Abstract
Leiomyosarcoma (LMS) in the sinonasal tract (SNT) is a rarity that has been firstly described in 1958. Since then, there have been only a few articles about this entity. Most of the data available about LMS in the SNT is derived from case reports. We believe that our case will support the data set and help guiding the management of this rare condition. A 84-year-old female presented with nasal airway obstruction on the left side. She experienced several episodes of epistaxis from her left nostril, what made her to seek medical care. A rhinoscopy revealed an obstructing mass in the left nasal cavity. Computed tomography (CT) scan of the paranasal sinuses revealed a homogenous mass occupying the left nasal cavity, bone destruction of the left middle, and inferior nasal turbinates. An infiltration of the left nasolacrimal duct was also present. The patient refused to undergo open surgery and the mass was removed during an endoscopic approach. The histopathological analysis combined with immunohistochemistry was consistent with LMS. The resection margins were positive for tumor cells. A staging with CT-neck-thorax, abdomen ultrasound, and MRI of the head ruled out metastases. She underwent a second endoscopic tumor resection surgery with positive resection margins and obtained adjuvant radiotherapy. On 9 months of follow-up, there was no recurrence or metastases.
Keywords
Introduction
Leiomyosarcoma (LMS) is an aggressive malignant neoplasia derived from smooth muscle cells which is rarely found in the sinonasal tract (SNT).1,2 It represents about 2.3% of all non-epithelial neoplasia of the nose, paranasal sinuses, and nasopharynx.1,3 Most of the data available about the management and prognosis of LMS of SNT were obtained from published case reports due to the rarity of this finding. 1
Case report
Presentation and Medical History
A 84-year-old female patient presented with nasal obstruction on the left side. The obstruction had been gradually increasing over the last 6 months. She experienced several episodes of epistaxis on the affected left side, what finally encouraged her to seek medical care. She did not have a history of irradiation or familial history of malignancy. There was no occupational carcinogenic exposure such as wood dust or textiles. She had no other systemic disease and had never smoked.
Clinical Examination
A rhinoscopy revealed a polypous pale pink–colored mass totally occupying the left side of the nasal cavity.
Imaging Studies
A computed tomography (CT) of paranasal sinuses and nasal cavity (Figure 1A and B) shows a mass (4.3 × 2.5 × 2.6 cm3) in the left nasal cavity with bone destruction of the middle and inferior nasal turbinates. An infiltration of the left nasolacrimal duct and bone destruction in the anterior and posterior ethmoidal cells as well as the septum were detected for further characterization of the mass Houndfield units were determined (approximately 30 HU) which were compatible with mucous or soft tissue.
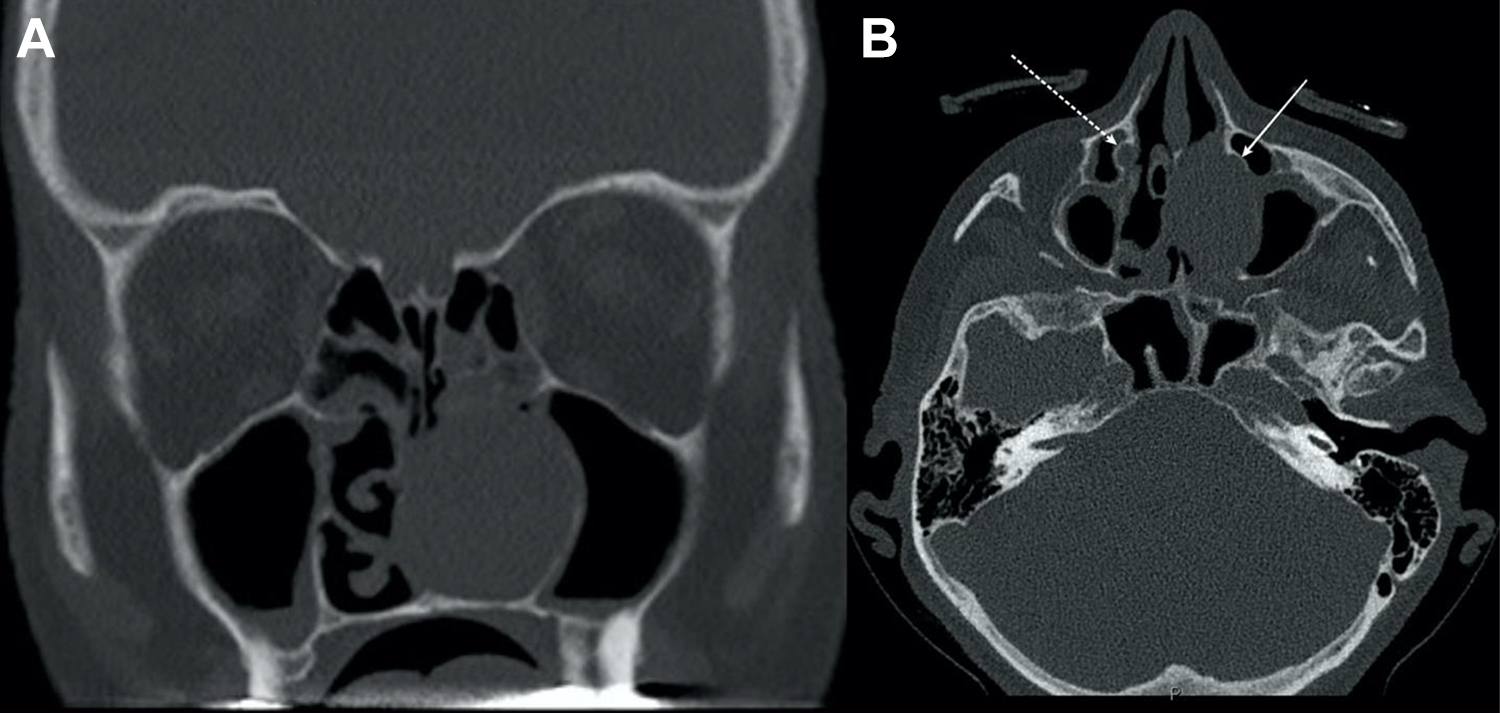
A and B, CT of paranasal sinuses and nasal cavity before the first FEES demonstrate a mass (4.3 × 2.5 × 2.6 cm3) in the left nasal cavity with bone destruction of the middle and inferior nasal turbinates. An infiltration of the left nasolacrimal duct (white straight arrow in B, the dotted arrow indicates normal nasolacrimal duct on the right side). CT indicates computed tomography.
Surgery, Staging, and Treatment
As the patient refused the suggested open surgery approach, an endoscopic endonasal approach with ethmoidectomy and maxillary sinus surgery was conducted to remove the mass. Intraoperative endoscopy of the nose revealed a non-pediculated tumor occupying the whole inferior nasal meatus to its cranial parts. After removing the 6 × 5 × 1.8 cm3 mass, the whole inferior and parts of the middle turbinate were noticed to be destructed by the mass. The lacrimal crest was destructed, and the lacrimal sac was free from bone. The anterior ethmoid cells were as well destructed by the neoplasia. We opened the nasolacrimal canal and removed suspicious tissue that was surrounding it. Furthermore, we took biopsies from the maxillary sinus, the ethmoidal sinus and the nasolacrimal duct to map the expansion of the tumor. The histopathology of the mass and the biopsy of the ethmoidal sinus was consistent with LMS with positive margins at the lacrimal portion. The other biopsies were free of any malignancy.
A staging with MRI (Figure 2A and B) of the head and neck, CT thorax, and abdomen besides ultrasound of the abdomen postoperatively showed no evidence of local or distant metastases.
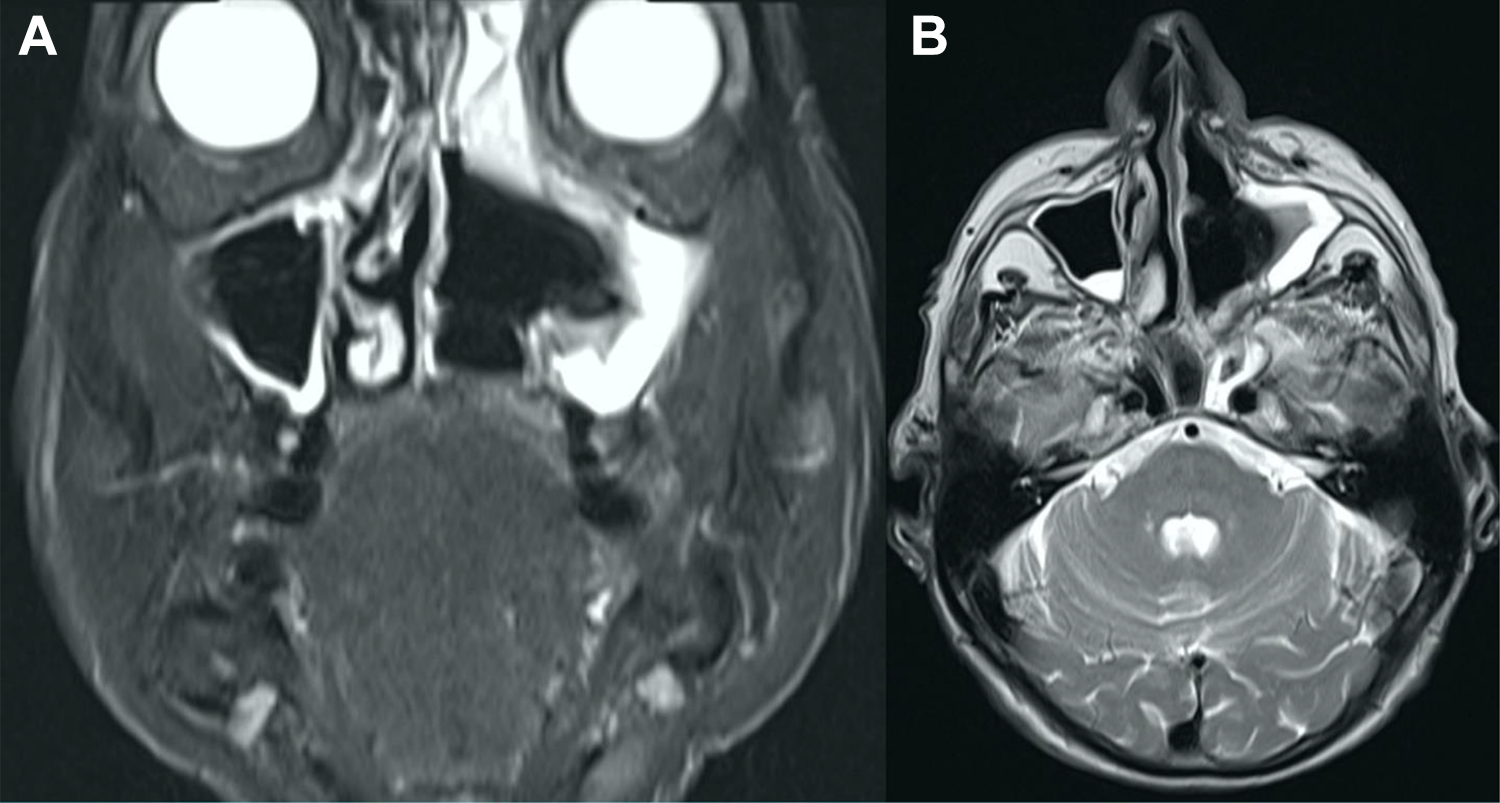
A and B (MRI coronal and axial views) demonstrates the resection defect and the mucosa swelling 1 week after the first endoscopic tumor resection.
A second endoscopic resection was done to radically remove the ethmoidal cells, the affected parts of the nasal turbinates and nasal septum, nasolacrimal duct, the lacrimal sac, and the suspicious tissue surrounding it. Besides that, we took multiple biopsies of the nasal septum, the anterior ethmoid, the posterior ethmoid, the maxillary sinus, and the lateral wall of the nose. The histopathology of the nasolacrimal duct and sac was consistent with LMS. The patient refused a third surgery to assure clear margins. She obtained adjuvant radiotherapy with entire dose of 52.5 Gy/64.5 Gy in 2 fields on the left side the paranasal sinuses and the periorbital medial, respectively.
Follow-Up
A clinical exam and MRI 9 months post-surgery showed no recurrence or metastases (Figure 3 A and B). The patient reported absence of the taste sensation and unclear visual impairment. These both could be attributed to the irradiation.4-6
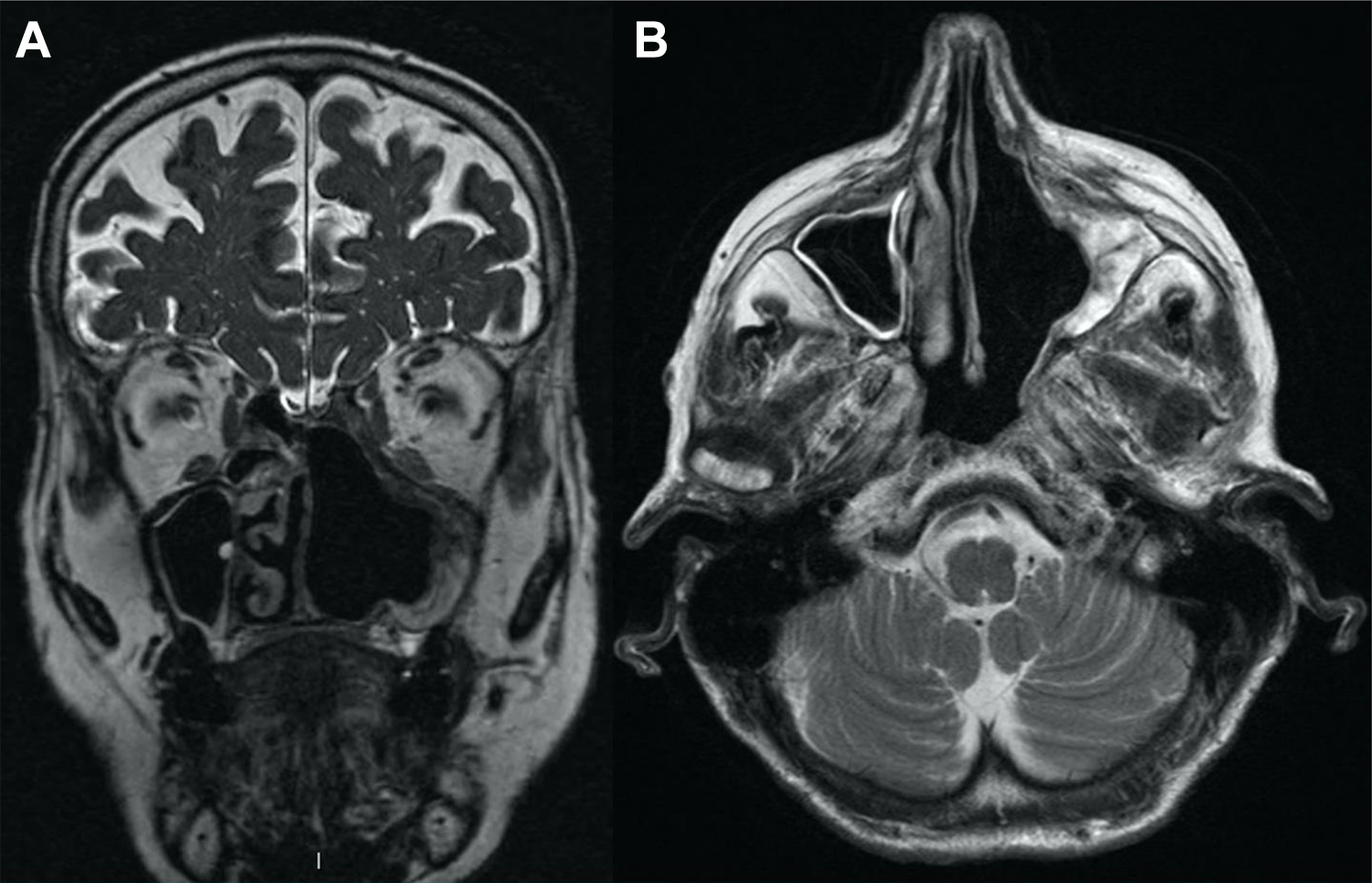
A and B (MRI coronal and axial views) demonstrates the follow-up on 9 months after the operation and the radiotherapy.
Discussion
Leiomyosarcoma is a malignity that is mostly located in the gastrointestinal tract, uterine, and retroperitoneum. 1 It is a scarcity to encounter it in the SNT. It contributes to only 2.3% of all non-epithelial neoplasia of the nose, paranasal sinuses, and nasopharynx.1,3 The most encountered localization of LMS within the SNT is the nasal cavity. 1 The origin of LMS in SNT is believed to be the tunica media in the blood vessels. 7 An association with occupational exposure to leather and textile dust, formaldehyde, chlorophenol, and cutting oils has been noticed.7,8 Another association with previous therapeutic irradiation and hereditary retinoblastoma has been well described.1,2,8
The presenting symptoms of LMS in the SNT depend on the localization of the LMS within the SNT. While involvement of the nasal cavity, as in our case, presents usually with unspecific symptoms like nasal stiffness and epistaxis, 7 involvement of the paranasal sinuses and skull base may present with a range from unspecific symptoms to sever functional impairment of the neighboring cranial nerves.1,9
Leiomyosarcoma tends to have aggressive local behavior characterized by local displacement of the neighboring structures rather than invasion.1,2 It metastasizes hematogenously and tends to be prone for local recurrence.1,2 The development of LMS in usual sites has been described in immunocompromised patients. 2
Computed tomography imaging of the paranasal sinuses will help determining the localization of the LMS and the involvement neighboring structures prior to surgical intervention. The definitive diagnosis is made by histopathology combined with immunohistochemistry.
Even though metastasizing is not very common in the LMS of SNT, 1 workup must include imaging studies involving the lung, brain, and liver as LMS metastasizes preferably to these organs.2,10 There is no agreed mainstay for the modality of imaging studies for LMS of SNT, but authors have used CT, MRI, and positron emission tomography imaging to define the presence of metastases.1,2,7,9 In our case, MRI of the head and CT of the sinuses was used to define the local involvement of the very tight intersecting bone and soft tissues in the head region. We excluded further metastases with CT-thorax-abdomen.
If the primary tumor is not operable, neoadjuvant chemotherapy to shrink the tumor followed by palliative surgery 1 or primary radiotherapy with or without chemotherapy has been tried.1,9,10 The mainstay of the therapy is excisional surgery with or without adjuvant radiotherapy if the primary tumor is amenable to the surgery.1,2,7,10,11 There is no clear recommendation for adjuvant radio-or chemotherapy though.1,10,11 In our case, we recommended the adjuvant radiotherapy because of the positive margins in the first surgery, the positive biopsy of the nasolacrimal duct and sac in the second surgery, the huge size of the tumor, and the anatomical localization of it, which does not allow reliable safety margins. If the endoscopic tumor excision is not expected to result in clear margins excision, we would always recommend choosing an open approach especially due to the better controlled removal of the nasolacrimal duct. However, in this case, the patient refused the open approach.
The factors that affect the prognosis of LMS of SNT are the size and localization of the tumor as well as the histological grade. The higher the grade the poorer the prognosis.1,2,10,11 The prognosis in general is poor.1,7 Eppsteiner et al reported in retrospective-based study on LMS of the head-neck region an 87.6% 5-year disease-specific survival for well-differentiated LMS and 52.7% for poorly differentiated LMS. 11
Ulrich et al reported a 38.24 months of disease-specific mean survival of 66% in a review of the LMS of SNT and skull base. 1 They did not stratify based on the grade or localization (presence of involvement of the skull base or not for example).
Conclusion
Leiomyosarcoma of the SNT is a rarity that must be included in the differential diagnosis of endonasal mass. Occupational exposure history, malignancy history, and irradiation history must be obtained due to the described association with LMS of the SNT.
The excisional surgery with or without adjuvant radiotherapy is the mainstay of the management. The indication for the adjuvant radiotherapy should be evaluated individually for each case as this can have sequels that decrease the quality of life for the patient. The prognosis is poor in general. Furthermore, detailed follow-up data of the patients over longer periods is needed to determine the survival.
Footnotes
Authors’ Note
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
