Abstract
Basaloid squamous cell carcinoma and large cell neuroendocrine carcinoma are not common in head and neck, these tumors rarely occur in the larynx but both have highly aggressive clinical behavior and a high mortality rate. The diagnosis is complicated by these tumors’ atypical clinical and pathological features. This case details a coexistence of basaloid squamous cell carcinoma and large cell neuroendocrine carcinoma of a woman in the larynx. The patient underwent endoscopy- and coblation-assisted transoral microsurgery to achieve hyoid horizontal epiglottidectomy and has no recurrence after 12 months of follow-up.
Introduction
Two histologically distinct tumors do not frequently coexist in the same anatomical site. Basaloid squamous cell carcinoma (BSC) is a rare cutaneous cancer, the pathological characteristics of BSC tend to reflect a subtype of basal cell carcinoma, although BSC has a higher likelihood of recurrence and greater potential for metastasis than squamous cell carcinoma. 1 Large cell neuroendocrine carcinoma (LCNEC) was previously considered an atypical carcinoid tumor, 2 which is usually associated with rapid growth, a poor prognosis, and a high mortality rate. These 2 tumor types are rarely involved in the larynx and often misdiagnosed. We report the case of a middle-aged woman who presented with a neck mass, but no esophageal or nasal symptoms. Careful immunohistochemical evaluations ultimately supported a diagnosis of coexistence of BSC and LCNEC in the larynx.
Case Presentation
A 54-year-old woman presented with a 6-month history of a rapidly swelling neck mass, albeit without any complaints regarding dysphagia, hoarseness, or nasal congestion. The patient had a history of smoking 30 to 40 cigarettes per day, but reported no alcohol consumption. A physical examination revealed metastatic neck lymph nodes on both sides (the left-side mass was the largest, 5.2 cm × 4.1 cm), but no evidence of distant metastasis. Enhanced magnetic resonance imaging of the neck and larynx revealed thickening of the laryngeal side of the epiglottis, as well as several enlarged lymph nodes in both sides of the neck (Figure 1A and B). Laryngoscopic examination also revealed an irregular mass on the laryngeal side of the epiglottis (Figure 1C).
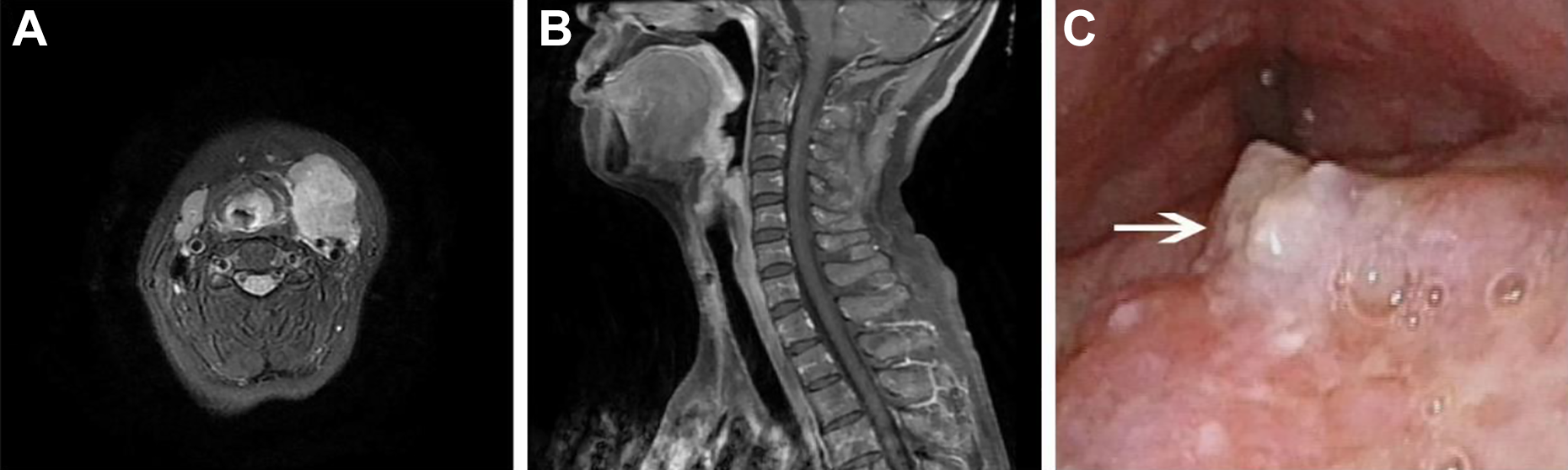
Axial (A) and sagittal (B) enhanced magnetic resonance imaging revealed thickening of the epiglottis, contrast enhancement at the epiglottis, left-side contrast-enhanced fused lymph nodes (5.2 cm × 4.1 cm), and right-side fused lymph nodes (2.2 cm × 1.0 cm). (C) Laryngeal endoscopy revealed a bulging lesion at the laryngeal side of the epiglottis, with observable necrosis on the tumor surface (white arrow).
A biopsy specimen was taken from the epiglottic lesion and the pathological examination revealed severe atypical hyperplasia of the squamous epithelium. A second biopsy was taken, and the results confirmed the presence of squamous cell carcinoma (cT1N2M0). The patient underwent endoscopy- and coblation-assisted transoral microsurgery to achieve hyoid horizontal epiglottidectomy. Intraoperative evaluation revealed that the lesion was confined to the epiglottis, although fused lymph nodes on the left side were closely adhered to the left internal jugular vein. Thus, we simultaneously performed radical left neck dissection and selective right neck dissection via the external cervical approach and without tracheotomy. The postoperative pathological diagnosis was coexistence of LCNEC and BSC, and the tumor consisted of small crowded cells and large malignant cells (Figure 2A and B) that invaded the epiglottis cartilage.
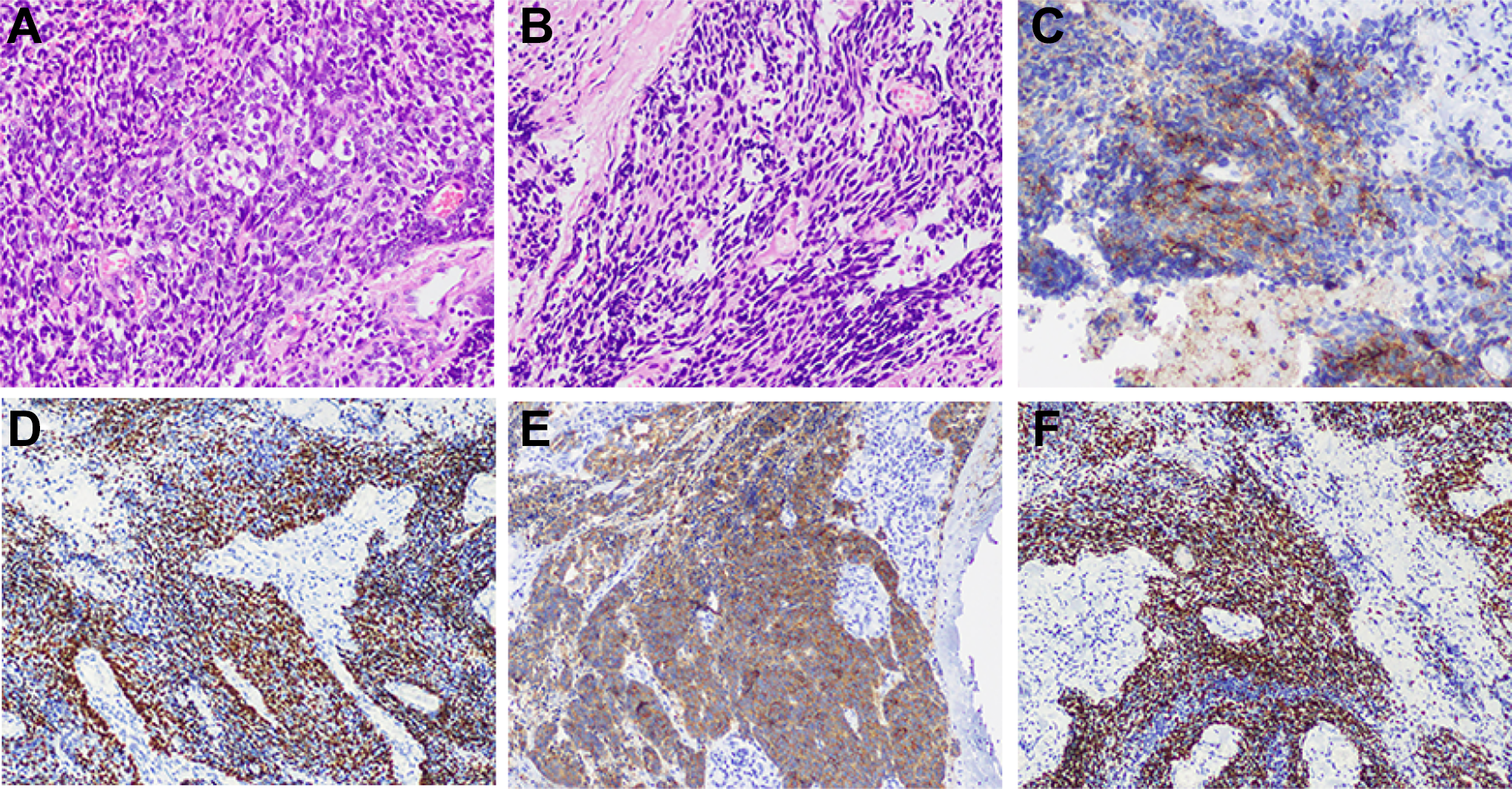
(A) A microphotograph revealed a large cell neuroendocrine carcinoma component with prominent nucleoli and high mitotic activity (hematoxylin and eosin, ×200). (B) A microphotograph revealed an invasive basaloid squamous cell carcinoma component (hematoxylin and eosin, ×200) that consisted of small crowded cells with minimal cytoplasm and hyperchromatic nuclei. Immunohistochemical staining revealed that the large cancer cells were positive for CD56 (C) and synaptophysin (E), while the small cells were positive for P63 (D) and P40 (F).
Immunochemistry Findings
A further immunohistochemical evaluation revealed that the LCNEC component was positive for cluster of differentiation 56 (Figure 2C), P63 (Figure 2D), synaptophysin (Figure 2E), and chromogranin A. The BSC component was positive for P63 (Figure 2D), P40 (Figure 2F), and cytokeratin 5/6. The tumor exhibited a high Ki-67 index (80%) but no expression of Epstein-Barr virus-encoded small RNAs. Pathological evaluation of the left lymph node revealed only metastasis of the LCNEC component.
Subsequent Treatment
The patient recovered well and exhibited no signs of wound infection at her discharge. However, 1 month later, the patient developed progressive dyspnea and endoscopic examination revealed laryngeal edema, which prompted us to perform tracheotomy. Given the high malignant potential of BSC and LCNEC, the patient was also treated using adjuvant chemotherapy (2 cycles of paclitaxel [175 mg/m2] plus cisplatin [90 mg/m2]) and local radiotherapy (63 Gy to the primary site and 66 Gy to the bilateral neck areas in 30 fractions). The patient has no signs of recurrence at 12 months after the surgery.
Discussion
Basaloid squamous cell carcinoma is a rare tumor with histopathological characteristics of both basal cell carcinoma and squamous cell carcinoma. 3 During the early stage, BSC can mimic a benign lesion or conventional squamous cell carcinoma; thus, BSC has unclear histological and behavioral characteristics that can confound a diagnosis. 4 Although BSC requires careful attention because of its potential for rapid growth, invasion, and distant metastasis. 5 Given these challenges, large deep biopsies are recommended because the features of BSC may only be present in the deep lesion layers near the bone, nerves, or blood vessels. 6 In the present case, the initial pathological examinations were unable to support a clear diagnosis, as the tumor’s location made it difficult to obtain a subsurface specimen. Therefore, a further pathological examination is often necessary to distinguish BSC from basal cell carcinoma with local aggressive features. 7
The literature indicates that complete tumor resection plays an important role in reducing local recurrence. For example, micrographically oriented histographic surgery (Mohs surgery) can reduce the rate of recurrence to 4.1% of cases. 4 Chemotherapy and/or radiotherapy are potential adjuvant options for managing BSC,2,8 but patients with late-stage BSC do not seem to respond to surgery and/or radiotherapy in most cases. 9 Although there is no current standardized treatment protocol for BSC, 10 the 5-year overall survival rate is only 48%, 8 and recurrence may be related to positive surgical margins, tumor diameter of >3 cm, and perineural, lymphatic, or bone invasion. 7 Wermker et al 1 analyzed the characteristics and outcomes of 89 primary BSC cases involving the head and neck and reported that local recurrence tended to be associated with vascular invasion, muscle invasion, bone infiltration, and advanced T classification (T3 or T4). Furthermore, local tumor recurrence was significantly associated with the maximum depth of vertical infiltration, tumor depth, and incomplete resection status. Therefore, local recurrence or regional lymph node metastasis from head and neck BSC appear to be related to incomplete tumor resection, larger tumor size, greater tumor depth and vertical infiltration, muscle invasion, and vascular invasion.
The 2017 World Health Organization classification of head and neck tumors lists 3 types of head, neck, and laryngeal neuroendocrine cancers. Laryngeal LCNEC has recently been reclassified as a grade III subtype of poorly differentiated neuroendocrine carcinoma, along with small-cell neuroendocrine carcinoma. 11 Smoking seems to be associated with the development of these large cell carcinomas. Although LCNECs are rare, they are extremely aggressive and have a high mortality rate, 12 and a meta-analysis of 29 LCNEC cases revealed a 5-year disease-specific survival rate of only 15%. 13 Moreover, most patients develop distant metastatic disease and die within 2 years. 14 However, LCNEC is usually difficult to diagnose and requires careful cytological, morphological, and immunohistochemical examination. 15
There is also no established treatment for LCNEC, although the literature suggests that complete resection is appropriate for early-stage tumors. 15 However, surgery alone does not appear to be sufficient, given the high risk of local recurrence and/or distant metastasis, 16 and the preferred adjuvant therapy consists of chemoradiotherapy.2,13 Adjuvant chemotherapy using platinum plus etoposide may prevent recurrence in patients with completely resected LCNEC, although it remains unclear whether advanced-stage LCNEC responds to chemotherapeutic regimens in a manner similar to small-cell lung cancer or non-small cell lung cancer. 17 The rapid enlargement of the cervical lymph nodes in the present case may be related to the highly malignant pathological type.
Simultaneous occurrence of BSC and LCNEC in the larynx is rare and difficult to diagnose. Both of these tumors are aggressive and prone to metastasis at an early stage, and an accurate diagnosis is complicated by the atypical clinical and pathological features. It appears that complete tumor resection and adjuvant chemoradiotherapy may be a potentially curative strategy in these cases, as our patient remains without signs of recurrence at 12 months after surgery, although long-term follow-up is clearly needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
