Abstract
Introduction:
The anterior ethmoidal artery (AEA) demonstrates anatomic variability relative to its descent from the anterior skull base. Our study’s objective was to assess for correlation of AEA descent and laterality, in addition to correlation of AEA descent and the presence of supraorbital ethmoid cells (SOEC) and concha bullosae (CB).
Method:
A retrospective study was performed at a tertiary rhinology center from January 2019 to January 2020. Noncontrast maxillofacial computed tomography scans were examined independently by 2 fellowship trained rhinologists. The vertical distance from both left and right AEAs to the ipsilateral skull base were compared and correlated with the presence of ipsilateral SOEC and CB.
Results:
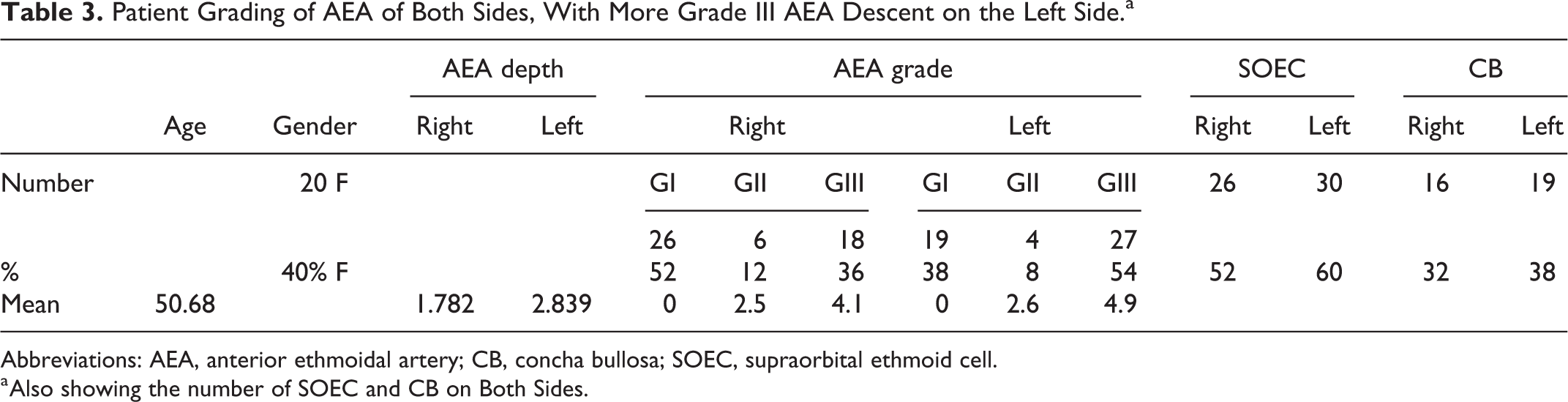
Computed tomography scans from 50 subjects were included. Mean age was 50.68 years (40% females). The distance of AEA to the skull base was greater on the left when compared to the right (62% vs 48%) (P < .05). The left AEA had an average descent of 2.84 mm versus 1.78 mm on the right (P < .05). An SOEC was present in 56% of cases. Thirty-eight percent of subjects had both SOEC and AEA descent on the right, while 52% of subjects had both on the left. This reached a statistical significance on both sides (P < .05). Concha bullosa was present in 35% of cases, with both AEA descent and CB present in 16% on the right, and 32% on the left.
Conclusion:
The AEA displays variability in vertical descent from the skull base, with greater variability on the left. These findings implore vigilance with evaluation of preoperative imaging and during sinus surgery, especially in the presence of SOEC and CB.
Introduction
The anterior ethmoidal artery (AEA) demonstrates anatomic variability relative to its inferior displacement, or descent, from the anterior skull base at the orbital plate of the frontal bone (OP). The trans-ethmoid position of the AEA is influenced by the artery’s relationship to the OP, lateral lamella of the cribriform plate (LLCP), supraorbital ethmoid cells (SOEC), and concha bullosae (CB) when present. The position of AEA in the ethmoid sinus has been reported to be very variable even between 2 sides of 1 individual. In terms of its distance from skull base, the artery may be within the skull base, particularly when the roof of ethmoid is low, or courses freely below skull base within a bony canal and being connected to skull base by a thin bony mesentery. Grading systems exist to describe its course. The AEA may transit the ethmoid sinus within the skull base (grade 1), adjacent but inferior to, described as prominent from, the inferior surface of the skull base (grade 2), or freely suspended in a mesentery inferior to the skull base (grade 3). This relationship of AEA with skull base is important because the chance of injuring the artery during surgery is higher when it lies freely below skull base. 1
The AEA is regarded as one of the anatomic features deemed “high-risk” in endoscopic sinus surgery (ESS). It is particularly dangerous because if transected at its lateral end, retraction may occur within the orbit leading to a retro-orbital hematoma. This complication may lead to compression of optic nerve and ultimately blindness. Conversely, a medial injury where the artery enters the LLCP may result in CSF leak or cerebrovascular insult or injury. 2
Lannoy-Penisson et al 3 and Ko et al 4 outlined a classification used to describe the course of AEA variation relative to skull base. The location of AEA is categorized as grade I when it is found within the skull base. It is grade II when it courses under skull base and considered as prominent. Grade III classification refers to AEA that travels freely at a distant from skull base. This classification is useful, as the presence of grade III AEA may warn the surgeons to exercise caution during surgery. The purpose of this study is to highlight the incidence of AEA descend between sides and its correlation with the presence of CB and pneumatized SOECs.
Methods
All aspects of this study were reviewed and approved by the general institutional review board of Baton Rouge (IRB00005439). An electronic database was queried for patients seen at Sinus and Nasal Specialists of Louisiana who obtained a sinus computed tomography (CT) scan from January 2019 to January 2020. Our exclusion criteria included patients under the age of 18 years, those with primary ciliary dyskinesia, immunodeficiencies, sinonasal polyposis, neoplasia, and also those with previous history of surgery to the nose and sinuses. Medical charts were reviewed and demographic data were collected, including current age, sex, and prior medical and surgical interventions. All available clinical data were recorded.
Computed tomography scans were performed using a Xoran 0.2 mm slice multi-detector CT scanner. Noncontrast maxillofacial cone beam CT scans (bone windows) were examined using a picture archiving and communication system independently by 2 fellowship trained rhinologists blinded to clinical information. Images were assessed using bone windows in the coronal plane with the palate fixed parallel to the Frankfort horizontal line. The vertical distance at the midpoint from the most inferior point of descent of the AEA to the inferior border of the ipsilateral orbital plate of the OP was digitally measured in mm on the left and right sides. The images were also assessed for the presence of SOECs and CB. The distances from both left and right AEAs to the ipsilateral skull base were compared within the cohort and correlated with the presence of SOEC and CB ipsilateral to the measured side.
Statistical analyses were performed using SPSS version 22 (SPSS Statistics for Windows, version 22.0; IBM). Descriptive data are presented as percentages and means ± SD. Kendall tau-B was used for ordinal values. χ2 analysis was used for relationships of nominal variables. The prevalence of related comorbidities and frequency of previous surgical procedures was also compared across treatment cohorts using Pearson χ2 analysis for relationships of nominal variables. Student t test (2-tailed) was used for comparisons of parametric data. Results were deemed significant with a P value of <.05.
Results
A total of 50 patients, 50.68 (range 19-88 years) years of age and 40% females were assessed. Of these patients, 6% were smokers and 16% were diagnosed with asthma. The prevalence a descended AEA was greater on the left than the right with 31 cases on the left side versus 24 cases on the right side (62% vs 48%; Figure 1) with a P < .05. We evaluated for significance between the 2 sides using the paired t test, and there was statistical significance between both sides (P = .00004). The average depth of descent (mm) was 2.84 on left side versus 1.78 on the right side (Table 1, Figure 2). We categorized the AEA descend into different grades, grade I the artery is within the skull base, grade II is prominent from skull base, and grade III is suspended below the skull base (Table 2, Figure 3). When evaluating those with AEA descent (grades II and III), the average depth of descent (mm) was 4.58 on the left and 3.7 on the right.
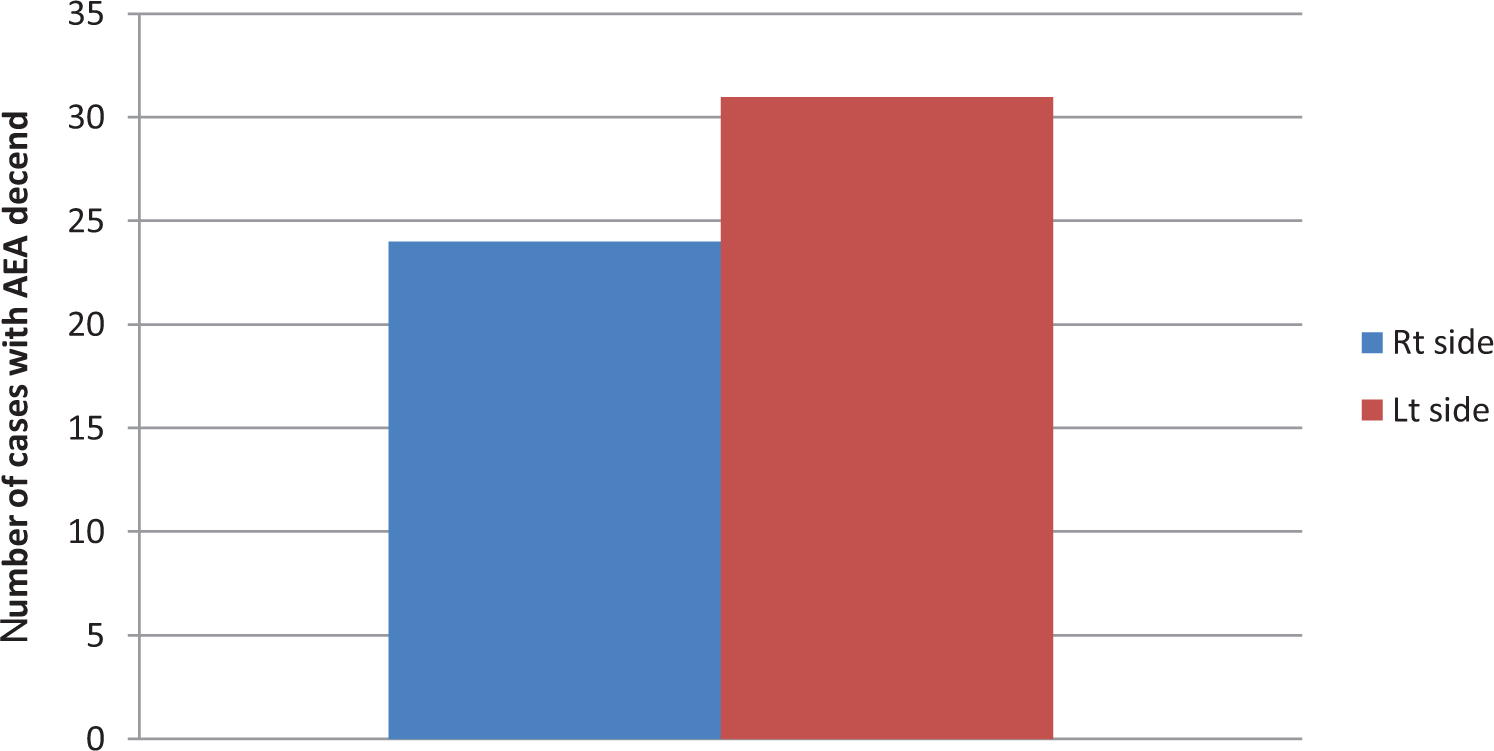
Number of cases with anterior ethmoid artery descent.
Number of Cases With Anterior Ethmoid Artery Descend and Mean Distance of Descent From the Orbital Plate of the Frontal Bone (mm).
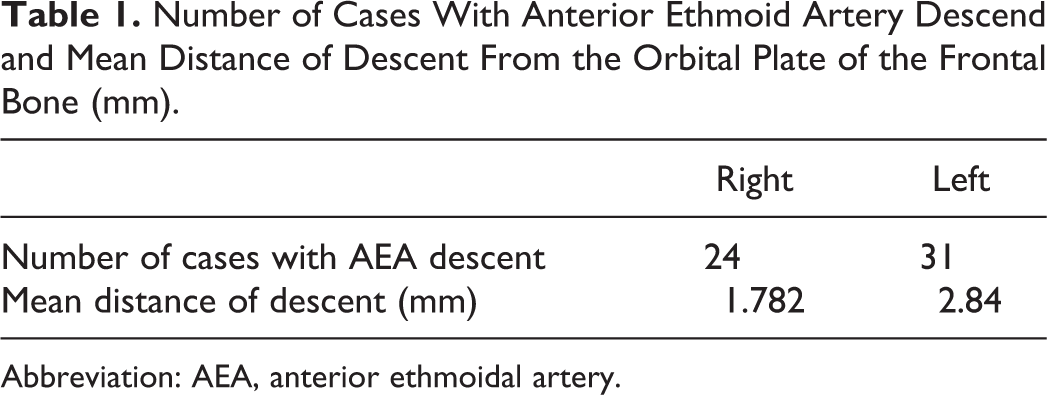
Abbreviation: AEA, anterior ethmoidal artery.
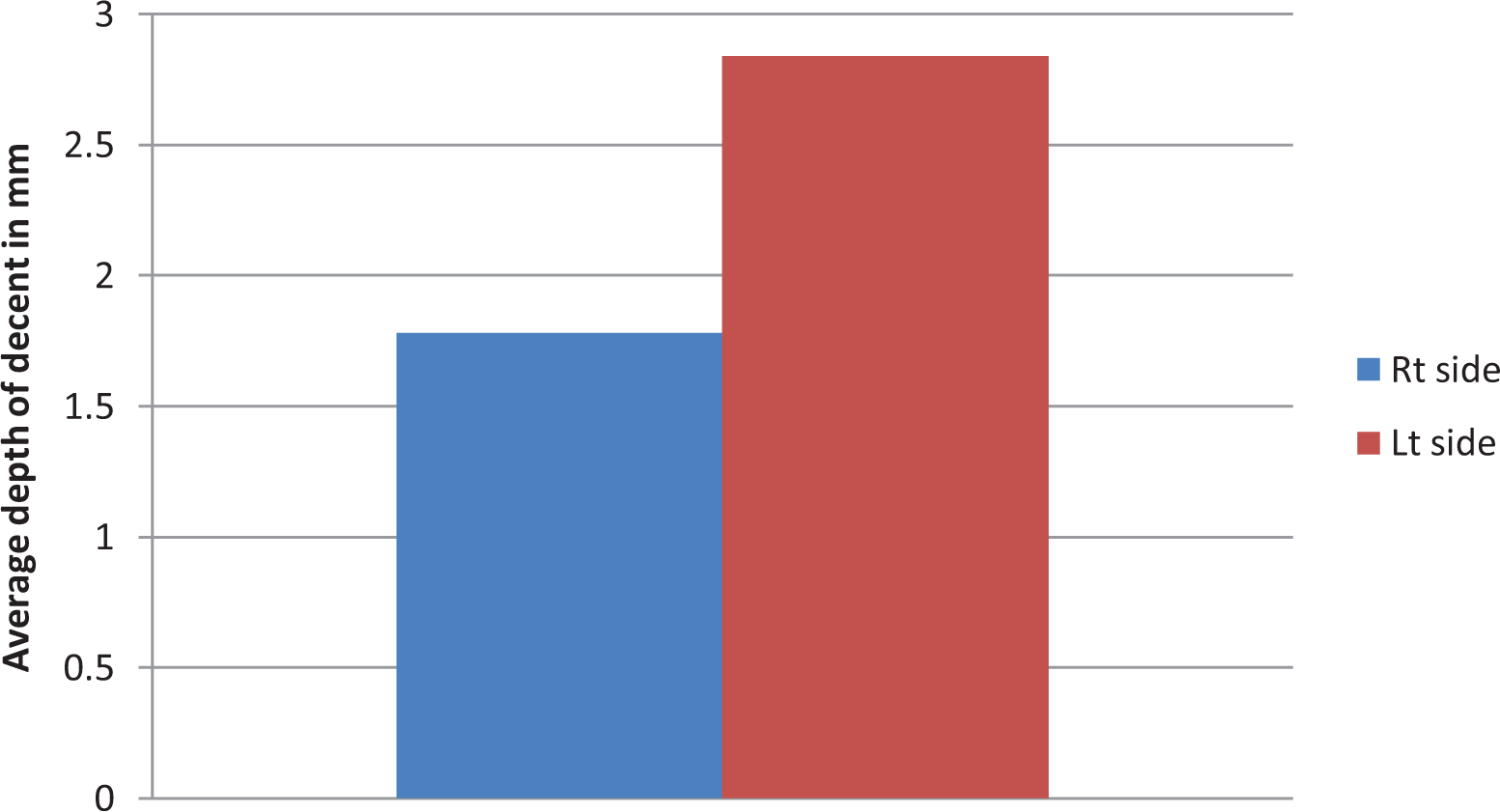
Mean distance of descent from skull base (mm).
Grade of Anterior Ethmoid Artery Relation to Skull Base

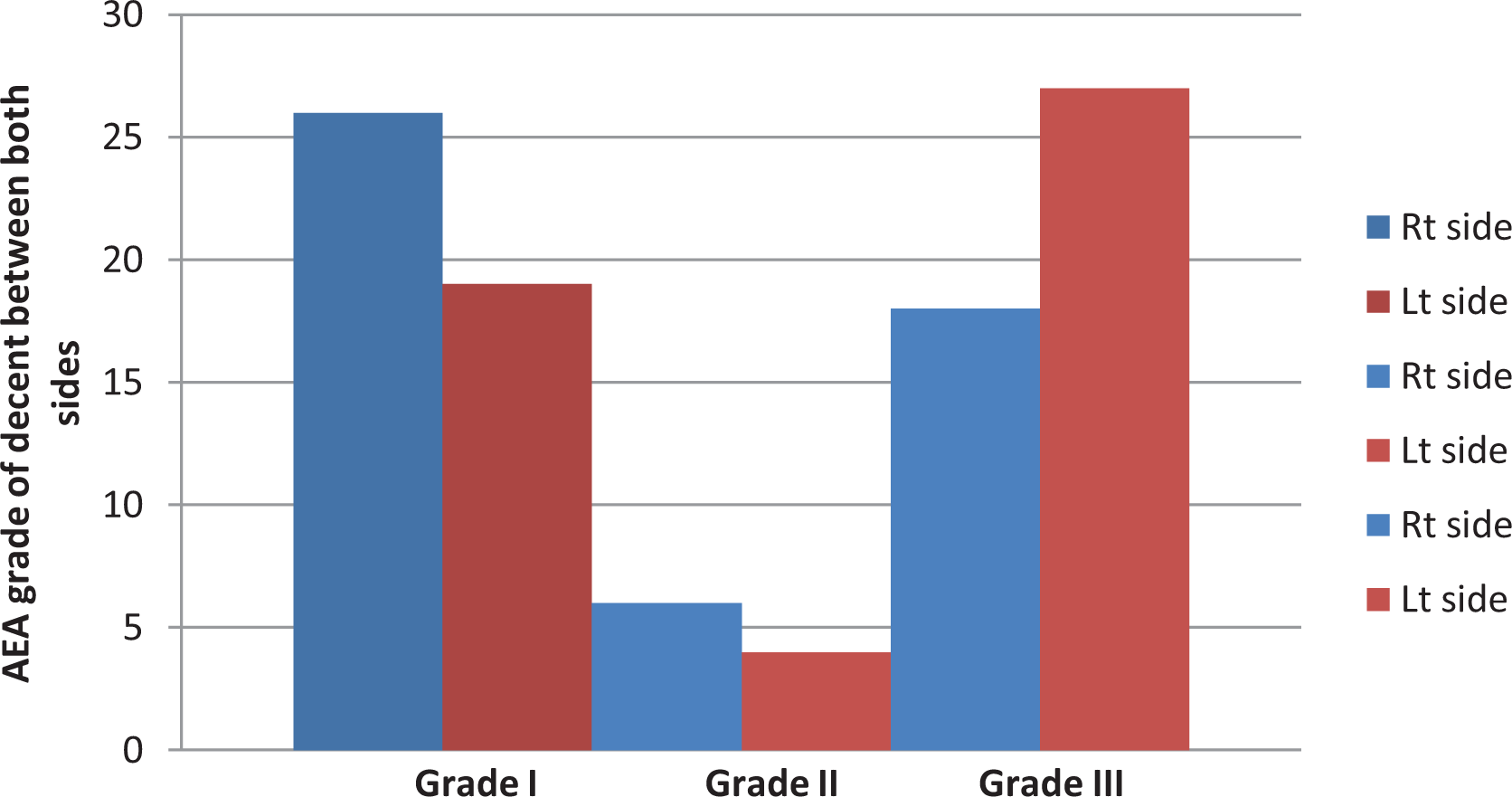
Grade of anterior ethmoid artery descent.
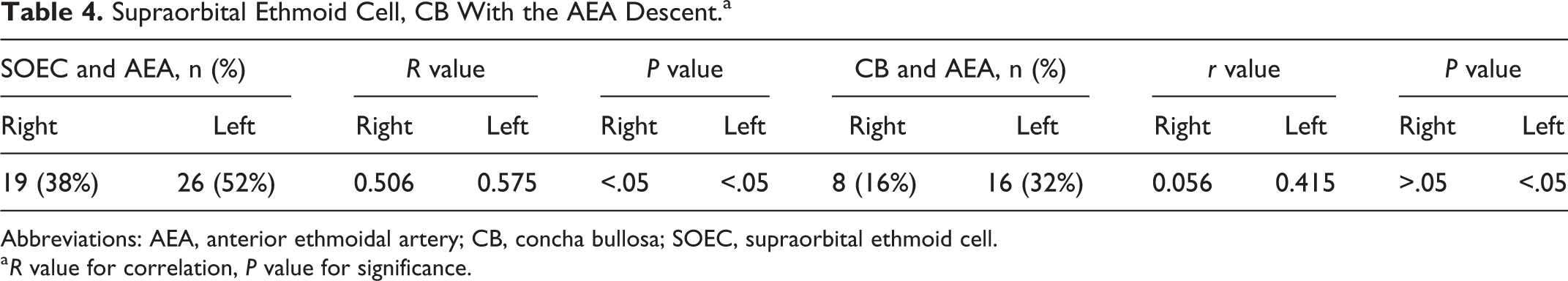
Supraorbital ethmoid cell was present in 56%, with 52% on the right and 60% on the left. We found that 38% of patients have both SOEC and AEA descent on the right and 52% patients on the left. There was a moderate correlation between the presence of SOEC and AEA descent on each side, with r = 0.506 on the right (P = .0002) and 0.575 the left (P = .00001; Figure 4).
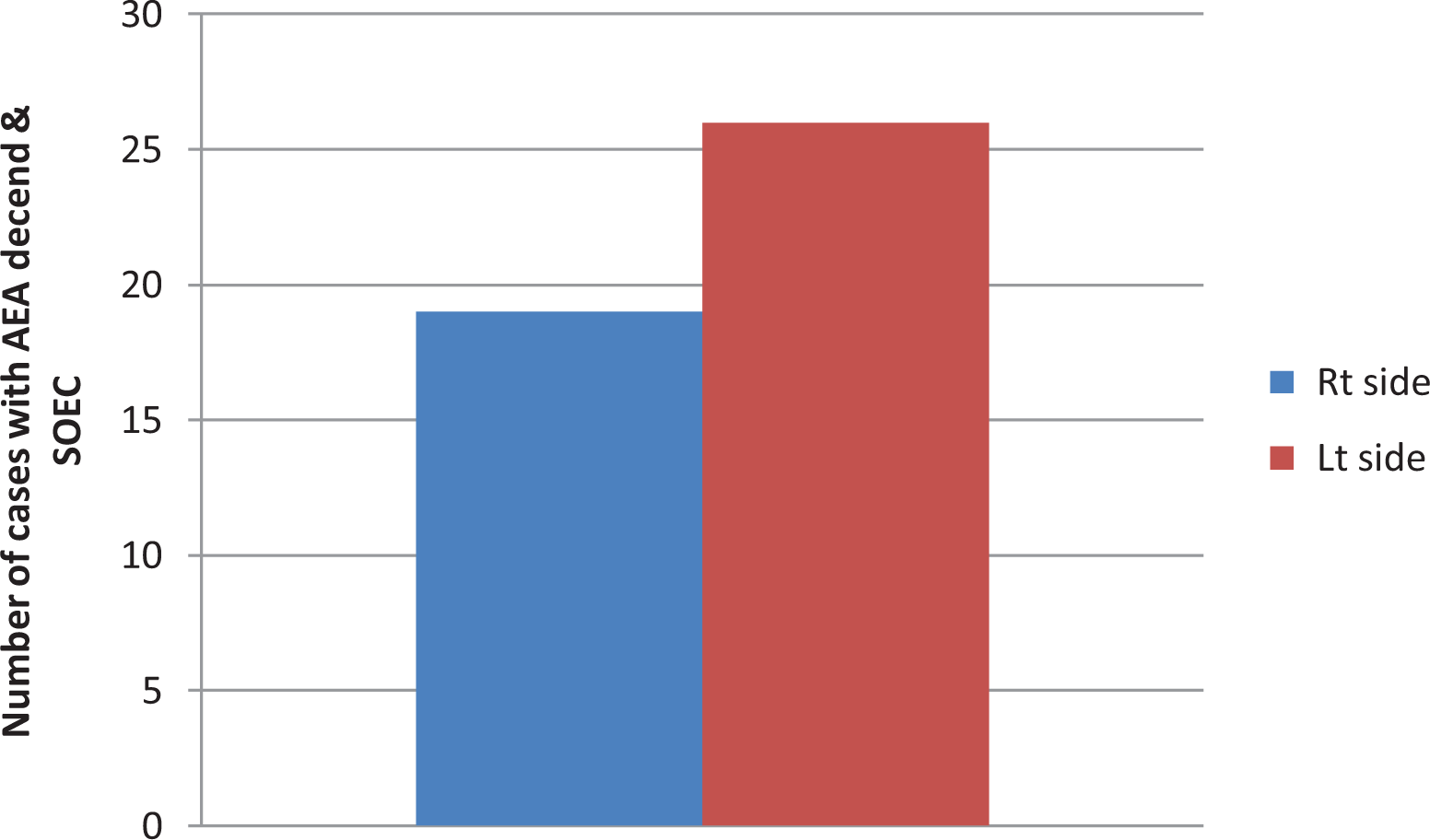
Correlation of anterior ethmoid artery descent and supraorbital ethmoid cell.
Concha bullosa was present in 35% of cases, 32% on the right, and 38% on the left. Patients with both AEA descent and CB were 16% on the right, while 32% had both on the left. There was a moderate correlation between the presence of CB and AEA descent on the left (r = 0.415; P = .003), while there was a weak correlation on the right (r = .056), which did not reach statistical significance (P = .7; Figure 5, Tables 3 and 4). Figure 6 is displaying the way we used to measure the depth of suspension of AEA from skull base, where it was measured at the orbital exit at the lamina papyracea, with the left-side AEA is more suspending than the right-side AEA.
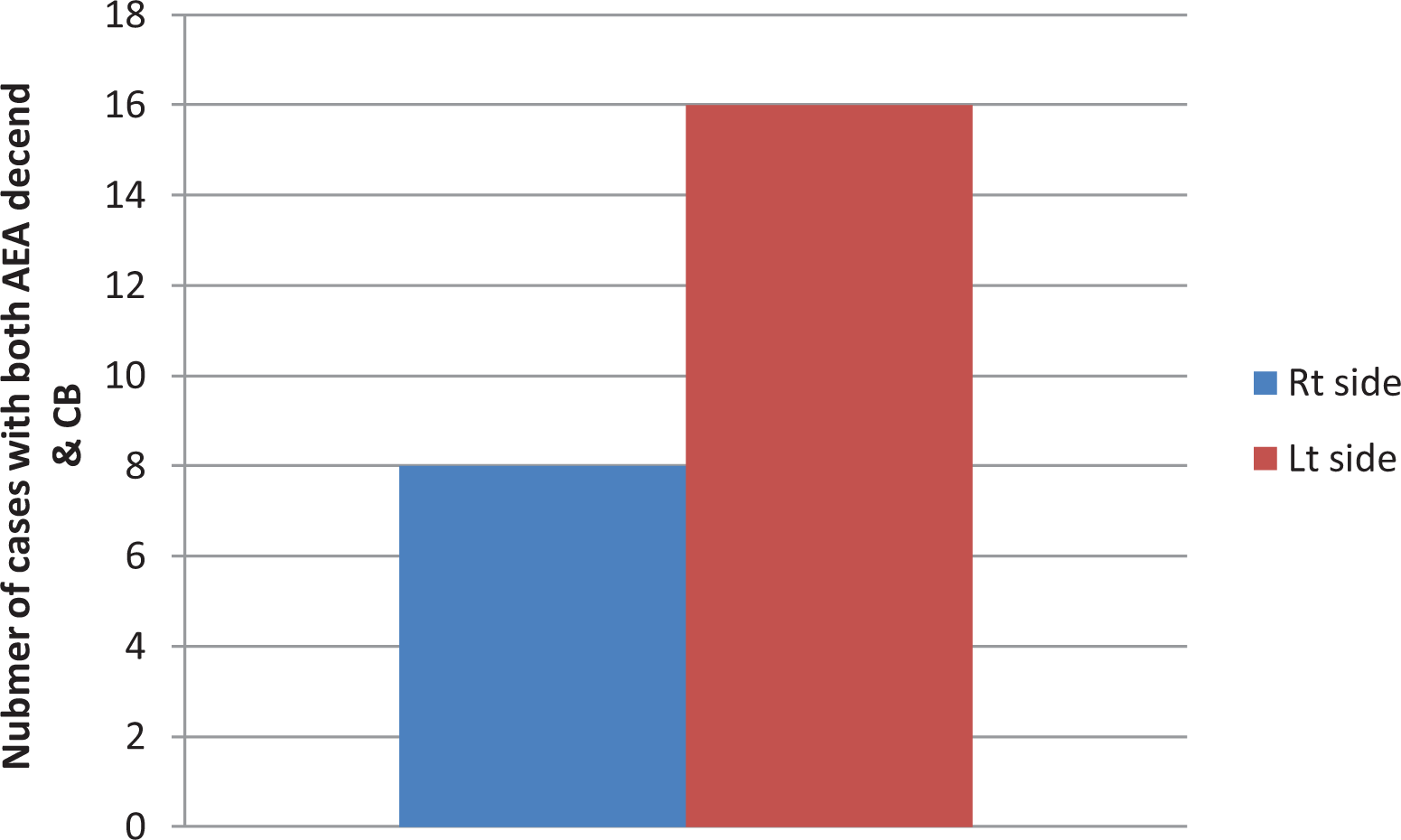
Correlation of anterior ethmoid artery descent and concha bullosa.
Patient Grading of AEA of Both Sides, With More Grade III AEA Descent on the Left Side.a
Abbreviations: AEA, anterior ethmoidal artery; CB, concha bullosa; SOEC, supraorbital ethmoid cell.
a Also showing the number of SOEC and CB on Both Sides.
Supraorbital Ethmoid Cell, CB With the AEA Descent.a
Abbreviations: AEA, anterior ethmoidal artery; CB, concha bullosa; SOEC, supraorbital ethmoid cell.
a R value for correlation, P value for significance.
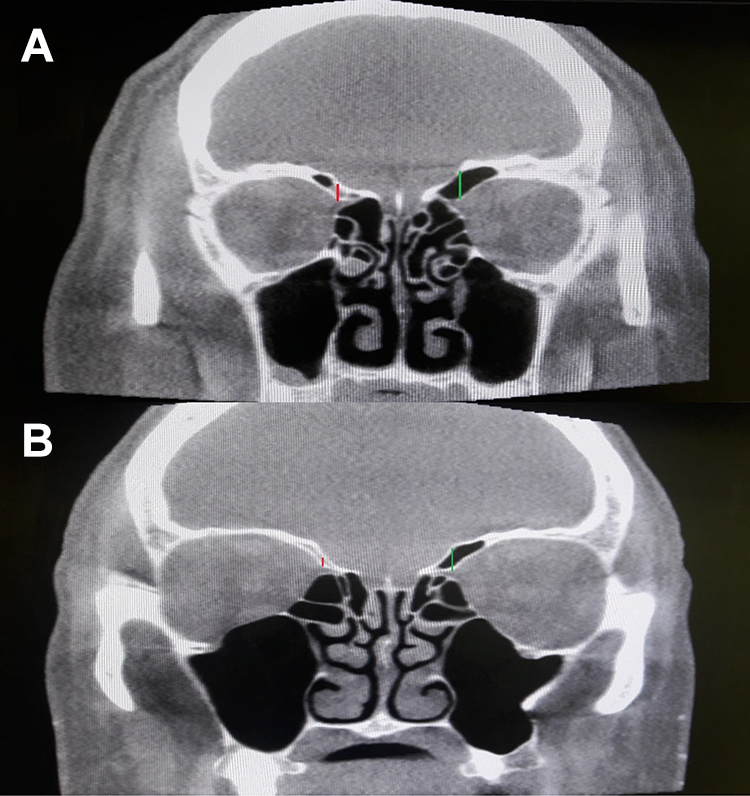
A and B, CT scan of 2 patients showing the AEA suspension from skull base. Measured from the point of orbital exit at the lamina papyracea. AEA indicates anterior ethmoidal artery; CT, computed tomography.
Discussion
The AEA arises from the ophthalmic artery, a branch of internal carotid artery. In its course from the orbit to the olfactory fossa and back into the nasal cavity, the artery crosses 3 cavities of the skull; the orbit, the ethmoid sinus, and the anterior cranial fossa. The AEA enters anterior ethmoidal canal through the medial wall of the orbit after piercing the frontoethmoidal suture line. It courses across ethmoid sinus together with anterior ethmoidal vein and anterior ethmoidal nerve inside a bony canal. The artery runs obliquely from posterolateral to anteromedial direction to reach the LLCP and enters the olfactory fossa. After intracranial entry into the olfactory fossa, AEA turns anteriorly forming the anterior ethmoidal sulcus in LLCP. The artery then reaches the nasal cavity through the anterior ethmoidal orifice of the CP and divides into anterior septal branches and anterior lateral nasal branches. 5
In our study, the prevalence of a descended AEA was greater on the left than the right, with the average depth of descent from skull base of the left AEA was found to be more than its equivalent on the right side, which showed statistical significance. Surgeons should use this as a warning for the likely possibility of the left AEA being descended from skull base. This variability has not been previously described in the literature, but some authors noted the AEA descent from skull base without consideration to the variability between both sides of the same patient.
Several authors analyzed the relationship of AEA with skull base. Lisbona Alquezar et al 6 viewed a total of 542 AEA from different articles, where he stated that in 361 (66.6%) cases, the artery was located at the level of skull base, while in 181 cases (34.4%), the artery was below the level of skull base. In 2 other studies, Moon et al 7 and Araujo et al 1 reported that AEA was embedded in skull base in 85.7% and 83.3% of cases, respectively. In 34 specimens studied by Simmen et al, 2 he mentioned that the AEA was closely placed or in the skull base in 22 (64.7%). In the remaining 12 specimens, the mean vertical distance from the skull base was 3.5 mm (range 1-8 mm). This demonstrates the wide variability in the horizontal and vertical position of the AEA. On the contrary, other authors as Cankal et al, 8 Joshi et al, 9 Ko et al, 4 and Yang et al 10 found that AEA was suspended from skull base within a mesentery connecting it to skull base in most cases.
In our study, we found that the prevalence of SOEC was greater on the left side, and there were more cases that had both SOEC with a descended AEA on the left, which reached statistical significance bilaterally. The prevalence of CB was also greater on the left, in addition to the finding of both CB and descended AEA. This correlation reached statistical significance on the left side but not on the right. This is clinically relevant in that patient with a CB will have a higher likelihood of a unilateral descended AEA.
The SOEC can be defined as pneumatization of the orbital plate of the OP lateral to the most medial plane of the lamina papyracea. Presence of SOECs also varies greatly across ethnicities. Previous studies have shown that 5.4% of Chinese patients, 11 2.4% of Korean patients, and 64.4% of patients of Western ethnicities have SOECs. 12 Joshi et al, 9 Jang et al, 13 Monjas-Canovas et al, 14 Souza et al, 15 and Yenigun et al 16 indicate that the prevalence of SOECs varies widely between 15% and 55.8% in their studies. This variability signifies the importance of identifying the different patterns of pneumatization between different patients. Joshi et al, 9 Jang et al, 13 and Yenigun et al16 further analyzed the effect of pneumatization of SOECs on the distance of AEA from skull base, and all authors agreed that there exists a strong correlation between the presence of SOECs and the distance of AEA from skull base. A well pneumatized SOEC causes AEA to be located further below skull base making it more susceptible to injury during surgery. Considering this observation, Jang et al 13 proposed that SOEC may serve as a consistent landmark for locating AEA. In his study, Simmen et al 2 correlated pneumatization of SOEC with the location of the AEA at the level of the skull base. Of the 34 specimens in his study, he found 10 cases with large SOEC pneumatization, and the AEA was located 3.7 mm below the level of the skull base on average (range 1-8 mm). In 6 cases, there was a small cell with less pneumatization, and the AEA was located at 0.2 mm from the skull base on average (range 0-1 mm). In the remaining 18 cases, there was poor pneumatization of SOEC and the AEA was at the level of the skull base, except in one case where it was hanging at 1 mm below the skull base.
Yenigun et al 16 found the pneumatization of the medial concha, at a rate of 32.6%, with the right (33.2%) and left (32.1%) being similar. No statistically relevant correlation was found between the presence of concha pneumatization and AEA visualization (right: P = .79, left: P = .72).
Keros 17 published his analysis of skull base variations within the ethmoid roof by creating 3 classifications depending on the length of the LLCP: type 1 between 1 and 3 mm, type 2 between 3.1 and 7 mm, and type 3 greater than 7 mm. His classification scheme has become widely adopted and is universally used by sinus surgeons to identify patients with skull base configurations at risk of injury during sinonasal surgery. Li et al 18 evaluated patients with and without SOECs and found an increase in the distance between the skull base and AEA with higher Keros classification. The presence of SOEC is also associated with a further increase in the distance and an increase in the prevalence of AEA descent from the skull base in all Keros types. However, these findings were only statistically significant in patients with Keros type 2. Poteet et al 19 further described the position of the AEA with respect to the skull base in a retrospective CT study of 101 maxillofacial scans; they found that The AEA was noted to be below the skull base with an increasingly greater frequency with increasing skull base height and Keros classification. In 55.0% of Keros type 3 cases, the AEA was found below the skull base, which is decreased to 29.5% of Keros type 2 measurements and 0.0% of Keros type 1 measurements for either sex.
Çomoğlu et al 20 investigated the relationship between the location of the AEA in the skull base and the depth of the olfactory fossa. This study found the depth of the olfactory fossa measured 5.88 ± 1.66 mm in individuals in whom the AEA coursed below the skull base on at least 1 side, and 4.99 ± 1.40 mm in individuals in whom it coursed within the skull base bilaterally. Floreani et al 21 demonstrated that on both sides, the mean height of the lateral lamella in those cases in which the artery coursed with the skull base was 4 mm. He applied it clinically to the Keros classification such that if the lateral lamella is classified as Keros grade 2 or 3 (greater than 4 mm long), then there is a 17.3 times greater likelihood that the anterior ethmoidal canal will be found suspended within a mesentery. A strong correlation was found between the increasing height of the lateral lamella and the incidence of the anterior ethmoidal canal being suspended within a mesentery.
A potential confounding variable of this study is that the patients presenting to a tertiary rhinology center are potentially more likely to have a variable anatomy, often associated with the issues leading to medical consultation, than the general population. However, the exclusion criteria were attempted to account for this possibility.
Our study helps to better understand the variability and complex anatomy of the AEA and confirms the importance of vigilant preoperative assessment of these parameters on CT imaging to avoid unnecessary injury during ESS.
Conclusion
The AEA has a very variable position even within the same individual. In our study, we detected that the left AEA is more likely to be descended from the skull base with an average descent of 2.84 mm. These findings emphasize the importance of evaluation of preoperative imaging and care taken during ESS, especially in patients with CB and SOEC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
