Abstract
Sinonasal small cell neuroendocrine carcinoma (SNEC) is an extremely rare and aggressive neoplasm that can arise in the sinonasal region. These tumors are associated with high morbidity and mortality, are difficult to diagnose, and are hard to treat. We describe 2 cases of this poorly understood malignancy and review imaging, pathology, and treatment decisions. A 41-year-old male and a 64-year-old female presented to a tertiary center in 2019 after developing nasal obstruction and were found to have sinonasal masses on imaging. Both biopsies showed strong expression of pancytokeratin with dot-like reactivity and expression of neuroendocrine markers chromogranin and synaptophysin. The findings were diagnostic of SNEC. Staging positron emission tomography/computed tomography and brain MRI were performed, and patients were discussed at a multidisciplinary tumor board. Neither had distant metastatic disease at presentation. One patient had no intracranial or orbital disease and underwent a subtotal endoscopic resection with adjuvant chemoradiation. The other patient demonstrated middle cranial fossa, dural, and orbital involvement as well as cranial nerve V palsy. This patient was treated with induction chemotherapy, followed by concurrent chemoradiation. Both patients are presently alive at 4 months follow-up, but one with persistent local disease and the other distant metastasis. Sinonasal small cell neuroendocrine carcinoma is a rare and poorly understood malignancy with an aggressive clinical course. Continued careful review of pathology and study of molecular features are needed for improved understanding of SNEC, and particularly for head and neck SNEC, to establish a staging system and better formulate treatment protocols.
Keywords
Introduction
Sinonasal small cell neuroendocrine carcinoma is a rare, aggressive tumor of the nasal cavity and paranasal sinuses with fewer than 100 total cases described in the English literature. 1 While neuroendocrine carcinomas typically occurs in the lungs, 2.5% to 5% of cases are extrapulmonary, with a subset occurring in nasal cavity and paranasal sinuses. 1 Patients may present with a myriad of symptoms, including but not limited to nasal obstruction, headache, pressure, and epistaxis.1,2
Sinonasal SNEC is often considered to have been first described in 1965 as an “Oat-cell carcinoma.” 2 The histopathology of sinonasal SNEC mirrors the pulmonary equivalent with dense cellular tumors arranged in ribbons, cords, and sheets with cells with high nuclear-to-cytoplasmic ratios and very brisk mitotic activity. 1
Understanding of this malignancy is very limited and treatment regimens are largely based on anecdotal evidence and extrapolation from lung small cell carcinoma patients. Its rarity and paucity in the literature represents a major barrier to further advancements in treatment. Based on available evidence, sinonasal SNEC appears to have very poor overall survival, with 5-year survival rates as low as 13%. 3 Treatment modalities vary greatly and include chemotherapy, radiotherapy, surgery, or a combination of modalities. 1 Given the rarity of this tumor, variety of treatment protocols, and poor survival, new information is critical in developing standard of care management strategies. In this case series, we describe the clinical features, pathology, and treatment courses of 2 patients with sinonasal SNEC who presented to our institution in 2019.
Case Presentations
Case 1
A 61-year-old female presented with right-sided nasal obstruction, facial pressure, hyposmia, and hypogeusia. Non-contrast sinus computed tomography imaging identified a large nasal mass, centered at the right olfactory cleft with erosion of the middle turbinate. Nasal endoscopy with biopsy confirmed these findings. Histopathologic examination identified SNEC based on the findings of blue tumor nests in a fibromyxoid stroma with cells with high nuclear to cytoplasmic ratios, molding, hyperchromasia, and abundant apoptosis and mitosis. Immunohistochemistry showed the tumor cells to be positive for cytokeratin AE1/AE3 with a dot-like pattern, extensive positivity for synaptophysin, and focal, dot-like positivity for chromogranin. The tumor was negative for desmin, calretinin, p40, CD99, SOX-10, and S-100 (Figure 1A-D). Magnetic resonance imaging (MRI; Figure 2) demonstrated a large right nasal mass extending into the cribriform plate and a positron emission tomography (PET) scan demonstrated no metastatic disease (T3N0M0).
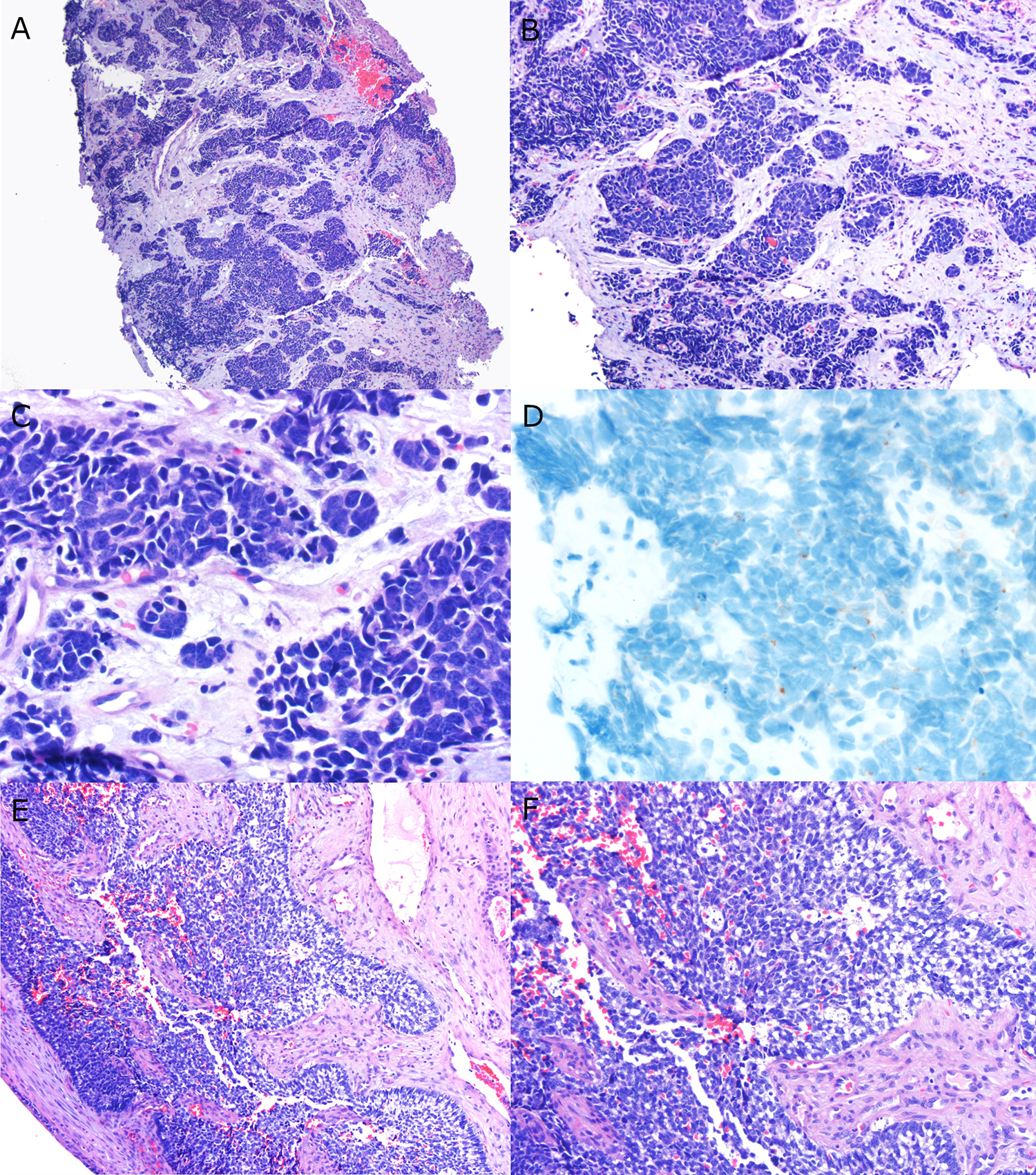
Pathologic features of the 2 cases. A, Low power H&E features for patient 1 showing nests of blue tumor in a prominent fibromyxoid stroma (4× magnification). B, Medium power showing the circumscribed nests and lack of specific differentiation such as squamous or glandular (10× magnification). C, High power showing the hyperchromatic cells with minimal cytoplasm, nuclear molding, and prominent apoptosis (40× magnification). D, Immunohistochemistry for chromogranin showing scattered foci of punctate brown staining (40× magnification). E, Low power view for patient 2 showing circumscribed nest of blue cells with peripheral palisading (10× magnification). F, Medium to high power view showing the angulated nuclei, minimal cytoplasm, peripheral palisading, and prominent apoptosis (20× magnification).
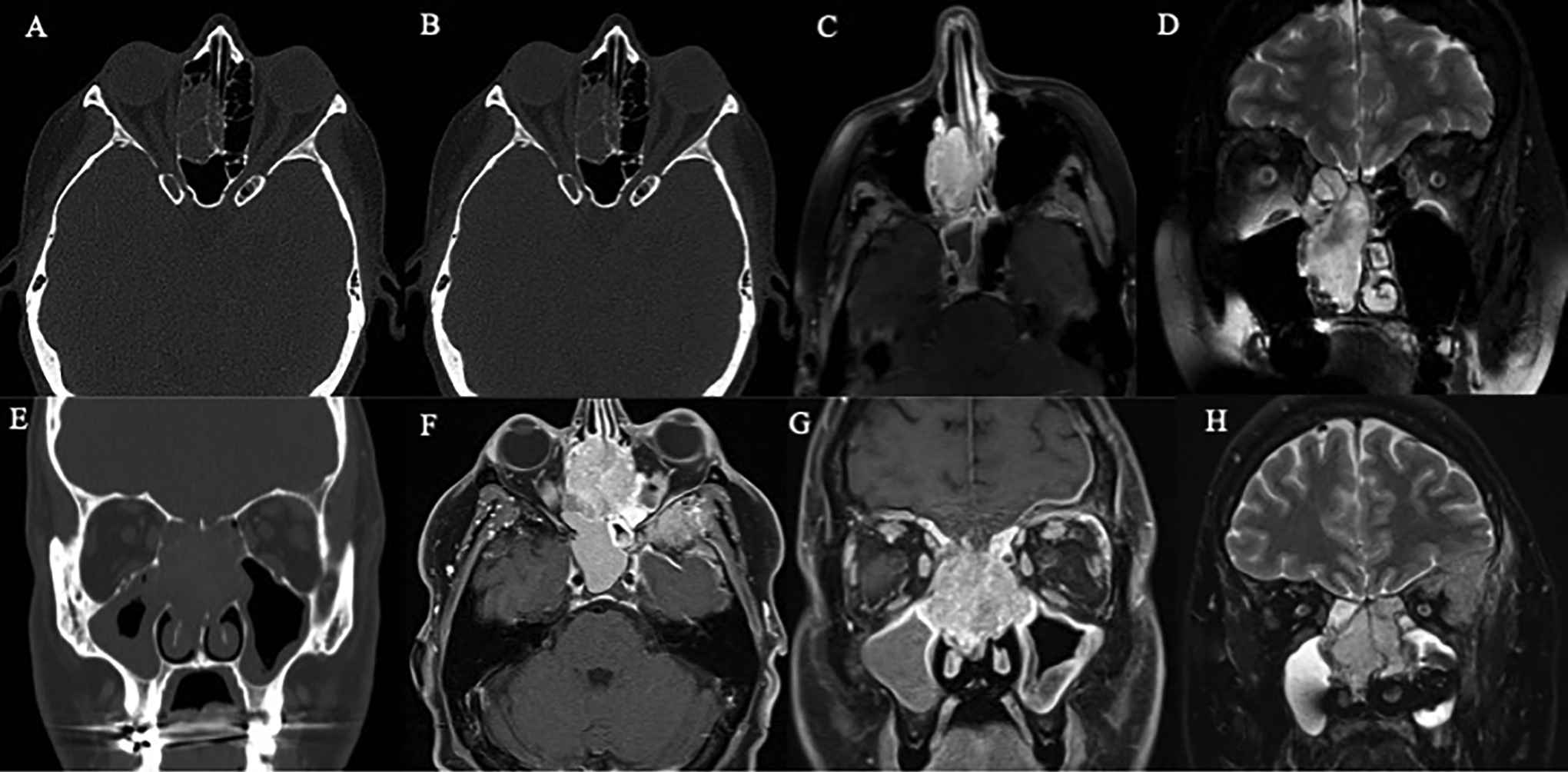
Case 1 images: CT (A and B) and post-contrast MRI (C and D) demonstrating large right nasal mass with extension into cribriform plate. Case 2 images: CT (E) and MRI (F-H) demonstrating a large SNEC with extension into the left orbit and middle cranial fossa. CT indicates computed tomography.
A right endoscopic maxillary antrostomy and sphenoethmoidectomy was performed along with resection of the middle turbinate up to the skull base. Subtotal resection was achieved as the tumor was invading the nasopharynx and pterygopalatine fossa. The patient subsequently underwent intensity-modulated radiation therapy (IMRT), receiving 65.1 gray (Gy) over 31 fractions. Systemic chemotherapy was offered to the patient but was declined.
While her local symptoms improved after treatment, a liver metastasis was identified 4 months following completion of radiotherapy. At last follow-up a month later, the patient’s primary site continues to show no evidence of disease. Chemoembolization is currently being pursued to treat the metastatic disease focus.
Case 2
A 41 year-old male was referred to our institution with a presumptive diagnosis of esthesioneuroblastoma with extension into the left orbital apex and middle cranial fossa. The patient had rapidly progressing facial numbness, epistaxis, and left-sided proptosis and diplopia. Positron emission tomography imaging demonstrated no distant metastasis. Histopathologic review of the biopsy diagnosed the tumor as SNEC based on large, irregular, blue tumor nests in a fibrous stroma. The tumor nests showed peripheral palisading and cells with high nuclear to cytoplasmic ratios, hyperchromatic, angulated nuclei, and abundant apoptosis and mitosis (Figure 1C and D). Immunohistochemistry showed the tumor cells to be positive for EMA, cytokeratin AE1/AE3 with a dot-like pattern, synaptophysin, and chromogranin with a focal, dot-like pattern. The tumor was focally positive for calretinin (5%-10%) and was negative for desmin, calretinin, p40, CD99, SOX-10, CD45, SALL4, TTF-1, Glypican 3, AFP, and p63. INI-1 was positive (retained). Surgery was canceled due to the new pathologic diagnosis and involvement of the middle cranial fossa (T4bN0M0). The patient underwent 3 cycles of chemotherapy followed by IMRT (60 Gy over 30 fractions).
Although symptoms improved initially, the patient developed rapid recurrence 4 months following completion of radiotherapy. He is currently receiving palliative immunotherapy. Due to intractable pain, a bilateral cingulotomy is currently being considered.
Discussion
Sinonasal small cell neuroendocrine carcinoma is a rare tumor with variable presentation. Treatment is largely patient-centric given the lack of substantive data, lack of well-defined treatment regimens, differences in precise tumor location and extension, patient preference, and comorbidities. Surgery, chemotherapy, and radiotherapy have all been used independently or in combination for multimodal therapy. Five-year survival outcomes for sinonasal SNEC are historically low, though further understanding and advanced treatment modalities may be able to increase survival and decrease morbidity.
The pathology typically demonstrates infiltrative nests of blue tumor in a fibrous stroma. The nests vary significantly in size from case to case, being well defined in some patients but poorly defined in others, and nuclear palisading at the periphery is common. Case 1 exhibited dense fibrous stroma separating the nests (Figure 1) within a background of nuclear crush artifact while case 2 showed nuclear palisading. The tumor cells of SNEC have minimal cytoplasm and are 2 to 3× the size of mature lymphocytes. 4 The nuclei are round to irregularly shape with tapering and teardrop shapes and dense or finely granular nuclear chromatin. Nucleoli are absent and nuclear molding is present in each case.
Cytokeratin AE1/AE3, cytokeratin CAM5.2, and neuroendocrine markers synaptophysin and chromogranin are positive, typically with a dot-like, perinuclear pattern. Synaptophysin can be strong and diffuse or patchy, and chromogranin is typically patchy as seen in patient #2 (Figure 1). There is a newer marker of neuroendocrine differentiation Insulinoma-associated protein 1 (INSM-1) which is a transcription factor that shows nuclear staining in neuroendocrine tumors. It is typically positive in head and neck SNEC. 5 In our case, both cases were negative for CD99, SOX10, desmin, and p40 immuno-helping to rule out the many blue cell tumors that arise in the sinonasal tract and are in the differential diagnosis.
Unfortunately, both patients described had poor prognosis after initial diagnosis and developed recurrence or metastatic disease. In the first case, the primary disease appears to have been well treated. Nevertheless, despite negative initial PET imaging, the patient ultimately developed metastatic disease. This may demonstrate a need for trimodality in treatment of this rare, aggressive disease. The second patient had extensive disease that made resection untenable given associated risks.
This series adds to a limited body of literature and highlights the tumor’s distinctive pathologic appearance as well as factors that should be considered when designing a treatment plan. Treatment decisions should involve a multidisciplinary team in consultation with the patient. Trimodality therapy of surgery, radiation, and chemotherapy is likely key to minimize risk of regional recurrence and distant metastatic spread. Given the high mortality rate and potential comorbidities associated with treatment options, patient goals of care must be strongly considered and discussed in any course of management.
Conclusion
Sinonasal small cell neuroendocrine carcinoma (SNEC) is a rare tumor that poses a diagnostic challenge due to the myriad of presenting symptoms and difficult pathologic diagnosis. Future advancements are needed to identify and standardize effective treatments.
Footnotes
Peer Review Statement
The Editor of the ENT Journal is an author of this paper, therefore, the peer review process was managed by alternative members of the Board and the submitting Editor had no involvement in the decision-making process.
Authors’ Note
Nikita Chapurin and Douglas J Totten denote equal contribution. This study was approved by the Institutional Review Board of Vanderbilt University Medical Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
