Abstract
Bilateral sensorineural deafness and unilateral cochlear ossification have rarely been described in patients with chronic myeloid leukemia (CML). A 21-year-old man presented to a hospital with right-sided sudden hearing loss and tinnitus. He was diagnosed with CML. Five days later, sudden hearing loss appeared in the other ear. Abnormality of the right-sided inner ear structure was revealed by preoperative magnetic resonance imaging; honeycomb-like cochlear ossification was observed during cochlear implant surgery in the right ear. The patient’s auditory performance exhibited significant improvement after bilateral cochlear implantation in our hospital. Hematological disorders must be considered in patients with sensorineural hearing loss. Cochlear implantation is feasible in patients with CML who exhibit sensorineural deafness, but cochlear ossification should be carefully evaluated by means of preoperative imaging examinations.
Introduction
Chronic myelogenous leukemia (CML) is a clonal myeloproliferative disorder that causes malignant transformation of hematopoietic stem cells. The fusion BCR-ABL oncogene, which results from reciprocal translocation between chromosomes 9 and 22, has been recognized as the molecular etiology of CML. 1,2 Fewer than 50% of patients with CML are symptomatic. 3 Common symptoms of CML include myeloid hyperplasia, leukocytosis, and splenomegaly. 4,5 Severe leukocytosis might result in blood hyperviscosity, causing reduction in microcirculation blood flow (ie, leukostasis). 1 Organ dysfunction secondary to leukostasis in patients with CML mainly involves brain, lung, heart, and kidney. 3 Otologic manifestations as initial symptoms (eg, sudden hearing loss, tinnitus, and vertigo) have been described in several patients with CML. 6,7 However, cochlear ossification has rarely been reported as a clinical symptom in a patient with CML. In this case report, we describe bilateral sensorineural deafness and unilateral cochlear ossification in a 21-year-old man with CML.
Case Report
A 21-year-old man presented to his local hospital with right-sided sudden hearing loss and tinnitus. Laboratory examinations showed white blood cell count of 626 × 109/L, hemoglobin level of 67 g/L, and platelet count of 358 × 109/L. Abundant mid-late immature granulocytic cells were observed upon microscopic examination of a peripheral blood smear. The results of bone marrow aspiration and biopsy, cytogenetic analysis, and quantitative reverse transcription polymerase chain reaction specific for the BCR-ABL fusion transcript confirmed the diagnosis of CML. Infiltration of the central nervous system (CNS) by hematologic or lymphoid malignant cells was not observed via cerebrospinal fluid cytology. After 2 days of oral hydroxyurea treatment (2 g, 3 times per day), the patient complained of sudden hearing loss on the left side. Bilateral profound sensorineural deafness was confirmed by audiological diagnosis. Subsequently, the patient received oral hydroxyurea treatment for an additional 6 days; he then received imatinib (0.4 g, once daily) for 8 months. The patient’s hearing performance did not improve and he was admitted to our hospital. Pure tone audiometry showed that the pure tone averages of the right and left ear for air conduction were 118.75 and 102.5 dB, respectively. Bilateral distortion product otoacoustic emissions were absent. A computerized tomography (CT) scan of the patient’s temporal bones showed no cochlear abnormalities. However, magnetic resonance imaging results revealed a reduced signal in the right-sided cochlear basal turn near the round window, which indicated abnormal cochlear structure (Figure 1). Two days after admission, the patient underwent right-sided cochlear implantation. Ossification of the round window membrane and honeycomb-like bone in the scala tympani of the cochlear basal turn were observed (Figure. 2). A cochlear-implant electrode was successfully inserted into the smooth scala tympani when part of the bone on the outer lateral wall of the cochlear basal turn had been removed; all honeycomb-like bone was removed from the scala tympani. Five months later, cochlear implantation was successfully performed in the patient’s left ear. The patient’s mean hearing threshold was 30 to 40 dB after both operations had been completed.
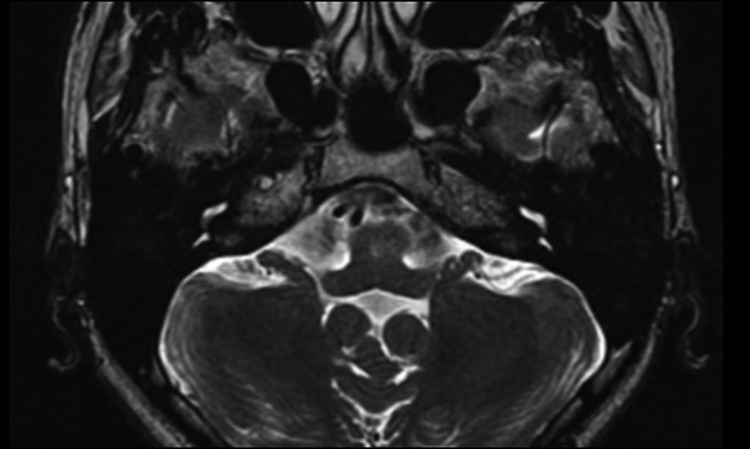
Inner ear magnetic resonance imaging (MRI) with strongly T2-weighted sequences and high spatial resolution depicted a reduced signal in the right-sided cochlear basal turn near the round window.
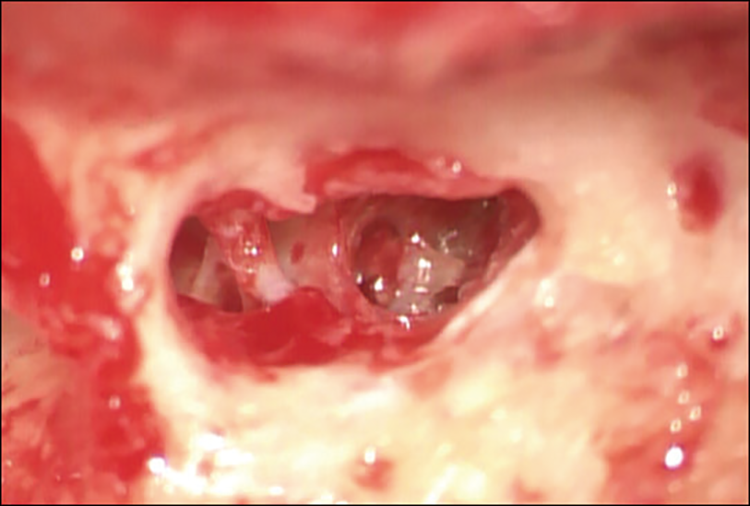
Honeycomb-like bone was found in the scala tympani of the cochlear basal turn during right-sided cochlear implantation.
Discussion
Otologic manifestations affect 15% to 40% of patients with leukemia. 1 The pathogenesis of leukemic otologic symptoms appears to be complex and involves multiple mechanisms: leukemic infiltration, hemorrhage of middle and inner ear, leukostasis, or infections. 9 Higher numbers of white blood cells are unable to deform adequately to traverse the microcirculation; thus, they are likely to form obstructive thrombi. The accumulation of leukemic white blood cells increases oxygen consumption, thereby contributing to localized hypoxia and infarction. 10 The inner ear receives blood supply from the labyrinthine artery; temporary stasis of leukocytes is sufficient to cause permanent damage to the delicate inner ear structures. 11 In our patient, the white blood cell count findings supported the diagnosis of leukostasis. Notably, CNS infiltration can be successfully treated by CNS-directed chemotherapy followed by allogenetic stem cell transplantation, whereas sensorineural hearing loss does not respond to this therapy. 12 In our patient, no CNS infiltration was observed; his hearing performance was not improved by hematological remission, suggesting irreversible inner ear damage due to leukostasis. However, current evidence is insufficient to determine the cause of the patient’s otologic symptoms; future histological studies in other patients are required to identify this etiology.
Our patient presented with right-sided hearing loss and tinnitus as initial manifestations of CML. He was diagnosed with CML based on observations of hearing impairment and tinnitus. Our findings highlight the need for workup regarding the underlying systemic causes of unilateral hearing loss. In addition, a reduction of signal in cochlear structure was observed by preoperative imaging examinations, while unilateral cochlear ossification was confirmed during cochlear implantation. Cochlear ossification as a manifestation of CML has rarely been reported. Labyrinthine artery occlusion and inner ear infection are well-recognized risk factors of cochlear ossification. 8,13,14 Therefore, we speculate that inner ear infection or leukostasis might be involved in the formation of cochlear ossification, although additional evidence is needed to identify the specific etiology.
Cochlear implantation, a well-established method for auditory restoration, is feasible in most patients with cochlear ossification. 13,15 However, cochlear ossification affects the process of cochlear implant surgery and creates challenges in cochleostomy. 13,16 Preoperative imaging examinations are essential for surgeons when planning cochlear implantation. 17,18 Magnetic resonance imaging indicated a reduction of signal in the right-sided cochlear basal turn. However, CT did not reveal clinically significant anatomical abnormalities in the inner ear; cochlear fibrosis and early-stage ossification might have interfered with diagnosis based on CT scan findings. 19 Therefore, our findings suggest that the possibility of cochlear ossification should be carefully evaluated in patients with CML who exhibit sudden hearing loss. Despite the surgical challenges, cochlear implantation was successfully performed in our patient. Improvement in terms of auditory performance suggested that the patient’s central auditory pathways were not impaired.
In conclusion, hematological diseases should be assessed in patients with sudden hearing loss. Potential intracochlear abnormalities should be carefully evaluated before cochlear implantation is performed in patients with CML who exhibit sensorineural deafness.
Footnotes
Acknowledgment
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Guangdong Province, China (General Program: 2020A1515010237); Shenzhen Key Medical Discipline Construction Fund (No.SZXK039); and Shenzhen Science and Technology Innovation Commission, China (Free Exploration Projects: JCYJ20180305163353862 and JCYJ20180305163259711).
