Abstract

Salivary duct stenosis most frequently involves parotid ducts and presents during the fourth through sixth decades of life with recurrent, painful swelling of involved salivary glands, often related to food intake.1-3 Etiologies include recurrent sialadenitis, sialolithiasis, trauma, radiation exposure, and inflammatory conditions such as Sjögren syndrome.1,2,4 Diagnostic imaging options for suspected ductal stricture include ultrasound, conventional sialography with contrast infusion, magnetic resonance sialography, and sialendoscopy.1,2 Sialography, which provides detailed information on ductal anatomy (visualization of the entire ductal system) and also enables assessment of ductal function (clearance rate of infused contrast after sialagogue administration), is the most sensitive modality for characterization of ductal stenosis.5,6 Sialendoscopy, although most frequently used as a therapeutic tool, also provides diagnostic information on the intraluminal appearance(s) of the ductal system.2,4 We correlated findings of conventional sialography and sialendoscopy with histopathology in a case of fibrous proximal ductal stricture that was refractory to dilation and ultimately required parotidectomy. Our observations provide insights into pathogenesis and prognosis of proximal ductal stricture.
A 42-year-old female presented to the otolaryngology clinic with 5 years of intermittent, painful right parotid swelling, aggravated by meals and relieved with antibiotics. Previously attempted distal dilation, performed close to the puncta by transoral endoscopic access, had been unsuccessful and symptoms persisted despite tympanic neurectomy. There was no history of xerophthalmia, xerostomia, or autoimmune disease. Computed tomography scan showed unremarkable parotid parenchyma and ducts. Right parotid sialogram, performed at our institution, revealed a single, localized stenosis in the proximal parotid duct, 6 cm from the duct orifice, with prestenotic duct dilatation and evidence for parenchymal damage (Figure 1A). Sialendoscopy confirmed the presence of a significant fibrotic stricture (Figure 1B) which was endoscopically dilated (with limited success), followed by insufflation with intraductal corticosteroid. Although frequency of parotid swelling decreased, pain persisted, and parotidectomy was therefore performed. At 4-month follow-up, the patient reported complete symptom resolution.
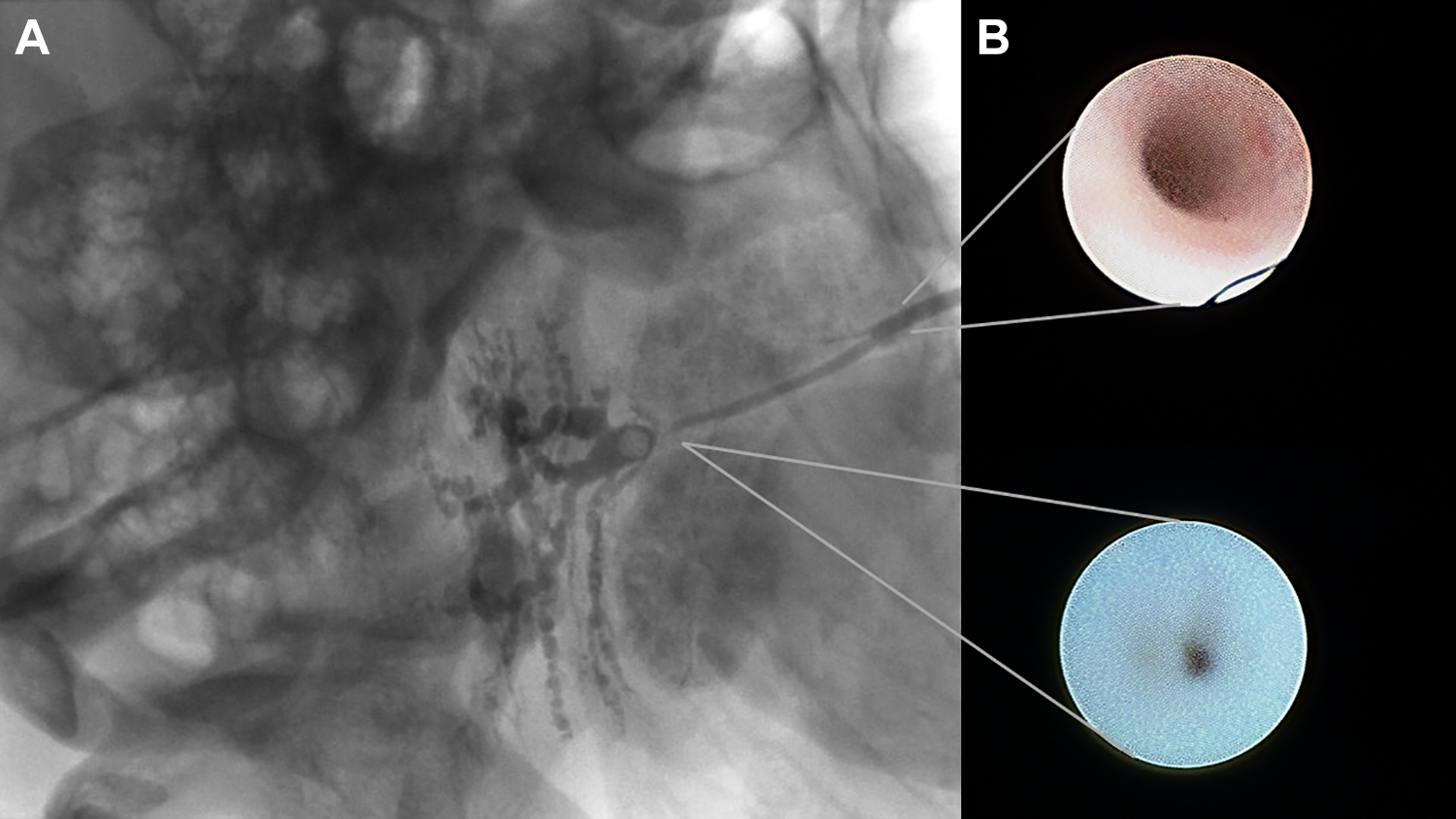
Respective appearances of normal parotid duct and ductal stricture on (A) conventional sialography and (B) sialendoscopy.
Histopathologic examination of the excised gland revealed dense interlobular fibrosis with acute and chronic inflammation proximal to the narrowing (Figure 2). Parotid tissue distal to the stenosis was normal. Within the stenotic region itself, the degree of stromal fibrosis and chronic inflammation was similar to that detected more proximally, but, in addition, squamous metaplasia was noted extending into the ductal lumen (Figure 3).
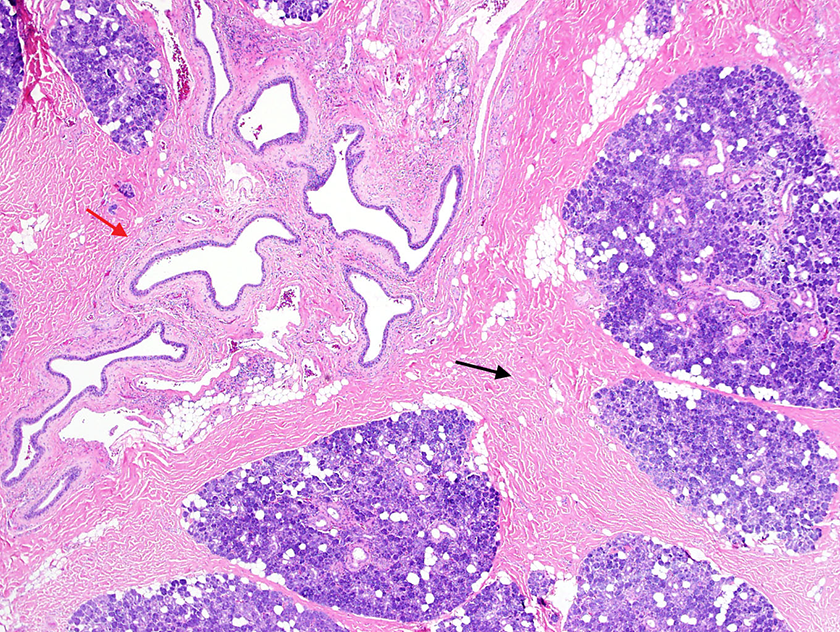
Right superficial lobe of the parotid gland with dense, hyalinizing interlobular fibrosis (black arrow) and chronic inflammation surrounding the parotid ducts (red arrow; ×40).
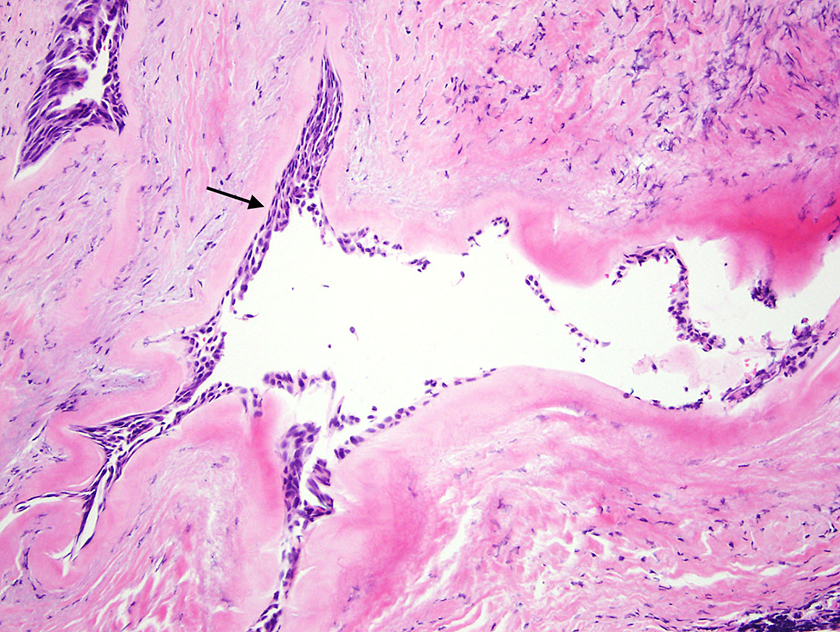
Parotid salivary duct showing squamous metaplasia (arrow), dense stromal fibrosis, and chronic inflammation. (×100).
Correlation of the histopathology of an excised ductal stricture with preoperative sialographic and sialendoscopic findings has not, to our knowledge, been described previously. In a study of 26 patients with parotid duct stricture, Qi et al reported the presence of duct epithelial cells, neutrophils, lymphocytes, and acidophils in fluid obtained by ductal irrigation during sialendoscopy. 7 Koch and Iro used endoscopy to develop a classification system of ductal strictures ranging from “inflammatory” (type 1) to “purely fibrotic” (type 3). 8 Neither study characterized the relation between anatomic stricture and underlying histopathology of duct and adjacent salivary gland tissue.
It has been reported that primary success rates of local therapies for ductal stenosis are lower, and recurrence rates higher, when stenosis is predominantly fibrotic rather than inflammatory.2,3,8,9 Moreover, Plonowska et al found that proximal salivary duct stenoses were less responsive to sialendoscopic dilation and sialendoscopy-assisted surgical procedures than distal ones. 10 These findings are concordant with the failed sialendoscopic dilatation and subsequent histopathologic confirmation of dense proximal ductal fibrosis noted in our patient. Choi et al have attributed this failed response to therapy to irreversible loss of mucosal function after fibrosis has occurred. 9
Parotidectomy has historically been the procedure of choice for recurrent sialadenitis refractory to conservative treatment—including sialadenitis associated with ductal stricture.11-13 Less invasive surgical treatment options are available, including duct ligation with the goal of inducing atrophy of the gland. Although long-term outcomes of ligation are not extensively reported, associated complications of duct rupture and sialocele have been documented.11,13,14 Percutaneous botulinum toxin injection into the salivary gland parenchyma has been studied in small cohorts and may be more effective than endoscopy-guided interventions.3,4,12,15,16 More recently, intraductal infusion of botulinum toxin has yielded promising results. 17
The correlation between sialographic/sialendoscopic findings and histopathology in our patient may have therapeutic implications. Salivary gland and ductal tissues distal to the stenosis were spared from the inflammatory process that was most intense at the site of the stenosis and that appeared to propagate proximally. Partial parotidectomy that conserves the segment of normal glandular tissue distal to the stenosis might preserve parotid function. It is conceivable that early targeted intervention to relieve the obstruction might also limit progression of inflammatory sequelae including proximal parotid damage and development of the dense fibrosis that renders endoscopic dilation untenable.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Henry T. Hoffman is a research consultant for COOK Medical and IotaMotion and contributing author for UpToDate.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
