Abstract
Extramedullary plasmacytomas represent a rare group of B-cell malignancies that arise outside the bone marrow and their disease process is still poorly understood. Here, we will describe a case of a 76-year-old patient who presented with a large chest wall and subglottic mass causing airway compromise and stridor. Biopsies showed atypical plasma cells with prominent nucleoli which were in keeping with an extramedullary plasmacytoma. Disease progressed despite surgical debulking, targeted radiotherapy, and multiple chemotherapy regimens. Although response to treatment is classically good, patients occasionally present with aggressive disease.
Introduction
Plasmacytomas represent tumours formed by abnormal plasma cells and can either be found in bone marrow or soft tissues. Solitary extramedullary plasmacytomas are diagnosed in absence of systemic disease but have the potential of evolving into multiple myeloma.1,2
Case Report
A 77-year-old male presented with a history of worsening dyspnoea, inspiratory stridor over several days on a background of insidious anorexia, and weight loss. On examination, he had a tender sternal mass with associated skin changes which had developed over the past 6 weeks. Nasendoscopy revealed a left vocal cord palsy and a mass involving the hypopharynx and subglottic larynx. There was no palpable cervical adenopathy.
Computed tomography (CT) delineated the lesion which involved the left piriform sinus, postcricoid hypopharynx, upper esophagus, cricoid cartilage, and subglottic larynx (Figure 1), and a separate sternal mass extending between the anterior thoracic wall and the superior mediastinum. Fluorodeoxyglucose positron emission tomography CT (FDG-PET/CT) confirmed avidity of these masses and of the posterolateral pleura and left atrial appendage node.
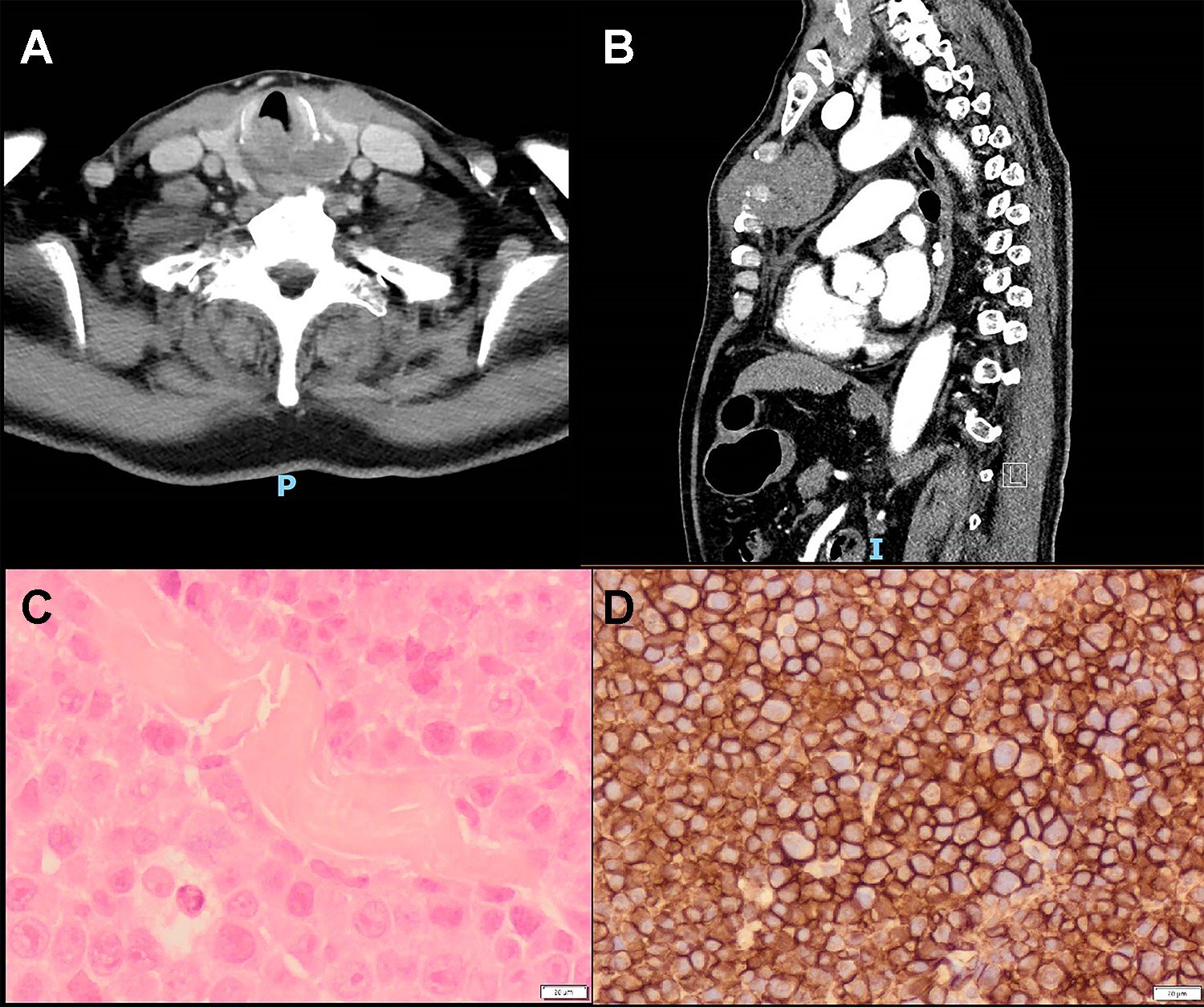
Computed tomography demonstrating (A) the lesion in the left piriform sinus and postcricoid hypopharynx eroding the cricoid cartilage, (B) the mass in the sternum, and photomicrographs showing (C) atypical plasma cells with prominent nucleoli, (D) atypical plasma cells expressing CD138.
The subglottic airway was debulked with an 8 W superpulse CO2 laser. Suspension direct laryngoscopy with jet ventilation was used and the supraglottic mass extending inferiorly from the posterior commissure was debulked at 50% to maintain his airway. Samples were sent fresh for histopathology and flow cytometry.
An ultrasound guided biopsy of the sternal mass was also performed. Histopathology revealed an undifferentiated malignant tumour composed of small round cells with hyperchromatic nuclei positive for CD38, MUM1, EMA, CD99, and CD79a. There were medium-sized ovoid cells showing mildly pleomorphic round/oval nuclei with prominent nucleoli, moderately pale cytoplasm, and occasional multinucleation. Cells were plasmacytoid with a mitotic count >15/10 hpf and atypical mitotic figures. There was lambda light chain restriction and cyclin D1 positivity. Additional work-up revealed strong and diffuse MYC expression, variable p53 expression, >80% Ki67 expression, and negative BRAF. Both blood and urine immunoglobulin D lambda paraproteins were high, and bone marrow flow cytometry was negative.
Following the initial management with intravenous corticosteroids, adrenaline nebulizers, and CO2 laser debulking, he was commenced on systemic chemotherapy. Following a single cycle of VTD (bortezomib-thalidomide-dexamethasone), he was changed to VCD (bortezomib-cyclophosphamide-dexamethasone) due to thalidomide intolerance, which he continued for a further 2 cycles. In view of progression of the sternal mass, local radiotherapy was delivered at 20 Gy in 5 fractions with significant symptomatic improvement. Biopsies confirmed highly proliferative plasmacytoma. In addition, a repeat PET showed significant disease progression with new multiple chest wall deposits, as well as an increase in size and FDG-avidity of the pericardial, pleural, pulmonary, sigmoidal, and skeletal disease.
He was commenced on RCD (cyclophosphamide-lenalidomide-dexamethasone), but despite further chemotherapy he developed a 12 mm pericardial effusion with tamponade a month later. This was drained percutaneously to reveal positivity for CD 138 suggestive of malignant effusion. A targeted immunomodulatory regime with RD (revlimid-dexamethasone) was trialled with no response. This was changed to BP (bendamustine-prednisolone) which showed minimal improvement. The patient passed away 8 months following diagnosis due to disease progression.
Discussion
Plasma cell neoplasms are categorized as multiple myeloma, plasma cell leukaemia, solitary plasmacytoma of bone, and solitary extramedullary plasmacytoma (EMP). Plasmacytoma arises in bone in 70% of patients and soft tissue in the remainder and accounts for 5% of plasma cell neoplasms. 3 Solitary plasmacytoma of bone can accompany multiple myeloma and suggests a more aggressive phenotype. Extramedullary plasmacytoma is a clinically separate entity and is diagnosed in the absence of hypercalcemia, renal function impairment, anaemia, and bone involvement.1,4 Up to 25% of patients with solitary EMP develop multiple myeloma later on. 5
Solitary EMP usually presents between the fifth and seventh decades and 90% arise in the upper aerodigestive tract, especially the nasal cavity and maxillary antrum. Solitary EMP may present with nasal discharge, epistaxis, swelling, pain, or obstruction. Solitary EMP has been reported in the pharynx and larynx, where it may present with dyspnoea and stridor. 6 The etiology is still unclear, but chronic irritation and viral infection may have a role in pathogenesis. 7 To differentiate EMP from multiple myeloma, reactive plasmacytosis, immunoblastic lymphoma, and plasma cell granuloma, the analysis of bone marrow aspirate may be needed in addition to histopathological analysis. Bence-Jones protein is present in 16% of cases of EMP and 25% will have a positive myeloma screen. 5 The presenting lesion is usually imaged by CT or magnetic resonance imaging and FDG-PET/CT is mandatory for staging given the potential for multifocality. 8 It is important to note that about 20% of patients have lymph node metastases at presentation. 5 The monoclonal hyperproteinemia is monitored and resolution within the first year reflects more favorable outcomes. 9 Chemotherapy is indicated for tumours larger than 5 cm and high grade or recurrent disease. 10 Furthermore, radiation therapy for the primary lesion has been shown to be superior to both chemotherapy and surgery. 11 Surgery alone plays little role in the management of EMP. 12
Recurrence rates are low and up to 70% of patients are disease free at 10 years. However, we present a difficult case of multifocal, aggressive EMP, despite nonprogression to multiple myeloma. Salvage chemotherapy with the DT-PACE regime (dexamethasone, thalidomide, cisplatin, doxotubicin, cyclophosphamide, and etoposide) has shown promising results in B-cell malignancies but is associated with high mortality and for this reason was not used in our patient. 13 Targeted IRD treatment (ixazomib-lenalidomide-dexamethasone) was also discussed but the patient did not meet the high eligibility criteria. 14
Plasma cell neoplasms are a rare cause head and neck masses. Otolaryngologists should remain cognizant of the possibility of systemic malignancy when lesions are found in the upper aerodigestive tract.
Learning Points
Extramedullary plasmacytoma is a rare B-cell malignancy arising in the upper aerodigestive tract in 90% of cases
Diagnosis must include histopathology, flow cytometry studies, molecular analysis of bone marrow aspirate, and FDG-PET/CT to rule out occult lesions
Managing these patients involves surgery and radiotherapy, although chemotherapy is necessary in patients with multifocal disease
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
