Abstract
Sinonasal lymphoepithelial carcinoma (SLEC) is an extremely rare malignancy. We present a case of SLEC in a 77-year-old man who presented with nasal congestion and persistent sinusitis. Imaging revealed a large right nasal mass involving right paranasal sinuses along with bulky bilateral cervical lymphadenopathy. In addition, there was a fluorodeoxyglucose avid L1 vertebral lesion. Biopsy of nasal mass and cervical lymph nodes showed syncytial growth of tumor cells in a lymphoplasmacytic background. Immunohistochemical stains showed positivity for pankeratin, CK5/6, epithelial membrane antigen, p40 (focal), and p63 (focal). An Epstein-Barr virus-encoded RNA by in situ hybridization was strong and diffusely positive. Based on these pathologic findings and considering the location of tumor, diagnosis of SLEC was rendered. L1 vertebral body lesion was clinical and radiologically considered to be a metastasis. Correlation with radiology to determine the exact location of tumor is extremely important for correct diagnosis due to its histopathologic similarities with relatively more common undifferentiated type of nasopharyngeal carcinoma. No standard treatment protocol has been established for this tumor yet. To our knowledge, this is first ever report of SLEC presented with clinical stage IV disease.
Introduction
Sinonasal lymphoepithelial carcinoma (SLEC) is a rare malignancy with approximately 40 cases documented with over 90% of lesions positive for the Epstein-Barr virus (EBV).1,2 Sinonasal lymphoepithelial carcinoma is found worldwide but is most common in Southeastern Asia.2-5 The World Health Organization defines SLEC as “a squamous cell carcinoma morphologically similar to nonkeratinizing nasopharyngeal carcinoma, undifferentiated subtype”. 2 Most of the cases usually present with locally aggressive disease with or without regional lymph node metastases. Distant metastases has not been reported at the time of presentation. 2 Sinonasal lymphoepithelial carcinoma typically responds well to radiation therapy with or without surgery and/or chemotherapy.6-8 Radiation is recommended even when there is lymph node involvement. 8 In this report, we present a unique case of SLEC that presented with distant metastasis. We reviewed the pathological features, immunohistochemistry, discussed important differential diagnosis, and treatment options available based on review of previously published cases.
Case Report
A 77-year-old Caucasian man with a remote history of smoking presented with sinusitis and was treated with amoxicillin-clavulanic acid. Later that month, he presented again with protracted cough associated with sinus congestion, pressure, and discomfort. At this time, he was diagnosed with right lower lobe pneumonia and treated with cefdinir for 10 days. Chest X-ray at this time demonstrated bronchial wall cuffing at the lung bases and probable chronic obstructive pulmonary disease. Despite the aforementioned antibiotics, the patient did not have resolution of his sinusitis and was now treated with levofloxacin and prednisone. Despite completing the prescription regimen(s), the patient reports no improvement in his symptoms including nasal congestion, yellow drainage, and pain/pressure in the left cheek.
Subsequently, the patient underwent nasal endoscopy which demonstrated a large tumor in the right nasal cavity arising from the medial to middle turbinate and obstructing the posterior nasal cavity. There was thick mucus draining around the tumor. The tumor was clinically suspected as being an inverted papilloma. Computed tomography scan and magnetic resonance imaging (Figure 1) demonstrated a right sinonasal mass with obstruction of the right maxillary sinus, right sphenoid sinus, right ethmoid air cells, and the right frontal sinus. Bulky bilateral cervical lymphadenopathy was also noted. Furthermore, Positron emission tomography scan suggested a possible metastasis to the L1 vertebral body.
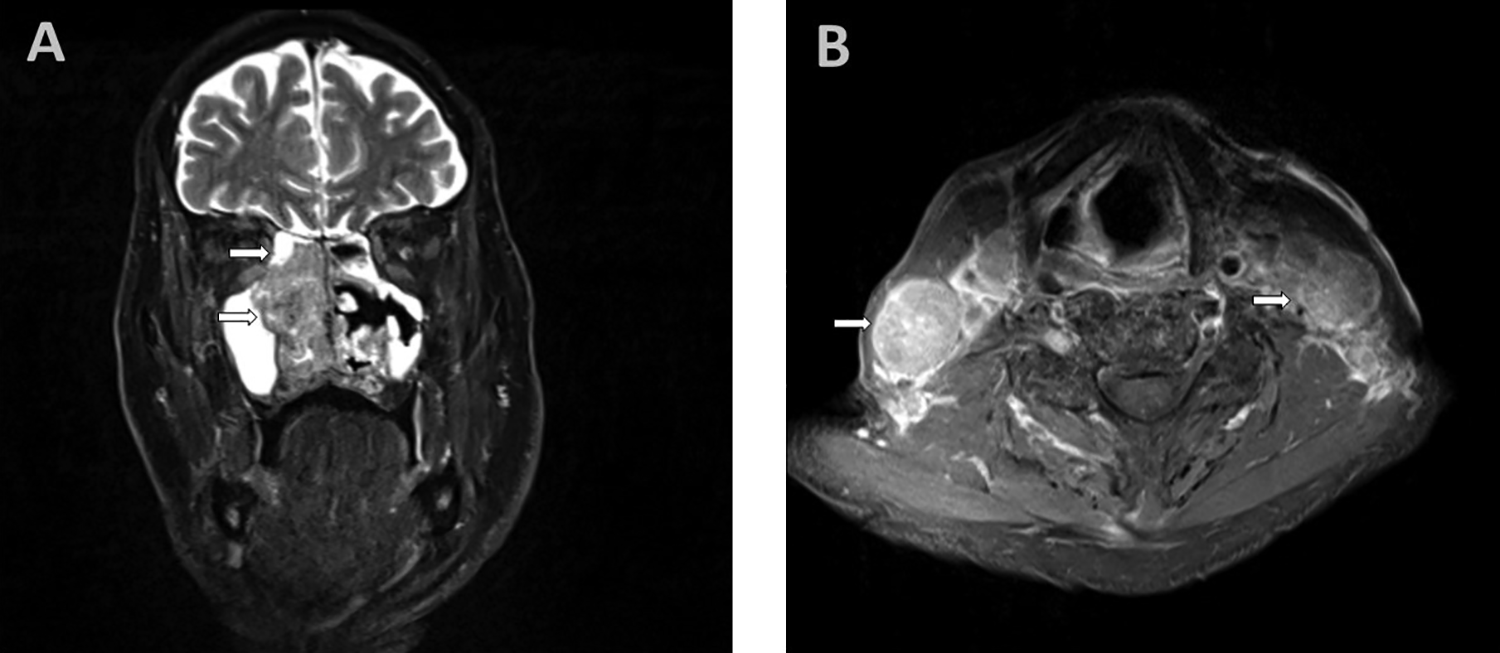
A, Right sinonasal mass (arrow) involving mid and posterior right nasal cavity with some involvement of ethmoid air cells and right maxillary sinus. (MRI T2, coronal section). B, Bulky bilateral lymphadenopathy (arrows; MRI T2, axial section). MRI indicates magnetic resonance imaging.
The patient was subsequently taken to the operating room wherein 3 superficial left posterior triangle lymph nodes were removed. Subsequently, a biopsy of the right nasal mass was also obtained. Histopathological examination (Figure 2) demonstrated sheets and nests of tumor cells separated and overrun by significant lymphoplasmacytic infiltration in some areas, whereas there was more fibrotic background in other foci. Nuclei of tumor cells were large round to oval with vesicular chromatin and prominent nucleoli. Foci of necrosis were seen (Figure 2C). Immunohistochemical study (Figure 3) showed tumor cells that stained positive for pankeratin, CK5/6, and epithelial membrane antigen (EMA). There was focal positive staining for p40 and p63. Epstein-Barr virus-encoded RNAs (EBER) staining via in situ hybridization was strong and diffusely positive. After pathology–radiology correlation, it was concluded that bulk of tumor is in the sinonasal area and therefore a diagnosis of SLEC was rendered. Left neck lymph node tissue was completely replaced by metastatic lymphoepithelial carcinoma (LEC). The metastatic deposit was 4.0 cm. No definitive evidence of extranodal extension was seen.
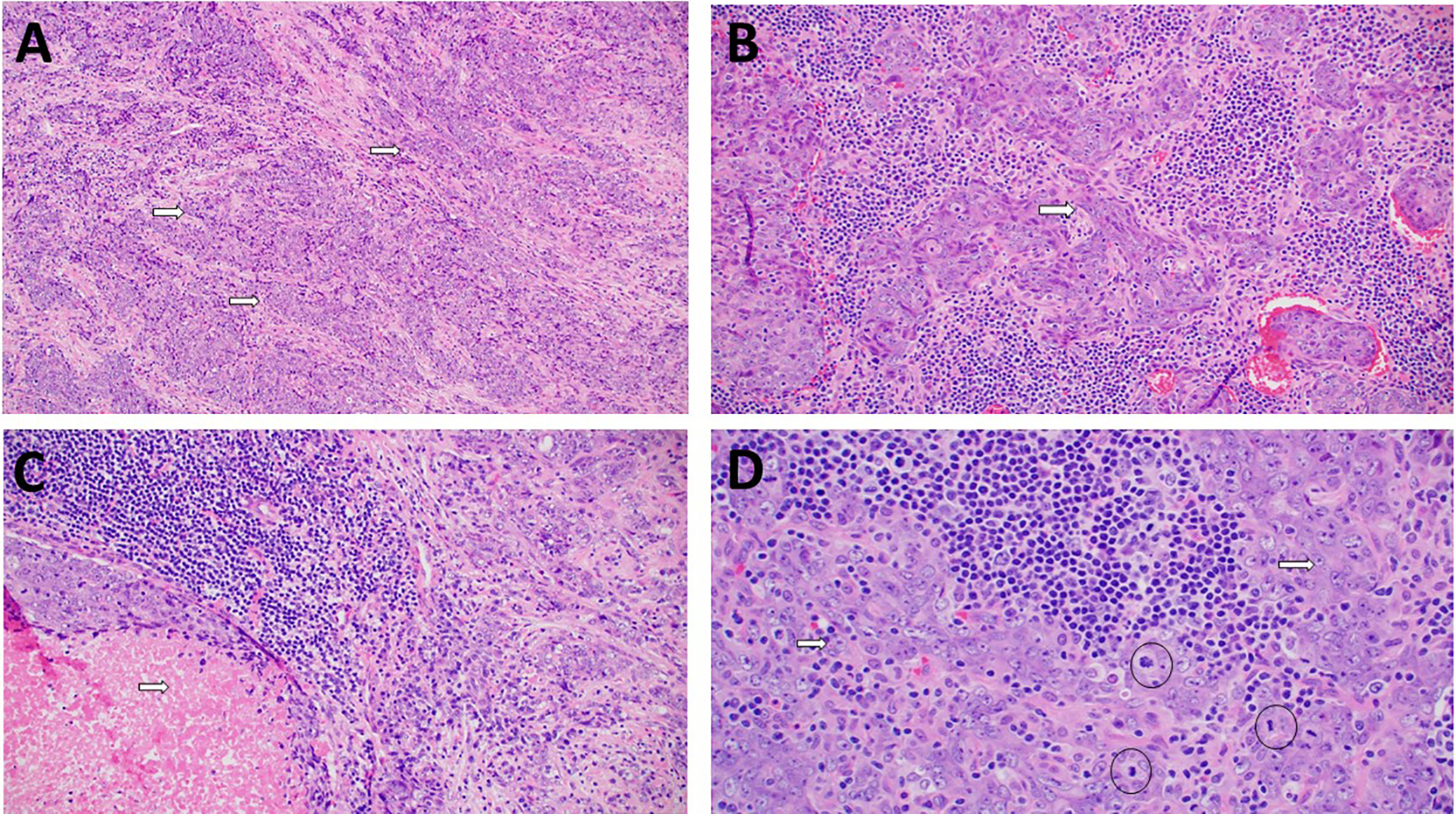
A, Cohesive clusters of tumor cells (arrows) in a more fibrotic background. (hematoxylin and eosin, magnification ×200). B, There are focal areas with fasicular growth with spindle shaped cells (arrow) in a lymphoplasmacytic background (hematoxylin and eosin, magnification ×200). C, Foci of necrosis (arrow) were identified (hematoxylin and eosin, magnification ×200). D, High power view of tumor showing large vesicular nuclei, prominent eosinophilic nucleoli (arrows) with indistinct cell borders creating a syncytial growth. Few mitotic figures (circles) are also present (hematoxylin and eosin, magnification ×400).
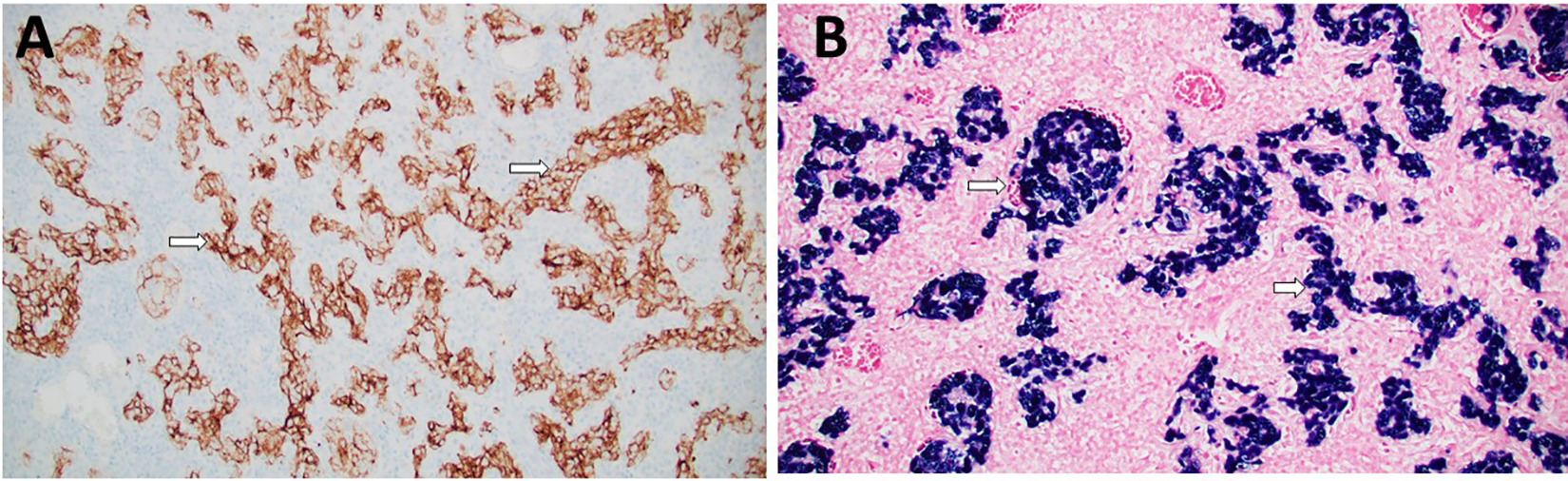
A, The lesion cells are positive (arrows) for CK5/6 immunohistochemical stain. (CK5/6 antibody, magnification ×200). B, In situ hybridization for Epstein-Barr-encoded RNA (EBER) is strong and diffusely positive (arrows) in tumor cells (EBER in situ hybridization, magnification ×200).
Due to the metastatic nature (clinical stage IVC T4a N2c M1) of the patient’s disease, confirmed metastases to the cervical lymph nodes and presumptive metastasis to the lumbar spine, definitive surgery and chemoradiation were deferred at this time in lieu of induction chemotherapy. Hematology–oncology recommend completing 2 to 3 cycles of cisplatin and docetaxel with subsequent scans. Radiotherapy was deferred until the patient’s response to chemotherapy can be assessed. Patient showed excellent radiographic response after 2 cycles of induction chemotherapy with cisplatin and docetaxel (Figure 4). He then refused additional chemotherapy. PD-L1 testing by immunohistochemistry was performed and showed 40% of tumor cells positive (membranous staining) using combined positive score. Therefore, he is currently undergoing Keytruda treatment.
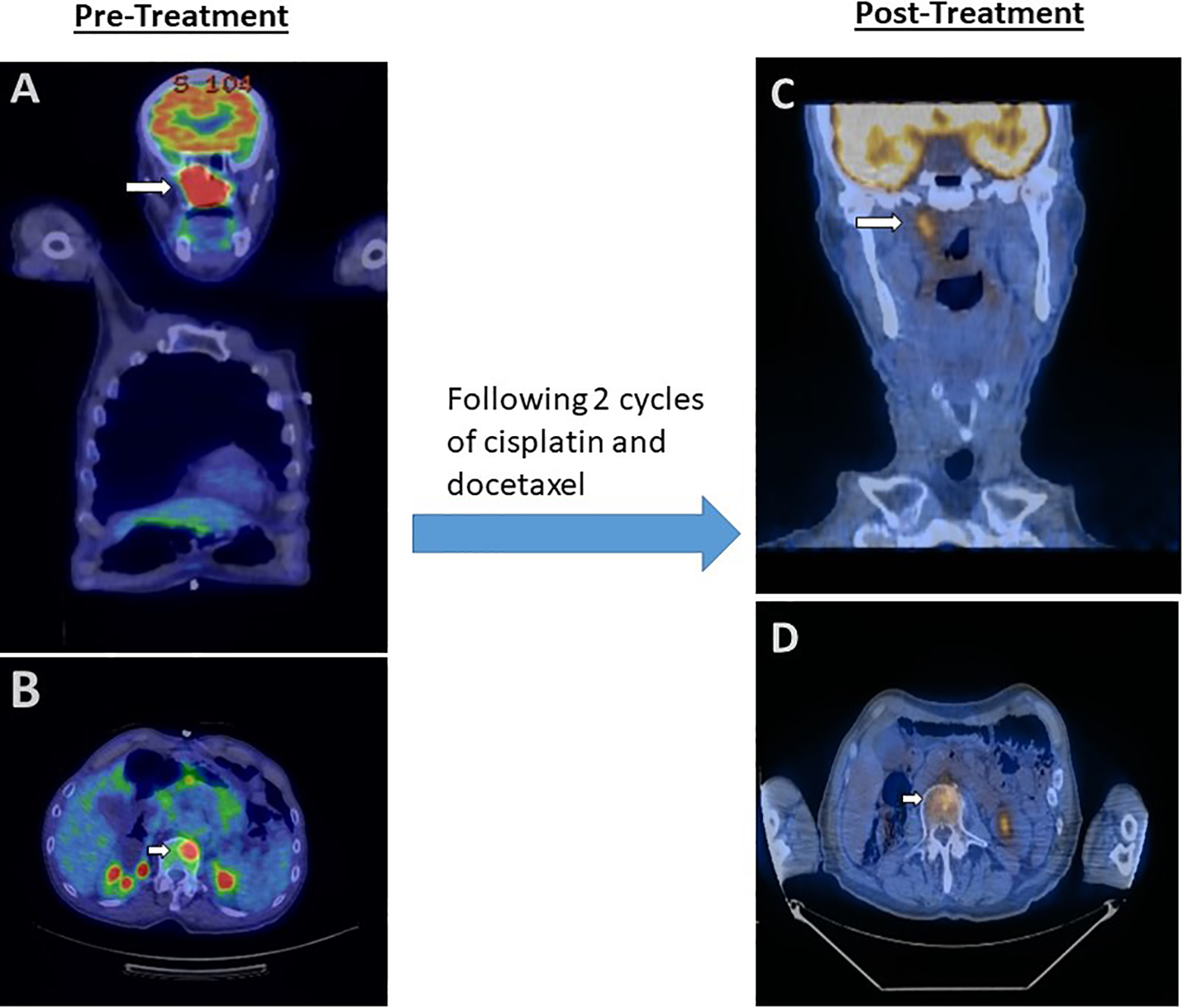
A. Pretreatment sinonasal mass showing increased FDG uptake (arrow) with an SUV of 11.3. (PET scan, coronal section). B. Pretreatment L1 vertebral body metastasis with FDG uptake (arrow) with an SUV of 9.4. (PET scan, axial section). C and D, Treatment with cisplatin and docetaxel showed significant reduction in tumor size and FDG uptake with a maximum SUV of 4.3 in sinonasal region and 4.1 in vertebral metastasis (PET scan, coronal section, and axial section, respectively). FDG indicates fluorodeoxyglucose; PET, Positron emission tomography; SUV, standard uptake value.
Discussion
Lymphoepithelial carcinoma was first described by Schminke and Regaud in 1921.9,10 In the head and neck region, LEC frequently occurs in the nasopharynx, salivary glands, and larynx. However, it occurs extremely rarely in the sinonasal area. Approximately, 40 documented cases have been reported so far with 7 cases in the last 20 years (Table 1). 2 It has a strong predilection for men in the fifth to seventh decades of life. 2
Review of Clinic-Pathologic Features and Follow-Up of Cases With Sinonasal Lymphoepithelial Carcinoma Reported in Last 20 Years.
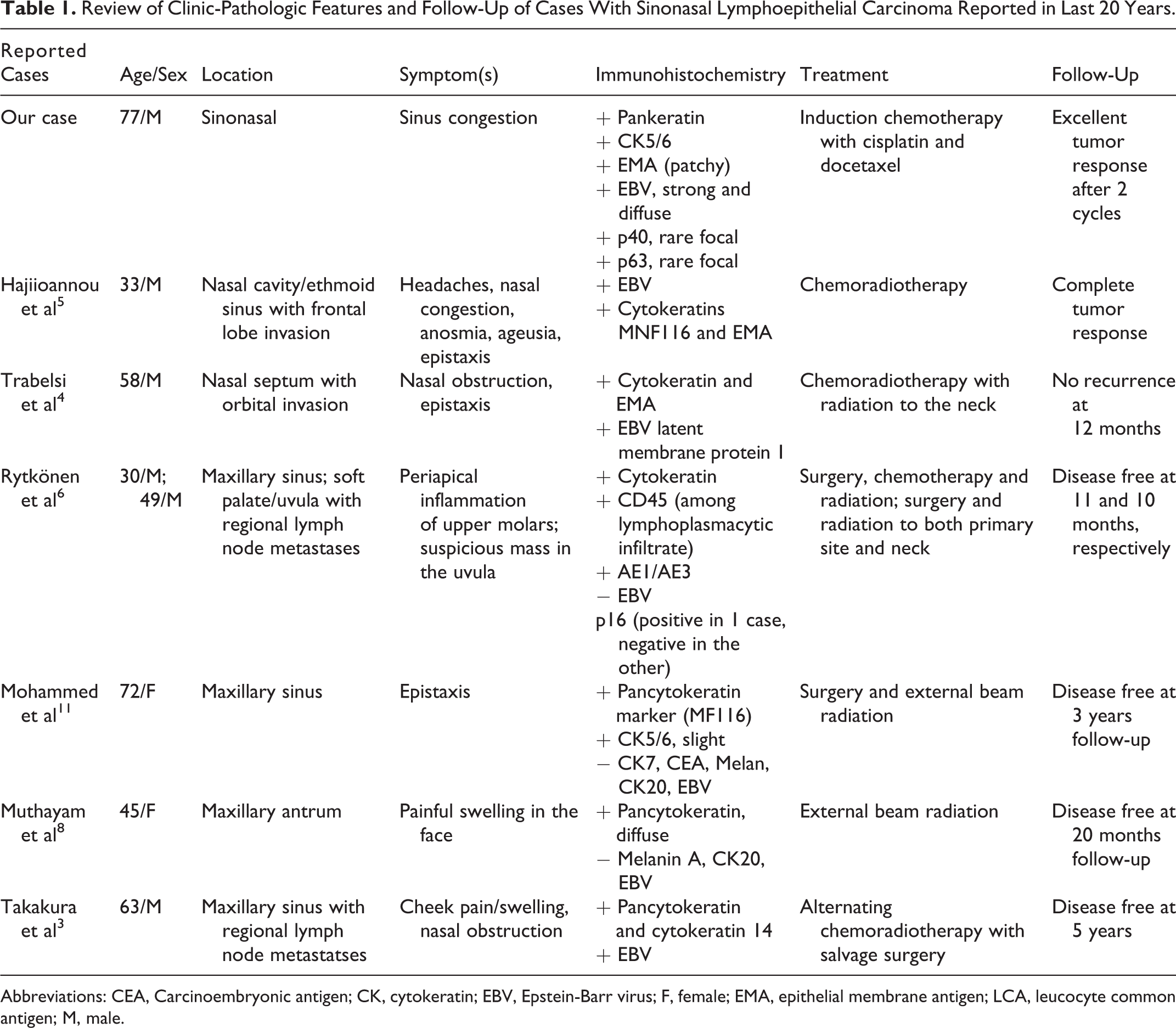
Abbreviations: CEA, Carcinoembryonic antigen; CK, cytokeratin; EBV, Epstein-Barr virus; F, female; EMA, epithelial membrane antigen; LCA, leucocyte common antigen; M, male.
Sinonasal lymphoepithelial carcinomas more commonly occur in the nasal cavity than the paranasal sinuses. 12 The majority of cases present with nasal obstruction, epistaxis, facial pain and, in the presence of invasive growth, proptosis and cranial nerve palsies.3-5,11,12 One case in our literature review also presented with anosmia and ageusia, which is due to invasion of tumor to the olfactory bulb, frontal lobe, and probably brain stem. 5
As compared to nasopharyngeal carcinoma wherein almost all cases involve EBV, SLECs are associated with EBV in approximately 90% of cases. 2 In our literature review, 3 of the 7 published cases with SLEC were negative for EBV.6,8,11 Smoking, chewing tobacco, human papillomavirus, and alcoholism do not seem to be associated with LEC. 13
Nasopharyngeal carcinoma, undifferentiated type shares the same histopathologic features as SLEC. Some authors have reported a relatively less prominent lymphoplasmacytic infiltration in SLEC which we noticed in our case as well (Figure 2A and C). 14 However, the main distinction relies on tumor location. In our case, the bulk of the tumor was located in the right nasal cavity, maxillary sinus, sphenoid sinus, ethmoid sinus, and frontal sinus with a subtle thickness of the nasopharyngeal mucosa. After discussion with clinical and radiology providers, it was concluded that the probable origin of the tumor was in the sinonasal mucosa leading to the diagnosis of SLEC.
Immunohistochemical analysis is not very helpful in distinguishing SLEC from nasopharyngeal carcinoma as both are positive for pancytokeratin, CK5/6, p40, p63, and usually positive for EBER by in situ hybridization. 2 Since a small number of cases with SLEC are sometimes negative for EBV, the absence of EBER can be of some assistance in an LEC of indeterminate origin in distinguishing it from nasopharyngeal carcinoma.
Other differential diagnoses of this tumor include lymphoma, melanoma, and sinonasal undifferentiated carcinoma (SNUC). Sinonasal lymphoepithelial carcinomas show an absence of immunoreactivity for hematolymphoid markers, melanoma-related markers, and neuroendocrine/neuroectodermal markers. Sinonasal lymphoepithelial carcinoma also typically stains positive for EMA but not for synaptophysin.2,7
Sinonasal undifferentiated carcinoma is a highly aggressive neoplasm and is usually considered to be a diagnosis of exclusion. Histologically, it has increased mitotic activity and necrosis compared to LEC.7,15,16 Additionally, SNUC is not associated with EBV.4,6,15,16
Mucosal melanomas can be distinguished from SLEC in that they usually lack syncytial growth, vesicular nuclear chromatin and, instead, tend to have a plasmacytoid appearance. 7 Melanoma markers such as SOX10, S100, HMB-45, Melan A, tyrosinase, and MITF-1 are further helpful in distinguishing these tumors from SLEC. 2
Extranodal natural killer/T-cell lymphoma (ENKTL) shares similarities with SLEC including high association with EBV and often presents with nasal obstruction and/or epistaxis. 2 However, ENKTL often stains positive for CD3 and CD56 while stains negative for cytokeratin.2,14 In cases where a lymphoproliferative process is suspected, a separate specimen for flow cytometry should be submitted for adequate evaluation.
Due to the limited number of cases, there is no standardized treatment policy reported to date for SLEC. 11 However, LEC is known to be radiosensitive.2-4,7,8,11 Yet, the initial treatment for maxillary sinus tumors has traditionally been surgery. In our literature review, cases that were limited to the sinuses received surgery with radiation; whereas the cases with regional lymph node metastases, orbital or brain invasion, at the time of presentation received chemotherapy in addition to radiation (Table 1). Takakura et al 3 suggested alternating chemoradiotherapy with 5-fluorouracil and nedaplatin followed by salvage surgery as an effective treatment for SLEC. Nonetheless, more research data are needed regarding treatment of this tumor in order to establish a standardized treatment protocol. Treatment response has been reported to be very good with recently published cases showing complete response with no evidence of disease recurrence up to 5 years of follow-up.2-4,7,8,11 Our case is unique in that our patient presented with clinical vertebral metastases along with regional lymph node metastases. In light of these metastases, it was decided after a tumor board discussion to start the patient on chemotherapy while radiotherapy and surgery were deferred.
Overall, prognosis of SLEC is favorable for patients with localized disease with a 5-year survival rate of approximately 78% and progression free survival rate of 69%.2,3 The presence or absence of EBV does not appear to offer prognostic value in diagnosing LEC of the maxillary sinus; rather its clinical utility is in diagnosing tumors that are suspected as being metastases. 8
Conclusion
To our knowledge, this is the first report of an extremely rare case of SLEC presented with stage IV disease (distant metastasis to L1 vertebra). Owing to its histopathologic similarities with its nasopharyngeal counterpart, correlation with clinical and radiologic findings to determine the location of the primary tumor is very important to assure an accurate diagnosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
