Abstract

A 94-year-old female was referred to our hospital with slow-growing mass on the mouth floor mass for 3 months. Physical examination revealed about 2.5 × 2 cm sized firm round submucosal mass at the right mouth floor that was palpable without pain. Ultrasonography-guided core needle biopsy was performed to identify the type of tumor on the first visit. Pathologically, the tumor cells were small with poorly defined borders, bare round or oval nuclei, and sparse cytoplasm. Immunohistochemical (IHC) analysis demonstrated that the tumor cells were positive for cluster of differentiation (CD)56, synaptophysin (SYN), and chromogranin but negative for thyroid transcription factor (TTF)-1 and cytokeratin 20 (CK20). Therefore, small cell neuroendocrine carcinoma (SNEC) in the head and neck was suspected. Positron emission tomography/computed tomography (PET/CT) and CT scan were performed to confirm whether it was a metastatic lesion, and fluorodeoxyglucose-18 uptake was noted at the right sublingual region and colon hepatic flexure (Figure 1). Based on colonoscopic biopsy, the abdominal lesion was diagnosed with colorectal adenocarcinoma. Wide resection of the right sublingual region including the sublingual gland and laparoscopic right hemicolectomy were performed simultaneously. Finally, histopathological findings confirmed that SNEC originated from the sublingual gland, and the colon specimen was diagnosed with adenocarcinoma. Thus, the patient was diagnosed with 2 malignancies: primary SNEC originating from the sublingual gland and colorectal adenocarcinoma. Unfortunately, the patient refused adjuvant radiation therapy, due to old age and poor general condition. The patient is currently undergoing monthly follow-up, and recurrence or metastasis has not been observed for 5 months.
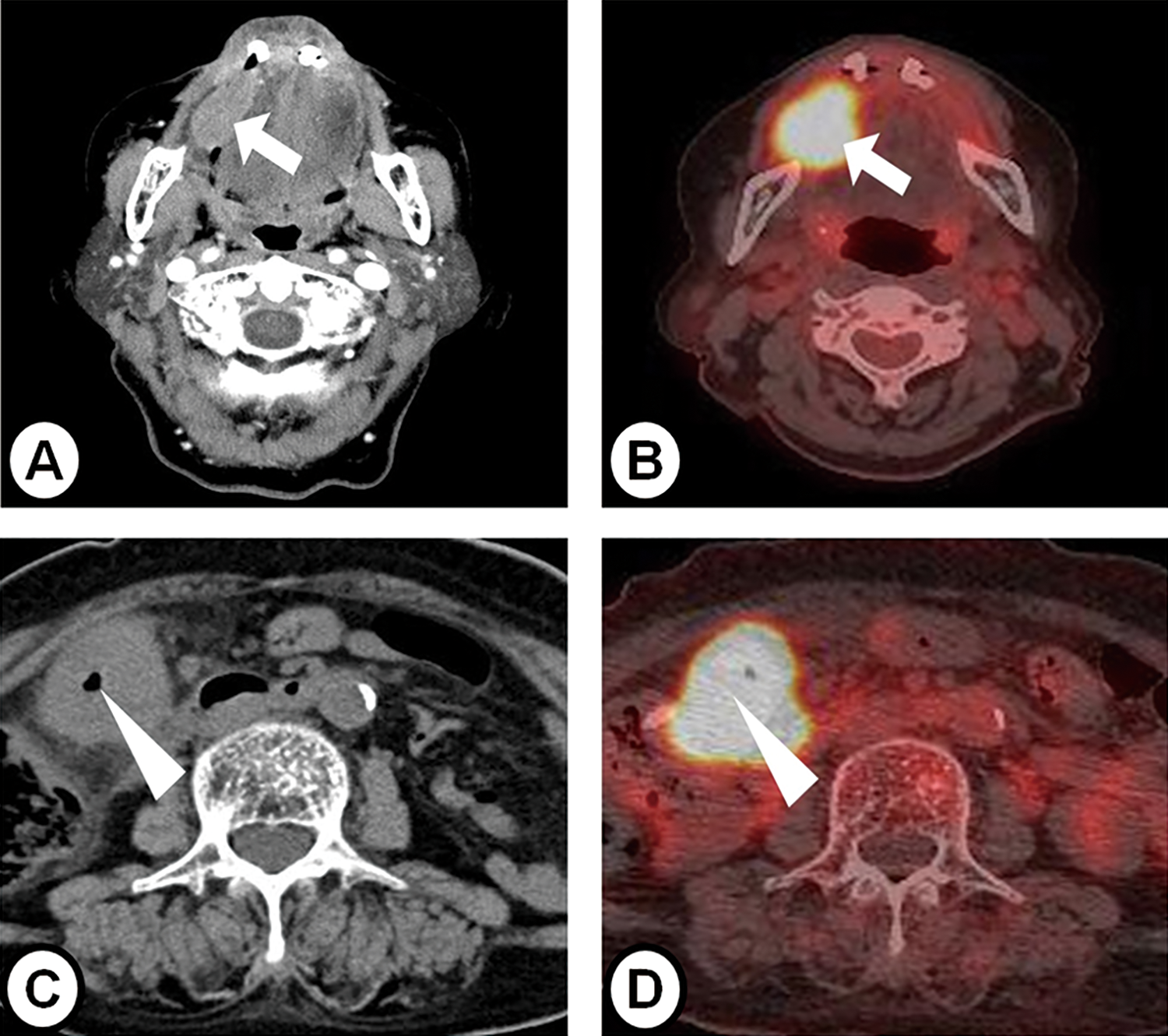
Computed tomography (CT) and positron emission tomography (PET)/CT scans: (A) Axial contrast-enhanced neck CT scan shows a 2.7 cm × 1.5 cm sized well-marginated isodense lesion on the right side of the sublingual region(arrow). (B) PET/CT scans show a fluorodeoxyglucose (FDG) uptake in the same place (arrow). (C) Axial abdominal CT scan shows diffuse wall thickness at the colon hepatic flexure (arrow head). (D) PET/CT scans shows increased FDG uptake in the abdominal lesion (arrow head).
Small cell neuroendocrine carcinoma originates from the neuroendocrine cell. There are many pulmonary neuroendocrine cells in the bronchial epithelium. Therefore, SNEC mainly occurs in the lung. The extrapulmonary form accounts for only 0.1% to 0.4% of all SNEC cases. 1 Furthermore, SNEC rarely originates in the salivary gland and accounts for <1% of all tumors in the salivary gland.2-7 Most cases of SNEC in the salivary glands occur in the parotid gland. 2 This is the first report of SNEC of in the sublingual gland.
Neuroendocrine tumors (NETs) are stratified into well-differentiated NETs and poorly differentiated neuroendocrine carcinomas (NECs). Well-differentiated NETs are further separated into 2 subgroups: grade 1 and grade 2. Neuroendocrine carcinomas are subclassified as large and small cell types. Small cell neuroendocrine carcinoma in the salivary gland may be difficult to diagnose as it is extremely rare. Small cell neuroendocrine carcinoma is diagnosed based on histological and IHC findings and may be misdiagnosed in small tissue samples. Therefore, core needle biopsy is preferred instead of fine-needle aspiration. Histologically, SNEC comprises tumor cells that are larger than mature small lymphocytes and have round or oval shape, consisting of solid sheets and nest of tumor cells. Additionally, IHC staining is essential for a definitive diagnosis. In NETs, the tumor cells express at least one of the neuroendocrine markers including SYN, chromogranin, CD56, CD57 (Leu-7), and neuron-specific enolase. Further, specific markers such as insulinoma-associated protein 1 and islet-1 are more sensitive and specific for NETs. 8 Immunohistochemical staining also helps to rule out the differential diagnosis of a primary SNEC in salivary gland from metastatic carcinomas. Thyroid transcription factor-1 is a sensitive marker for pulmonary or thyroid neoplasms. If tumor cells show negative for TTF-1, metastatic carcinoma from the lung or thyroid may be excluded. However, TTF-1 is also positive in pulmonary and extrapulmonary small cell carcinomas. Therefore, TTF-1 expression should not be used to distinguish primary from metastatic small cell carcinomas in extrapulmonary sites. 9 In this case, the possibility of Merkel cell carcinoma should also be considered. Merkel cell carcinoma is a rare NEC of the skin. CK20 negativity practically help to rule out Merkel cell carcinoma. 6 In our case, IHC staining was positive for CD56, SYN, and chromogranin and negative for TTF-1 and CK20 (Figure 2). Furthermore, physical examination and imaging studies including CT and PET/CT have been performed to exclude metastatic SNEC.
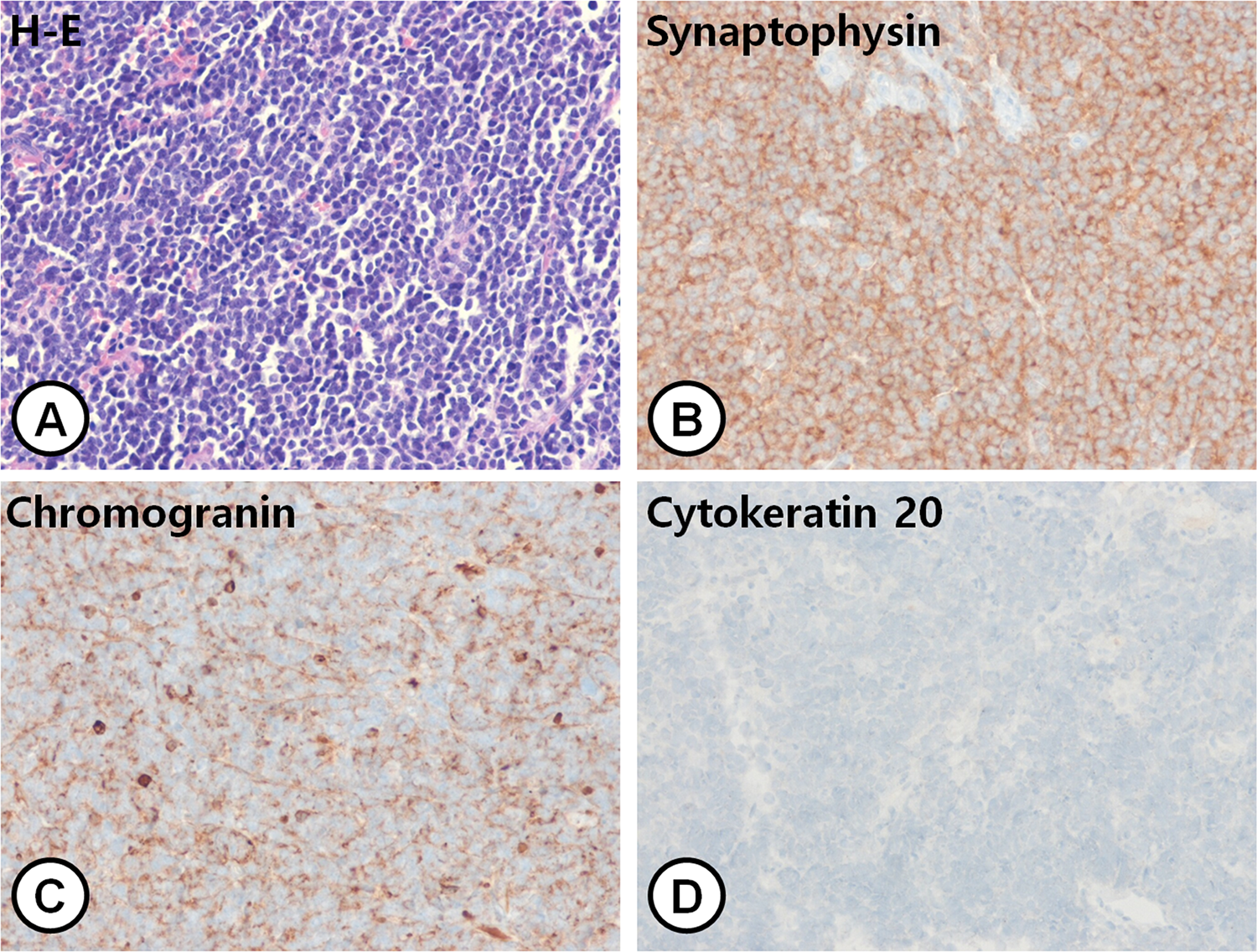
Hematoxylin-eosin (H-E) stain and immunohistochemical stain (original magnification ×200). (A) H-E stain shows monomorphous round tumor cells with bare nuclei and sparse cytoplasm. The shape of nuclei was round or oval. (B-D) Immunohistochemical staining shows strong positive for synaptophysin and chromogranin and negative for cytokeratin 20.
Old age, large tumor size, and lymph node metastases were related to poor prognosis. Servato et al reported that age was a significant prognostic factor, and the 5-year survival rate (5YSR) of younger patients (<65 years) was almost 6 times (63.5% vs 11.5%) higher than that of older patients (>65 years). Moreover, tumors <3 cm have a better prognosis (5YSR: 43.3% vs 11.25%) than large tumors. 3 In the largest cohort study that analyzed 198 patients with SNEC in salivary glands by Bai et al, the survival time was longer in lymph node negative patients than positive patients, but there was no difference in prognosis between N1 and N2 patients. 2 Surgery with or without adjuvant radiotherapy is the mainstay of treatment for tumors in the head and neck region. However, there is currently no definite treatment regimen for primary SNEC in the salivary gland as it is rare.1,2 In our case, since the patient was old with a small tumor (<3 cm) and no lymph node metastases, she had a good prognosis after surgery.
There are several types of salivary gland tumors, and rarely, tumors such as SNEC can occur in older patients. These tumors are difficult to diagnose histologically by fine needle biopsy. Therefore, it may be necessary to perform core needle biopsy or punch biopsy to obtain sufficient tumor tissues. In this case, SNEC in the sublingual gland was quickly confirmed as core needle biopsy was performed at the first visit. Therefore, SNEC was successfully controlled despite her age.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2018 Yeungnam University Research Grant.
