Abstract
Objective:
Immunity is associated with acute low tone hearing loss. However, the exact pathophysiology of immunity-mediated acute low tone hearing loss remains unknown. In this study, we evaluated the presence, therapeutic effectiveness, and immunopathological mechanisms of anti-endothelial cell autoantibodies (AECEs) in patients with acute low-frequency hearing loss.
Material and Methods:
Forty-nine patients who were treated as inpatients having acute low-frequency hearing loss and additional symptoms, such as ear fullness, tinnitus, dizziness, or hyperacusis, were enrolled in this study. Serum samples from these patients were collected for laboratory serum autoimmunity detection, including AECAs, antinuclear antibodies, immunoglobulin, and circular immune complex. Therapeutic responses to combination therapy in short-term outcome and serum cytokine levels were compared between AECA-positive and AECA-negative patients.
Results:
Anti-endothelial cell autoantibodies–positive patients tended to show significantly less response to standard therapy compared with AECAs controls (P < .05). Moreover, some serum cytokine levels elevated in both AECAs− and AECAs+ groups. Positive ratio of interleukin-8 and concentrations of macrophage inflammatory protein-1α were found higher in AECAs+ groups (P < .05).
Conclusion:
The results supported that AECAs might wield influence on the short-term outcome of acute low-tone hearing loss (ALHL) treatment. Furthermore, AECA-mediated acute low-frequency hearing loss possibly involved dysregulation of inflammation process and release of cytokines.
Keywords
Introduction
Sudden sensorineural hearing loss (SSNHL) is a type of sudden hearing loss in which at least 3 consecutive frequencies are affected and decrease more than 30 dB occurring within a 72-hour window. 1 Acute low-tone sensorineural hearing loss (ALHL) is a type of idiopathic SSNHL in which frequencies at 500 Hz and below are not distinguished. Patients with ALHL often complain of tinnitus, ear fullness, dizziness, and hyperacusis. 2 Although there are several causes of ALHL, the specific etiology is still unknown. Previous studies have suggested that genetic causes (eg, mutations in the Wolfram Syndrome gene 3 -5 and Mondini dysplasia 6 ), central neural system lesions, 7 aging, 8 viral infections, renal failure, 9 and early warnings of Meniere disease 10,11 all play roles in the development of ALHL.
Autoimmune inner ear disease or immune-mediated inner ear disease is an autoimmune process that leads to inner auditory dysfunction, thereby contributing to cochlear and vestibular symptoms. Low-frequency hearing loss may coexist with autoimmune diseases such as Susac syndrome (SS) and systemic lupus erythematosus (SLE), suggesting a possible role of autoimmunity in the inner ear. Anti-endothelial cell antibodies (AECAs) have been shown to be involved in the pathophysiology of SS. 12,13 Hitherto, some studies have examined the relationship between autoimmunity and sensorineural hearing loss (SNHL), 14 -17 and SNHL can be caused by autoimmune disorders of the inner ear or subordinate to systemic immune diseases. 12,14,15 Notably, the levels of several autoantibodies such as AECAs are elevated in patients with SNHL. 16 -18
In the basic research, stress response and inflammation were viewed as one of the important pathology on SNHL. 19 -24 Inflammatory-mediated upregulation of cytokines, in serum or in the cochlea, enhanced the noise-induced, drug-induced ototoxicity as well as age-related hearing loss. 23,25 -27 Several clinical investigations revealed that systematic stress and immunological response were involved in the SNHL. 28 -31 Elevated serum inflammatory biomarkers may correlate with the severe onset and poor prognosis of SNHL. 28 Imbalance of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio led to endothelial dysfunction by means of microvascular disturbances and inflammatory processes, which might play an important role in the pathophysiology of hearing loss and thereby influencing the patients’ prognosis in recovery. 29
ALHL is most often treated using a combination of diuretics and steroids. 32 Factors involved in the prognosis of ALHL include the initiation of treatment after the onset of the disease and accompanying symptoms (eg, tinnitus, vertigo, hearing thresholds before treatment initiation, and younger age). 32 In addition, female sex and definite ALHL also affect prognosis. 2,33 Previous reports have shown that AECAs may affect the type and severity of SNHL. 16,17 However, the impact of AECAs on therapeutic responses to combination therapy remains unknown, and the cellular mechanisms involved in these processes have not been determined yet.
Accordingly, in this study, we evaluated the roles of AECAs in ALHL in patients receiving a combination of diuretics and steroids. Additionally, we examined the pathophysiological mechanisms through which AECAs mediated ALHL.
Methods
Study Participants Materials
In this retrospective study, we enrolled 49 patients diagnosed with ALHL between July 1, 2016, and December 31, 2017. Patients visited the outpatient clinic at the Eye, Ear, Nose, and Throat Hospital of Fudan University. Patients who were treated as inpatients having ALHL and additional symptoms, such as ear fullness, tinnitus, dizziness, or hyperacusis, were enrolled in this study. Serum samples from these patients were collected for laboratory serum autoimmunity detection, including AECAs, antinuclear antibodies, immunoglobulin, and circular immune complex (CIC).
All patients included in this study met the following criteria 1,34,35 : main symptoms of acute or sudden onset of cochlear symptoms, including ear fullness, tinnitus, and hearing loss; low-frequency hearing loss; no vertigo; and unknown cause. Audiometric criteria for low-frequency hearing loss were as follows: the average of hearing levels at low frequencies of 0.125, 0.25, and 0.5 kHz was 30 dB or more; the sum of hearing levels at high frequencies of 2, 4, and 8 kHz was 60 dB or less; cochlear symptoms may be recurrent; may progress to Meniere’s disease; may be accompanied by light dizziness; and may be bilateral. For definite ALHL, all of the main symptoms were present, including the 2 first audiometric criteria listed above. For probable ALHL, all of the main symptoms were present, as well as the first audiometric criterion above, and patients exhibited the same hearing levels at high frequencies of 2, 4, and 8 kHz as the contralateral ear.
Patients who were diagnosed as having low-frequency hearing loss met all the following inclusion criteria: no history of ocular or vestibular disease, no medical treatment within 14 days of onset, and availability for long-term follow-up. All participants had normal acoustic impedance and admittance tests (define as the presence of a tympanometric curve type “A”) and a contralateral acoustic reflex threshold in merely one of the analyzed frequencies. In addition, all participants had normal middle ear function and no abnormal otological endoscopy findings. The exclusion criteria were as follows: history of any other ear disease, such as genetic hearing loss or dysfunction of vestibular system including vestibular Schwannoma; the presence of abnormal middle ear function suspected of conductive hearing loss; and history of ascertained autoimmune disease.
We ultimately included 23 patients with positive AECAs serum findings; we then chose sex- and age-matched 26 controls (negative AECA group) also matched for duration before onset of treatment relative to that in the positive AECAs group (Table 1).
Profile of the Patients Between the 2 Groups.a
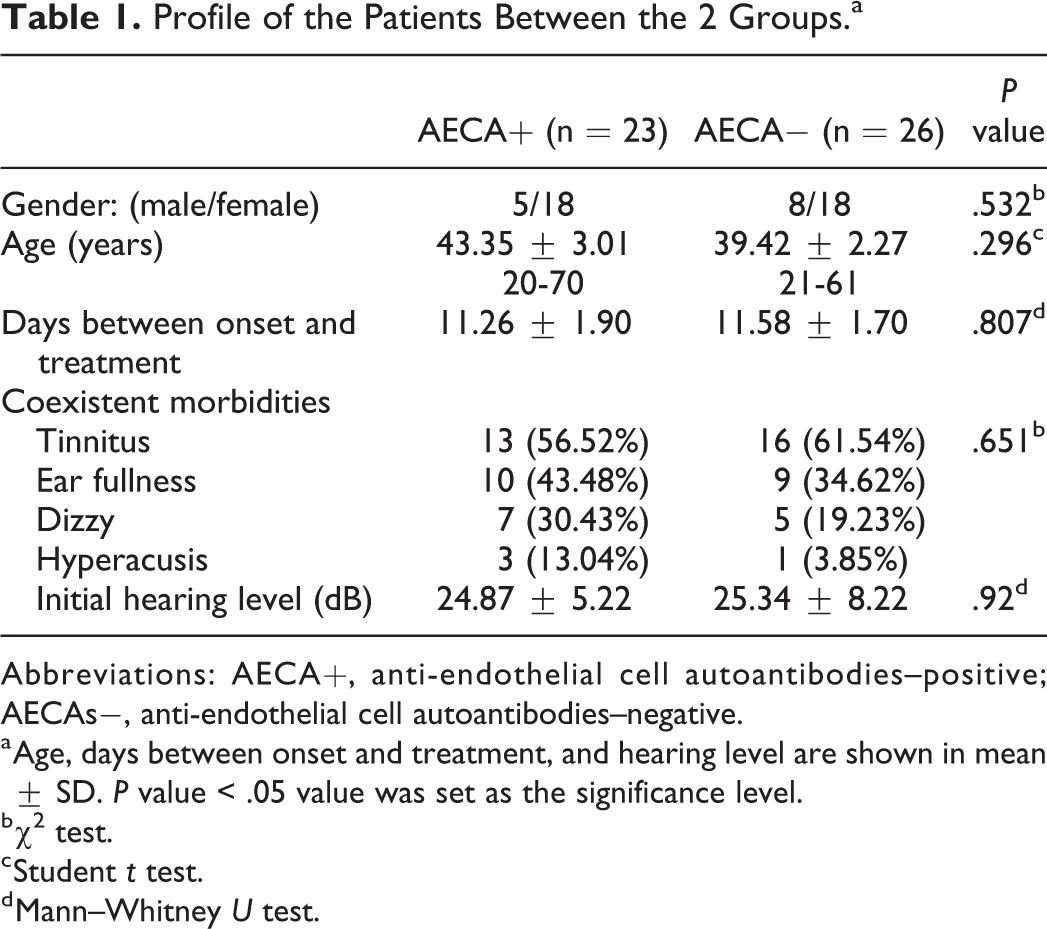
Abbreviations: AECA+, anti-endothelial cell autoantibodies–positive; AECAs−, anti-endothelial cell autoantibodies–negative.
a Age, days between onset and treatment, and hearing level are shown in mean ± SD. P value < .05 value was set as the significance level.
b χ2 test.
c Student t test.
d Mann–Whitney U test.
All patients were treated with combined therapy, including steroid and diuretics. Corticosteroid therapy was comprised of oral prednisone 1 mg/kg/d for 7 days, gradually tapered off with or without methylprednisolone intravenous injection. None of the patients had received intratympanic steroid injection. At the same time, patients were treated with 20% mannitol 250 mL once daily for 3 or 4 days in the first week.
Pure tone audiometry (PTA) was initially performed immediately before ALHL treatment and was repeated 1 week after combined dehydration and corticosteroid therapy. The pure tone hearing threshold was calculated as one-fourth the sum of hearing at 500 Hz, 1, 2, and 4 kHz; and the mean low-frequency hearing threshold was calculated as the average hearing threshold at 125, 250, and 500 Hz. The primary audiological response to the therapeutic regimen was classified into 4 grades 35 based on the most recent guidelines. Briefly, complete recovery was assumed when all 3 low frequencies (125, 250, and 500 Hz) of final audiograms were 20 dB or less or improvement was observed to the same degree of hearing in the unaffected ear. Partial recovery was assumed when the mean hearing level of 3 low frequencies improved by at least 10 dB, but not to the degree of complete recovery. No change was assumed when the mean hearing level of 3 low frequencies was improved by less than 10 dB. Finally, deterioration was assumed when the findings did not meet any of the above criteria. Based on these criteria, patients were separated into a response group, which included patients with complete recovery and improvement, and a nonresponse group, which included patients without change or with the deterioration of hearing loss.
To testify the relevance of endolymphatic hydrops in our patients, thus dehydration test was performed amid part of the patients. Eleven patients underwent dehydration tests following intravenous administration of 20% mannitol (250 mL) for 2 hours. Pure tone audiometry was performed immediately after intravenous 20% mannitol administration and at 1 and 2 hours after treatment. Results were considered positive when there was at least 10 dB improvement in average pure tone threshold at 2 or more low frequencies (125, 250, 500 Hz).
Anti-Endothelial Cell Autoantibodies Detection
All blood samples from 49 patients were obtained the first day at the clinic and their sera were stored at −80 °C. Anti-endothelial cell autoantibodies detection was performed with the kit (F180726AA; EUROIMMUN) based on an indirect fluorescence technique. Purified antigen from human umbilical vein endothelial cells were coated onto the biological slides to make solid-phase antigens. Negative (AECA negative) and positive (AECA, IgG) human controls were also provided with the kit. Each human serum was firstly diluted with phosphate-buffered saline buffer (pH 7.2), and then brought to combine with the antigen substrate to form an antigen–antibody complex. The antigen–antibody complexes were then labeled with FITC(Fluorescein isothiocyanate)-labeled sheep antihuman IgG. Positive result was defined as single serum titer over 1:100 as well as the observation of green fluorescence of the sections under the microscope (Figure 1).
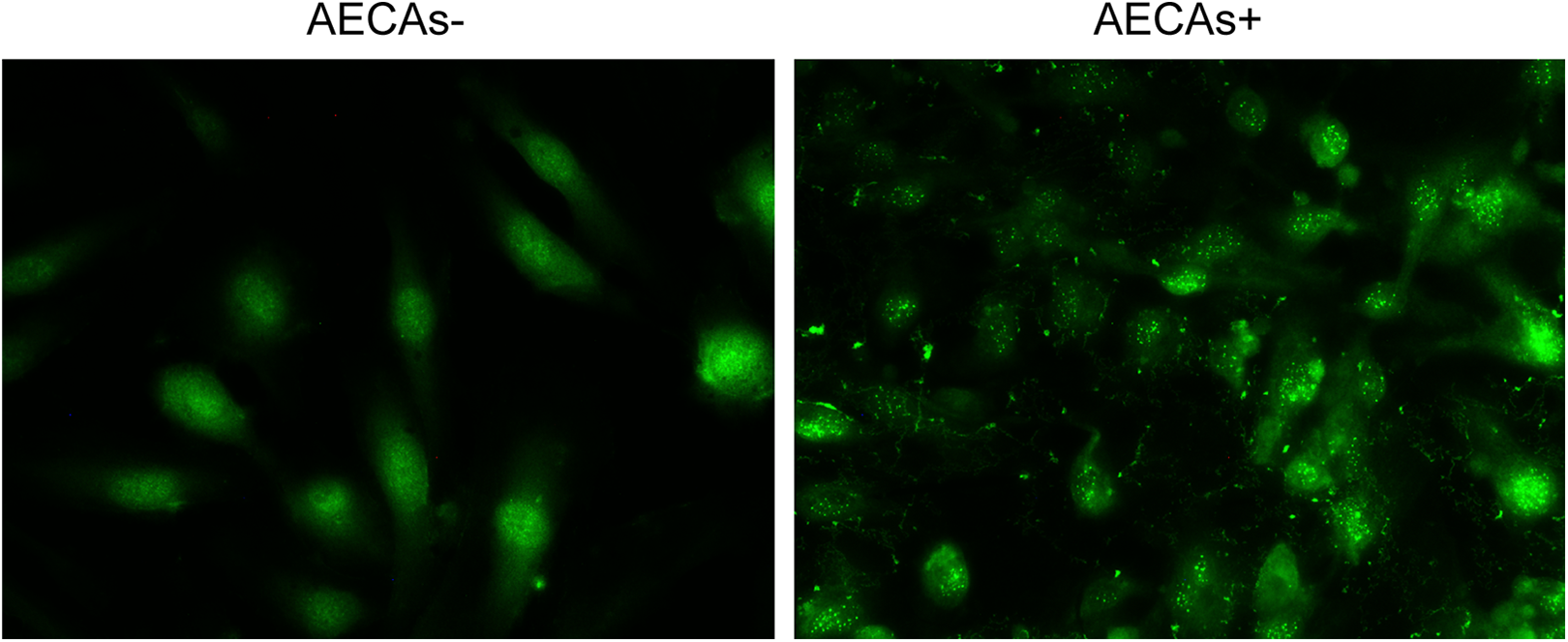
Immunofluorences of AECAs from patients. Specific granular green fluorescence localizing to cytoplasm of the specimen from AECAs+ obviously different from AECAs−. AECA+ indicates anti-endothelial cell autoantibodies–positive; AECAs−, anti-endothelial cell autoantibodies–negative.
Cytokine Immunoassay
The protein content of each sample was quantitatively detected using a multiplex immunoassay developed by Abcam based on FirePlex microgel technology. The assay was carried out as follows. For each sample, at least 37.5 µL of 1× Human Assay Diluent was added to 12.5-µL sample (all samples were diluted at least 1:4; for affluent sample analytes, the actual operation was diluted using the recommended ratio from Abcam). For capture, 75 µL of 1× Capture Particle Mix Solution was added to each well of a 96-well filter plate and filtered. The microgel was rinsed once by adding 175 µL of 1× Wash Buffer and then 50 µL of the diluted sample or reference sample. The plates were incubated for 60 minutes at 750 rpm with shaking at room temperature. For detection, wells were rinsed twice with 175 µL of 1× Wash Buffer, and 50 µL of 1× Biotin Detector Antibody Mix was added to each well. The plates were incubated for 60 minutes at 750 rpm with shaking at room temperature. After rinsing twice with 175 µL of 1× Wash Buffer, freshly prepared 1× Reporting Mix was added to each well. The plates were incubated for 30 minutes at 750 rpm with shaking at room temperature. Wells were again rinsed twice with 175 µL of 1× Wash Buffer, and 175-µL Run Buffer was added to each well. Samples were finally scanned using an EMD Millipore 6HT or 8HT flow cytometer.
Statistical Analyses
Statistical analyses were performed using IBM SPSS Statistics software. Whether the data were parametric or nonparametric was ascertained using Shapiro-Wilk normality tests. Statistical significance of data, including age, duration of symptoms before treatment, and hearing threshold, was analyzed using t tests in cases of parametric data and Mann–Whitney U tests in cases of nonparametric data. The χ2 tests and Fischer exact tests were used for cross-table analysis. Results with P values of less than .05 were considered statistically significant. The FirePlex Analysis Workbench or R software was used to analyze streaming output results (http://www.abcam.com/FireflyAnalysisSoftware).
Results
Profile of Patients
We evaluated 49 patients in this study. Female predominance was observed among our group. The average age in AECAs+ patients was 43.35 ± 3.01 years (range: 20-70 years), whereas that in AECAs− patients was 39.42 ± 2.27 years (range: 21-61 years). No significant differences between the 2 groups were observed in terms of duration of onset or treatment. Common complaints included tinnitus, ear fullness, dizziness, and hyperacusis. Initial hearing levels were comparable between the AECAs+ and AECAs− groups (Table 1).
Comparison of Responses to Combined Therapy Between the AECAs+ and AECAs− Groups Within 1 Week
Primary evaluation of PTA was performed 7 days after corticosteroid therapy (methylprednisolone 1 mg/kg/d) combined with dehydration therapy (20% mannitol 250 mL once daily for 3 or 4 days). Responses to the combined therapy were significantly different between the 2 groups (Figure 2, Table 2). Only 4 AECAs+ patients showed significant recovery. Most AECAs+ patients did not respond effectively to combined therapy compared with AECAs− patients (P = .005).
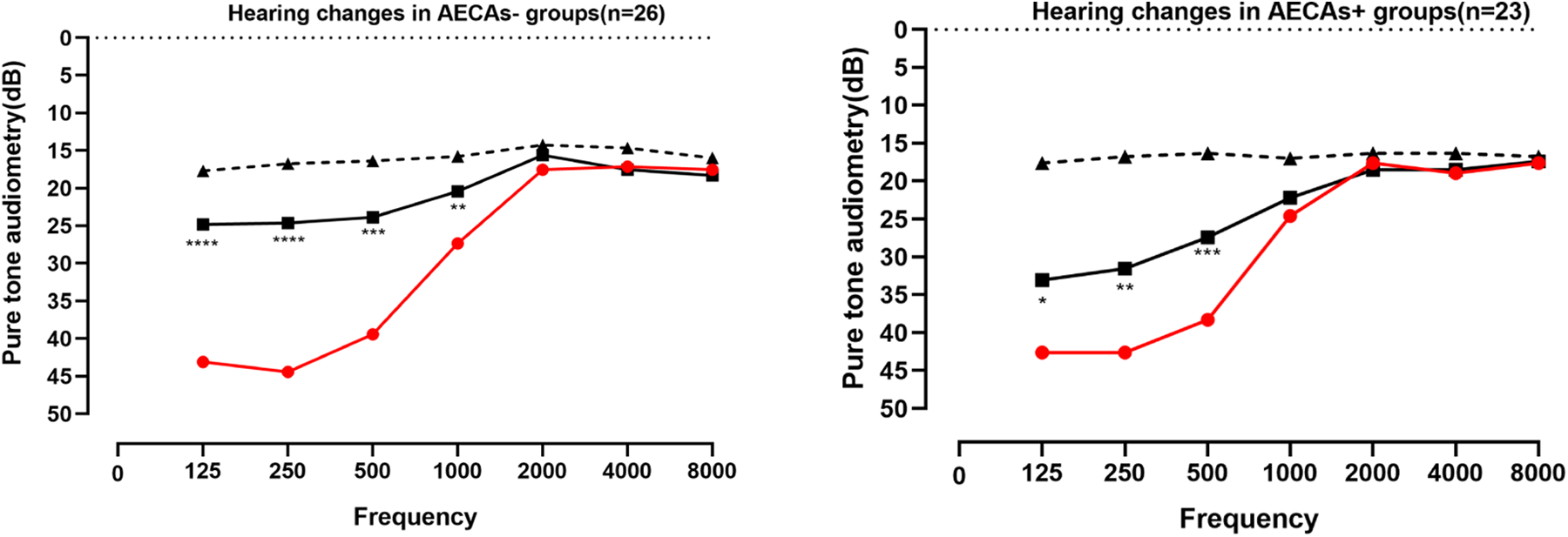
Threshold changes in pure tone audiometry before and after treatment between the 2 groups are shown above. Hearing threshold significantly decreased after treatment in both groups. However, average low-frequency hearing (125, 250, and 500 Hz) recovery in AECAs− (17.88 ± 13.93 dB) was significantly more obvious than that in AECAs+ (10.51 ± 11.65 dB) groups. Black bold line represented the hearing threshold after treatment; Red bold line represented the hearing threshold before treatment; black dotted line is the hearing threshold of the contralateral ear. Data are presented as means ± SD; n = 23 and n = 26 in AECAs+ and AECAs− groups, respectively; *P < .05, **P < .001, ***P < .0001, ****P < .00001, paired t test. AECA+ indicates anti-endothelial cell autoantibodies–positive; AECAs−, anti-endothelial cell autoantibodies–negative.
Comparison of the Therapeutic Response Toward the Combined Therapy Within 7 Days
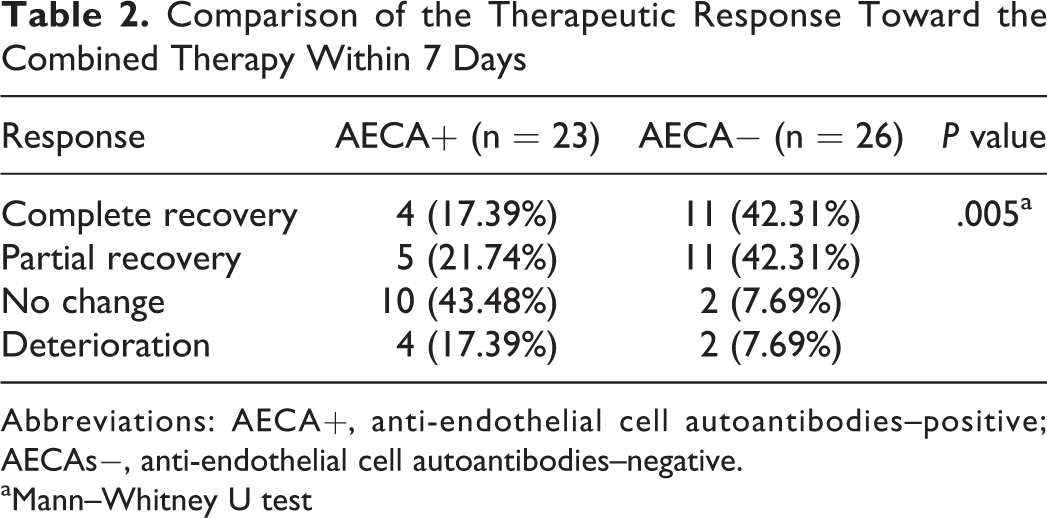
Abbreviations: AECA+, anti-endothelial cell autoantibodies–positive; AECAs−, anti-endothelial cell autoantibodies–negative.
aMann–Whitney U test
Mann–Whitney U test was used to assess the significance of therapeutic outcomes between the 2 groups. A P value < .05 was considered statistical significant. Eleven patients randomly selected to undergo dehydration tests. Nine patients showed significant elevation of the auditory threshold by more than 10 dB at 1 to 2 hours after 20% mannitol injection, while 2 patients were not (Figure 3, Table 3).
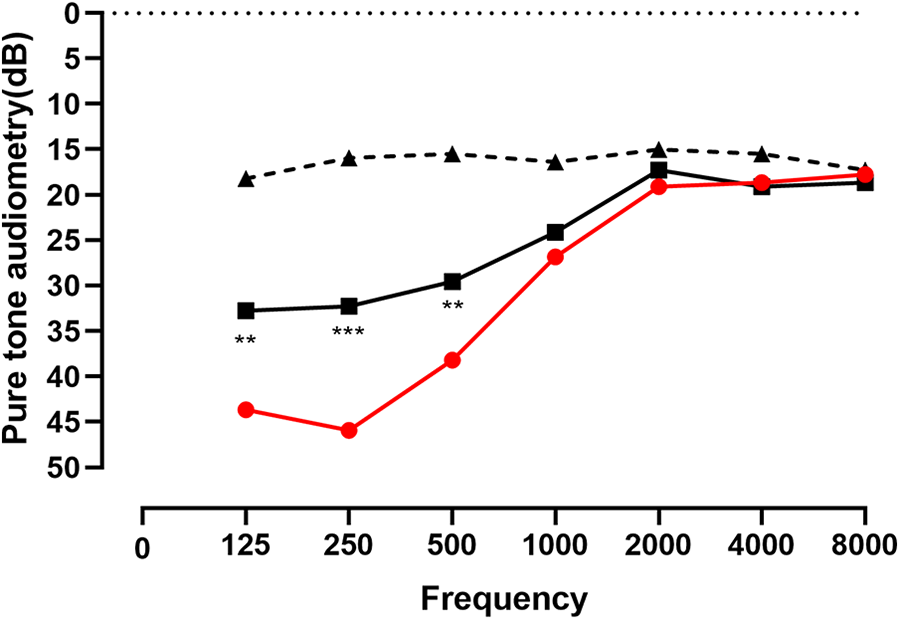
Threshold changes in pure tone audiometry before and 2 hours after 20% mannitol injection among 11 patients are shown above. Black bold line represented the hearing threshold after treatment; Red bold line represented the hearing threshold before treatment; black dotted line is the hearing threshold of the contralateral ear. Data are presented as means; n = 11; **P < .001, ***P < .0001, paired t test.
Hearing Responses to the Dehydration Test Among 11 Patients.a
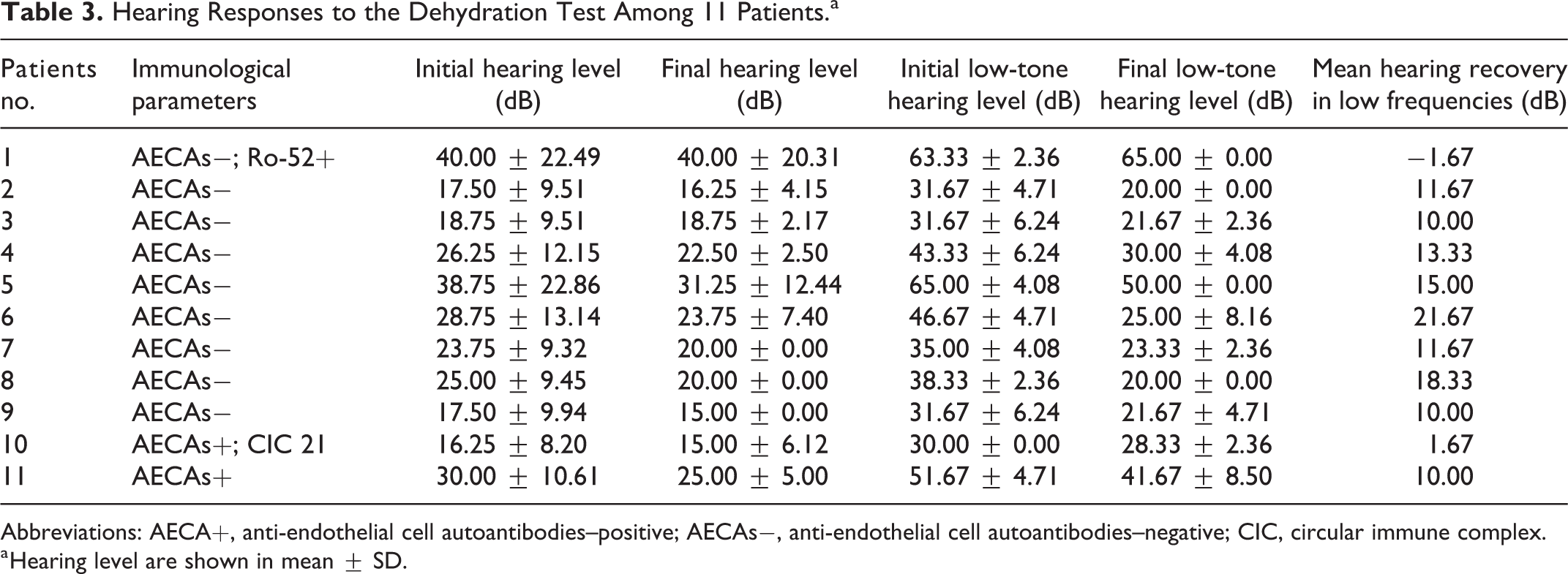
Abbreviations: AECA+, anti-endothelial cell autoantibodies–positive; AECAs−, anti-endothelial cell autoantibodies–negative; CIC, circular immune complex.
a Hearing level are shown in mean ± SD.
Changes in Inflammatory Mediators in Patients With ALHL
We hypothesized that abnormality in autoimmunity was concomitant with dysregulation of inflammatory process in our patients. Next, 43 serum samples were randomly selected for the analysis of cytokine detection by immunoassays for granulocyte macrophage colony-stimulating factor, interferon (IL)-γ, IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-9, IL-10, IL-12p70, IL-13, IL-17A, monocyte chemotactic protein (MCP)-1, macrophage inflammatory protein (MIP)-1α, MIP-1β, and tumor necrosis factor (TNF)-α (Table 4). Interleukin-8 (>0.51 pg/mL), MCP-1 (>1.52 pg/mL), MIP-1α (>0.508 pg/mL), and MIP-1β (>0.508 pg/mL) were defined positive in patients from the 2 groups, positive rate of IL-8 differed between the 2 groups (Table 5, *P = .01). Meanwhile, concentration of MIP-1α (P < .001) differed significantly among groups (Figure 4, *P = .016).
Comparison of Associated Cytokines Between the 2 Groups.a
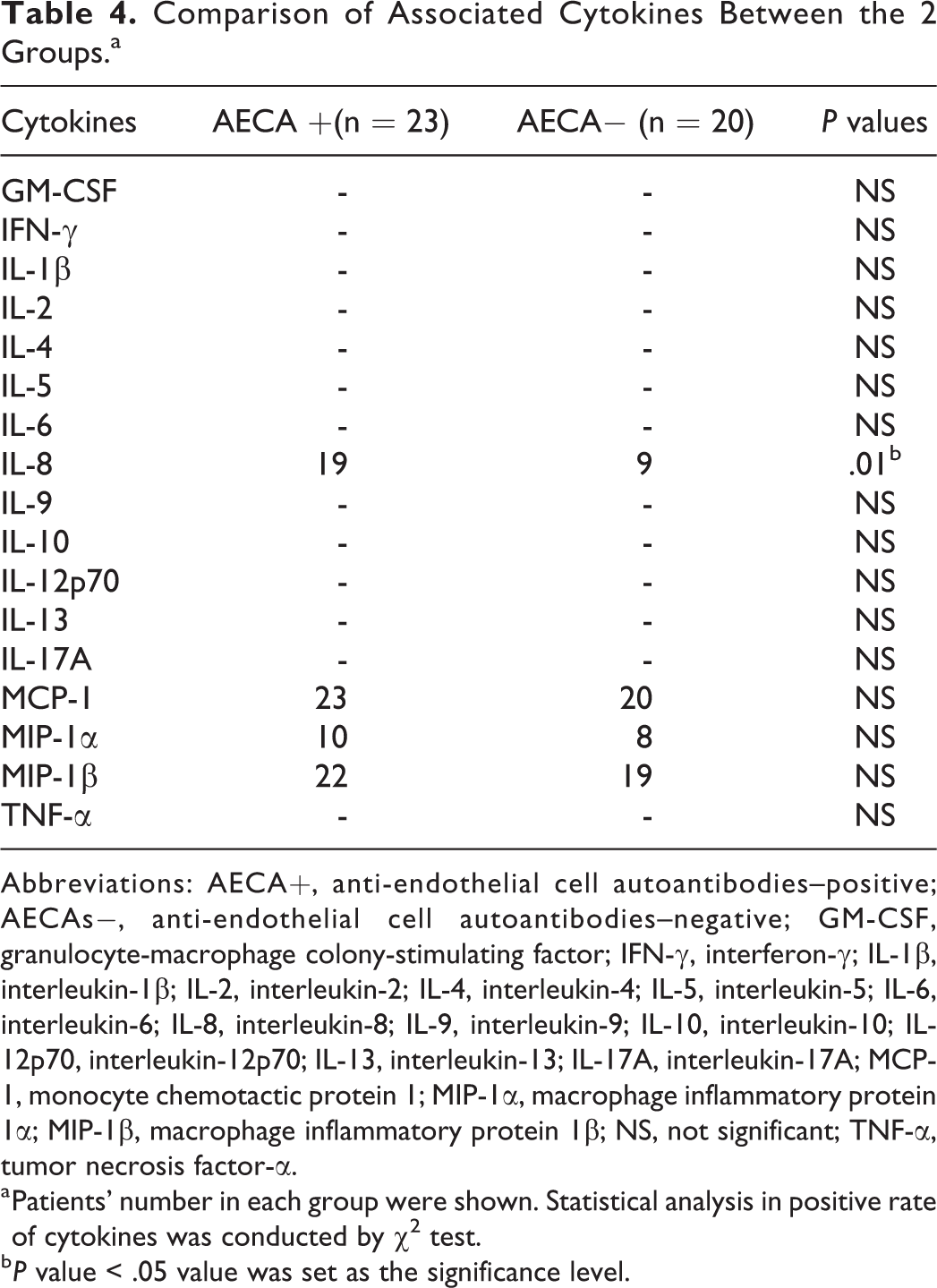
Abbreviations: AECA+, anti-endothelial cell autoantibodies–positive; AECAs−, anti-endothelial cell autoantibodies–negative; GM-CSF, granulocyte-macrophage colony-stimulating factor; IFN-γ, interferon-γ; IL-1β, interleukin-1β; IL-2, interleukin-2; IL-4, interleukin-4; IL-5, interleukin-5; IL-6, interleukin-6; IL-8, interleukin-8; IL-9, interleukin-9; IL-10, interleukin-10; IL-12p70, interleukin-12p70; IL-13, interleukin-13; IL-17A, interleukin-17A; MCP-1, monocyte chemotactic protein 1; MIP-1α, macrophage inflammatory protein 1α; MIP-1β, macrophage inflammatory protein 1β; NS, not significant; TNF-α, tumor necrosis factor-α.
a Patients’ number in each group were shown. Statistical analysis in positive rate of cytokines was conducted by χ2 test.
b P value < .05 value was set as the significance level.
Profile of Patients With Other Immunological Abnormality.a
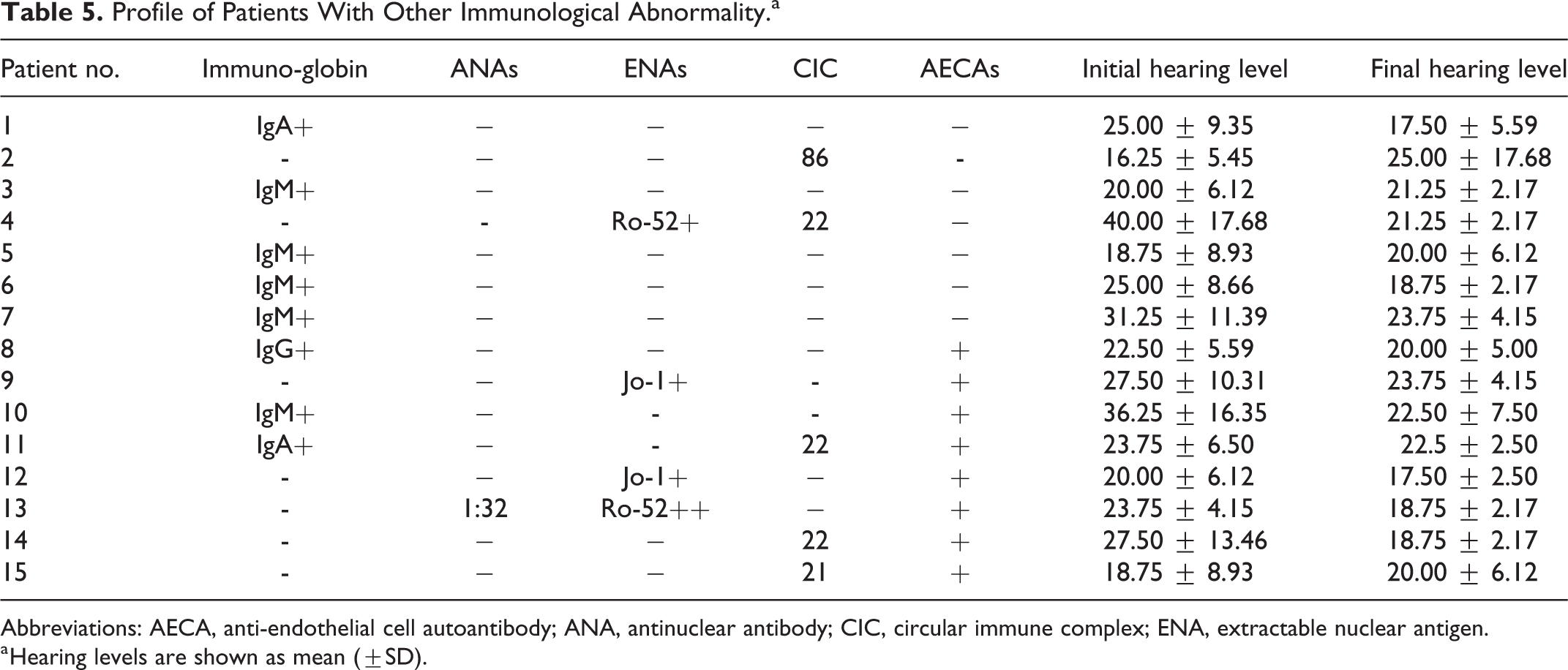
Abbreviations: AECA, anti-endothelial cell autoantibody; ANA, antinuclear antibody; CIC, circular immune complex; ENA, extractable nuclear antigen.
a Hearing levels are shown as mean (±SD).
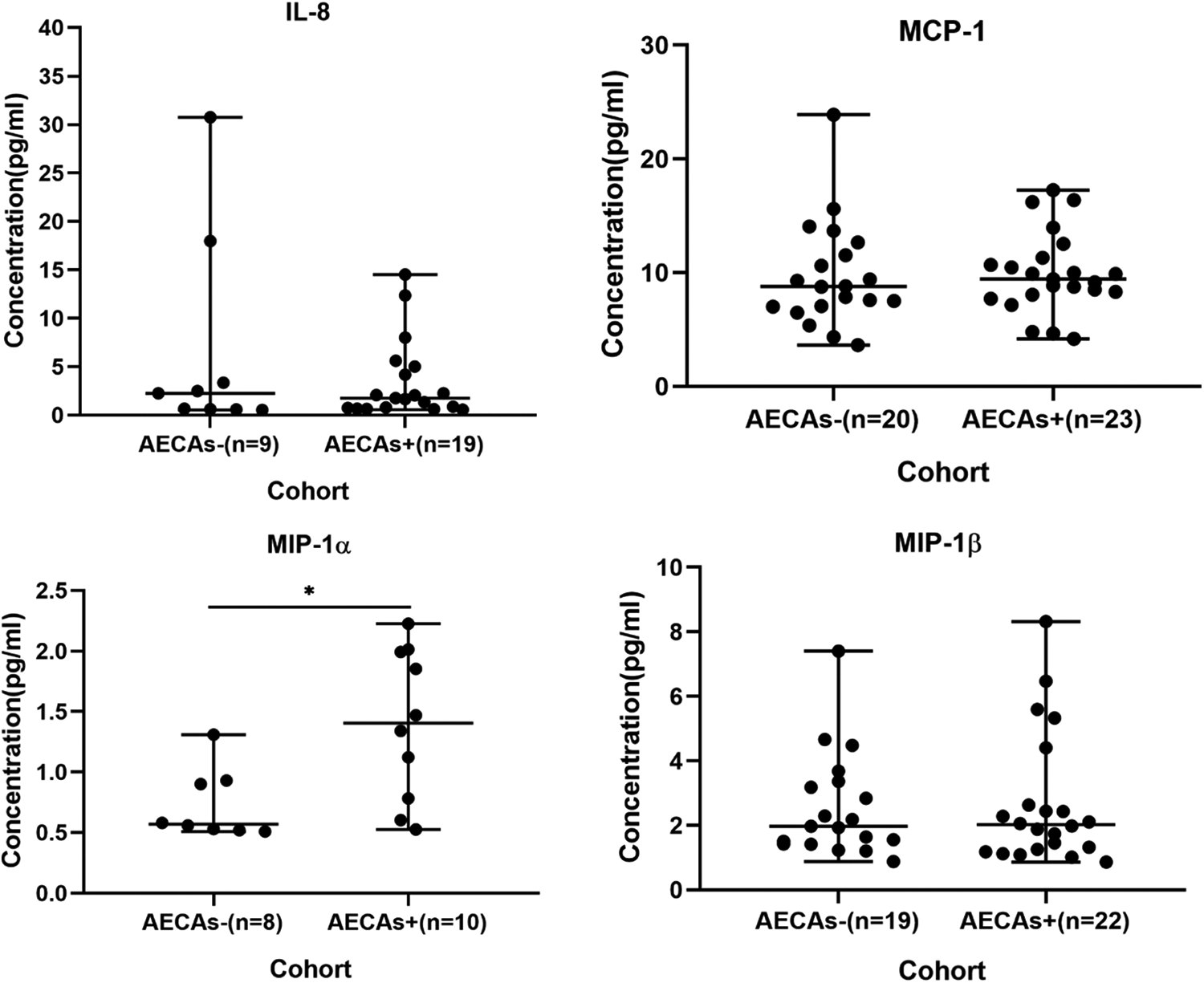
Concentrations of IL-8, MCP-1, MIP-1α, and MIP-1β in serum from AECAs- and AECAs+ patients. The concentrations of IL-8 is 6.58 ± 10.02 in AECAs−, 3.45 ± 3.96 in AECAs+; MCP-1 is 9.76 ± 4.52 in AECAs−, 9.92 ± 3.44 in AECAs+; MIP-1α is 0.73 ± 0.27 in AECAs−, 1.39 ± 0.59 in AECAs+; MIP-1β is 2.57 ± 1.57 in AECAs−, 2.68 ± 1.98 in AECAs+. Results are reported in pg/mL and were obtained by flow cytometry and shown in mean ± SD. The lines represent medians and ranges. *P < .05. AECA+ indicates anti-endothelial cell autoantibodies–positive; AECAs−, anti-endothelial cell autoantibodies–negative; IL-8, interleukin-8; MCP-1, monocyte chemotactic protein; MIP, macrophage inflammatory protein.
Discussion
In the otologic clinic, patients with ALHL account for a large proportion of total patients. However, the pathophysiology of this disease has not yet been conclusively elucidated.
Our current study showed that patients with AECAs+ showed insufficient responses to therapy and the positive rate of IL-8 along with the concentration of MIP-1α in serum samples from patients were significantly different between the 2 groups, indicating that they might induce the poor prognosis in AECAs+ groups. Previous studies have proposed an association between autoimmunity and SNHL. 16,17,36 Although the specific pathophysiology has not been fully elucidated, endolymphatic hydrops, vasculitis, basement membrane immune complex deposition in labyrinthine vessels, and atrophy of the stria vascularis have been proposed to be involved. 12,14,15,30,37 Laboratory examination of serum autoantibodies may provide an intriguing marker in the diagnosis of autoimmune inner ear disease. Anti-endothelial cell autoantibodies are associated with systematic vasculitis, such as SS. 12,13,37,38 Indeed, vascular pathogenesis has been reported to be one vital factor in SNHL. Another research revealed that a vascular treatment strategy may be effective in patients with SNHL. 39 Vascular endothelial dysfunction, as evaluated by measurement of omega-3 polyunsaturated fatty acid levels, has also been reported in sudden and idiopathic SNHL. 40 -43 Researches has supported the correlational hypothesis that endothelial cells were related to the inflammation process and the signaling pathways. 44 The presence of AECAs and their role in stria vascularis injury and vascular damage may also affect immune-mediated sensorineural deafness 16,17 by modulating some adhesion molecules and increasing the secretion of inflammatory cytokines. 30 Some researchers articulated that some of the molecules or cytokines demolished the integration of the blood–liquid barrier. 45 The breakdown of the blood–liquid barrier and substance changes in lymph fluid may cause ALHL. 46 Moreover, inflammatory biomarkers 28 was inextricably intertwined with the prognosis of SNHL.
Endolymphatic hydrops is one theory of pathophysiological changes observed in patients with ALHL and involves expansion of the membranous labyrinth initiating from the apical turn of the cochlea. 11,47 -50 Acute hydrops is responsible for ALHL owing to the susceptibility of the apex of the cochlea to pressure changes. In patients with ALHL, there is a relatively high incidence of spontaneous hearing recovery and recurrence or progression to Ménière’s disease. 51,52 The involvement of endolymphatic hydrops in ALHL is supported by the clinical course, results of electrocochleographic examinations, positive results in glycerol tests (performed in patients who are skeptical of endolymphatic hydrops), vestibular-evoked myogenic potential, and magnetic resonance imaging results. 10,47,48,53,54 Our results were consistent with the previous researches that ALHLs relied upon endolymphatic hydrops in part, but the number of cases was probably limited to explore the effect of AECAs and other immunological abnormality in this process.
We also detected other abnormal immunological parameters between the 2 groups. Among 23 patients in the AECAs+ group, CIC was positive in 4 patients. Antinuclear antibodies were positive in one of 23 patients. Jo-1, an autoantibody belonging to the extractable nuclear antigen (ENAs) family, was positive in 2 of 23 patients. Among 26 patients in the AECAs− group, 2 patients showed elevated CIC, and one patient showed increased ENAs. Immunoglobulin (Ig) was positive in 3 of 23 patients in the AECAs+ group and 5 of 26 patients in the AECAs− group. IgM antivirus was detected in 4 patients from the AECAs− group and only 1 patient from the AECAs+ group (Table 4). Certainly, patients with thyroid autoimmune disease, such as Hashimoto’s thyroiditis, are prone to hearing impairments owing to the existence of circulating antithyroid antibodies. 55 In another study, the low-frequency auditory threshold in patients with anti-Tg-positive hypothyroidism was found to be higher than that in patients with anti-Tg-negative hypothyroidism. 56 Hearing loss in patients with SLE is often accompanied by tinnitus with involvement of low and middle frequencies. 14 Low- and mid-frequency hearing loss in SS may be caused by autoimmune-mediated pathology of microvessels, including cochlear and vestibular arterioles, 12,37 suggesting the susceptibility of the apical turn of the cochlea to poor blood supply. 38 These results further illustrated that no less autoimmunity than immune response contributed to the initiation of ALHL.
Limitations
Our experiment explored the possible effect of AECAs on ALHL. Further study could be designed to investigate profoundly the short-term and long-term effect of AECAs on ALHL and propose effective therapeutic regimen in the future. Ultimately, the detailed cellular mechanism needed to be determined between the dysregulation of immune response and initiation of ALHL.
Conclusion
In conclusion, we demonstrated the involvement of autoimmunity in ALHL and reported that AECAs+ patients with ALHL might be less responsive to combined therapy. Furthermore, cytokine detection provided insights into the potential cellular mechanisms through which AECAs mediate ALHL, suggesting the possible pathophysiology of ALHL mediated by AECAs and inflammatory cytokines IL-8 and MIP-1α release. Our findings provide important insights into the selection of an appropriate therapeutic regimen, including specific immunosuppressive treatments in patients with SNHL in the future. However, the exact cellular mechanisms between the AECAs and these interleukins had not been determined yet. Further experiments are needed to clarify their interactions in this process.
Footnotes
Authors’ Note
Yusu Ni contributed to design, authorship, editorial, and made substantial contributions to the conception and design of work. Diyan Chen, Zhujian Wang, Gaogan Jia, and Huanyu Mao contributed to authorship, review, and made substantial contributions to the conception and design of work. Diyan Chen, Zhujian Wang, Gaogan Jia, and Huanyu Mao contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant 17ZR1404600 from the Nature Science Foundation of Shanghai Science and Technology Committee.
