fingolimod.
Introduction
Multiple sclerosis (MS) is a chronic autoimmune disease of the central nervous system affecting mainly young adults. 1 In around 85% of patients, the disease begins with its commonest form—relapsing-remitting MS (RRMS)—which manifests with inflammatory activity—recurrent clinical relapses and new brain and spinal cord lesions in magnetic resonance imaging (MRI). 1 Within 15 years of RRMS onset, around half of patients convert to secondary-progressive multiple sclerosis (SPMS) with the prominent neurodegenerative process, clinically resulting mainly in progressive gait and cognitive disturbances. 2 Although inflammatory activity dominates in RRMS, the neurodegenerative clinical and radiological features are also found at the early stage of the disease. 3
Previous studies concerning olfaction in MS were performed with comprehensive but time-consuming tests 4 and showed contradictory results. Some of them revealed that olfactory alterations were present early in the disease course, 5 correlated with disability progression, 6 and cognitive deficits.7,8 Impaired olfactory identification was also associated with the third ventricle width perceived as a marker of central brain atrophy. 9 On the other hand, few studies showed no difference in odor identification between patients and control group 10 and no correlation with the Expanded Disability Status Scale (EDSS) and disease duration. 11 Therefore, the aim of the present study was to compare olfactory function between patients with RRMS and controls with a short screening tool—the Sniffin’ Sticks Identification Test (SSIT). Assuming that the hyposmia was more common in RRMS, we also searched for association between olfactory dysfunction in SSIT and clinical and radiological features of the disease, such as a number of relapses, MRI characteristics, cognitive function, and results of cerebrospinal fluid (CSF) analyses.
Material and Methods
Consecutive patients with RRMS who fulfilled all the inclusion criteria, did not meet any of the exclusion criteria and gave informed consent, were recruited in comprehensive MS center in the University Hospital in Krakow, Poland, between January and March 2018. All patients were treated with disease-modifying therapies (DMTs)—18 with injectables (8 with interferon β-1a, 7 interferon β-1b, and 3 with glatiramer acetate) and 12 with oral drugs (8 with dimethyl fumarate and 4 with fingolimod). Control group comprised healthy volunteers without a history of neurological and rhinological disorders. Inclusion criteria were the diagnosis of RRMS according to the revised 2017 McDonald criteria, age of 18 to 65 years, EDSS score of 0 to 6.0, and current DMT treatment. Exclusion criteria were as follows: signs of upper respiratory tract infection within the last 6 weeks, otorhinolaryngological disorders affecting the sense of smell (chronic rhinosinusitis, rhinitis), clinical relapse in the preceding 3 months, current steroid treatment, and history of severe head trauma.
Both in patients and controls, on the basis of the medical interview, we collected the data regarding the presence of hypertension, diabetes mellitus, and thyroid disease. We additionally reviewed the list of medications and history of smoking. In patients, the data concerning a number of previous relapses, current EDSS as of 2018, and number of new T2 and gadolinium-enhancing (Gd+) lesions on recent brain MRI scan performed ±12 months in relation to olfactory assessment were collected. Relapses were confirmed by a neurologist and defined as described previously. 12 Cerebrospinal fluid analyses at the time of MS diagnosis, including cytosis, protein concentration, and the presence of oligoclonal bands (OCBs), were also collected.
The patients were followed up for 12 months after olfactory testing, and the data regarding new clinical relapses, new brain MRI lesions, and worsening of EDSS were gathered. Cognition and fatigue were assessed 24 months after olfactory examination with the Symbol Digit Modalities Test (SDMT) 13 and Fatigue Scale for Motor and Cognitive Functions (FSMC), 14 respectively. To exclude depression, the Beck Depression Inventory-II was applied. 15
Olfactory function was assessed with the 4-minute screening procedure—Polish version of SSIT—consisted of 8 pens with different scents familiar to Polish population (orange, cinnamon, mint, lemon, coffee, carnation, pineapple, fish). 16 The comprehensive version of all 3 domains of Sniffin’ Sticks Test—Identification, Threshold, and Discrimination—was adapted for Polish patients. 17 Patients and controls chose 1 of 4 possible options for each stick. Hyposmia was defined as the score of ≤6 points (below 90th percentile of normosmic individuals). 18
Third ventricle width on recent brain MRI was measured on T1 axial scans by an experienced neuroradiologist blinded to the patients’ clinical evaluation. Measurements of both thalami were made manually on 3-dimensional T1 MRI, AW Server 3.2, GE, by a blinded assessor.
The study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of Jagiellonian University in Krakow. Written informed consent was obtained from each patient and member of the control group.
Continuous data were presented as mean and standard deviation, median, and interquartile range. Categorical data were presented as counts and percentages. Continuous variables were tested for normality with Shapiro-Wilk test and compared by Student t test or by Mann-Whitney U test, as appropriate. The Pearson or Spearman rank correlation coefficients were calculated to evaluate linear dependence between variables, with normal or non-normal distribution, respectively. To identify independent predictors of SSIT, multivariate linear regression was performed and included variables which showed association with SSIT in the univariate model (P < .2) and did not correlate substantially with other independent variables (r > 0.5). Models were adjusted for age and sex. A P value of .05 (2 sided) was considered statistically significant. All statistical analyses were performed using STATISTICA version 13.
Results
The study included 30 patients with RRMS and 30 controls matched for age (mean 38.2 ± 9.8 vs 39.3 ± 12.3, P = .69) and sex (females 63.3% vs 53.3%, P = .43). There were no significant differences between patients and controls according to the hypertension (10.0% vs 10.0%, P = 1.0), diabetes mellitus (6.7% vs 3.3%, P = 1.0), thyroid disease (10.0% vs 23.3%, P = .30), and ever-smoking (26.7% vs 13.3%, P = .33). Mean periods of time from the first MS symptoms and MS diagnosis were 8.3 ± 4.9 and 5.5 ± 3.6 years, respectively. Patients with RRMS had a nearly 2-fold higher risk of hyposmia in comparison to healthy controls (66.7% vs 36.7%, OR = 1.82, 95% CI, 1.10-3.67, P = .02; Figure 1).
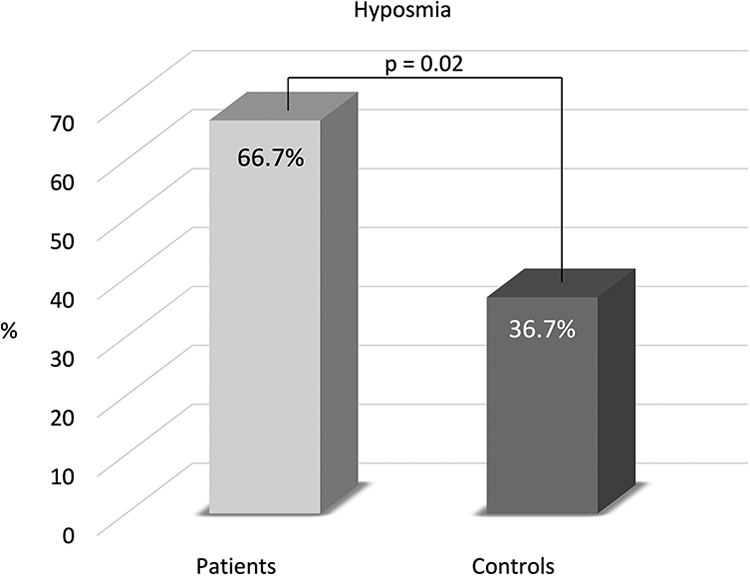
The frequency of hyposmia in patients with relapsing-remitting multiple sclerosis and control group.
The SSIT score did not correlate with disease duration, length of DMT use, and number of previous DMTs (Table 1). There was no difference in the SSIT score in patients with and without OCBs in the CSF (5.8 ± 1.2 vs 5.0 ± 2.0, P = .51). Neither inflammatory (number of previous relapses, number of new T2 or Gd+ lesions on recent brain MRI) nor neurodegenerative (current EDSS, SDMT, and FSMC scores; thalamic volume; third ventricle width) MS features did not show any correlation with olfactory dysfunction (P > .05). None of these variables predicted hyposmia in the multivariate model (Supplemental Table 1).
Correlation Between Sniffin’ Sticks Identification Test Score and Inflammatory and Neurodegenerative Features of Multiple Sclerosis Related to Current Disease-Modifying Treatment.a,b
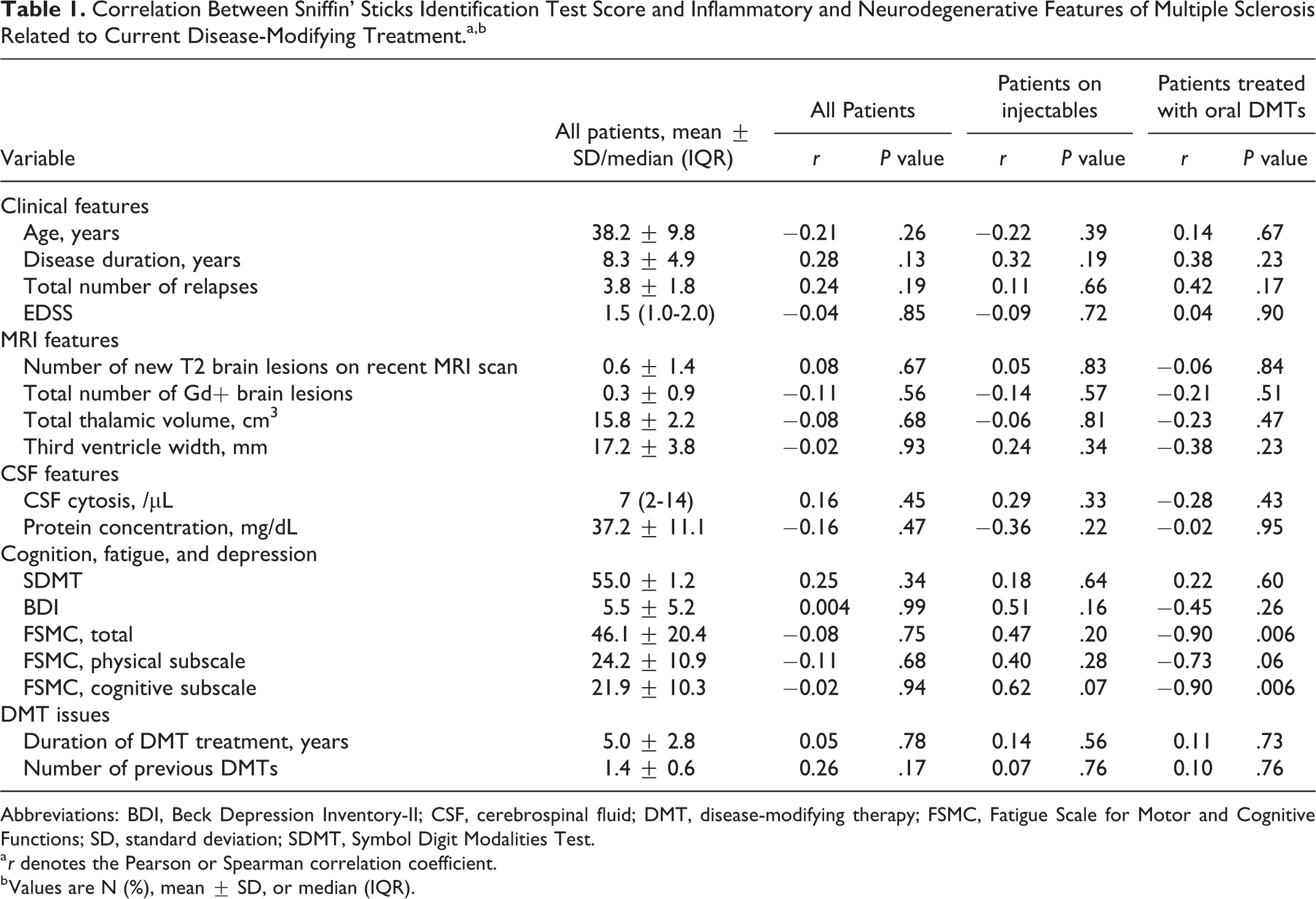
Abbreviations: BDI, Beck Depression Inventory-II; CSF, cerebrospinal fluid; DMT, disease-modifying therapy; FSMC, Fatigue Scale for Motor and Cognitive Functions; SD, standard deviation; SDMT, Symbol Digit Modalities Test.
a r denotes the Pearson or Spearman correlation coefficient.
b Values are N (%), mean ± SD, or median (IQR).
Hyposmia did not correlate with new disease activity within the next 12 months after olfactory evaluation in relation to new clinical relapses (r = 0.20, P = .28), new T2 (r = 0.17, P = .36) or Gd+ brain MRI lesions (r = −0.07, P = .71), and worsening of EDSS independent of clinical relapses (SSIT scores 5.9 ± 1.9 vs 6.0 ± 1.0, P = .42).
In patients treated with oral DMTs, olfactory dysfunction strongly correlated with FSMC score (r = −0.90, P = .006), mainly with its cognitive subscale, 24 months after evaluation with SSIT (r = −0.90, P = .006; Table 1). Such a correlation was not observed for patients receiving injectables.
Discussion
Our study demonstrated that patients with RRMS evaluated with a simple screening tool—SSIT—more often had hyposmia than healthy controls. This finding stayed in accordance with previous papers showing that olfactory dysfunction was more prevalent in patients with MS than controls, regardless of the tool used for olfaction evaluation.5,7,9 However, in most studies, the comprehensive but time-consuming tests were performed, such as The Connecticut Chemosensory Clinical Research Center olfactory test 5 and The University of Pennsylvania Smell Identification Test, 7 whereas in a recent study, olfaction was assessed with the short tool similar to the one used in our center and consisting of 12 different smells—the Odor Stick Identification Test for the Japanese. 9 Besides olfactory dysfunction is more common in progressive MS subtypes7,19,20, our study showed that hyposmia is also present at early stage of a disease, that is in patients with RRMS with relatively modest neurological deficit.
We found no correlation between hyposmia and inflammatory MS features, such as the number of previous relapses and new T2 or Gd+ brain MRI lesions. Our findings resembled the data coming from previous research and showing that identification of different smells was not associated with the number of clinical relapses. 9 In our study, hyposmia in patients with RRMS correlated with neither of neurodegenerative MS markers, including EDSS, SDMT, and MRI atrophy surrogates such as third ventricle width and thalamic volume. Previous research found that olfactory dysfunction might be associated with current EDSS,6,9 although this effect seemed to be mild 9 ; however, the olfactory event-related potential method was used, and the time of prospective observation of patients was longer (3 years) than that in our research (12 months). 6
Previous studies concerning cognition and olfaction produced contradictory results.8,9 One large study including 146 patients with MS found that mean olfactory bulb volume did not correlate with neuropsychological testing, 8 which was similar to the results of our study. On the other hand, Carotenuto et al 7 found that olfactory dysfunction was associated with cognitive impairment; however, it was more prominent in patients with SPMS. Finally, in a small study of 40 patients with RRMS, olfactory disturbances correlated not only with lower processing speed and working memory on neuropsychological testing but also with third ventricle width. 9 To our knowledge, our study is the second that evaluated the association between olfactory dysfunction and third ventricle width but produced negative findings. It was quite astonishing taking into account that in comparison to the previous study, 9 our patients had longer disease duration (8.3 vs 3.5) and higher median EDSS (1.5 vs 1.0), suggesting that the neurodegenerative process could be more prominent in their MS course. On the other hand, 10% of patients from the study of Okada et al was treatment naive, whereas all our patients were under the influence of DMTs. To resolve this discrepancy, we searched for association with thalamic volume since the atrophy of this structure was seen in early MS stages 21 ; however, we found no correlation with olfactory dysfunction.
When we analyzed patients according to treatment, we found that those receiving dimethyl fumarate or fingolimod and without concomitant hyposmia did better on FSMC, which was mainly driven by the FSMC cognitive subscale. The FSMC allows to quantify cognitive and motor fatigue separately 14 and lately, the scores in FSMC cognitive subscale were related to the performance of neuropsychological tests. 22 Therefore, the results of our study may indirectly confirm previous findings concerning the association between olfactory and cognitive dysfunction in MS. This correlation present only in patients treated with oral DMTs could be explained by the ability of fingolimod 23 and dimethyl fumarate 24 to slow the brain atrophy rate more significantly than injectables. 25
The sensation of olfaction is transmitted through the olfactory bulb and tract to the brain cortex. 4 This complicated olfactory network is disrupted in patients with MS by multifocal demyelinating brain lesions, mainly in the frontal and temporal lobes. 26 Furthermore, the neurodegenerative process resulting in neuronal death and irreversible axonal injury affects both the olfactory tract and the cortex in patients with MS, which is also found in other diseases with coexisting neurodegeneration and hyposmia, such as Parkinson or Alzheimer disease. 27 Both gray and white matter lesions in MS contribute to olfactory network damage. 28
Our study has important limitations. First, the patient group was small, and analyses according to the current treatment need to be interpreted with caution. Additionally, due to a small window for patient recruitment, there is a risk of selection bias. Second, we did not perform a comprehensive olfactory examination as a possible control for the screening test. Third, we did not monitor for change in olfaction and thalamic volume with time. Moreover, cognitive functions were assessed only once and 24 months after olfactory examination.
In conclusion, our study demonstrates that hyposmia in patients with RRMS is nearly twice as common as in controls. Olfactory dysfunction in patients with RRMS treated with dimethyl fumarate or fingolimod correlated with fatigue level 24 months after evaluation with SSIT. However, future studies performed on greater patient population treated with different DMTs are needed to confirm this finding.
Supplemental Material
Supplemental Material, sj-pdf-1-ear-10.1177_0145561320973777 - Olfactory Dysfunction in Patients With Relapsing-Remitting Multiple Sclerosis Treated With Disease-Modifying Therapies
Supplemental Material, sj-pdf-1-ear-10.1177_0145561320973777 for Olfactory Dysfunction in Patients With Relapsing-Remitting Multiple Sclerosis Treated With Disease-Modifying Therapies by Marcin Wnuk, Leszek Drabik, Monika Marona, Joanna Szaleniec, Amira Bryll, Paulina Karczk, Justyna Kolasinska, Monika Kolasinska, Maciej Ziekiewicz, Jacek Skladzien, Tadeusz Popiela and Agnieszka Slowik in Ear, Nose & Throat Journal
Footnotes
Authors’ Note
Marcin Wnuk contributed to conceptualization, data curation, draft writing, and editing; Leszek Drabik did conceptualization, formal analysis, and draft editing; Monika Marona contributed to conceptualization, data curation, and draft editing; Joanna Szaleniec did conceptualization, methodology, and data curation; Amira Bryll and Paulina Karcz contributed to methodology and data curation; Justyna Kolasinska, Monika Kolasinska, Maciej Ziekiewicz did data curation; and Jacek Skladzien, Tadeusz Popiela, and Agnieszka Slowik supervised and reviewed draft.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the grant from Jagiellonian University Medical College (number N41/DBS/000206).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
