Abstract
Methotrexate-associated lymphoproliferative disorders (MTX-LPD) frequently involve the extranodal organs throughout the body. Among the extranodal occurrences of MTX-LPD, pulmonary involvement is most frequent. In contrast, there are only a few reports of MTX-LPD in the nasal cavity or paranasal sinuses. Moreover, there are no previous reports of MTX-LPD mimicking granulomatosis with polyangiitis (GPA) in imaging examinations. We describe a case of a 53-year-old woman with MTX-LPD mimicking GPA in the nasal cavity and lungs. She complained of left nasal obstruction and discharge, general fatigue, and continual fever for 2 months. The patient had been diagnosed with rheumatoid arthritis and received methotrexate (MTX) for over 10 years. Contrast-enhanced computed tomography revealed unenhanced masses in the nasal cavity and multiple masses with cavitary changes in the bilateral lungs, suggesting GPA. However, histological examination of the nasal lesion and a history of MTX treatment indicated a diffuse large B-cell lymphoma type MTX-LPD. Two weeks after MTX withdrawal, prominent improvements in both lesions were observed. Complete regression of the nasal lesion was observed 3 months after discontinuation of MTX. Thus, MTX-LPD may mimic GPA in imaging examinations.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic multisystemic disease characterized by permanent bilateral and symmetrical synovitis of the peripheral joints. 1 Currently, methotrexate (MTX) is widely used as a disease-modifying antirheumatic drug (DMARD). 2 It is an antimetabolite that is structurally analogous to folic acid, and its mechanism of action is based on the alteration of DNA synthesis through inhibition of dihydrofolate reductase. 1 The risk of lymphoma in patients with RA treated with DMARDs is reported to be higher, with patients treated with MTX experiencing the highest risk. 3 Methotrexate-associated lymphoproliferative disorders (MTX-LPD) are lymphoproliferative diseases or lymphomas in patients treated with MTX for autoimmune diseases. 4 Therefore, patients with RA treated with MTX can develop MTX-LPD that has characteristics similar to lymphomas occurring in immunosuppressed patients. 2 Methotrexate-associated lymphoproliferative disorder is classified as “other iatrogenic immunodeficiency-associated LPDs” according to the revised fourth edition of the World Health Organization classification of tumors of hematopoietic and lymphoid tissues. 5
Methotrexate-associated lymphoproliferative disorder frequently involves the extranodal organs throughout the body.2,6 Among the extranodal occurrences of MTX-LPD, pulmonary involvement is most frequent.7,8 In contrast, there are only a few reports of MTX-LPD in the nasal cavity or paranasal sinuses.6,7,9-11 Moreover, there are no previous reports of MTX-LPD mimicking granulomatosis with polyangiitis (GPA) in imaging examinations. Herein, we report a rare case of MTX-LPD mimicking GPA in the nasal cavity and lungs.
Case Report
A 53-year-old Japanese woman presented to our hospital with complaints of left nasal obstruction and discharge, general fatigue, and continual fever for 2 months. The patient was 164 cm tall and weighed 52 kg. She had lost 4 kg during the last 6 months. The patient had been treated for RA for 10 years and 6 months and had taken MTX at a total dose of 4578 mg (4-12 mg/wk) for 10 years and 3 months. She was also taking folic acid at 5 mg/wk for the past 8 years and 6 months. In addition, oral prednisolone (0-5 mg/d) and loxoprofen sodium hydrate (a maximum dose of 120 mg/d) were administered.
Endoscopic findings revealed that the left inferior turbinate had a marked swelling with an irregular surface (Figure 1A).The right-sided nasal septum was partially swollen. Otoscopy revealed left-sided otitis media with effusion due to nasal obstruction. No abnormalities were detected in the other laryngopharyngeal regions. Laboratory data revealed elevated levels of serum C-reactive protein (CRP, 0.85mg/dL; normal, 0-0.3mg/dL), soluble interleukin-2 receptor (sIL-2R, 1950 U/mL; normal, 145-519 U/mL), and lactate dehydrogenase (LDH, 240 U/L; normal, 119-229 U/L). The white blood cell count was within normal limits (7000/µL; normal, 3100-9200/μL), and differential white blood cell counts were as follows: neutrophils, 66.5% (normal, 40%-67%); basophils, 0.4% (normal, 0%-1%); eosinophils, 1.8% (normal, 1%-5%); lymphocytes, 27.3% (normal, 24%-48%); and monocytes, 4.0% (normal, 3%-9%). Epstein-Barr virus (EBV)-specific immunoglobulin M to the viral capsid antigen was negative, immunoglobulin G was positive, and proteinase 3 antineutrophil cytoplasmic antibody was negative.
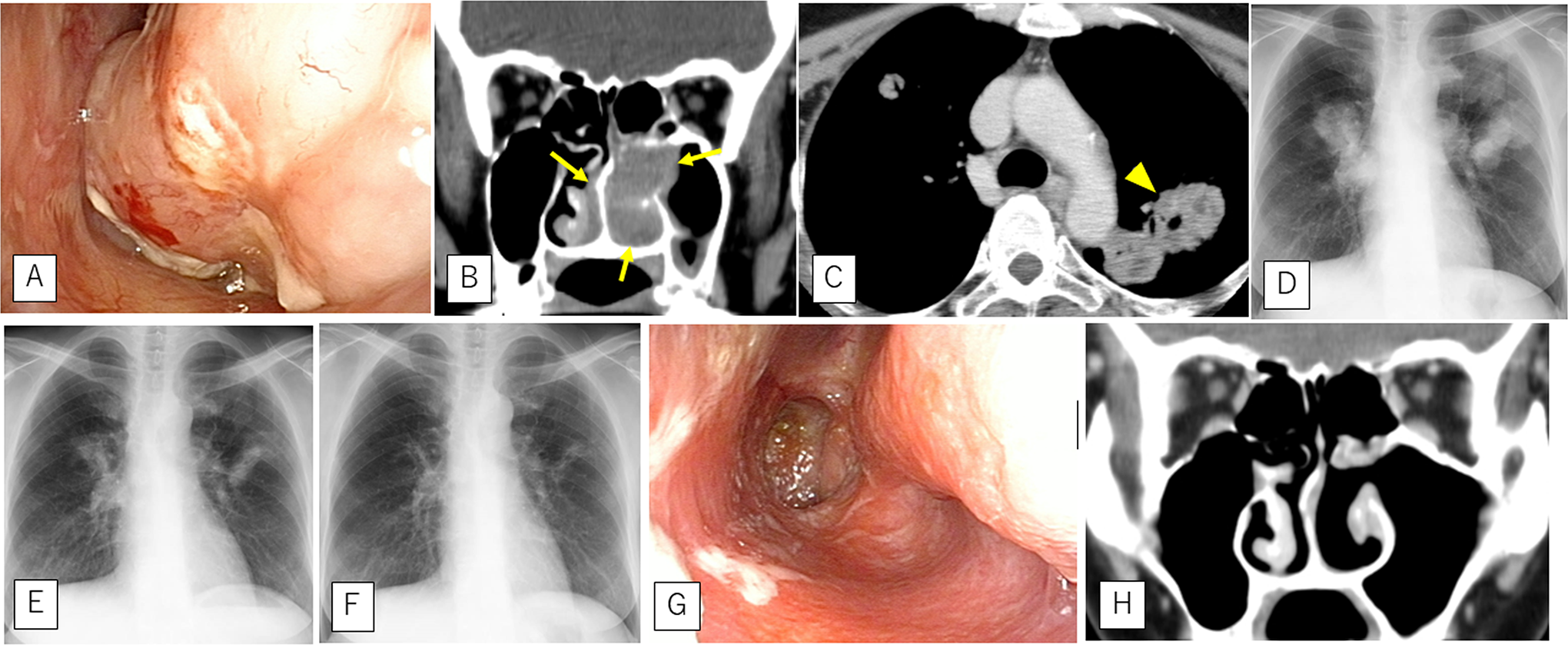
The left inferior turbinate shows a marked swelling with an irregular surface (A). Contrast-enhanced computed tomography (CT) shows unenhanced masses in the bilateral nasal cavities (B), indicated by arrows. Chest CT and radiography show multiple masses with necrotic changes in the bilateral lungs. Arrowheads indicate cavitary nodules (C and D). Two weeks after simple methotrexate (MTX) withdrawal, the lung lesions have reduced on chest radiographs (E). Continual chest radiography performed 2 months after MTX withdrawal shows almost complete disappearance of pulmonary lesions (F). Three months after MTX withdrawal, complete regression of nasal lesions can be observed (G), and paranasal computed tomography shows disappearance of the lesion with bone destruction (H).
Paranasal contrast-enhanced computed tomography (CT) revealed unenhanced masses in the bilateral nasal cavities (Figure 1B). Chest CT and radiography showed multiple masses with necrotic changes in the bilateral lungs, with some of the masses showing cavitary changes (Figure 1C and D). These findings suggested GPA.
A biopsy specimen from the nasal lesion showed dense, diffuse infiltration, and proliferation of large atypical lymphoid cells in the mucosal connective tissue (Figure 2A and B). Immunohistochemical analysis showed that the atypical lymphoid cells were positive for CD20 (Figure 2C) and negative for CD3 (Figure 2D). An in situ hybridization study revealed that almost all these cells were also positive for EBV-encoded small RNA (EBER; Figure 2E). Although histological findings were consistent with EBV-positive diffuse large B-cell lymphoma (DLBCL), the patient was diagnosed with MTX-LPD because she had been treated with MTX.
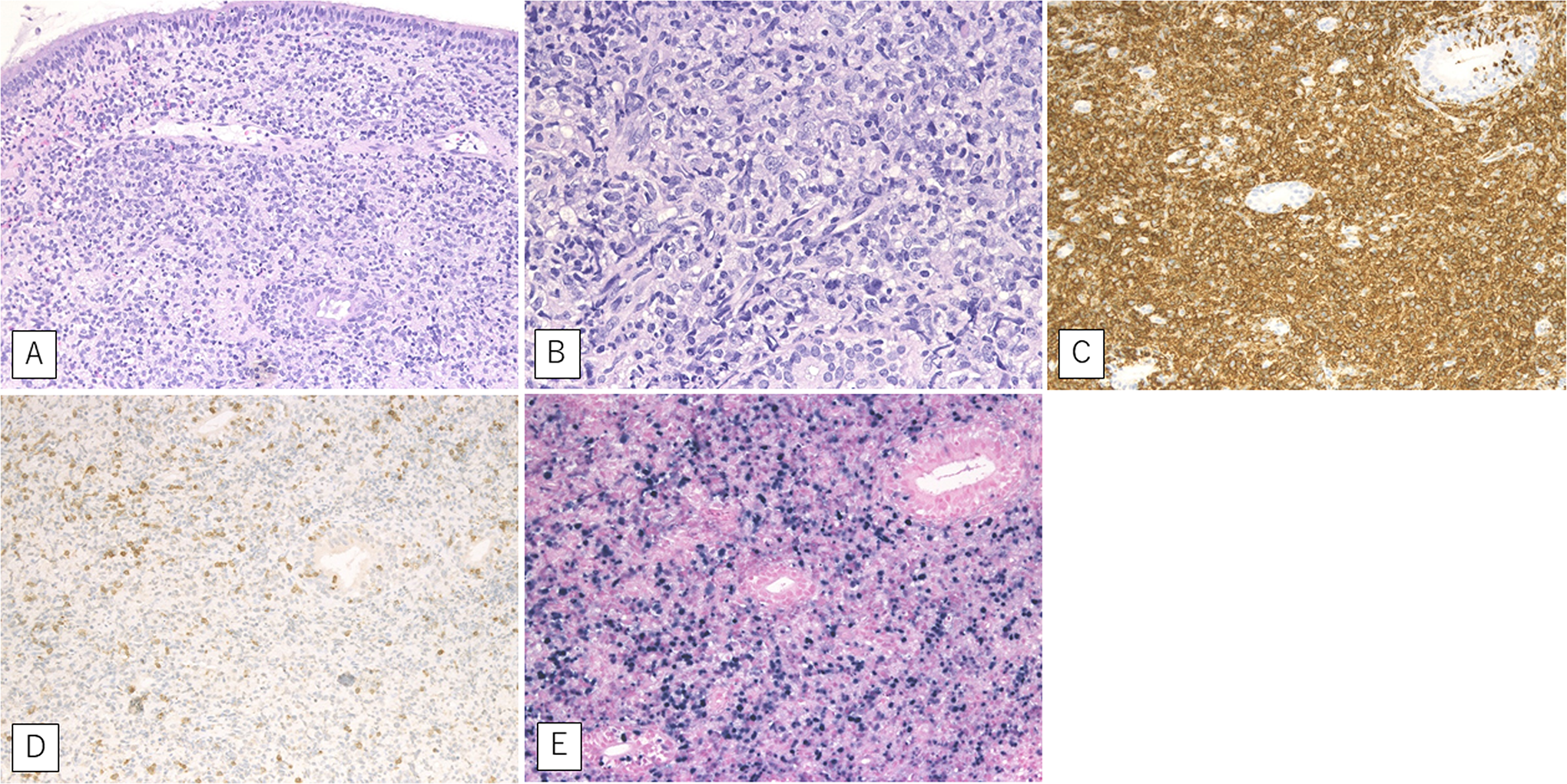
A biopsy specimen from the pharyngeal lesion shows dense, diffuse infiltration of large atypical lymphoid cells in the mucosal connective tissue (A and B). The atypical lymphoid cells show positivity for CD20 (C) and negativity for CD3 (D) in immunohistochemical analysis. In situ hybridization shows that almost all these cells are also positive for Epstein-Barr virus-encoded small RNA (E).
The patient was advised to discontinue MTX. Two weeks after simple MTX withdrawal, the nasal mucosa showed necrotic changes with prominent crusty deposits in the nasal cavity. Improvement of pulmonary lesions was observed simultaneously on a chest X-ray (Figure 1E). Serum levels of CRP, sIL-2R, and LDH decreased to 0.48 mg/dL, 1210 U/mL, and 207 U/L, respectively. Chest radiography showed that the pulmonary lesions had almost disappeared 2 months after MTX withdrawal (Figure 1F). Three months after MTX withdrawal, complete regression of the nasal lesions was observed (Figure 1G and H), and laboratory findings revealed normal levels of CRP (0.09 mg/dL), sIL-2R (448 U/mL), and LDH (183 U/L). Further,18F-deoxyglucose positron emission tomography/CT (FDG-PET/CT) conducted at 8 months after discontinuation of MTX revealed no significant FDG uptake in the nasal cavity and lungs. Eight years after the discontinuation of MTX, there were no signs of recurrence. Therefore, we believe MTX-induced LPD and spontaneous remission occurred after the withdrawal of MTX.
Discussion
The ability of MTX to inhibit cellular proliferation may hinder normal physiologic regeneration of the nasal epithelium and this effect may predispose patients to septal cartilage ischemia, necrosis, and eventually perforation. 12 Previously, there have been 2 cases of RA complicated with nasal septum perforation due to MTX-LPD9,10; the histological type was peripheral T-cell lymphoma in 1 case and natural killer/T-cell lymphoma (NK/T-cell lymphoma) in the other. Although B-cell LPD is the most common among MTX-treated patients,2,8,11 the histological type in the 2 previously reported cases of nasal MTX-LPD, excluding the nasal septum, was also NK/T-cell lymphoma.7,11 To the best of our knowledge, the present case is the first report of nasal MTX-LPD with the histological characteristics of DLBCL.
The nasal symptoms of both malignant lymphoma and GPA, known as Wegener granulomatosis, can be very similar, including recurrent sinusitis, nasal discharge, tenderness in maxillary areas, nasal obstruction, and nasal deformities. 13 In the present case, the lesions were found in both the nasal cavity and the lungs. Additionally, multiple nodules with cavity formation in the lungs were observed on CT images; this suggested a necrotic change in tissue. It is extremely rare for nasal DLBCL to be accompanied by pulmonary lesions simultaneously. 14
Imaging findings such as heterogeneous/unenhanced masses in the nasal cavity and multiple cavitary masses in the lungs suggested necrosis and angiocentric/angiodestructive features. Granulomatosis with polyangiitis frequently involves these organs, with solid or cavitary nodules or masses throughout the lungs. Central cavitation typically occurs in nodules measuring >2 cm in diameter. 15 Therefore, GPA is consistent with the findings in our patient. Diffuse large B-cell lymphoma can represent various imaging findings, including heterogeneous masses, air bronchograms, and necrosis. 16 Furthermore, necrosis in DLBCL can be a poor prognostic factor. 17 There are several reports about the imaging findings for MTX-LPD. Recently, Makihara et al reviewed MTX-LPD cases with multiple pulmonary nodules. 18 In their review, imaging findings for pulmonary lesions of MTX-LPD, not limited by DLBCL-type LPD, were varied and included nodular density, infiltrative shadows, and ground-glass patterns. To the best of our knowledge, there are few reports regarding imaging-based prognostic factors for MTX-LPD.
In the present case, the nasal lesion showed EBER positivity. Although angioinvasion was not apparent because of the small size of the material, our case should be classified as DLBCL-type MTX-LPD. Lymphomatoid granulomatosis is also a rare EBV-positive B-cell LPD. Characteristic pathological findings include angioinvasion/angiodestruction by neoplastic cells and various degrees of necrosis. 19 In a clinicopathological study, angioinvasion was observed in 14 of the 34 patients (41%) with EBV-positive MTX-LPD (EBV-positive mucocutaneous ulcer), with a particularly high frequency in cases with EBER-positive atypical lymphoid cells. 20 Although both DLBCL and DLBCL-like MTX-LPD can share the same histopathological features, DLBCL-like MTX-LPD showed a better prognosis. 20 Therefore, necrotic changes in imaging due to DLBCL-type MTX-LPD might not have a poor prognosis.
This report has some limitations. First, FDG-PET/CT was not performed prior to MTX withdrawal in this patient. Therefore, it remains unclear whether the lesions were present only in the nasal cavity and lungs. In this regard, because nasal and pulmonary lesions disappeared with MTX withdrawal alone, we speculate that similar changes were observed in all lesions even if lesions were present in other regions before MTX withdrawal. In the present case, FDG-PET/CT at 8 months after MTX withdrawal confirmed not only the disappearance of the nasal and pulmonary lesions but also the absence of other organ involvement. Second, histological examination was not performed for lung lesions. Therefore, we cannot completely rule out the possibility of GPA for lung lesions. A previous study reported that 2 cases with mild forms of GPA showed complete remission and disappearance of pulmonary opacities without specific treatment during several years. 21 In this regard, a negative proteinase 3 antineutrophil cytoplasmic antibody was confirmed on blood tests. In addition, as the lung lesions showed the same reduction after MTX withdrawal as the nasal lesions, it seemed reasonable to consider lung lesions with the same histological characteristics as those of the nasal cavity.
Conclusion
We encountered a rare case of DLBCL-type MTX-LPD in the nasal cavity. Nasal and pulmonary imaging findings suggested GPA. Complete regression of both lesions was observed after simple discontinuation of MTX. Thus, MTX-LPD may mimic GPA in imaging examinations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
