Abstract
Objectives:
Pneumatic compression garment therapy (PCGT) has been established as treatment for postradiotherapy lymphedema, and its use in head and neck patients is becoming more common. Although effects on interstitial edema of the cervical soft tissues have been studied, effects on internal laryngopharyngeal edema, as well as associated symptoms of dysphagia and dysphonia, have yet to be published.
Methods:
We surveyed 7 patients treated with radiation for head and neck cancer (HNC) who had also been prescribed PCGT for cervical lymphedema. Patients were asked about subjective experience with the device, and also administered the Eating Assessment Tool-10 (EAT-10) and Voice Handicap Index-10 (VHI-10) surveys regarding their symptoms after using PCGT. Laryngoscopy videos from these same periods were also reviewed and scored using a validated tool for assessing laryngopharyngeal edema.
Results:
85% of patients reported at least some improvement in dysphagia and dysphonia following PCGT. Average EAT-10 score after PCGT was 11.4 and average VHI-10 score after PCGT was 8.7. These compare more favorably to historical scores for the same questionnaires in similar patient populations. Laryngeal edema scores on endoscopic examination were not significantly different after at least 3 months of therapy (pre: 20.15, post: 20.21, P = .975); however, the utility of this result is limited by a low inter-rater reliability (Krippendorff α = .513).
Conclusions:
While we are unable to show any difference in objective assessment of laryngopharyngeal edema on endoscopic examination in this small pilot study, patients report substantial subjective improvement in postradiotherapy dysphagia and dysphonia following cervical PCGT that warrants more formal investigation.
Keywords
Introduction
Radiation therapy is a well-recognized treatment modality in the management of head and neck cancer (HNC). While this modality is considered to be quite effective, it is often accompanied by numerous complications and toxicities that range from early manifestations in the immediate posttreatmentperiod to late effects that can develop and persist years after treatment has been completed.1,2 Of these late toxicities, lymphedema is a relatively common, yet often underrecognized complication. Lymphedema represents a spectrum, where in its earliest stages, patients can experience a heaviness or fullness in the affected tissues without any appreciable functional deficit. As chronic lymphostasis persists, tissue inflammation occurs, inevitably leading to increased fibroblast proliferation and associated impairments in function of the affected tissue.2,3 For patients with HNC treated with radiation, this equates to difficulties with neck mobility, speaking, swallowing, and breathing; not to mention the associated physical deformities and cosmetic changes that can accompany chronic lymphedema.3-6
Complete decongestive therapy (CDT) is generally regarded as the gold standard treatment for lymphedema today.3,7 However, it is time consuming, initially requiring intensive work with a massage therapist trained in manual lymphatic drainage (MLD), followed by long-term daily maintenance work by patients themselves.3,7 With the goal of simplifying the patient’s self-care, pneumatic compression garment therapy (PCGT) was developed as a way of combining the MLD component with the compressive bandaging used in CDT. In 2016, the Flexitouch advanced pneumatic compression system (Tactile Medical) was the first such device to be approved by the Food and Drug Administration for use in patients with head and neck lymphedema. The system utilizes a combined garment for the head, neck, and torso to administer sequential, pneumatic compression to the affected area (Figure 1). Patients are advised by the company to use the device for half an hour on a daily basis to achieve maximal therapeutic effect. There are a number of studies demonstrating the benefits of advanced PCGT, particularly with regard to the overall improvement in economic burden to the health care system when compared to CDT.7-9 However, only one paper has looked at feasibility and outcomes of the device in the head and neck population. In addition to subjective assessments of overall patient satisfaction with the device, this paper by Mayrovitz et al exclusively used external facial and neck measurements as its principle objective outcome. While all the outcomes assessed in the article were generally significantly favorable for the use of PCGT, there was no assessment of the device’s impact on internal manifestations of head and neck lymphedema. 7

Photo of Flexitouch system, provided by, and with permission from Tactile Medical. Left and middle images show the frontal and profile views of the device in use; image on the right shows pneumatic pump for the device.
Our institution has independently made efforts to utilize PCGT for many of our patients with HNC in recent years, primarily because of difficulties with access to adequate massage and physical therapy services for patients who are often traveling from distant rural communities for their care. While we are generally prescribing the device to treat the external manifestations of cervicofacial lymphedema following HNC treatment, we have noted several anecdotal responses from individual patients who have reported improvements in their dysphagia and dysphonia following use of PCGT. Given these few but positive reports, as well as the absence of any data in the literature regarding the effects of PCGT on the laryngopharyngeal manifestations of lymphedema, we sought to develop a pilot study to investigate the topic more directly. For HNC patients with posttreatment lymphedema, we hypothesized that administration of patient reported outcomes measures (PROMs) pertaining to symptoms of dysphagia and dysphonia would demonstrate overall symptomatic improvement in these domains following treatment with PCGT. We simultaneously hypothesized that objective assessment of laryngopharyngeal edema on routine endoscopic examinations would show an overall improvement during that same period of treatment.
Methods
This pilot study was approved by the Institutional Review Board of LSU Health Science Center. Patients were selected for inclusion based on a history of having undergone radiation therapy as part of their original treatment for HNC, as well as having been prescribed PCGT for postradiation cutaneous cervical lymphedema. The specific form of PCGT utilized in all patients was the Flexitouch Head and Neck System (Tactile Medical). All patients had undergone at least 6 months of intermittent treatment with PCGT prior to participation in this study. A formalized regimen of CDT was not utilized in any of these patients. Patients having undergone surgical treatment for their cancer, including laryngectomy, were not included. Patients meeting the above criteria were surveyed about the frequency with which they used their PCGT, as well as their overall sense of subjective symptomatic improvement in the domains of dysphonia and dysphagia. Additionally, participants were given the Voice Handicap Index-10 (VHI-10) and Eating Assessment Tool-10 (EAT-10), with responses corresponding to their current symptoms after a minimum of 6 months of PCGT. Mean scores were calculated for descriptive purposes only, as pretreatment scores were not able to be collected. Nonstatistical, qualitative comparisons to historically published data for similar patient populations were then performed for this descriptive information.
As a second part of this study, laryngoscopy videos from before and after initiation of PCGT were reviewed and scored using the validated Patterson scale for assessing laryngopharyngeal edema. 10 All pre- and posttreatment videos were randomized before being shown to a mixed audience that included 2 speech pathologists and 4 otolaryngologists working at an academic medical center. All reviewers completed Patterson assessments for each video being reviewed. Inter-rater reliability was subsequently assessed using Krippendorff’s α, and a paired T test was used to compare mean Patterson scores before and after PCGT.
Results
Fifteen patients at our institution were treated using PCGT, though 6 of these patients had previously undergone total laryngectomy, and 2 patients reported using their devices for less than 3 months in total. The remaining 7 patients met inclusion criteria for this study. All 7 were given the aforementioned questionnaires; however, only 6 of these patients were included in the endoscopic evaluation, as 1 patient did not have her pretreatment laryngoscopy examination recorded. Six patients were female. The average age at completion of cancer treatment was 49 (median 52, range 32-57), while the average age at initiation of PCGT was 50 (median 53, range 33-58). Mean duration to initiation of PCGT from completion of radiation therapy was 1.5 years. See Table 1 for patient tumor and treatment data. Figure 2 includes patient self-reported data on frequency of use of PCGT.
Tumor and Treatment Data.

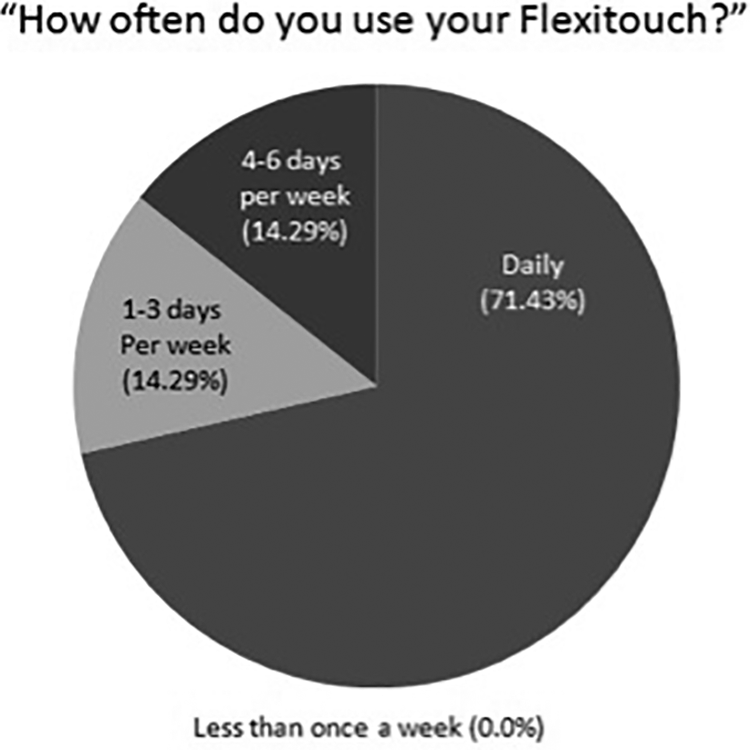
Collective responses by patients regarding frequency of PCGT usage. PCGT indicates pneumatic compression garment therapy.
Figures 3 and 4 show patient self-reported changes in dysphagia and dysphonia, respectively, following PCGT. Regarding EAT-10 results, the average posttreatment score was 11.4 (standard deviation [SD] 4.8). Regarding VHI-10 results, the average posttreatment score was 8.7 (SD 9.5). Nonstatistical comparisons between these scores and those of historically published data are included in the discussion below.
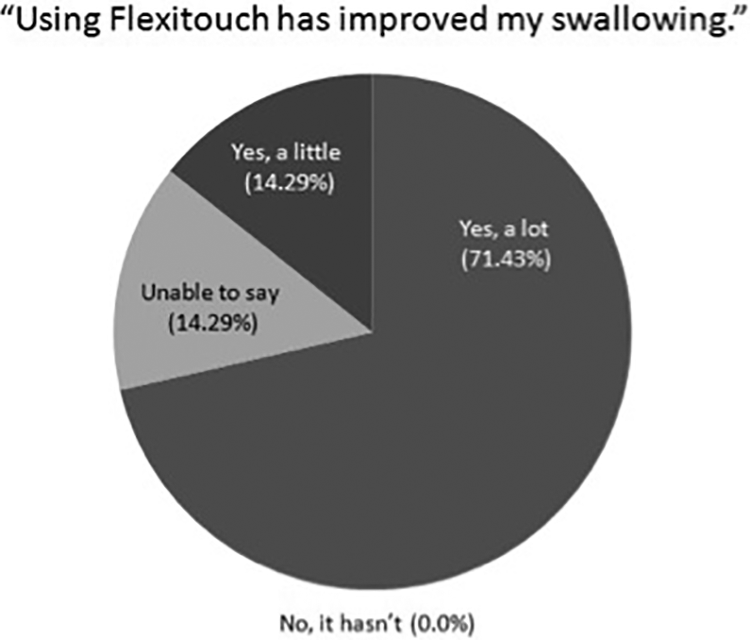
Collective responses by patients regarding subjective improvement in dysphagia.
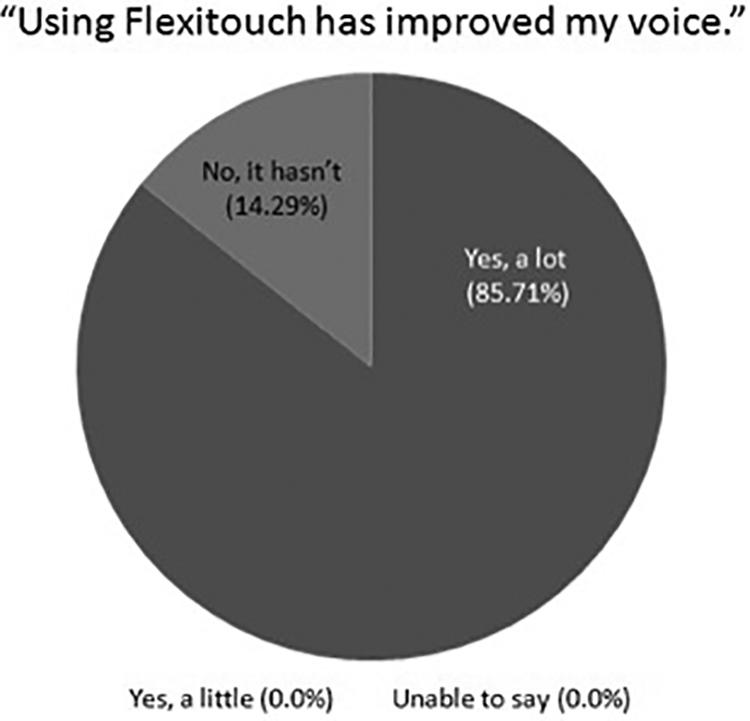
Collective responses by patients regarding subjective improvement in dysphonia.
Assessments of laryngopharyngeal edema on endoscopy using the Patterson scale revealed an average pretreatment score of 20.15 and an average posttreatment score of 20.21. These results were not statistically significant (P = .975), though inter-rater reliability for these assessments was also noted to be quite poor, with a Krippendorff’s α of .513 (95% CI: 0.442-0.581).
Discussion
Our understanding of the biomechanical basis and treatment principles for lymphedema is relatively new in the history of medicine. Building on the work of osteopaths Andrew Still and C. Earl Miller in the early 20th century, Emil Vodder is generally credited for developing and publishing his MLD technique in 1967 as the first formal therapy for improving lymphatic flow.3,11,12 Nearly 30 years later, Michael and Ethel Foldi combined Vodder’s techniques with the use of compressive bandaging, skin care, and therapeutic exercise to develop CDT, which as previously mentioned has traditionally been regarded as the gold standard treatment for lymphedema.3,7
Access to formally trained CDT therapists near our institution has been rather limited, and PCGT has become a notable asset for many of our HNC patients with symptomatic, cervical lymphedema. Working under the theoretical assumption that the benefits of PCGT may also extend to laryngopharyngeal edema, we have found empirical success in treating patients with dysphagia and dysphonia using this modality. This admittedly limited pilot study was conducted to help broaden the awareness of PCGT as an alternative to CDT, while simultaneously serving as a tool to help guide further, more rigorous, prospective investigation.
To our knowledge, this is the first and only study to evaluate the effects of PCGT on postradiotherapy dysphonia and dysphagia attributable to chronic laryngopharyngeal edema. Pneumatic compression garment therapy is well established for the treatment of lymphedema in general, though this has most often been applicable to the trunk or extremities.13-15 There are few, but convincing studies that have served as the basis for the Flexitouch system’s use in head and neck lymphedema patients, 7 and it is currently the only available PCGT device for use in this specific subset of patients. This device has been marketed and approved for the treatment of head and neck lymphedema based on data that look exclusively at external measures of lymphedema 7 ; however, no efforts have yet been made to assess the internal laryngopharyngeal lymphedema that may also be benefitting from overall improvements in lymphatic circulation. It is with this idea in mind, and the known association between laryngopharyngeal edema and symptomatic dysphagia and dysphonia,4,5,16 that we chose to pursue this investigation.
Regarding our patient demographics, most characteristics seem to be fairly heterogeneous. However, it is worth mentioning that our final study population seems to be heavily female (85%) when compared to the known predominance of HNC in men. While our initial study population prior to removing those patients who did not meet inclusion criteria was more gender balanced (54% female), this nevertheless, remains an outlying variable.
As seen in Figure 2, our PROM data demonstrated notable compliance with the prescribed daily use of PCGT in the 7 patients studied. It is reasonable to assume that such compliance is at least somewhat tied to a perceived symptomatic benefit, and this is supported by the most notable of our results, where with the exception of a single patient, we observed nearly universal improvement in both subjective dysphagia and dysphonia following the use of PCGT.
Our VHI-10 and EAT-10 data are solely meant to be descriptive in nature, as no pretreatment metrics were able to be collected for statistical comparison. Nevertheless, there is some positive encouragement here. For the EAT-10, a score of 3 or greater is generally associated with swallowing dysfunction, and a score of 11 or greater on the VHI-10 suggests some degree of voice handicap. 17 While our average EAT-10 score is certainly above this cutoff, our average VHI is notably below. Soldatova and Mirza looked at long-term voice and swallowing outcomes for oral and oropharyngeal cancer following primary or adjuvant chemoradiation. They found average VHI-10 and EAT-10 scores of 13.5 and 14.5, respectively. 17 Given our lower and more favorable averages of 8.7 and 11.4, respectively, the numbers from our pilot study seem to encourage future prospective evaluation for the purpose of statistical comparison.
The endoscopic arm of our investigation was much less rewarding and did not yield any significant differences between pre- and post-PCGT examination findings. Moreover, we demonstrated a low inter-rater reliability as evidenced by our Krippendorff’s α value of .513 (reliability is traditionally favored at values greater than 0.8). The Patterson scale has been previously utilized with good inter-rater reliability in other publications looking at laryngopharyngeal edema in patients with HNC.2,10,16,18 Rater experience for this study was deemed to be adequate by virtue of their training and practice location in a high volume cancer center. We can only suspect that our poor inter-rater reliability is a possible reflection of our small sample size during this early pilot investigation.
Aside from the sample size limitations that have already been discussed, it is worth mentioning that our mean time to initiation of therapy may have been longer than ideal for maximization of benefit. Smith et al in their 2015 article found that patients who were referred for lymphedema therapy 6 weeks to 5 months after their cancer treatment were significantly more likely to respond to therapy. 5 This relative delay very well might be playing a role in our difficulties observing an objective difference on endoscopic evaluation. Going forward with prospective investigations, this information could be used to develop a treatment referral protocol that maximizes our patients’ likelihood of successful response.
Finally, neurosensory issues associated with radiation and posttreatment dysphagia have been reported extensively in the literature with mixed results. 19 As such, the possibility remains that our patients’ ability to accurately report their own degree of symptomatic dysphagia on basic subjective PROMs is limited. As this investigation is taken toward future prospective endeavors, graded evaluations of pre- and posttherapy-modified barium swallow studies would be one way to notably improve our objective assessment of the impact of PCGT on these patients’ dysphagia.
Conclusion
Overall, while we were unable to show any difference in objective assessment of laryngopharyngeal edema on endoscopic examination, the patients in this pilot study reported substantial subjective improvement in postradiotherapy dysphagia and dysphonia following the use of PCGT. Further investigation is warranted to help strengthen the validity of these results, as well as to help elucidate the exact mechanism underlying their occurrence. We hope that the preliminary findings of this pilot study will help guide more formal, prospective investigation, while simultaneously raising awareness of PCGT as a potential treatment for those patients with HNC who suffer from the effects of chronic lymphedema.
Footnotes
Authors’ Note
This article was presented at the 2019 Triological Society Combined Sections Meeting in Coronado, California.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
