Abstract
Background:
We evaluated the efficacy of medical treatment on thiol–disulfide balance despite ongoing allergic stimulation.
Methods:
The research design was a prospective observational study that included 35 persistent allergic rhinitis (AR) patients. All patients who were diagnosed with persistent AR were included. A skin prick test was applied to all patients, and the Sino-nasal Outcome Test-22 was used to evaluate sinonasal symptoms. Thiol/disulfide homeostasis balance parameters were measured using a novel automatic and spectrophotometric method and compared statistically. Serum total thiol (TT), native thiol (SH), disulphide (SS), disulphide/native thiol (SS/SH), disulphide/total thiol (SS/TT), and native thiol/total thiol (SH/TT) ratios were measured after the second month of the treatment.
Results:
The 35 patients included 20 (58%) females and 15 (42%) males. The mean age of the patients was 33.17 ± 9.9 years. Disulphide, SS/SH, and SS/TT ratios decreased significantly after the treatment (P < .05), while SH and SH/TT increased significantly (P < .05). The mean SH measurement increased significantly in the second month (P = .001), but TT mean measurements showed no difference after the treatment (P = .058). The mean SS measurements, on the other hand, decreased significantly in the second month (P = .003).
Conclusion:
Thiol/disulfide homeostasis may be used as a marker to evaluate the efficacy of persistent AR treatments. After the treatment, the increase in SH levels suggested the decrease in oxidative stress, even though allergen exposure continued.
Introduction
Allergic rhinitis (AR) is a common disease that affects 10% to 40% of the human population worldwide. Although AR is not a life-threatening disease, it can decrease the quality of life by causing sleep disorders, daily activity loss, and labor loss.1,2 Once an allergen enters the body, a series of response mechanisms occurs and mediators such as interleukins, histamine, and prostaglandin cause inflammation in the airways that can result in itching, runny nose, and nasal congestion.3,4
Previous studies have shown a relation between oxidative stress (OS) and inflammatory airway disease.5,6 Cytokines in the airway during the inflammatory process cause the formation of the reactive oxygen species (ROS) in AR. 7 Oxidative balance (OB) is the balance between the production and removal of free radicals in the body. By providing an OB, minimizing the damage caused by these free radicals is possible.
Thiols are important antioxidants that can remove free ROS by enzymatic and nonenzymatic ways. 8 The natural thiols in the body are glutathione, cysteine, homocysteine, N-acetylcysteine, and glutathione. These oxidants are critical for cell hemostasis as a part of normal metabolism. 9 Oxidative stress causes an increase in cytokines, which are the beginning of the inflammatory process.10,11 Allergic rhinitis and asthma cause a locally increased oxidative load because of mucosal inflammation, which disturbs thiol/disulfide hemostasis (TDH). 12
Thiol/disulfide homeostasis was first measured in 1979 using methods such as high-performance liquid chromatography and bioluminescence.13,14 Today, the levels of thiol and disulfide can be measured individually or simultaneously by the fully automated methods developed by Erel and Neselioglu. 15 Because a follow-up of OS may indicate the success of medical treatment, we evaluated the efficacy of medical treatment on TDH despite ongoing allergic stimulation.
Materials and Methods
Institutional review board approval was given by the Ethical Committee of the local hospital. A total of 35 patients (20 females and 15 males) with the diagnosis of persistent AR who applied to the outpatient clinic of the Otorhinolaryngology Department between April 2018 and August 2018 were enrolled in the study. Informed consent was received from all participants.
Persistent AR was diagnosed according to the Allergic Rhinitis and Its Impact on Asthma (ARIA) guidelines. Patients had more than one of the signs and symptoms of AR (eg, sneezing, watery rhinorrhea, nose blockage, and/or nasal pruritus), and their symptoms occurred more than 4 days per week and for more than 4 consecutive weeks [16]. A skin prick test was applied to all patients. Patients allergic to dust mites, molds, cockroaches, and animal dander were included in the study. The exclusion criteria were medication for persistent AR one month before the study, smoking, cardiovascular, rheumatologic, hypertensive and diabetic diseases, chronic sinusitis, septum deviation, nasal polyposis, asthma, and severe obstructive sleep apnea disorder.
All patients had complete ear, nose, and throat examinations. Venous blood samples were taken from patients before and after the treatment, and all the samples were placed into EDTA-containing tubes. Subsequently, the assay samples were centrifuged at 1500 × g for 10 minutes, and the sera were stored at −80°C. The samples were evaluated by spectrometric method using a Cobas c 501 analyzer (Roche Diagnostics). 15 The thiol/disulfide measurement was done by reducing the dynamic disulfide bonds (-Si, -S) to the free thiol groups (-SH) using sodium borohydride NaBH4 (10 mL). The unused NaBH4 was washed away with formaldehyde solution and reduced to 5,5′-dithiobis-(2-nitrobenzoic) acids (DTNB). The total thiol (TT) content in the samples was determined using a modified Ellman reagent after the reaction with DTNB. The TT (the sum of the native and reduced thiol) was measured, and the disulphide (SS) amount was calculated by subtracting native thiol (SH) from the TT level. Disulphide/total thiol (SS/TT), disulphide/native thiol (SS/SH), and native thiol/total thiol (SH/TT) ratios were calculated as a percentage.
Data were collected by means of a self-administered questionnaire. Sino-nasal Outcome Test (SNOT-22), which was validated by Hopkins et al 16 was used to evaluate sinonasal symptoms. The SNOT-22 is composed of 22 questions that are scored from 0 to 5; a lower score implies a better result.
All patients took a 10-mg rupatadine fumarate tablet once a day and sprayed 50 µg/puff beclomethasone dipropionate twice daily in both nostrils for 2 months, according to ARIA guidelines. 17 Patients were examined during the first week of the treatment to check for any side effects. At the end of the second month, the blood tests and SNOT-22 questionnaire were repeated.
Statistics
Statistical analysis was performed using SPSS version 21.0. The normal distribution was evaluated by the visual (histogram) and analytical methods (Kolmogorov-Smirnov/Shapiro-Wilk tests). A Student t test was used for the normally distributed parametric data, and the Wilcoxon test was used for nonparametric data. P values below .05 were set as statistically significant for the results. Although investigating the associations between non-normally distributed and/or ordinal variables, the correlation coefficients and their significance were calculated using the Spearman test. A 5% type-1 error level was used to infer statistical significance.
Results
Thirty-five patients were enrolled in this study. The mean age of the patients was 33.17 ± 9.9 years. Twenty (58%) were female while 15 (42%) were male. There was no significant difference in terms of age and sex characteristics (P < .05; Table 1).
Demographic Characteristics of Patients.
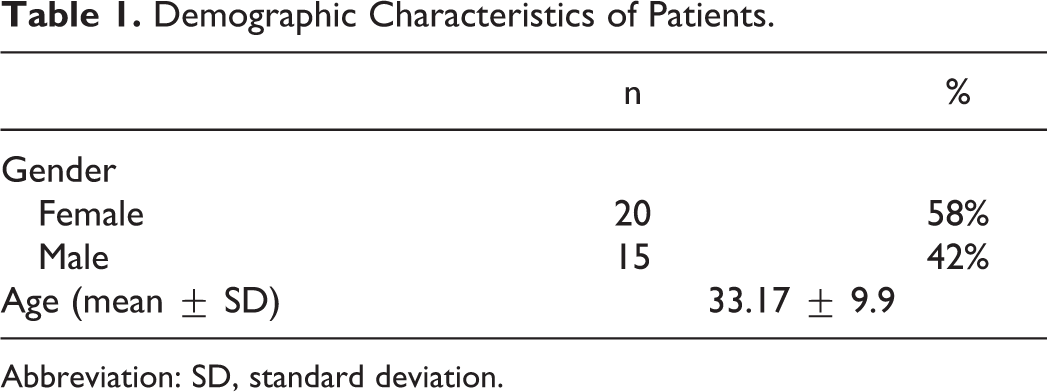
Abbreviation: SD, standard deviation.
Serum TT, SH, SS, SS/SH, SS/TT, and SH/TT ratios were measured after the second month of treatment. The mean SH measurement increased significantly in the second month (P = .001), but the TT mean measurements showed no difference after the treatment (P = .058). The mean SS measurements decreased significantly in the second month (P = .003).
Disulphide/native thiol and SS/TT ratios decreased significantly after the treatment, respectively (P = .007, P = .007). However, the SH/TT ratio showed no difference after the treatment (Table 2). The mean SNOT-22 decreased significantly after the treatment (P < .001; Table 3). There is a moderately strong significant correlation between SNOT-22 and disulfide (P < .05; Table 4; Figure 1).
Serum Thiol Values Before and After Treatment With SAR.a
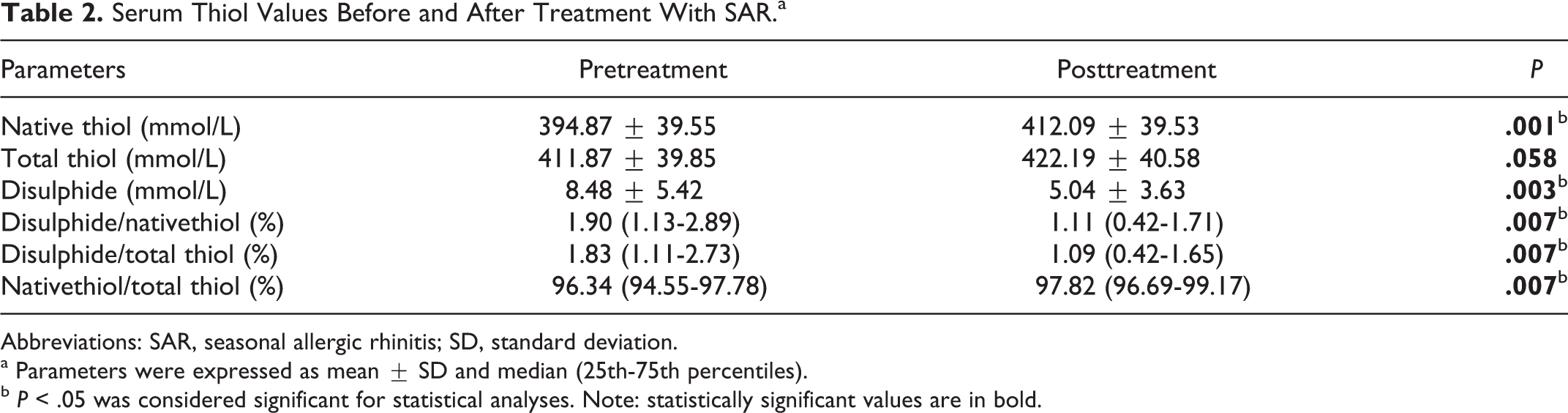
Abbreviations: SAR, seasonal allergic rhinitis; SD, standard deviation.
a Parameters were expressed as mean ± SD and median (25th-75th percentiles).
b P < .05 was considered significant for statistical analyses. Note: statistically significant values are in bold.
SNOT-22 Values Before and After Treatment.a
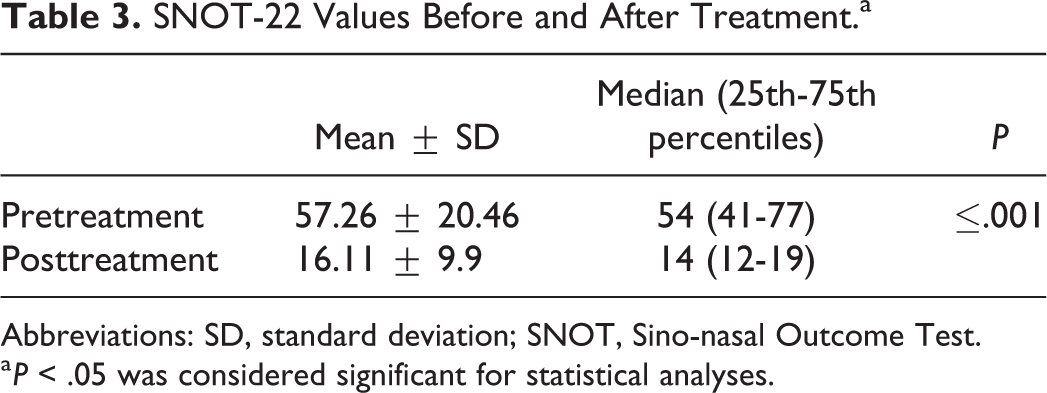
Abbreviations: SD, standard deviation; SNOT, Sino-nasal Outcome Test.
a P < .05 was considered significant for statistical analyses.
SNOT-22 and Disulphide Correlation.

Abbreviations: SD, standard deviation; SNOT, Sino-nasal Outcome Test. Note: significant values are in bold.
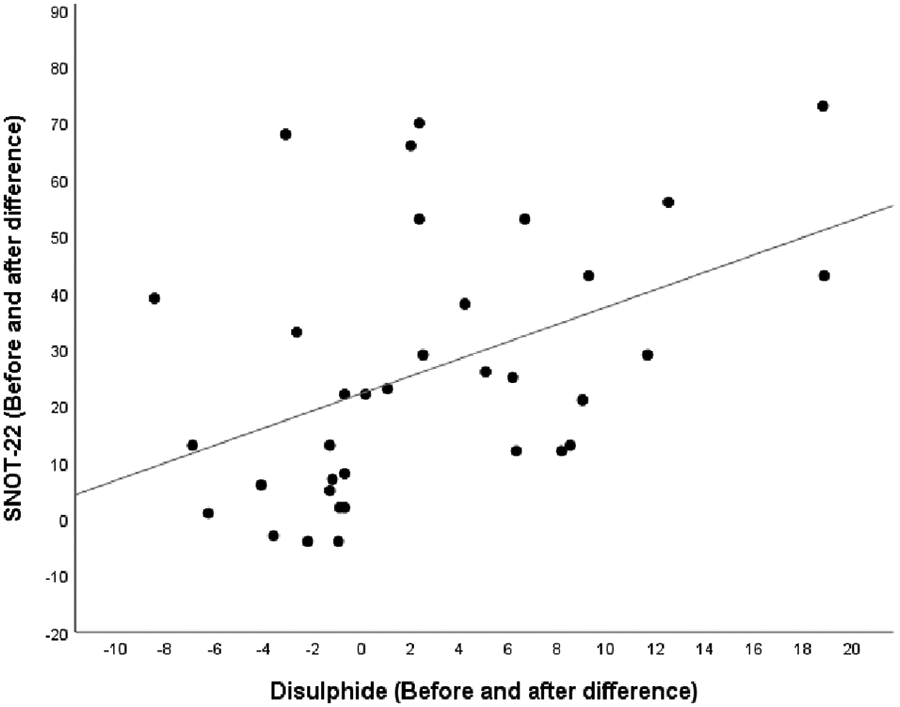
Sino-nasal Outcome Test (SNOT)-22 and disulphide difference.
Discussion
Thiol groups are known as mercaptans, an important component of the antioxidant system. Thiols are a class of organic compounds containing a sulfur atom and a sulfhydryl group (-SH) formed by a hydrogen atom bonded to the carbon atom. Thiol groups remove ROS, and TT levels can be used to evaluate the oxidation status. 18 The thiols can undergo an oxidation reaction with oxidants and form disulfide bonds. 15 In the oxidation process, it was observed that the level of SH decreased linearly. Cytokines involved in the inflammatory process of airways, such as AR, release ROS with various stimuli. 7 When the medium has an excess of oxygen radicals, the resulting SS bonds buffer the toxicity of the oxygen radicals. This creates dynamic TDH, an indicator of OS that is necessary for the regulation of detoxification, apoptosis, signaling pathways, and enzymatic reactions in the body.19,20 This balance deteriorates in favor of SS in many chronic diseases.21,22 The resulting disulfide bonds can be reduced back to the thiol groups. 18
The respiratory mucosa has developed a defense mechanism against oxidants. 12 Allergens cause an imbalance between ROS and antioxidant defense systems. This oxidative imbalance increases the formation of ROS, such as hydroxyl radicals, superoxides, and peroxides.23,24 Treatment for AR alleviates the inflammatory reaction. To evaluate the effectiveness of a treatment, subjective methods are used. Sino-nasal Outcome Test-22 is the most commonly used, and it correlates with the reduction of OS in the body (Table 4). An improvement in SNOT-22 can show the efficacy of treatment. In our study, we obtained a significant decrease in SNOT-22 values after the treatment (Table 3), which suggests a decreased OS in the body.
However, there is still a need for more objective methods to demonstrate the reduction of OS and the effectiveness of treatment. A decrease in ROS or an increase in antioxidants may reveal this relation. Gratziou et al 25 showed that the use of intranasal corticosteroids with seasonal allergic rhinitis (SAR) reduced OS markers but that nitric oxide concentration was not affected. Thiol/disulfide homeostasis may be used to demonstrate that relation. Ulusoy et al 26 compared the plasma thiol levels of patients with SAR during a period of AR exacerbation and during an asymptomatic period. They found that SS was significantly higher in patients with SAR during exacerbation periods and that SH was significantly higher in patients with SAR during asymptomatic periods. In our study, we evaluated these markers after the treatment. We found that SS, SS/SH, and SS/TT ratios decreased significantly after the treatment (P < .05). Furthermore, SH and SH/TT increased significantly (P < .05). This suggests that rupatadine fumarate and beclomethasone dipropionate decreased OS, resulting in decreased SNOT-22 scores. For this reason, SH and SS may be used to objectively evaluate the efficacy of the treatment.
In future, these markers may give information about the initiation and cessation of AR treatment or may allow to adjust treatment doses.
Conclusion
Thiol/disulfide homeostasis may be used as a marker to evaluate the efficacy of treatment. Total thiol levels did not change during the treatment; however, SS was higher in patients with persistent AR during exacerbation periods. After the treatment, the increase in SH levels suggested the decrease in OS even though the allergen exposure continued.
Footnotes
Declaration of Conflicting Interests
The author(s) declare they have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
