Abstract

Temporal bone meningoceles are uncommon malformations in which meninges protrude through a defect in the temporal bone while meningoencephalocele is a condition where brain tissue and meninges herniate through the bone defect. Bone defects can be acquired (trauma, surgery, tumor spread) or spontaneous (congenital). 1 Spontaneous temporal bone meningoceles and meningoencephaloceles are unusual findings and may present with hearing loss, headache, otorrhea, and recurrent acute otitis media or rarely by otogenic meningitis or even cerebral abscess due to the thinness of tegmen tympani which separates the middle cranial fossa from the tympanic cavity. Small temporal bone meningoceles can be difficult to differentiate from other solid or cystic temporal bone mass on high-resolution computed tomography (CT). However, magnetic resonance imaging (MRI) with high-resolution sequences is able to depict meningoceles as it presents as a cerebrospinal fluid (CSF) collection. Magnetic resonance imaging also can show spontaneous CSF fistulas between the subarachnoid space and middle ear or a defect in the osseous labyrinth. Magnetic resonance imaging is the main imaging technique in assessing complications of middle ear infections leading to CSF fistulas, meningitis, or cerebral abscess formation. 2
This case describes a 59-year-old white female patient who presented with a history of recurrent right-sided middle ear and mastoid infection and a previous episode of bacterial meningitis caused by Streptococcus pneumonia. Upon examination, the patient had normal colored tympanic membrane, without perforation and complained of right-sided otalgia. Pure tone audiogram suggested bilateral normal hearing threshold levels with smaller sensorineural hearing losses in higher hearing frequencies. Tympanometry revealed a normal type A tympanogram. Because of recurring otitis media and previous pneumococcal meningitis, the patient was scheduled for CT and MRI. Temporal bone imaging revealed bilateral temporal bone meningoceles, a left petrous apex cephalocele, and an arachnoid cyst in the middle cranial fossa. A high-resolution CT examination using a soft tissue algorithm revealed bone erosions at the cranial parts of both petrous bones showing soft tissue density (Figure 1) A bone defect with sharply defined osseous margin in the left petrous apex filled showing fluid density was also noticed. The fluid density collection was seen in the left middle cranial fossa with bone remodeling. Computed tomography with bone algorithm using axial and coronal images showed bulbous enlargement of the geniculate segments of the facial nerve canals with focal bone dehiscence (Figure 2). Magnetic resonance imaging with T2-weighted and fluid-attenuated inversion recovery (FLAIR) sequence showed signal intensities similar to CSF filled the enlarged facial nerve canals at the geniculate ganglion segments (Figure 3A and B). No contrast enhancement or mass lesions were detected along the course of facial nerves to suggest other lesions such as schwannoma (Figure 3C and D). Three-dimensional constructive interference in steady state (CISS) high-resolution MRI showed bulbous expansions of geniculate segments of the facial nerve canals with bone dehiscence and protrusions of meninges and CSF into the petrous bones suggesting bilateral meningoceles (Figure 4A). Magnetic resonance imaging also showed cystic left petrous apex lesion along posterolateral margin of Meckel cave filled with CSF fluid characteristic of a petrous apex cephalocele (Figure 4B). The CISS also showed left middle cranial fossa arachnoid cyst with fluid content that follows CSF intensity and bone remodeling of the adjacent skull (Figure 4B). No surgical reconstruction of temporal bone defects was performed since the patient did not have neurological symptoms or loss of function.
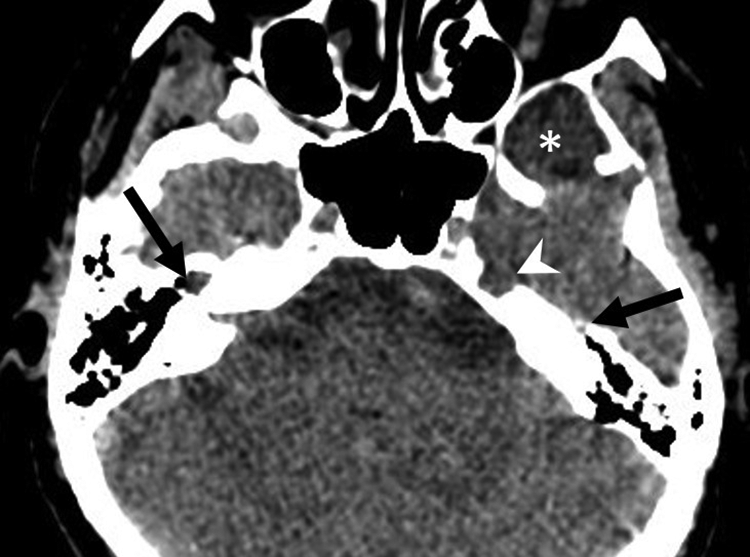
Axial CT image showing small bone erosions of petrous parts of the temporal bones (arrows). Bone defect in the left petrous apex with fluid density (arrowhead). The fluid density collection in the left middle cranial fossa with bone remodeling (asterisk). CT indicates computed tomography.
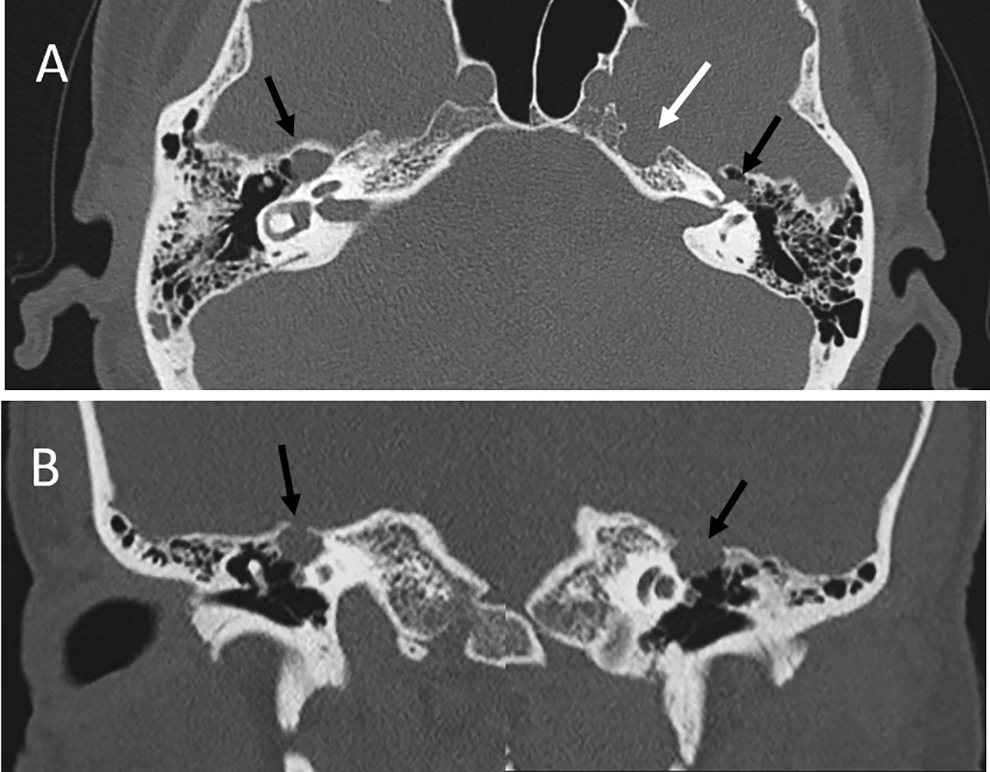
A, Axial CT image shows the soft tissue densities and bulbous enlargement of geniculate ganglion segments of the facial nerve canals (black arrows). Bone remodeling of the left petrous apex (white arrow). B, Coronal CT image shows the focal dehiscence of the temporal bone over the dilated geniculate segment of the facial nerve canals (black arrows). CT indicates computed tomography.
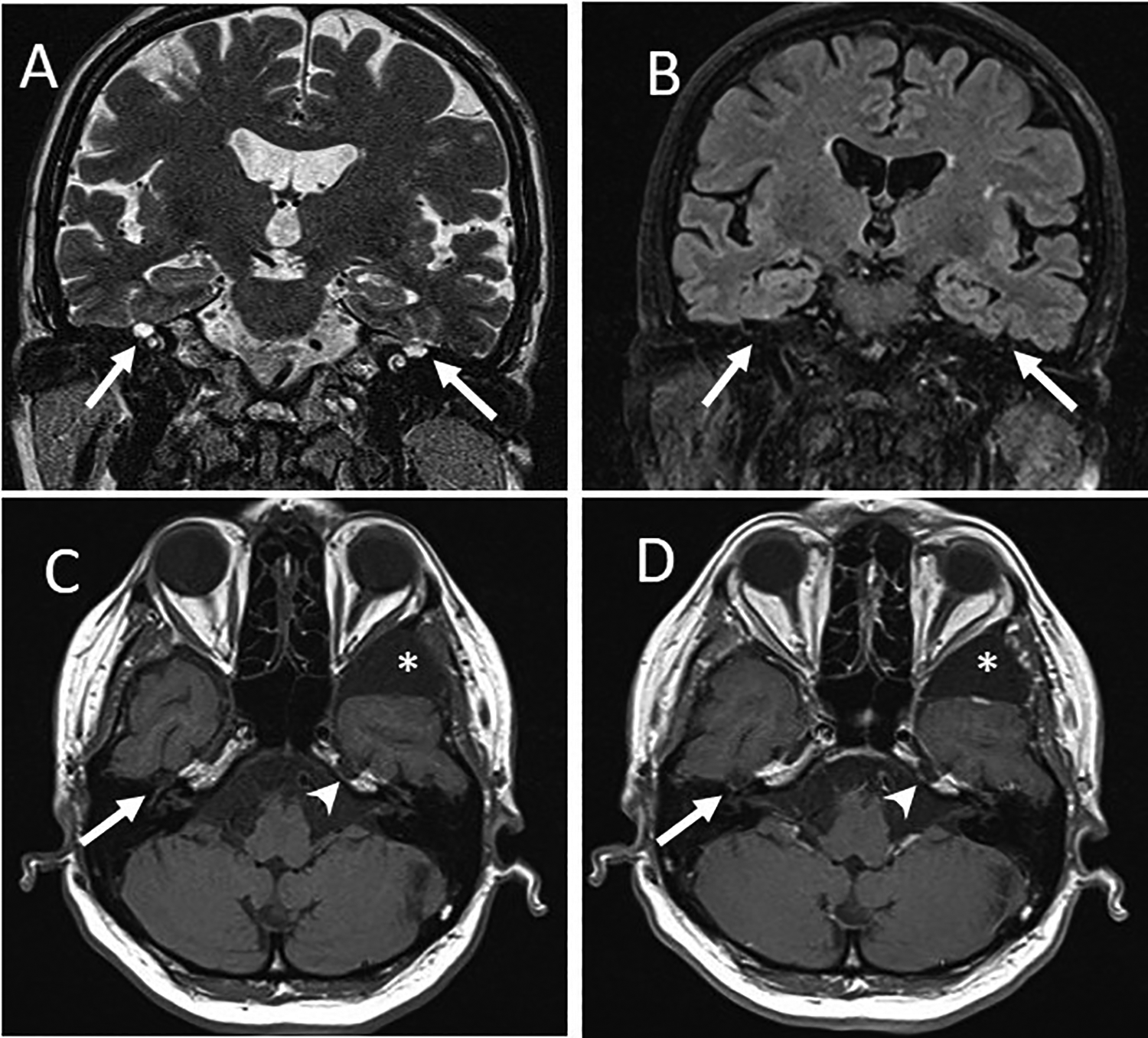
A and B, T2-weighted and FLAIR images show dilatation of the geniculate segments of the facial nerve canals filled with fluid that follows signal intensities similar to the cerebrospinal fluid. Coronal T1 MR image (C) and T1 contrast-enhanced (D) image show no enhancement or solid mass in the temporal bones. Left petrous apex bone defect (arrowhead) and cystic lesion in the left middle cranial fossa (asterisk) also show MR signals similar to the cerebrospinal fluid. FLAIR indicates fluid-attenuated inversion recovery; MR, magnetic resonance.
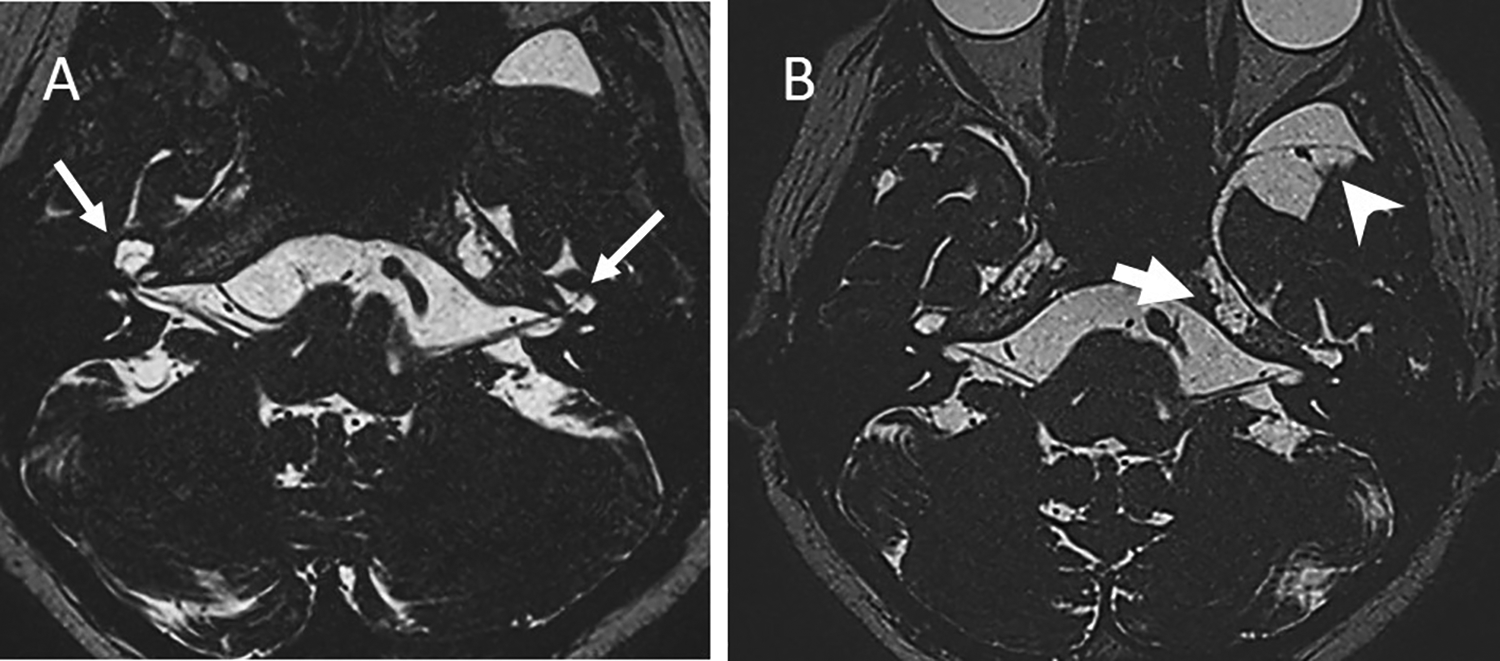
A, Axial 3D CISS MR images show expansion of the geniculate segments of the facial nerve canals filled with cerebrospinal fluid (white arrows) representing temporal bone meningoceles. B, There is a dilatation of subarachnoid space in the left petrous apex that communicate with Meckel cave representing petrous apex cephalocele (wide arrow). Cyst filled with cerebrospinal fluid in the left middle cranial fossa with bone remodeling represents arachnoid cyst (arrowhead). 3D-CISS indicates 3-dimensional constructive interference in steady state; MR, magnetic resonance.
Our case report was assembled following Case Report (CARE) case report guidelines, with written informed consent obtained from the patient and the institutional review board according to the Helsinki Declaration of 1983.
This case presents a rare combination of malformations of the skull base and meningeal membranes which affects both temporal bones and puts the patient at risk of otogenic infection spread and for developing meningitis. 3 Few authors have reported similar cases in which a connection between recurrent meningitis and meningoceles was established. Although some cases report trauma-related cephaloceles, the vast majority of the cases suggest congenital abnormalities. 4 -9 Cephaloceles are mostly accidental findings in patients who undergo radiological imaging for a number of conditions. Patients may present with symptoms which vary from headache, neck stiffness, photophobia, fever, hearing loss, rhinorrhea, otorrhea, and middle ear infection. 5 -7 A thorough literature review shows many patients showing a link between cephaloceles and recurrent meningitis, although our case is the first to describe a bilateral temporal bone localized meningocele in conjunction with previous meningitis episodes which originated from a middle ear infection. Because the tympanic and mastoid tegmen is usually thinner than the posterior fossa surface of the petrous pyramid, with larger spaces into which intracranial tissue can herniate, it is not surprising that meningoencephaloceles of the temporal bone more commonly arise from the middle fossa. 1 As reported, meningitis is the initial presentation in up to 90% of pediatric cases compared with 0% to 30% of adult cases. 8,9 Due to the complex anatomical region of the temporal bone and presence of meningoceles with CSF build up in the petrous bones, both CT and MRI were needed. Whereas CT excelled in revealing bone erosions, MRI with FLAIR was needed for the detection of CSF collections and CSF fistula between subarachnoid space and bone defects. Previous reports described meningoceles in various locations, some in the temporal bone region as well, but none have described bitemporal localization with facial nerve canal involvement. 1,8,9 Despite the large size and proximity of the meningocele to the facial nerve, our patient remained asymptomatic, showed no signs of nerve palsy and therefore did not require surgical treatment, but was scheduled for clinical observation, and the infections of the right middle ear subsided after antibiotic treatment, with the patient free from recurrence during a 10-month follow-up period.
Footnotes
Authors’ Note
Data sharing is not applicable to this article as no new data were created or analyzed in this study. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
