Abstract
Aim:
Pharyngodynia, nasal congestion, rhinorrhea, smell, and taste dysfunctions could be the presenting symptoms of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2. The aim was to perform a systematic review of current evidences on clinical presentation of COVID-19, focusing on upper airway symptoms in order to help otolaryngologists identifying suspected cases.
Methods:
We searched PubMed and Web of Science electronic databases.
Results:
We included 5 retrospective clinical studies for a total of 1556 hospitalized patients with COVID-19, 57.5% were male and mean age was 49.1 years. Pooled data revealed that pharyngodynia was present in 12.4% of patients, nasal congestion in 3.7%, and rhinorrhea was rare. No reports on COVID-19 and olfactory/gustative disorders matched inclusion criteria but preliminary evidences suggested they could be present. Common symptoms were fever (85.6%), cough (68.7%), and fatigue (39.4%). Frequent comorbidities were hypertension (17.4%), diabetes (3.8%), and coronary heart disease (3.8%); 83% of patients had alterations on chest computed tomography that were bilateral in 89.5% of cases. Ground-glass opacity was the most common finding (50%). Lymphopenia (77.2%) and leucopenia (30.1%) were common. Critical cases with complications were 9%, intensive care unit admission was required in 7.3%, invasive ventilation in 3.4%, and mortality was 2.4%.
Conclusion:
Otolaryngologists should know that pharyngodynia, nasal congestion, olfactory, and gustative disorders could be the presenting symptoms of COVID-19. Clinical presentation together with radiological and laboratory findings could help to identify suspected cases.
Introduction
A novel member of human RNA coronavirus was newly identified in Wuhan, China. International Committee on Taxonomy of Viruses 1 officially named it as severe acute respiratory syndrome-coronavirus 2 (SARS-CoV-2). It was established that the SARS-CoV-2 belonged to the β-coronavirus 2b lineage in the phylogenetic tree. By examining the full-length genome of SARS-CoV-2, it was discovered that this novel virus shared 87.99% identity sequencing with the Bat SARS-like coronavirus, and it shared approximately 80% identity nucleotide with the original SARS epidemic virus. 2 Based on the preliminary information of this novel virus, it was considered that SARS-CoV-2 represented the third zoonotic human coronavirus of the century. 3 World Health Organization (WHO) recently named the disease caused by SARS-CoV-2 as coronavirus disease 2019 (COVID-19). Clinical evidences have demonstrated that this virus was transmissible from person to person. 4 The transmission capacity was usually estimated using the so-called basic reproduction number or R0. Reliable estimates placed the R0 value of the COVID-19 in 1.4 to 2.5, similar to the R0 of the coronavirus SARS (SARV-CoV) at the beginning of the epidemic (2.2-3.7). The latter value was reduced to an R0 of 0.67 to 1.23 at the end of the SARS-CoV epidemic. By contrast, the coronavirus Middle-East respiratory syndrome has always remained at lower R0 values (0.29-0.80). 5 It seemed that the COVID-19 could be more easily transmitted than SARS. The SARS-CoV-2 cases increased rapidly in Wuhan and Hubei Province and they extended with transmission chains throughout China. Outside China, imported cases and secondary cases have been reported in many countries and territories, and WHO declared COVID-19 outbreak a pandemic on March 11, 2020. 6
Studies have shown that COVID-19 could induce fever, dry cough, dyspnea, and fatigue in infected patients. In more severe cases, infections caused viral pneumonia and could lead to severe acute respiratory distress syndrome (ARDS) and even death. 7 Pharyngodynia, nasal congestion, and rhinorrhea have been reported in patients with COVID-19. 8 Recently, the European Rhinology Society reported that “a significant part of the COVID-19 patients (20-60%) appear to have loss of smell. Loss of smell can be the presenting symptom before other symptoms like coughing/fever occur.” 9 Also Professor Claire Hopkins, the President of British Rhinological Society, said that “There is already good evidence from South Korea, China and Italy that significant numbers of patients with proven COVID-19 infection have developed anosmia/hyposmia”. 10 Consequently, an otolaryngologist could be the first to evaluate patients with COVID-19. Considering that the most likely route of transmission of SARS-CoV-2 is by contact and respiratory droplets (aerosols), over short distances (1.5 m), 2 ENT examination stands at high risk for transmission of the virus to health-care providers.
Real-time reverse-transcriptase polymerase chain reaction (RT-PCR) assays were used for detection of SARS-CoV-2 from respiratory secretions collected by nasal and oropharyngeal swabs. 11 High-false-negative rates (FNRs) have been reported for these tests. Results from RT-quantitative PCR could be affected by variations in viral RNA sequences or in viral loads in different anatomic sites during the disease natural history. 11 By estimate, FNR from one-time testing was 30% to 50% in real COVID-19 cases. 11 Considering 1014 COVID-19 cases, a recent report found that only 59% of patients had positive SARS-CoV-2 swabs at presentation, while chest computed tomography (CT) had higher sensitivity for diagnosis of COVID-19. 12 Clinical presentation is fundamental to identify suspected COVID-19 cases. To the best of our knowledge, no previous report focused on upper airway symptoms.
The aim of the present study was to review systematically the current evidences on clinical presentation of COVID-19, focusing on upper airway symptoms in order to help otolaryngologists identifying suspected cases.
Materials and Methods
Electronic Database Search
A search was run in the PubMed and Web of Science electronic databases for articles on COVID-19. The search term was “COVID 19.” The “Related articles” option on the PubMed homepage was also considered. We examined titles and abstracts of articles available in the English language. The identified full texts were screened for original data, and the related references were retrieved and checked manually for other relevant studies.
Inclusion and Exclusion Criteria
Studies were included when the following general criteria were met: (1) articles were original reports including at least 80 patients; (2) studies included only laboratory-confirmed cases; (3) studies reported detailed information about clinical presentation; (4) study design was not a case report, editorial, letter to the editor, or review; (5) the report was published in the English language.
Data Extraction
The 2 authors analyzed the data from the literature. Any disagreements were solved by a discussion among the study team members. Included studies were analyzed to extract all available data and assure eligibility for all patients. Descriptive data of patients, treatments, and outcomes were extracted and recorded for all included studies. Age, sex, clinical symptoms, associated chronic medical illness, radiology, and laboratory findings were extracted from electronic medical records. Only results of chest CT were considered. Data regarding complete blood count, C-reactive protein, procalcitonin, lactate dehydrogenase, and
Results
Retrieving Studies
Search was started on February 24, 2020, and the last search was March 1, 2020. A total of 823 titles were retrieved (627 from PubMed, 196 from Web of Science). After removing duplicates, title and abstract screening allowed us to identify 54 studies potentially relevant to the topic. The full-text screening of such articles led to exclusion of 49 studies that did not match inclusion criteria. The remaining 5 articles 7-8,13 -15 were considered eligible for entering the review. Figure 1 summarizes the selection process.
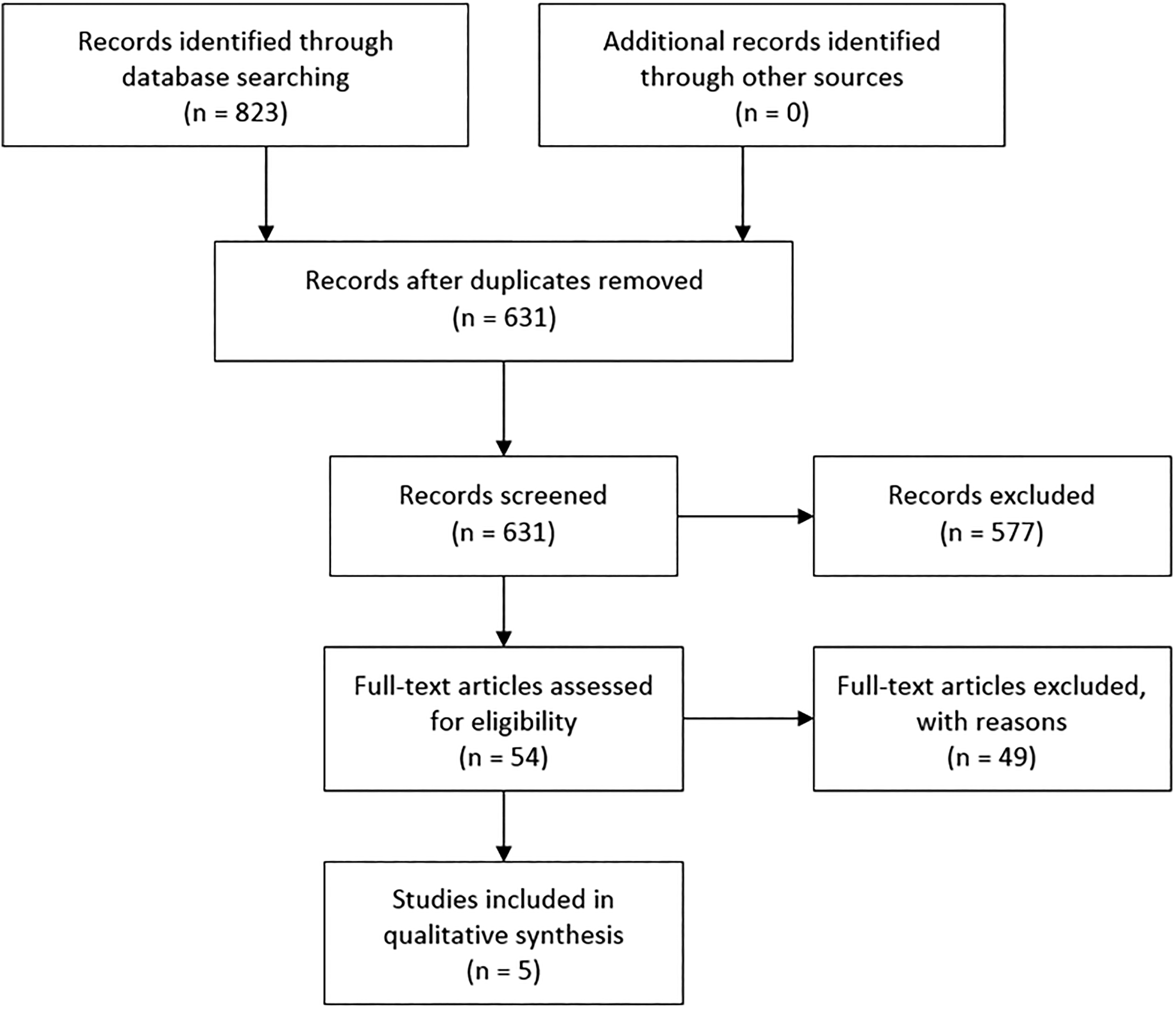
Flow diagram showing decision process that led to included studies.
Assessment of the Studies
All included studies had adequate relevance to the subject of this review. No study was a randomized controlled trial, but all retrieved articles were retrospective (5 cohort studies). Quality of the evidences was low, level 4, according to the Oxford Centre for Evidence-based Medicine for ratings of individual studies. 16 None used a control group. All included studies followed the COVID-19 diagnostic protocol proposed in the “Handbook of COVID-19 Prevention and Treatment” by the First Affiliated Hospital, Zhejiang University School of Medicine. 17 A confirmed case was based on epidemiological history (including cluster transmission), clinical manifestations (fever and respiratory symptoms), lung imaging, and results of SARS-CoV-2 nucleic acid detection. All studies used high-throughput sequencing or RT-PCR assays on nasal 7 or pharyngeal swab specimens. 7-8,13 -15 All studies followed the protocol established by the WHO for RT-PCR assay. 18 The 5 included studies reported only hospitalized patients.
Clinical Presentation
We included 1556 patients, 895 males (57.5%) and 661 females (42.5%). The median age in years was 47, 7 55, 8 56, 13 57, 14 and 44. 15 Pooled data regarding symptoms and comorbidities were reported in Table 1. In the included studies, internal medicine physicians or Centers for Disease Control and Prevention staff members conducted the clinical examinations. Considering upper airway symptoms, pharyngodynia was present in 12.4% of patients, while nasal congestion in 3.7%. Rhinorrhea was found only by Chen et al in 4% of patients. 8 None of the included studies reported about olfactory or gustative dysfunctions. One report did not found any upper airway symptoms. 14 Fever, cough, and fatigue were the most common symptoms (85.6%, 68.7%, and 39.4%, respectively). One investigation reported that fever occurred in only 43.8% of patients at initial presentation and developed in 87.9% of cases during hospitalization. 7 Frequent associated chronic medical illness were hypertension (17.4%), diabetes (3.8%), and coronary heart disease (3.8%). The median incubation period, when reported, was 3.0. 7 One study reported that the median durations from first symptoms to dyspnea, hospital admission, and ARDS were 5 days (interquartile range [IQR]: 1-10), 7 days (IQR: 4-8), and 8 days (IQR: 6-12), respectively. 13
Clinical Presentation of Patients With COVID-19.
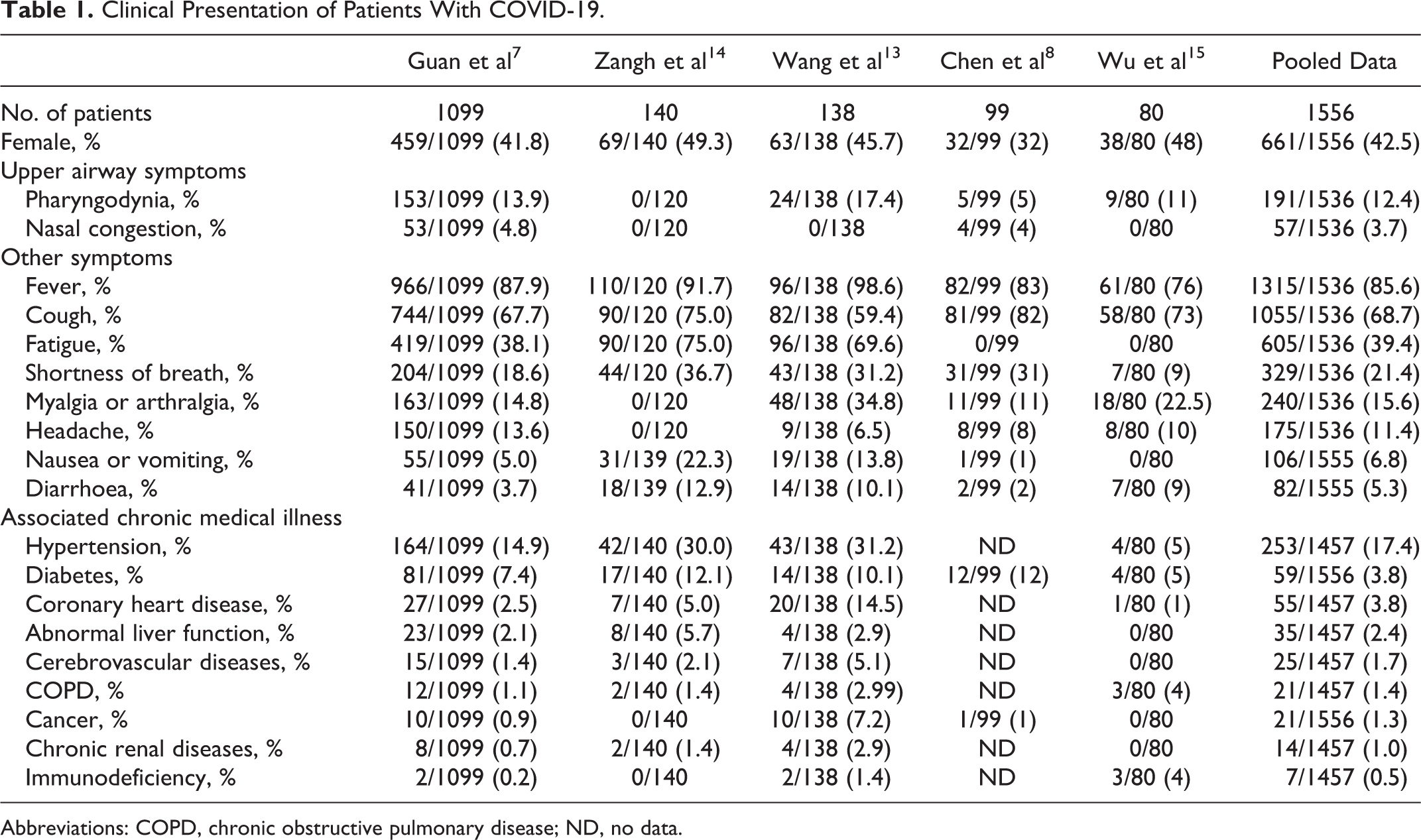
Abbreviations: COPD, chronic obstructive pulmonary disease; ND, no data.
Radiology
One investigation used chest X-ray in some patients (14%) and chest CT in the others, 7 while other reports used CT in all patients. 8,13 -15 Most of patients (83%) had alteration on chest CT that was bilateral in 89.5% of cases. Different studies considered different radiological characteristics; ground-glass opacity was reported by 3 authors and was present in almost 50% of cases. 7,8,14
Laboratory Findings
Two studies 13,15 reported only mean values of laboratory tests. The others 3 investigations used the same normal range for blood cell count allowing us to pool data (Table 2). For other parameters, ranges were dissimilar between studies. Lymphopenia (77.2%) and leucopenia (30.1%) were common.
Radiological and Laboratory Findings in Hospitalized Patients With COVID-19.
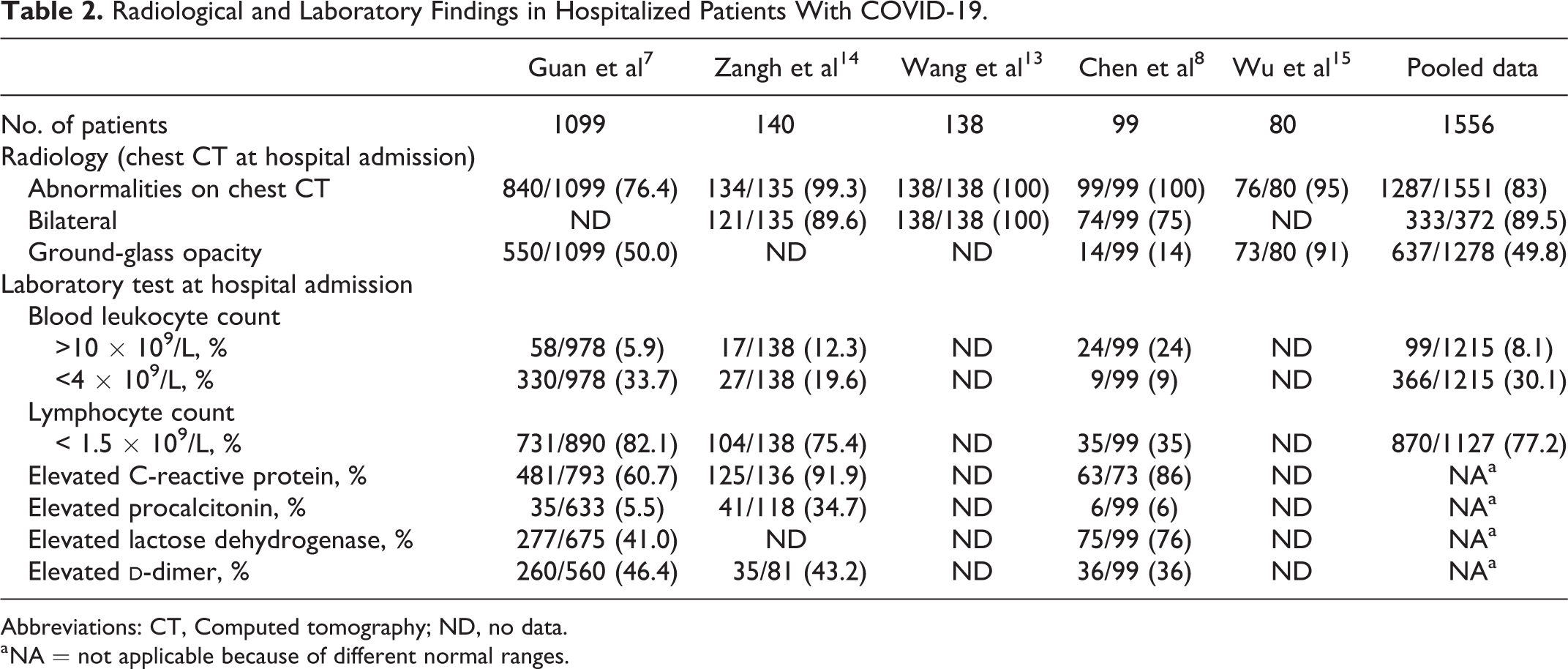
Abbreviations: CT, Computed tomography; ND, no data.
a NA = not applicable because of different normal ranges.
Severe Cases
Two studies classified patients at presentation in severe versus nonsevere. Guan et al 7 used international guidelines for community-acquired pneumonia 19 and found 15.7% of severe cases. Severe patients were older, had more associated comorbidities, radiologic abnormalities, lymphopenia, and leucopenia.
Zhang et al
14
defined severity of COVID-19 according to the diagnostic and treatment guideline for SARS-CoV-2 issued by Chinese National Health Committee.
17
This clinical classification includes: Mild cases (the clinical symptoms are mild and no pneumonia manifestations can be found in imaging); Moderate cases (patients have symptoms such as fever and respiratory tract symptoms, etc, and pneumonia manifestations can be seen in imaging); Severe cases (meeting any of the following criteria: respiratory distress with respiratory frequency ≥30/min; pulse oximeter oxygen saturation ≤93% at rest; or oxygenation index [artery partial pressure of oxygen/inspired oxygen fraction] ≤300 mm Hg); and Critical cases (meeting any of the following criteria: occurrence of respiratory failure requiring mechanical ventilation; the presence of shock; or other organ failures that require monitoring and treatment in the intensive care unit [ICU]).17
Zhang et al found 41% of severe cases on admission who were older, showed increased leukocyte number,
Complications, Supportive Treatments, and Outcome
Three studies considered the clinical course of 1336 patients during hospitalization (Table 3). Acute respiratory distress syndrome was the most frequent complications (6%). Most of patients needed oxygen therapy (44.8%), while ICU admission was required in 7.3%. Mortality was 2.4%, while 10% of patients were discharged from hospital at time of paper publication. Severe cases yielded significantly higher rates of any complications as compared with nonsevere cases and needed more supportive treatments.7 Patients who required ICU care were significantly older and were more likely to have underlying pathologies.13
Complications, Supportive Treatment, and Outcome of Hospitalized Patients With COVID-19.
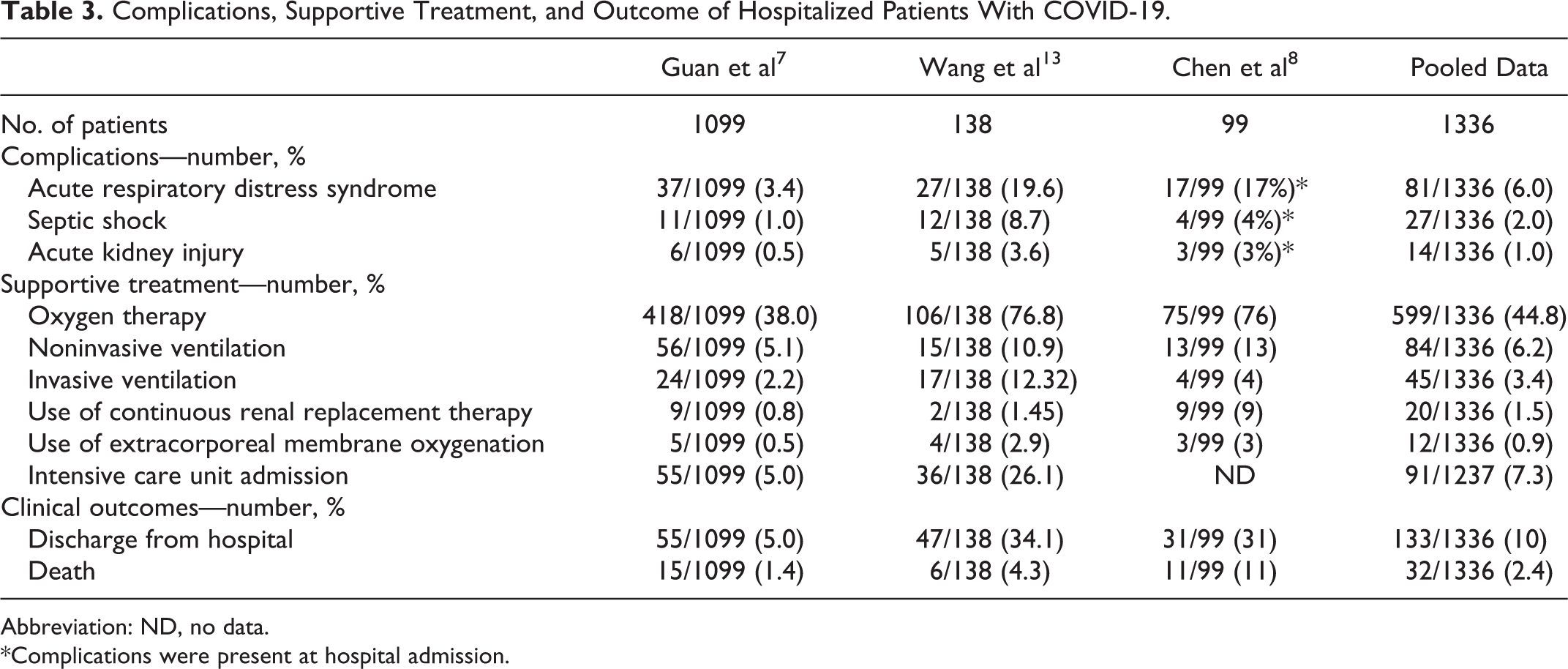
Abbreviation: ND, no data.
*Complications were present at hospital admission.
Discussion
Recently, the Chinese Center for Disease Control and Prevention published a large case series of COVID-19 in mainland China (72 314 cases, updated through February 11, 2020). 20 Among a total of 72 314 case records, 44 672 were classified as confirmed cases of COVID-19 (62%; diagnosis based on positive viral nucleic acid test result on throat swab samples), while others were suspected upon clinical characteristics. Most cases were classified as mild (81%; ie, nonpneumonia and mild pneumonia), and most common symptoms were cough and fever 20 but there was not a detailed description of other symptoms. Increasing number of reports claimed that negative nasal and oropharyngeal swab did not rule out COVID-19, and patients could transmit the infection. 11,12,21 Consequently, clinical presentation could be helpful to identify suspected cases. These patients should be isolated (use of individual rooms was recommended, with negative pressure if possible), and health personnel should use waterproof gowns, gloves, goggles, and surgical masks or FFP2 masks. 22,23 Pharyngodynia, nasal congestion, rhinorrhea, and anosmia were reported as presenting symptoms of COVID-19. 7 -10 The aim of the present investigation was to review systematically the current evidences on clinical presentation of COVID-19, focusing on upper airway symptoms in order to help otolaryngologists identifying suspected cases.
We found that pharyngodynia was quite common between patients with COVID-19 (12.4%), while nasal congestion was not frequent (3.7%), and rhinorrhea was rare. The most common symptoms were fever (85.6%), cough (68.7%), and fatigue (39.4%). Frequent associated chronic medical illness were hypertension (17.4%), diabetes (3.8%), and coronary heart disease (3.8%). A recent review and meta-analysis 24 confirmed that fever and cough were the most frequent symptoms but reported that sore throat was rare. The majority of included patients (48 981) in that review were extracted from studies that did not described accurately the clinical presentation of patients. 25 -27 In the present investigation, we excluded those studies 25 -27 because they were incomplete in symptom description and could confound the real clinical picture of COVID-19. Our review considered only hospitalized patients and this could influence the percentage of complaints. As previously mentioned, more than 80% of patients with COVID-19 had mild symptoms 20 and a consistent part of them could not require hospitalization. Consequently, our estimates on upper airway symptoms could be biased as we found only studies describing hospitalized patients. In patients with mild symptoms, differential diagnosis should be addressed. In fact, pharyngodynia with or without fever could be the presenting symptom of other respiratory infections (eg, tonsillitis or epiglottitis). Nevertheless, it is important to inform otolaryngologists that sore throat and nasal congestion could be present in patients with COVID-19.
Olfaction alterations remain an open issue. None of included reports considered smell reduction. On March 22, 2020, the American Academy of Otolaryngology—Head and Neck Surgery stated that anosmia and dysgeusia have been reported by patients ultimately testing positive for SARS-CoV-2 and proposed to add these symptoms to the list of screening tools for possible COVID-19 infection. 28 A first survey on olfactory and taste disorders was conducted on hospitalized patients with COVID-19 at Hospital Sacco (Milan, Italy) 29 but this report did not match inclusion criteria of the present review. Between 59 interviewed patients, 33.9% reported at least one taste or olfactory disorder and 18.6% reported both alterations. 29 Postviral anosmia was one of the leading causes of loss of sense of smell in adults, accounting for up to 40% cases of anosmia, 30 and it should not be a surprise if SARS-CoV-2 would also cause anosmia in infected patients. Previous studies have shown the ability of SARS-CoV to cause neuronal death in mice by invading the brain via the nose close to the olfactory epithelium. 31 The human coronavirus 229E has already been isolated in nasal discharge from 1 patient with postviral olfactory dysfunction 32 ; SARS-CoV-2 exploited the angiotensin-converting enzyme 2 (ACE2) receptor to gain entry inside the cells, similar to SARS-CoV. 33 The brain has been reported to express ACE2 receptors that have been detected over glial cells and neurons. 33 According to these preliminary evidences, we believed that smell and taste disorders should be considered manifestations of COVID-19.
In our review, lymphopenia (77.2%) and leucopenia (30.1%) were common. Many patients had also elevated C-reactive protein, lactose dehydrogenase, and
There is no specific treatment for COVID-19, although different experimental treatments with antiviral drugs (lopinavir/ritonavir; remdesivir) and interferon are being used. 34 Supportive treatments were frequently needed in our reviewed cases: Oxygen therapy (44.8%) and mechanical ventilation (noninvasive in 6.2% and invasive in 3.4%) were the most required. According to the large series from Chinese Center for Disease Control and Prevention, 14% of COVID-19 cases were severe (ie, dyspnea, respiratory frequency ≥30/min, blood oxygen saturation ≤93%, partial pressure of arterial oxygen to fraction of inspired oxygen ratio <300, and/or lung infiltrates >50% within 24-48 hours), and 5% were critical (ie, respiratory failure, septic shock, and/or multiple organ dysfunction or failure). 20 The overall mortality was 2.3%. 20 In our series, only 2 studies classified patients with severe COVID-19 but with different criteria. Guan et al 7 found 15.7% of severe cases, while Zhang et al 14 found 41%; this difference should be due to the different number of included pneumonia patients (79.1% vs 100%). 7,14 In our series, critical cases (ie, cases with complications) were 9%, and mortality was 2.4%, similarly to the larger series. 20 Most of reports agreed that severe cases were more frequent in older patients and in those with preexisting comorbid conditions (ie, cardiovascular disease, diabetes, chronic respiratory disease, hypertension, and cancer). 7,8,13,20 It has to be underlined that total number of cases with COVID-19 is likely higher due to inherent difficulties in identifying and counting mild and asymptomatic case. This means that many suspected and clinically diagnosed cases are not yet counted. Consequently, mortality estimates could not be realistic at this stage.
The main limitation of the present review was related to the low qualities of included evidences (all retrospective studies). Furthermore, we considered only hospitalized patients and this could not fully represent the clinical spectrum of COVID-19. We found no studies conducted by ENT specialists, and upper airway symptoms (eg, olfactory disorders) could have been underestimated.
In conclusion, otolaryngologists should be aware that pharyngodynia, nasal congestion, smell, and taste disorders could be presenting symptoms of COVID-19. The most common symptoms were fever, cough, and fatigue but fever could be absent in a significant part of patients at initial presentation. Lymphopenia and bilateral alterations at chest CT were very common between patients with COVID-19 and could be helpful in identifying suspected cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
