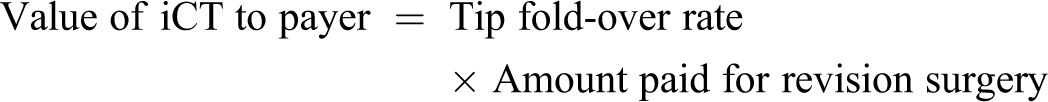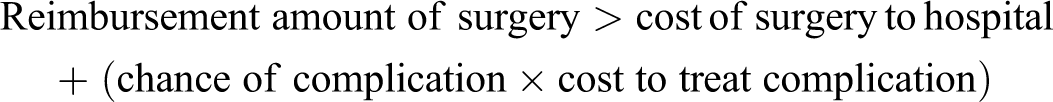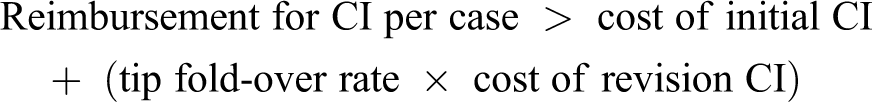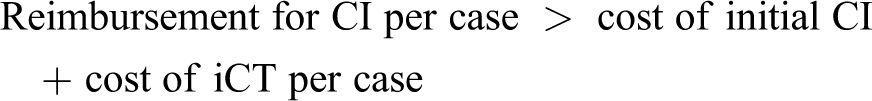Abstract
Objective:
Electrode array tip fold-over is a complication of cochlear implant surgery that results in poor hearing outcomes and often leads to revision surgery. However, tip fold-over can be corrected immediately if identified through intraoperative computed tomography, which also potentially provides information about final intracochlear positioning. Our objective was to provide the first economic analysis of intraoperative computed tomography by generating models in fee-for-service and bundled payment reimbursement structures of payer and institutional cost-effectiveness of this technology used in cochlear implantation over 1, 5, and 10-year time periods.
Methods:
Cost data specific to a commerically available intraoperative computed tomography machine was obtained from the manufacturer, Xoran Technologies. Institutional tip fold-over rate was obtained from already published data. Medicare reimbursement rate for cochlear implantation was obtained from institutional accountants. Private payer reimbursement for and cost of revision cochlear implantation were estimated based on available data.
Results and Conclusion:
At large volume centers, cost-effectiveness of this technology is possible in both fee-for-service and bundled payment reimbursement structures at various time points dependent on payer mix. Even low volume cochlear implantation centers (<150 per year) can financially benefit from intraoperative computed tomography in bundled payment models at 5- and 10-year periods regardless of payer mix. This model demonstrates key factors at play in determining cost-effectiveness of this technology including institutional factors and payer type and suggests this technology can align incentives both to improve patient care and outcomes with institutional and payer financial well-being.
Keywords
Introduction
In patients with sensorineural hearing loss, cochlear implantation (CI) can vastly improve quality of life in both children and adults. 1 During CI surgery, an electrode array (EA) is inserted ideally into the scala tympani (ST) component of the cochlea. 2 Insertion causes trauma to intracochlear structures such as the basilar membrane, osseous spiral lamina, and/or spiral ligament 3 and can lead to EA tip fold-over as well as scalar translocation from ST to scala vestibuli (SV). Tip fold-over occurs when an EA is advanced despite the tip being stuck and has an incidence of between 0.8% in straight (a.k.a. lateral wall) EA and 4.7% in precurved (i.e., perimodiolar) EA. 4 Data from one high-volume institution shows tip fold-over rates of 2% to 3%.5,6 More common than tip fold-over is scalar translocation in which the EA takes an unfavorable insertion trajectory which causes it to violate the basilar membrane and/or osseous spiral lamina and translocate from ST to SV. Translocation has been shown to lead to poorer audiological outcomes. 7 Scalar translocation rates are much higher than rates of tip fold-over ranging from 6.2% 7 to 22% 8 for straight EA and from 10% 7 to 32% 9 for precurved EA.
Several technologies have been used to detect tip fold-over within the operating room (OR) offering the chance for correction during the same operative procedure. These technologies include spread of excitation (SOE) and/or electric field imaging (EFI) as well as radiological options such as plain X-rays, fluoroscopy, or intraoperative computed tomography (iCT). The SOE and EFI are nonradiological techniques in which an individual electrode or pair of electrodes in the EA is stimulated and the resultant electrical field recorded on other electrodes.5,10 Responses in patients with fold-over are different than those without fold-over. The advantage of these techniques is that they can be done during routine audiological testing of the CI EA after implantation but prior to recovery from general anesthesia. The disadvantage, however, is that if an abnormal electrical signal is identified, the recommendation is to confirm presence or absence of tip fold-over with imaging.
Plain film X-ray and/or fluoroscopy have also been used effectively to identify tip fold-over. 11 Cost of either of these technologies is typically included in the facility fee of the surgical center and adds a small amount of time to the surgical procedure. 6 Intraoperative computed tomography, on the other hand, is potentially the ultimate radiographic imaging taking multiple flat plate images during rotation around the patient and reconstructing these to provide dramatically more information about the anatomy; regarding CI EA location, image processing can be applied to the images to predict scalar location of the EA (ST only, SV only, ST/SV translocation). 6 However, at present, cost of iCT is not included in the facility fee.
Whether a certain technology is ultimately integrated into clinical care relies not only on its proven efficacy but also the practicalities of whether an institution can afford the capital investment and ongoing variable costs. Thus, alongside the potential patient-centered benefits, cost-effectiveness is an important element in transitioning a new technology from a research-associated practice with a small number of patients to becoming a standard of practice. Although an increasing amount of imaging techniques are making their way into the OR, only a small number of studies have evaluated their cost-effectiveness. For example, intraoperative magnetic resonance imaging (MRI) has been shown to be cost-effective in simulated high-grade glioma resection surgery models by increasing progression-free survival and gross-total resection rates.12,13 Similar to the role of MRI in this study, iCT use in CI is capable of decreasing reoperation rates by identifying tip fold-over intraoperatively allowing immedicate correction and can likely lead to better quality of life with better hearing outcomes, 6 although no studies have evaluated its cost-effectiveness.
Cost-effectiveness itself has become an increasingly complex topic given the variety of reimbursement structures currently in use. 14 Two of the most common methods for surgical reimbursement, fee-for-service and bundled payments, offer differing incentives to institutions and payers; traditional fee-for-service models, in which institutions are paid per each service delivered to patients, stands in stark contrast to bundled payment models in which an institution is paid a lump sum to cover both the initial procedure and any other services given to treat complications. 15
Our objective of this study was to examine the cost-effectiveness of iCT during routine CI by generating models that demonstrate the thresholds of operative volume and tip fold-over rate needed to achieve cost-effectiveness over 1, 2, and 5-year time periods. Our goal was not to compare iCT directly to other tip fold-over prevention technologies but rather independently evaluate whether iCT could be simultaneously advantageous to key stakeholders—institutions, payers, and patients. Given the diversity of reimbursement models in use today, we devised 2 versions of our model to fit both fee-for-service and bundled payment model scenarios.
Methods
Cost data specific to the iCT used at our center was obtained from the manufacturer (XoranTechnolgoies LL) with a fixed cost of the machine of $410 000 (all figures are late 2018/early 2019 US dollars) including a 1-year warranty with comprehensive service and 1-day training on operation of the Windows-based interface. Based on department experience, we assumed in our model a $25 000 annual cost of maintenance for years 2 and on, as well as a $40 000 cost for iCT operator with $6000 of additional fringe benefits. We referenced recently published data which showed a tip fold-over rate of 3.1% and used 3% in these models for simplicity. 6 We assumed that all tip fold-over cases identified outside of the OR were revised and that all cases in which iCT was used had a final tip fold-over rate of 0% (if identified in OR and immediately corrected).
We obtained Medicare reimbursement rates for primary and revision CI ($30 080.35 for both) from institutional accountants. Data from the RAND Corporation specific to our institution showed that the ratio of Medicare payment versus private payer payment for the most recent reporting period (2015-2017) was 2.08. 16 Thus, we multiplied the Medicare reimbursement for CI by this factor to obtain an estimate of the amount a private payer would be expected to reimburse for CI ($62 567.13). We estimated the cost of a revision CI by referencing the American Hospital Association’s (AHA) most recent factsheet on the ratio of Medicare reimbursement to hospital cost. 17 We used a ratio of 0.87 and Medicare reimbursement for CI to estimate a Medicare revision cost of $34 575.12.
We first generated a model in which an institution would be reimbursed in a fee-for-service structure, such that any revision CI would be covered by payers at the same rate of initial CI. From the payer perspective, we calculated the value a technology capable of preventing tip fold-over, and then from the institutional perspective, the volume necessary to achieve cost neutrality at different reimbursement rates. We then generated a model in which an institution was paid one bundled payment for a CI, and if a revision was necessary, was not reimbursed further. Using our institution’s tip fold-over rate, we calculated the case volume necessary over varying time domains in order for iCT to be cost neutral or profitable from this institutional perspective. All formulas used to derive figures are described in the Results section. This study was approved by the Institutional Review Board at Vanderbilt University Medical Center.
Results
Fee-for-Service: Payer Perspective
In a fee-for-service reimbursement structure, if a tip fold-over was to occur and a patient thus needed reoperation, a payer would incur the additional cost of revision surgery. Thus, a technology preventing this situation would have a certain value to payers, calculated by the formula below and shown in Figure 1.
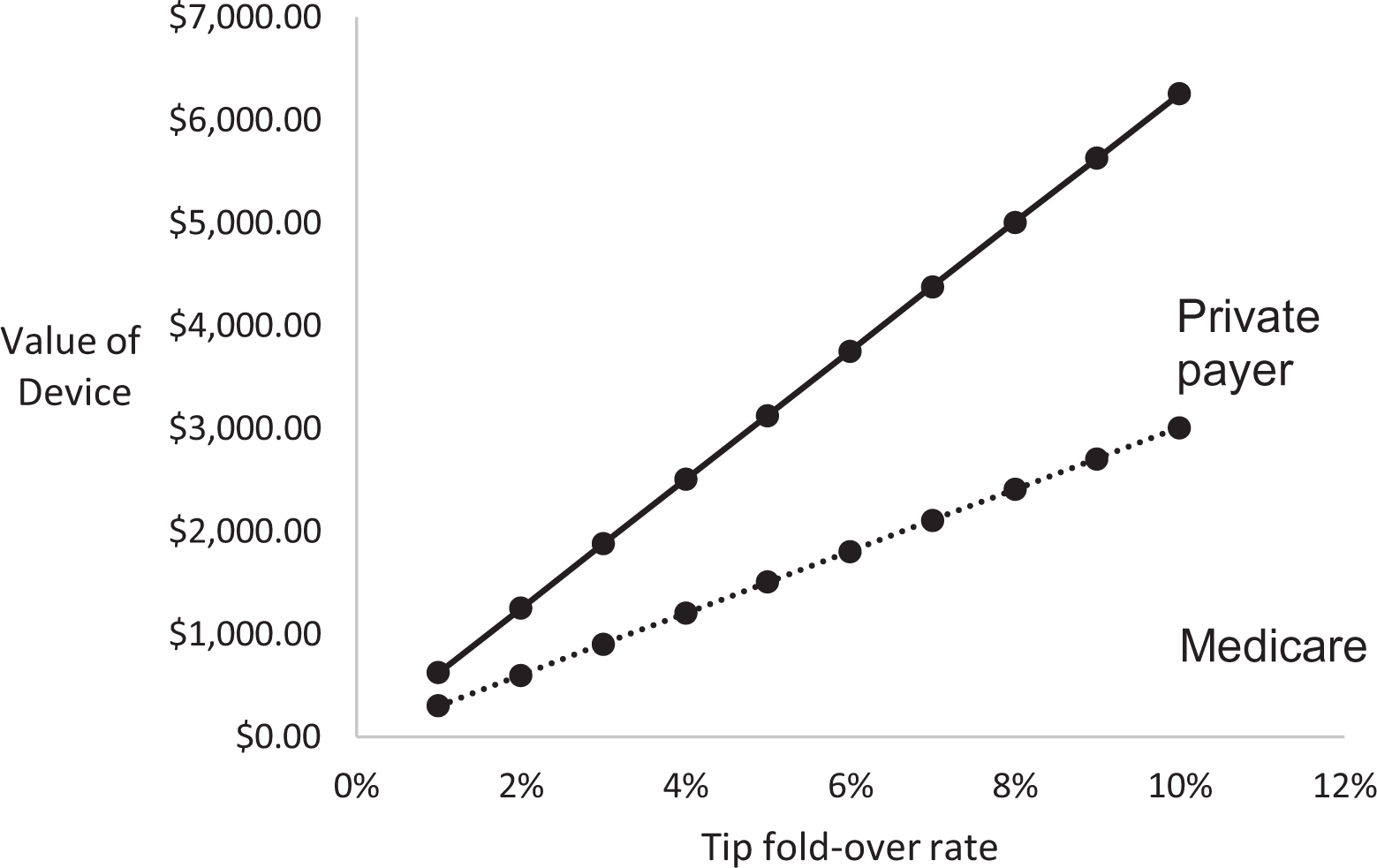
Value to payer of tip fold-over prevention device based on varying tip fold-over rates. Legend: Solid line represents value of iCT to private payer and dotted line represents value to Medicare. iCT indicates intraoperative computerized tomography.
As shown in Figure 1, even at relatively low rates of tip fold-over (such as 2% or 3%), the value of a tip fold-over prevention technology is markedly different between payers ($602 vs $1251, $902 v $1,877, respectively). At institutions (or with devices with) higher tip fold-over rates, the value of iCT is notably large (at 8%, $2406 and $5005).
Fee-for-Service: Institutional Perspective
Institutions in a fee-for-service model must weigh the cost of buying and operating an iCT machine versus the amount they are reimbursed for their services. If we assume payers would reimburse institutions an amount for performing iCT, this could offset these initial and operative costs. Figure 2 shows this scenario and demonstrates the operative volume of cochlear implants per year needed for an institution to breakeven depending on the reimbursement amount for iCT. These data points were calculated from:
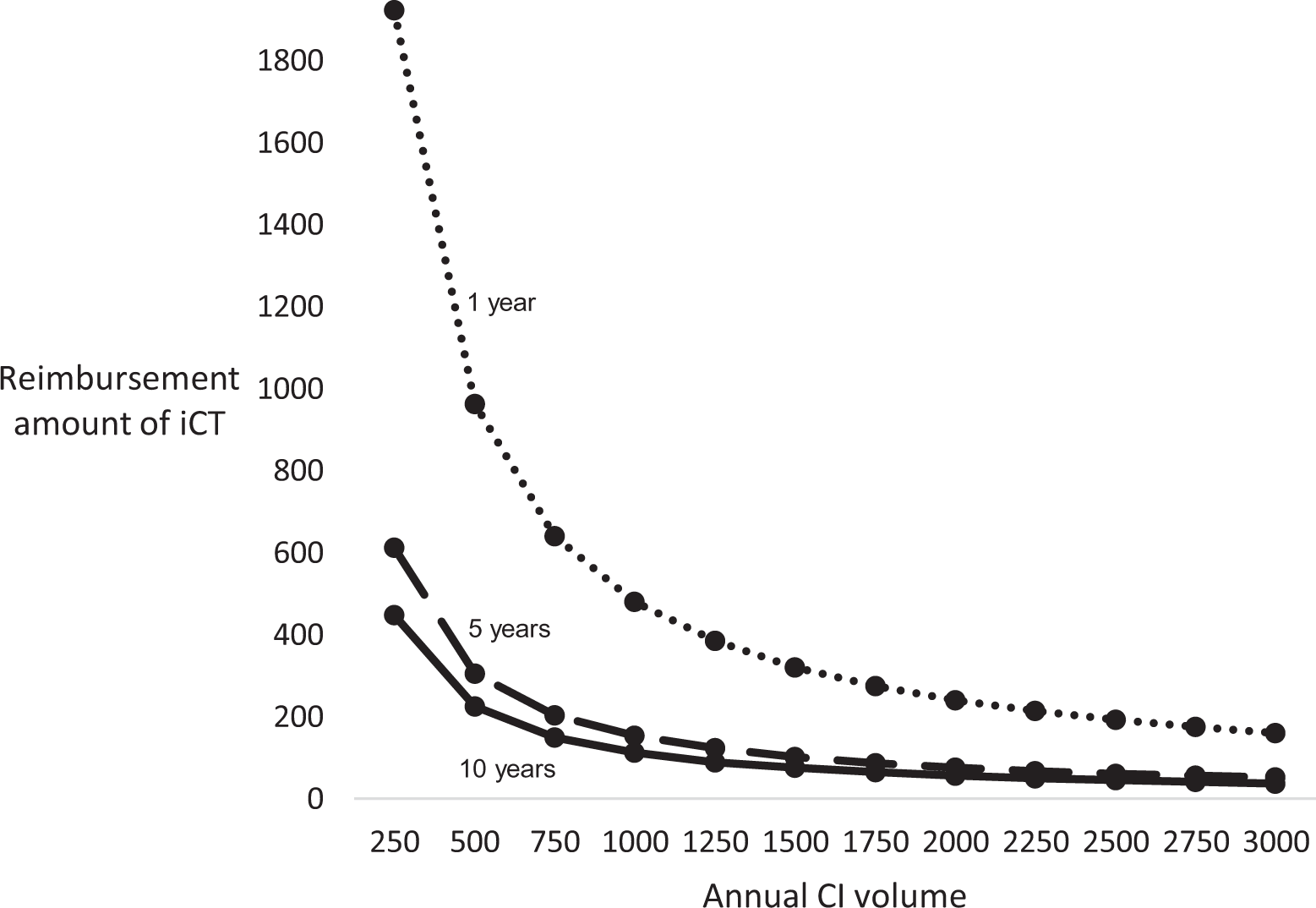
Operative volume necessary for institutional breakeven at 1, 5, and 10 years. Legend: Dotted line represents 1-year time period, and dashed and solid lines represent 5- and 10-year time periods, respectively.
At an institution such as ours with a high volume of CI (>250 a year), institutional breakeven at 5- and 10-year periods occurs at a low reimbursement amount for iCT (∼$300 or less) and for 1 year ($1000). According to Figure 1, payers (Medicare and a private payer) would value iCT at $902 and $1877, respectively, suggesting our institution would breakeven or financially benefit from iCT at 5- and 10-year periods at reimbursement rates significantly lower than the value to payers. Institutions with smaller operative volume (<250 CI per year) still reach breakeven at 5- and 10-year periods with reimbursement amounts <$1000, although their 1-year breakeven is estimated to be between $2000 and $3000.
Bundled Payment: Institutional Perspective
If an institution is reimbursed by a bundled payment option, then the economic value to the institution would be favorable if:
Applying this and simplifying by excluding complications other than tip fold-over:
Adding in iCT to eliminate tip fold-over changes this expression to:
The decision to use iCT is thus a comparison between the per case cost of iCT versus the amount saved or the tip fold-over rate multiplied by the cost of revision CI. Using our tip fold-over rate and estimated cost of revision surgery of $34 575.12, Figure 3 shows the net difference between the amount saved by performing iCT (tip fold-over rate multiplied by cost of revision surgery) and the per case cost of iCT at 1, 5, and 10 years. This model shows only at a very large operative volume (>500 cases) would our institution save money at 1-year, although at 5- and 10-year periods iCT would be money-saving even at lower operative volumes (>150 and >100 cases, respectively).
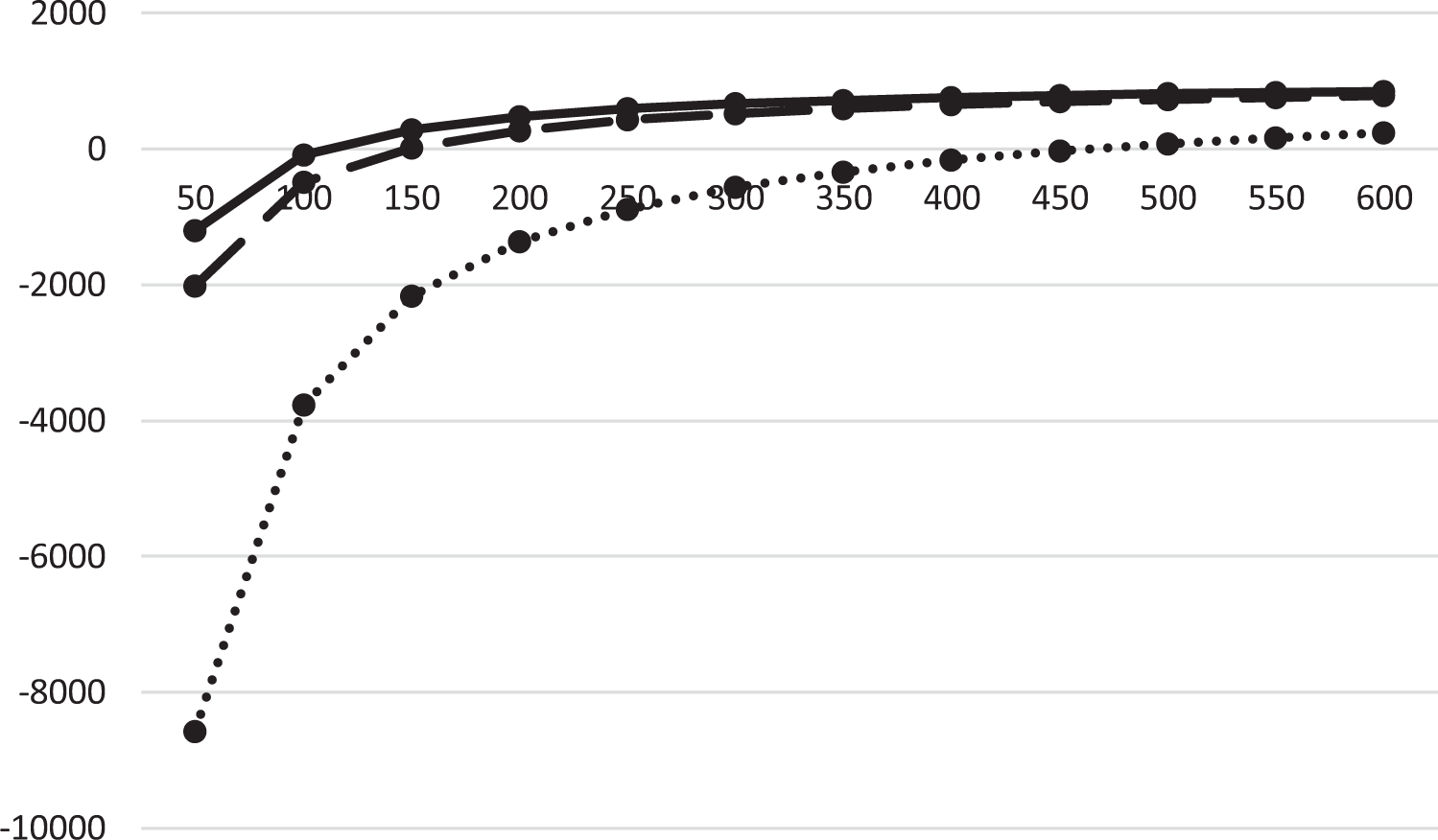
Net difference of money saved versus spent on iCT at 1, 5, and 10 years. Legend: Dotted line represents 1-year time period, and dashed and solid lines represent 5- and 10-year time periods, respectively. Note a positive value is indicative of money saved by the institution. iCT indicates intraoperative computerized tomography.
Discussion
Whether any technological advance is adopted within health care depends on many factors, but economic considerations are usually paramount. Within our study, we reduced the decision of whether to adopt iCT to strictly an economic standpoint evaluating cost-effectiveness from both payer and institutional perspectives. Our models were simplified immensely to generate a worst case scenario (i.e., iCT only used to rule out CI tip fold-over). The results of Figure 3 were surprising that even lower volume centers (150 CI per year) would break even at the 5- and 10- year time points, especially given that we did not factor in any additional economic benefit from the iCT such as use for routine sinus and temporal bone computed tomography scans outside of the OR.
Regarding the models used, first, these models demonstrate how cost-effectiveness of a single technology varies widely across payers and institutions depending on reimbursement rates, complication (tip fold-over) rates, operative volume, and payer mix. These models also demonstrate how reimbursement structures greatly influence the incentives an institution is facing. Importantly, these models could be generalized to other technologies that prevent surgical complications.
While other technology exists which can identify tip fold-over, namely fluoroscopy, comparing iCT to such is an “apples to oranges” comparison given the dramatic difference in the quality and quantity of information conveyed to the surgeon. Intraoperative computerized tomography allows surgeons to definitively confirm that the EA is within the cochlea, assess how far away the EA is from the modiolus, and detect scalar translocation. 6 The much less expensive fluoroscopy (C-arm machines range from $25 to $75 K) 18 conveys much less information—namely, only that the EA is coiled or not. Coiling implies intracochlear positioning but is also seen with coiling within the middle ear or even the superior semicircular canal.
Second, these models suggest possible scenarios for which iCT would be cost-effective for both payers and institutions. For example, as shown in Figure 2, in a fee-for-service model employed at institutions with high operative volume and low, but still existent, tip fold-over rates, institutions would breakeven at 5- and 10-year periods if they were reimbursed at significantly below the value given to the payer, even if all patients were Medicare patients. In other words, both the payer and payee economically benefit from routine use of iCT in CI. However, the logistics of billing for and obtaining reimbursement for iCT have yet to be explored at our institution because our iCT was funded in part from research dollars thus preventing us from billing for its use clinically. Nonetheless, the machine we use, the Xoran XCAT XL, is Food and Drug Administration cleared and has Current Procedural Terminology codes associated with its use.
As mentioned earlier, we specifically acknowledge oversimplifications of our models including the following. We are limited by complexity and lack of transparency of our health care system, in general, in setting and communicating prices for services and thus used available estimates. Reimbursement rates for procedures vary across geography and institutions, as does cost to an institution to perform these procedures. Even at a single institution, these costs vary depending on patient demographics, anatomy, complication rates, and area of delivery (e.g., inpatient vs outpatient surgery). The ratio we used from the RAND Corporation to estimate private payer reimbursement was a ratio calculated across all services delivered at our institution and is thus imperfect in this model. The estimate used for revision surgery cost calculated from AHA data faces the same challenge, as CI may be reimbursed above or below that average. In addition, we excluded some details for simplicity, like the possibility that a patient who has a tip fold-over may choose not to undergo repeat surgery, and that tip fold-over may have additional costs associated with it including additional audiology and physician appointments in dealing with poorer audiological outcomes associated with tip fold-over.
But there are also benefits to iCT which we did not quantitatively include in our models but are important to note. For example, improved hearing outcomes by better electrode placement likely occur.6,19 However, incorporation into models such as ours would require prospective studies of individuals with optimally and suboptimally placed EA. Moreover, intraoperative imaging gives near real-time feedback to surgeons who may adjust their technique based on that feedback, suggesting an additional role for CT scanning in temporal bone laboratory CI training. And, most importantly, from a patient perspective, the personal cost of revision surgery (including discomfort, time off work, and decreased productivity) could be quantified albeit subjectively.
In conclusion, these models suggest that iCT can align incentives for payers, institutions, and patients by being financially beneficial to the former and improving patient care and outcomes for the latter. Institutions and payers should evaluate iCT and similar technologies using models like these with their respective parameters, and future research should take into account the economic realities of such technologies in addition to patient outcome data.
Footnotes
Authors’ Note
Both RFL and MBM participated in all steps of data collection and analysis, manuscript writing, and revisions.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RFL is a consultant for Advanced Bionics and Spiral Therapeutics and the PI of an NIH grant which utilizes inCT scanning in image-guided, minimally invasive cochlear implantation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported by Award Number R01DC008408 from the National Institute on Deafness and Other Communication Disorders. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Deafness and Other Communication Disorders or the National Institutes of Health.