Abstract
Lymphoepithelial carcinoma (LEC) of the salivary gland is a rare malignancy which is identical to undifferentiated nasopharyngeal carcinoma. However, most patients are treated with surgery as primary treatment, which is impossible for some very locoregionally advanced patients. And there are few reports of patients treated by an induction chemotherapy (IC) and concurrent chemoradiotherapy (CCRT) approach. This report describes 3 cases of advanced stage LEC of the salivary gland. All patients presented with a palpable mass of variable duration and underwent induction CCRT. All cases were positive for Epstein-Barr virus–encoded small RNAs. After IC, all cases had reached partial response and all achieved complete response after CCRT. All patients remained local–regional recurrence-free after 6-month follow-up for case 1, 50-month for case 2, and 14-month for case 3 up to our last follow-up. No serious adverse events were found.
Keywords
Introduction
Lymphoepithelial carcinoma (LEC) is a rare malignant tumor, which occurs in multiple sites, including the skin, lung, oral cavity, and so on. 1 And the major salivary glands are the most common sites. It is a clinicopathological entity characterized by an undifferentiated nonkeratinizing squamous cell carcinoma with significant lymphocytic infiltration and is identical to undifferentiated nasopharyngeal carcinoma (NPC). Many studies have pointed out its strong association with Epstein-Barr virus (EBV) infection as well as sensitivity to irradiation or antitumor drugs 2 similar to NPC. For advanced NPC patients, induction-concurrent chemoradiotherapy or CCRT with/without adjuvant chemotherapy is the standard treatment and has resulted in satisfactory outcomes. However, surgery with postoperative radiotherapy (PORT) is the mainstay treatment paradigm for advanced LEC. 1 It is acknowledged that surgery has the disadvantage that it may cause facial depression deformity, paresthesia or dysesthesia, which can significantly affect patients’ quality of life.
Here, we report 3 cases of pathologically diagnosed LEC originating from the salivary gland, which were treated with induction chemotherapy (IC) and CCRT by a multidisciplinary team.
Case Report
Case 1
A 27-year-old man complained of a palpable and slowly growing mass in the right parotid area for 2 years, with ipsilateral facial paresis and xerostomia. Prior to admission, he had been treated using traditional Chinese medicine, but ultimately there was no response. Furthermore, he had no notable medical history. On clinical examination, a solid tumor mass was found (Figure 1A). Lymph nodes were found at ipsilateral levels I, II, III, IV, and VIII, with extranodal extension (ENE) and fusion in the intraglandular and upper nodes. Examination of the nasopharynx, oral cavity, oropharynx, and laryngopharynx found no lesions. An ultrasonography-guided fine needle aspiration (FNA) biopsy was performed, and the histopathological diagnosis was LEC. Based on the clinical, radiological, and pathological findings, a final diagnosis (T4N3M0) was made. In situ hybridization for EBV-encoded small RNAs (EBERs) was positive in the tumor cells, and quantification of EBV-DNA load was 7.70 × 103 copy/mL.
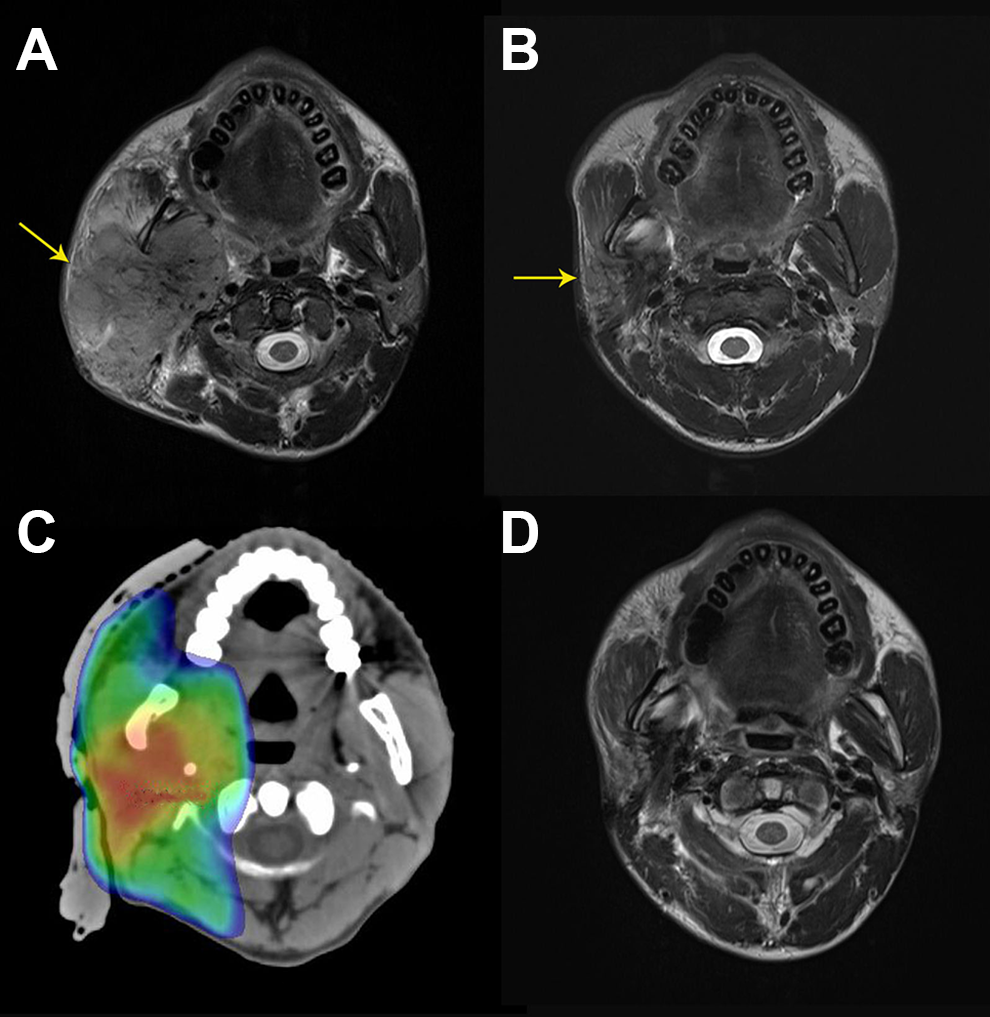
A, The salivary gland tumor (yellow arrow) invaded to the parapharyngeal space, masseter muscle, carotid sheath, and so on, (B) after induction chemotherapy (IC), partial response (PR) was achieved; (C) target volume delineation; (D) complete response (CR) was achieved after concurrent chemoradiotherapy (CCRT).
The patient was treated with IC and CCRT. The IC regimen was a combination of docetaxel, cisplatin, and fluorouracil, comprising intravenous infusion of docetaxel 60 mg/m2 on day 1, intravenous infusion of cisplatin 60 mg/m2 on day 1, and continuous intravenous infusion of 5-fluorouracil 500 mg/m2/d on days 1 through 5 for 120 hours, 3-weekly, for 4 cycles. 3 Concurrent chemotherapy consisted of nedaplatin 40 mg/m2 intravenous infusion on day 1, weekly, for 4 cycles. As for radiotherapy, the patient was treated with intensity-modulated radiotherapy techniques. The gross tumor volume (GTV) was defined as the residual primary tumor (including lymph node metastasis) after IC. The medium risk clinical tumor volume (CTV1) was defined as GTV plus a 0.5- to 1-cm margin and covering the primary tumor region before IC. The low risk CTV (CTV2) was defined as CTV1 plus a 5-mm margin together with the ipsilateral cervical selective lymph drainage areas. 3 The radiation dose schedule was as follows: the planning gross tumor volume (PGTV) was prescribed a dose of 70 Gy; PCTV1, 61 Gy; and PCTV2, 46 Gy3 (Figure 1C). After IC, a partial response (PR) was achieved (Figure 1B) and complete response (CR) was achieved after CCRT (Figure 1D). No serious adverse events were found. The plasma EBV-DNA load has remained 0 since 2 months after treatment. Six months after treatment, the patient showed no evidence of disease.
Case 2
A 33-year-old man was admitted with a painful, slowly growing mass in the left periauricular region and facial paresis for 6 months. According to the patient, the mass was first detected 1 year ago, and he had not experienced any discomfort associated with the mass at the time. Magnetic resonance imaging revealed a solid tumor mass, located across the deep and superfacial lobe of the parotid gland, and wrapping around the carotid sheath (Figure 2A). In addition, enlarged lymph nodes were found in ipsilateral levels II and III, with ENE and fusion (Figure 2B). He then underwent FNA biopsy and was histopathologically diagnosed with LEC with positive EBERs. The quantification of EBV-DNA load was 4.60 × 103 copy/mL. Clinical staging was T4N3M0. The patient was treated with the same IC and CCRT regimen as case 1. After CCRT, CR was reached in both primary tumor and lymph nodes (Figure 2C). No serious adverse events were found. The quantification of EBV-DNA load went down to 0 a month and a half after treatment. There was no evidence of recurrence or metastasis after 50 months.
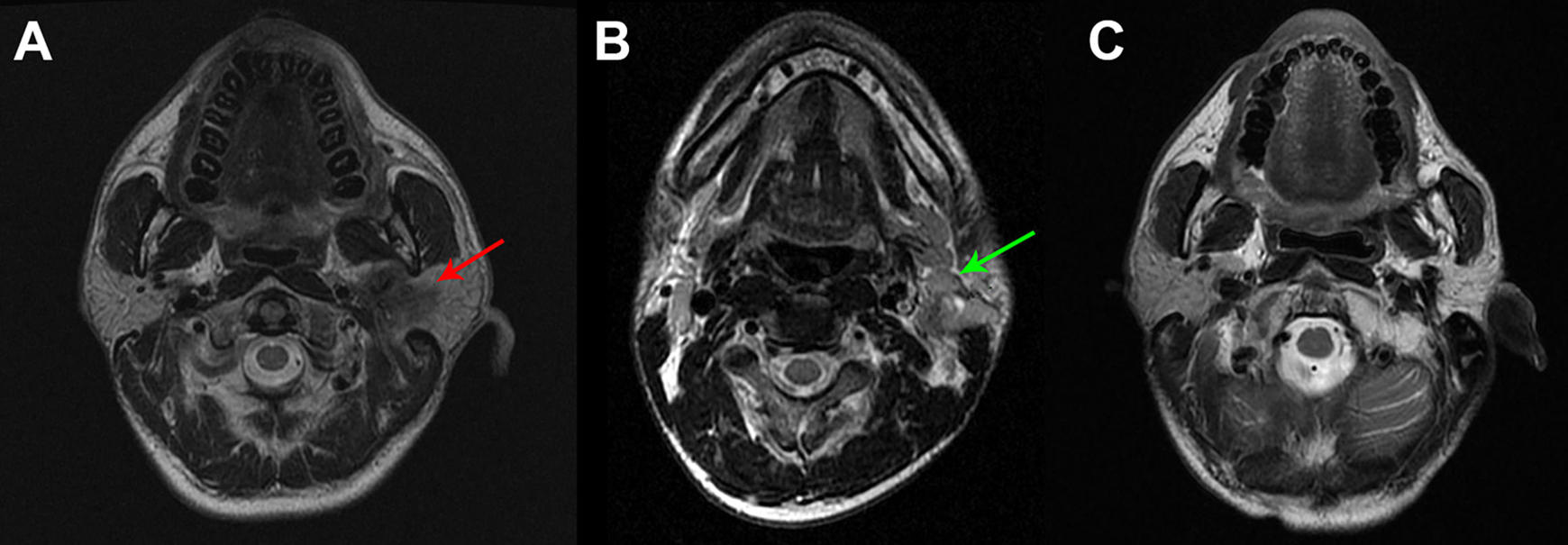
A, The primary tumor (red arrow) was detected in the left parotid; (B) the enlarged lymph nodes (green arrow) in ipsilateral side before the treatment; (C) after induction chemotherapy (IC) and concurrent chemoradiotherapy (CCRT), complete response (CR) was reached in the primary tumor.
Case 3
A 52-year-old man was referred to the Sun Yat-sen University Cancer Center for consultation due to a suspicious mass in the right submandibular region. According to the patient, the mass was first detected 1 year ago with no other clinical manifestations. However, it had enlarged rapidly after acupuncture and moxibustion treatment. Magnetic resonance imaging scans (Figure 3A) showed a solid mass, approximately 7 cm in diameter. After performing FNA biopsy, the histopathological report showed LEC. Lymph nodes were found in ipsilateral levels II, III, IV, and VIII. Clinical staging was T4N2M0. A positron emission tomography–computed tomography scan was performed to evaluate the primary malignancy. There was no evidence of distant metastases. In addition, the test for EBERs was positive, and quantification of EBV-DNA load was 4.19 × 104 copy/mL. The patient was treated with the same IC and CCRT regimen as case 1. After IC, the patient reached PR, and CR was achieved after CCRT. No serious adverse events were found. Fourteen months after treatment, the patient developed distant metastases to the liver, lung, spleen, and bones, with quantification of EBV-DNA load reaching up to 1.24 × 108 copy/mL. There was no evidence of locoregional recurrence.
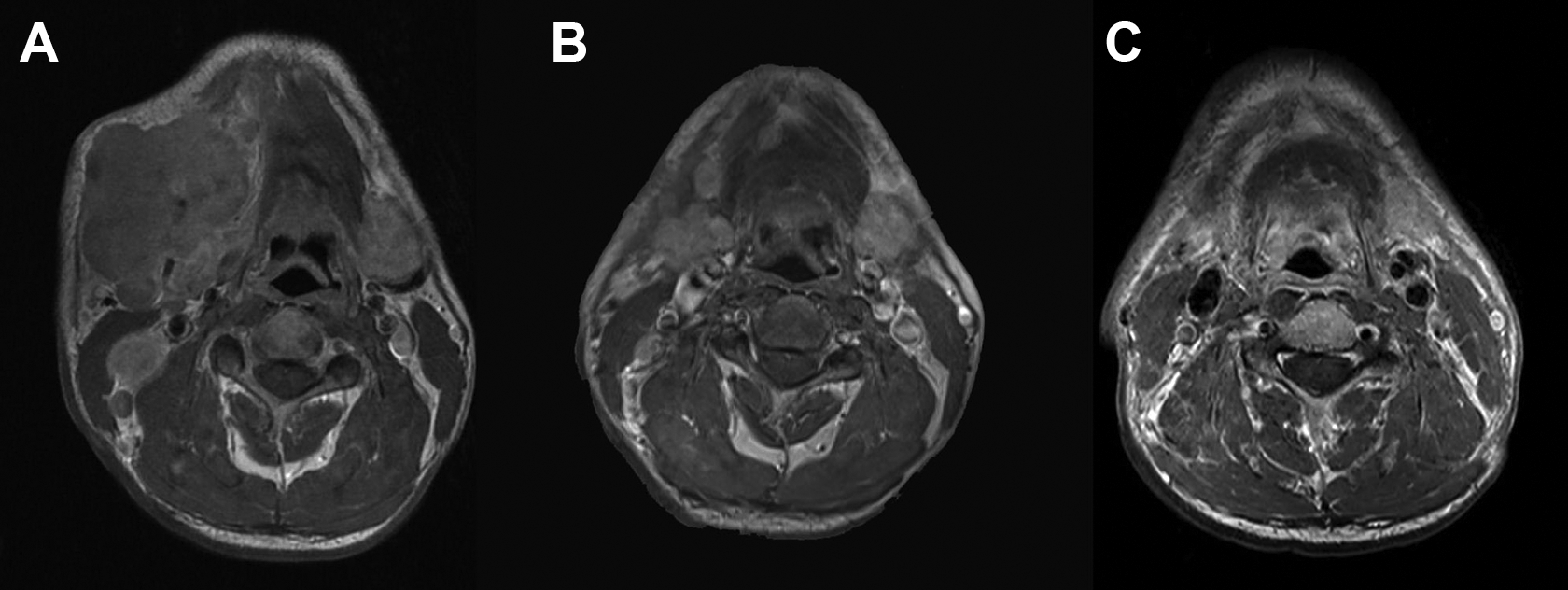
A, The mass invaded the right sublingual gland, masseter, and body of the mandible with enlarged lymph nodes in ipsilateral side. B, Both the gross tumor and lymph nodes reduced remarkably after induction chemotherapy (IC). C, Complete response (CR) was achieved after concurrent chemoradiotherapy (CCRT).
Discussion
Lymphoepithelial carcinoma is histologically indistinguishable from NPC. The histopathological finding is commonly found at the nasopharynx and it is very rare to occur at other sites, so no abnormal findings about nasopharynx have to be assured before diagnosis. Epstein-Barr virus plays an important role in the pathogenesis of LEC, and this association is much stronger in Asians than in Caucasians. 4 Determination of EBV status is valuable in diagnosing distant tumors which are suspected metastases, 4 and this was consistent with our study (case 3).
It’s acknowledged that chemoradiotherapy has been the first-line therapy in the treatment of advanced NPC. But combined therapy with surgery and postoperative radiation has been adopted for LEC for years. 1 We reviewed the literature concerning LEC of the salivary gland after 2000 (Table 1) and found scarcely any reports of patients treated without surgery. Only Maeda et al 2 reported a case of LEC in the parotid gland, effectively treated with CCRT, and with no locoregional recurrence or metastasis after 5 years from treatment. For other non-nasopharyngeal head and neck cancers, Muthayam et al 4 administered radiotherapy alone to LEC of the maxillary sinus in one case with no lymph node metastasis, and the patient remained disease-free at the 20-month follow-up. To the best of our knowledge, this is the first study to perform an induction-concurrent approach in patients with advanced LEC of the parotid gland.
A Summary of the Review of the Literature on Salivary Gland Lymphoepithelial Carcinoma.a
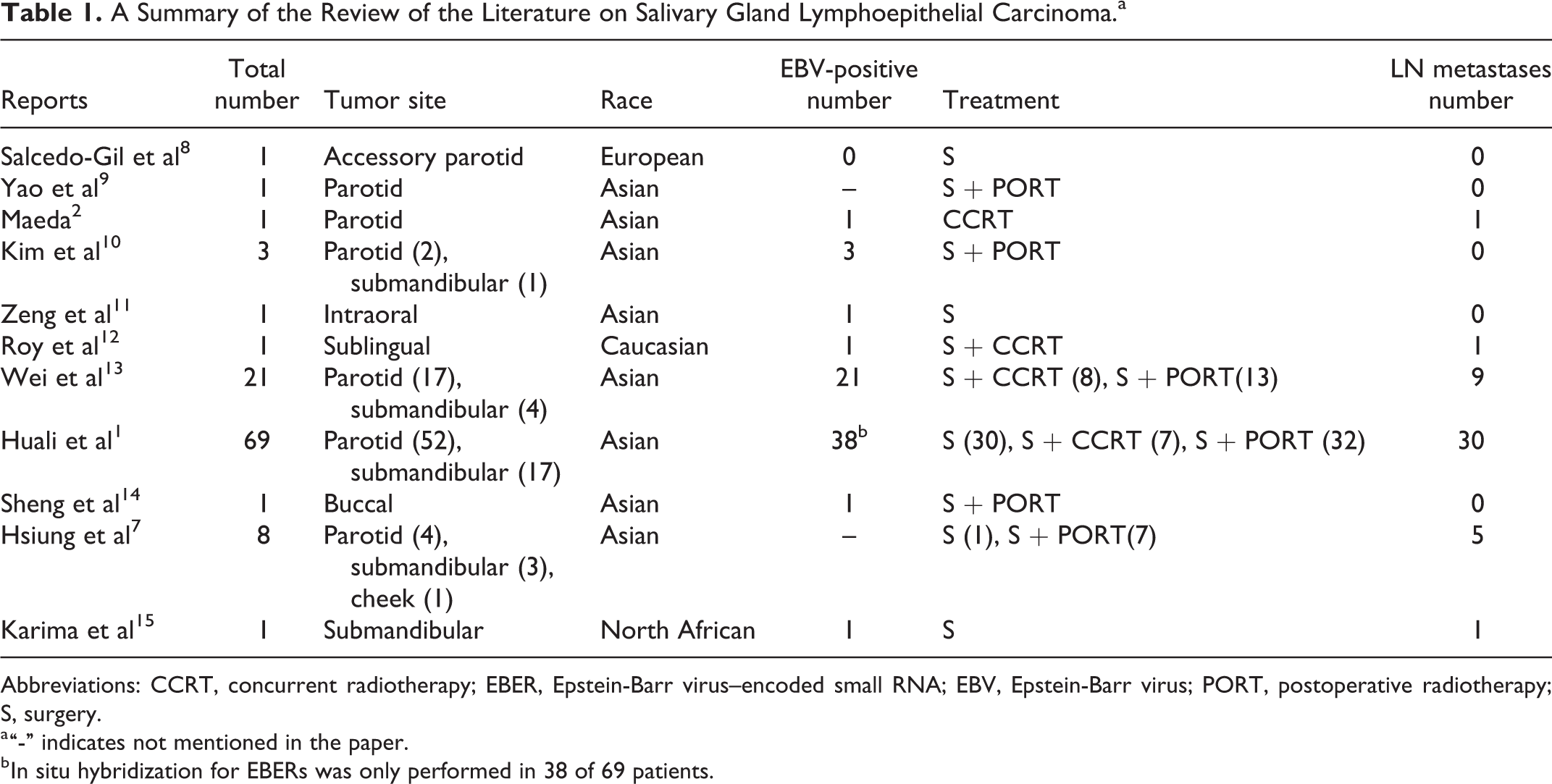
Abbreviations: CCRT, concurrent radiotherapy; EBER, Epstein-Barr virus–encoded small RNA; EBV, Epstein-Barr virus; PORT, postoperative radiotherapy; S, surgery.
a “-” indicates not mentioned in the paper.
b In situ hybridization for EBERs was only performed in 38 of 69 patients.
Multiple studies have reported a response rate of 73% to 93% with a significant tumor volume decrease in NPC after IC,3,5 which led to a remarkable reduction in target volume and corresponding toxicity. In addition, IC is considered to be more effective than adjuvant chemotherapy, which can be associated with improved patient compliance and early eradication of micrometastases, and can significantly increase failure-free survival and overall survival for advanced NPC; consequently, it has been adopted in National Comprehensive Cancer Network (NCCN) guidelines. 5 As a result, considering the similarity between LEC and NPC, the induction-concurrent approach was performed in our study and favorable outcomes were achieved. On the other hand, the tumors in the 3 cases mentioned above were all classified as category T4, which brought difficulties and challenges for operation. Surgery would likely cause significant contour defects, resulting in mild to severe disfigurement and irreversible damage to facial nerve function, which could decrease the patients’ quality of life and self-confidence. As in case 1, it was impossible to perform operation before IC, because the primary tumor had invaded almost all the right part of the head, extending to the temporal lobe, the carotid sheath and even the submandibular gland. The patient would definitely have facial depression deformity after surgery. Consequently, in this case, chemoradiotherapy would be a better choice. In our study, after treatment all 3 cases had their facial nerve intact and a normal appearance without reconstruction.
Cervical lymph node metastases are involved in up to 70% of patients, 6 a rate that is similar to NPC. In our study, all 3 advanced cases had lymph node metastases, distributed from the parotid nodes to the cervical nodes. Besides, ENE is always found in late-stage patients, which also brings difficulties and challenges for operation.
Chan et al evaluated 103 cases of salivary gland LECs with a 5-year overall survival (OS) of 80.8%, 6 while Ma et al identified 69 patients with 5-year OS and recurrence free survival (RFS) rates being 90% and 69%, respectively. 1 Considering the limited number of patients and the short follow-up duration in our study, the conclusion needs to be proven in a larger case study. However, the fact that there was no evidence of recurrence in any of our cases gave us confidence. As for distant metastases, Hsiung et al reported 2 cases of distant failure of 9 patients after PORT, the interval between surgery and distant failure being 6.3 months and 6.5 months, respectively, and all were classified as T3N2bM0. 7 However, in Ma’s research, there were also only 2 patients who experienced distant metastases of 69 patients after surgery, and the interval between surgery and distant failure was 82.5 months and 70.3 months, respectively. 1 The discrepancy may partly be due to the different compositions, with 100% advanced patients in Hsiung’s study and 30.4% in Ma’s. Many studies1,7 demonstrated that the main cause of treatment failure was distant metastasis, which occurred more frequently in patients with lymph node involvement. This is similar to Hsiung’s and our results, and more prospective studies with abundant samples will need to be carried out.
Conclusions
We applied the induction-concurrent approach in 3 cases, and after IC, tumor volumes had all decreased by up to 80% in our study, and all achieved CR after CCRT. All the patients avoided cosmetic or functional disturbance, and no patients developed locoregional failure. On account of its similarity to NPC, this approach has the potential to be widely recommended for advanced patients in future, but the findings need to be verified in a clinical trial with abundant samples.
Footnotes
Authors’ Note
S.L. and D.X. contributed equally to this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
