Abstract
Objective:
To review current pragmatic issues and controversies related to tympanostomy tubes in children, in the context of current best research evidence plus expert opinion to provide nuance, address uncertainties, and fill evidence gaps.
Methods:
Each issue or controversy is followed by the relevant current best evidence, expert insight and opinion, and recommendations for action. The role of expert opinion and experience in forming conclusions is inversely related to the quality, consistency, and adequacy of published evidence. Conclusions are combined with opportunities for shared decision-making with caregivers to recommend pragmatic actions for clinicians in everyday settings.
Results:
The issues and controversies discussed include (1) appropriate tube indications, (2) rationale for not recommending tubes for recurrent acute otitis media without persistent middle ear effusion, (3) role of tubes in at-risk children with otitis media with effusion, (4) role of new, automated tube insertion devices, (5) appropriateness and feasibility of in-office tube insertion in awake children, (6) managing methicillin-resistant Staphylococcus aureus acute tube otorrhea, and (7) managing recurrent or persistent tube otorrhea.
Conclusions:
Despite a substantial, and constantly growing, volume of high-level evidence on managing children with tympanostomy tubes, there will always be gaps, uncertainties, and controversies that benefit from clinician experience and expert opinion. In that regard, the issues discussed in this review article will hopefully aid clinicians in everyday, pragmatic management decisions.
Keywords
Introduction
Tympanostomy tube insertion, the most common ambulatory surgery performed in children in the United States, 1 has origins stretching back more than 200 years. Sir Astley Cooper, a British surgeon and expert anatomist, observed in the late 18th century that some patients with tympanic membrane perforations stabilized their middle ear disease. His attempts to duplicate this with paracentesis (myringotomy), however, were short lived because the perforations closed rapidly. Adam Politzer, in the late 19th century, realized that prolonged middle ear ventilation was needed for success, but it was not until 1954 that Armstrong resolved middle ear effusion (MEE) for a few weeks using a ventilating tube. Subsequent randomized controlled trials (RCTs), first undertaken in the 1980s, demonstrated tube efficacy for otitis media with effusion (OME) and recurrent acute otitis media (AOM). 2
Concurrent with a rise in tympanostomy tube use in the 1990s were allegations of tube overuse, based on proprietary indications and chart review. 3 In 2013, the first, national guidelines on tympanostomy tube indications were published, 4 with subsequent rates of adherence by clinicians estimate at 95%. 5 The current guidelines, however, are limited in scope and deal primarily with indications and global management issues, remaining silent, in many cases, on the pragmatic challenges and controversies encountered by clinicians. Moreover, attitudes toward tubes remain fluid and continue to evolve, leading to a recent position statement on tube insertion in office settings without general anesthesia. 6 This review offers clinicians pragmatic advice on current tube controversies, with the intent of promoting evidence-based care and optimal outcomes.
Methods
This review is based on current, best research evidence combined with the author’s reputation as an otitis media expert and 30-year experience in managing children with tympanostomy tubes. Current best evidence emphasizes RCTs, systematic reviews, and trustworthy clinical practice guidelines (CPGs) produced by professional medical societies. The author’s expertise in otitis media relates to authorship of all related national CPGs, 2,7,8 founder and inaugural president of the International Society for Otitis Media, 9 and the top-ranked expert worldwide (March 2020) in tympanostomy tubes (middle ear ventilation) and OME. 10
Each issue or controversy (Table 1) is followed by the relevant current best evidence, expert insight and opinion, and recommendations for action. The role of expert opinion and experience in forming conclusions is inversely related to the quality and consistency of published evidence, but interpreting any body of evidence, even with a low risk of bias, involves drawing some conclusions about the validity, consistency, and limitations of the aggregate evidence presented. These conclusions are combined with opportunities for shared decision-making with caregivers to recommend pragmatic actions for clinicians in everyday settings.
Tympanostomy Tube Controversies and Issues Discussed in This Review Article.

Abbreviations: AAO-HNSF, American Academy of Otolaryngology–Head and Neck Surgery Foundation; AOM, acute otitis media; otitis media with effusion.
Results
What Are the Appropriate Indications for Tympanostomy Tubes?
Background and context
The answer to this question might at first seem simple—recurrent infection, chronic OME, or structural changes of the tympanic membrane—but the nuances of articulating indications in clear, nonambiguous, evidence-based recommendations were not fulfilled until 2013 when the American Academy of Otolaryngology–Head and Neck Surgery Foundation (AAO-HNSF) published the first national CPG on tympanostomy tubes. 4 Prior to this, several studies questioned the need for tube insertion, 3,11,12 leading The Joint Commission and the American Medical Association to include tympanostomy tubes in 2012 on its list of 5 clinical areas of potential overuse. 13
Current best evidence
The AAO-HNSF CPG offers 6 evidence-based recommendations on indications for tympanostomy tube insertion (Table 2), 4 based on a trustworthy guideline development process that included otolaryngology, primary care, nursing, and allied health.
Current Evidence-Based Indications for Tympanostomy Tube Insertion.4 ,a

Abbreviations: AOM, acute otitis media; CPG, clinical practice guideline; OME, otitis media with effusion.
a Recurrent AOM is at least 3 episodes in the past 6 months or 4 in the past 12 months (with at least 1 in the past 6 months).
Expert insight and opinion
The validity of the statements (Table 2) is bolstered by the multidisciplinary nature of the guideline development group combined with trusted methodology and extensive internal, external, and editorial peer review. The guidance, however, is not comprehensive because it addresses major opportunities for quality improvement, not all potential indications for tubes.
Recommendations for action
Clinicians should follow the CPG recommendations, recognizing that tube insertion is nearly always elective with a large role for shared decision-making. Whenever uncertainty exists, it is best to reevaluate the child in a few months rather than coercing families into an “indicated” surgery for which they are uncomfortable. There are indications for tubes, however, not shown in Table 2, for which more urgent tube insertion would be appropriate, which may include tympanic membrane retraction, retraction pocket with keratin debris (precholesteatoma), or suppurative complications of AOM (eg, acute facial paralysis). Inserting tubes for retraction-type ear disease may require adjunctive procedures (eg, cartilage graft tympanoplasty) that are beyond the scope of this article.
Why Does the AAO-HNSF CPG on Tympanostomy Tubes State That Tube Insertion Is Not Recommended for Children With Recurrent AOM Who Do Not Have Effusion in One or Both Ears at the Time of Assessment for Tube Candidacy?
Background and context
Tympanostomy tubes have traditionally been recommended for recurrent AOM, including by the American Academy of Pediatrics (AAP) in the AOM CPG. 8 The AAO-HNS tympanostomy tube CPG adds nuance to this recommendation by suggesting that only children with recurrent AOM and persistent MEE in one or both ears are candidates for tube insertion.
Current best evidence
The natural history of recurrent AOM without persistent effusion is favorable, with more than 40% of children in RCT control groups having no additional AOM episodes in the next 6 months and only about 15% continuing to have recurrent AOM. 14 In addition, the only RCT of tubes for recurrent AOM that specifically excluded children with baseline MEE found no benefit of tube insertion for reducing subsequent AOM incidence. 15
Expert insight and opinion
When confronted with a child with recurrent AOM without persistent MEE who was referred explicitly for tube insertion, it can be tempting to offer surgery. The key question, however, relates not to how many AOM episodes the child already had in the past but to whether tube insertion will benefit the future. The existing evidence suggests they will not, which should limit enthusiasm for tubes regardless of external pressure.
Recommendations for action
Clinicians should monitor children with recurrent AOM for future episodes, after which persistent MEE may change the status of tube candidacy. Currently, about 70% to 80% of clinicians adhere to this recommendation, with reasons for nonadherence based primarily on caregiver preference. 16
Is It Ever Appropriate to Recommend Tympanostomy Tubes When OME Is First Diagnosed, Without a Period of Watchful Waiting for 3 Months, for Children Who Are At Risk for Speech, Language, or Learning Problems From Otitis Media?
Background and context
The concept of the “at-risk child with OME” (Table 3) was first introduced in the AAP 2014 CPG on OME and reaffirmed in an update and in the AAO-HNSF tympanostomy tube CPG. 4,7 This highlights a subgroup of children with OME at increased risk for developmental difficulties (delay or disorder) because of comorbid, baseline conditions. Despite a recommendation for tubes in at-risk children with OME unlikely to resolve quickly (Table 3, recommendation #6), some clinicians hesitate to insert tubes without 3 months, or longer, of watchful waiting.
The “At-Risk Child” With OME: Risk Factors for Developmental Difficulties.4
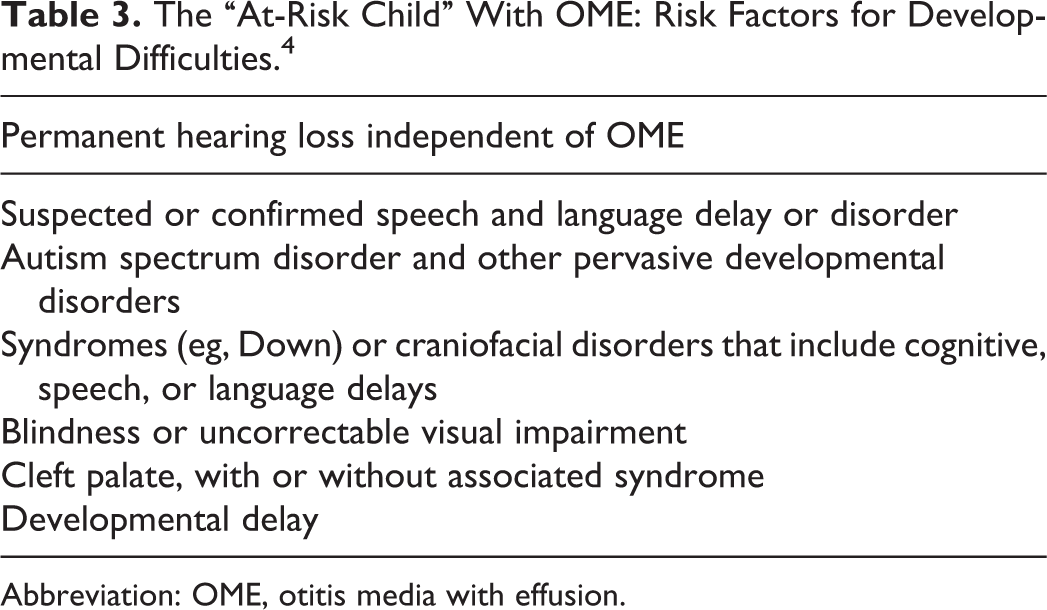
Abbreviation: OME, otitis media with effusion.
Current best evidence
Based on systematic review of cohort studies and control groups in RCTs, there is good evidence that OME is unlikely to resolve quickly (20%-30% in 6 months) when present for at least 3 months or of any duration with a type B (flat curve) tympanogram. 14 Evidence is limited, however, to show a beneficial impact of tube insertion for OME in the at-risk population, 17 primarily because at-risk children are excluded from nearly all RCTs of tube efficacy.
Expert insight and opinion
In the author’s opinion, tympanostomy tubes are underused for at-risk children because OME is often asymptomatic, an uncooperative child can be difficult to examine, and the narrow ear canal in children with Down and other syndromes or craniofacial disorders impairs visibility of the tympanic membrane. Even when OME is detected, clinicians may hesitate to recommend tubes without a period of watchful waiting, which can adversely impact children by delaying intervention with the likelihood of low spontaneous resolution.
Recommendations for action
Clinicians should offer tympanostomy tubes to at-risk children with chronic OME or a flat tympanogram, even when OME is first diagnosed. There is a role for shared decision-making with caregivers, but the role is proportionately reduced as the level of hearing impairment from the effusion increases.
What Is the Role of New, Automated Tympanostomy Tube Insertion Devices in Managing Otitis Media?
Background and context
Several device manufactures have developed automated tympanostomy tube insertion devices to facilitate rapid tube insertion, potentially without general anesthesia. Current devices include the Hummingbird TTS (Preceptis Medical), 18 Tula System (Tusker Medical), 19 and the Solo TTD (AventaMed). 20
Current best evidence
There is limited information (March 2020) to support the safety and efficacy of these devices, in general, beyond industry-funded studies with modest sample sizes, often conducted and published by authors receiving financial benefits from the device manufacturer. The Hummingbird TTS has been studied in 128 children under conscious sedation, 21 the Tula System in 292 children combined with iontophoresis in the office setting, 22,23 and the Solo TTD is only approved (March 2020) for clinical use in Europe.
Expert insight and opinion
The primary limiting factor for using these devices is the proprietary nature of the tympanostomy tubes that are preloaded. Little to no data are available on the median intubation period or incidence of otorrhea (postoperative, sporadic, or chronic), granuloma, medialization, early extrusion, plugging, or persistent perforation. Moreover, the devices are expensive without clear ways to recover the cost in a hospital- or office-based setting. Accidental discharge of a tube can further increase cost because it may be difficult, or impossible, to reload the tube into the delivery system.
Recommendations for action
Clinicians should remain alert to new publications on automated tube insertion devices, paying attention to populations studied, rates of successful intubation, complications, and tube performance characteristics. Individuals looking for ways to more rapidly insert tubes may find these devices of benefit, assuming they can recover the added purchase cost involved.
Is In-Office Insertion of Tympanostomy Tubes, in Awake Children Without General Anesthesia, Feasible and Appropriate for Managing Otitis Media?
Background and context
Interest in office-based alternatives to general anesthesia for tube insertion began in 2011 when the Food and Drug Administration (FDA) assessed a growing body of evidence, from animal research and observational studies, suggesting that anesthetic agents may harm developing brains with subsequent behavioral and cognitive deficits. 24 This was followed by an FDA safety communication in 2016 for a label change (safety warning) on inhalational anesthetics, plus midazolam and propofol, because the “exposure to these medicines for lengthy periods of time or over multiple surgeries or procedures may negatively affect brain development in children younger than 3 years.” 25 Given the high incidence of tube insertion in this age-group, plus the not infrequent need for tube replacement (or other surgery, such as adenoidectomy, tonsillectomy, or hernia repair), a desire to avoid general anesthesia is readily understood.
Current best evidence
Although the evidence regarding the neurocognitive impact of anesthesia in young children continues to evolve, 26 evidence regarding in-office tube insertion led the AAO-HNSF to issue a position statement in 2019 (Table 4) in response to an FDA-initiated request. 27 The decision to endorse in-office tube placement in appropriately selected patients was based, in part, on observational studies showing high rates of success, comparable outcomes with operating room insertion, and low rates of adverse events. 21,22,28 -30
AAO-HNS Position Statement on In-Office Placement of Tubes in Awake Children.26

Abbreviation: AAO-HNS, American Academy of Otolaryngology–Head and Neck Surgery.
Expert insight and opinion
The author has regularly performed in-office tube insertion in awake children since 2010, 27,28 using a papoose board for protective stabilization, premedication with acetaminophen for postprocedure pain relief, 31 and topical phenol for local anesthesia (children age 2 years or older). At the time of this writing (March 2020), I have done this in over 500 children ranging from age 3 months to 9 years, typically using a beveled fluoroplastic tube or occasionally a t-tube (narrow canal) with over 98% success, mean procedure time of 4 to 5 minutes (both ears), high caregiver satisfaction, and no significant complications.
Recommendations for action
Whether to undertake in-office tube insertion in awake children should be based on clinician experience, clinician ability to interact and reassure caregivers (who will be present and observing), caregiver preference, and judgment regarding the level of cooperation (or lack thereof) to be expected from a given child. The existing literature is too sparse to generalize conclusions about safety or efficacy. It is also too early to judge the potential impact, if any, of new devices for rapid, automated tube insertion (see preceding issue) on clinician willingness to embrace this approach.
Does Acute Tympanostomy Tube Otorrhea (TTO) Caused by Methicillin-Resistant Staphylococcus aureus Require Oral Antibiotic Therapy When Culture Shows Resistance to Fluoroquinolones?
Background and context
Acute TTO, also called an “ear infection with a tube,” is a common sequelae of tube insertion with a mean incidence of 26% and a range in observational studies of 4% to 68%. 32 Although most episodes respond promptly to topical fluoroquinolone eardrops, 4 some clinicians may culture the drainage and encounter methicillin-resistant Staphylococcus aureus (MRSA) that is deemed “resistant” to fluoroquinolones based on minimum inhibitory concentrations (MICs). This often results in oral antibiotics, most often trimethoprim–sulfamethoxazole or clindamycin.
Current best evidence
Resistance cut points based on MICs are irrelevant for topical therapy, which achieves a local concentration up to several thousand times higher than that achieved in serum after oral administration. 33 Fluoroquinolone otic drops (0.3%) have 3000 µg/mL concentration, which exceeds the MIC for all MRSA isolates but is reduced in the middle ear because not all solution passes through the tube. For most isolates, however, the concentration achieved should be more than sufficient to control nearly all MRSA otorrhea. 34
Expert insight and opinion
Acute TTO should routinely be treated with topical quinolone eardrops and culture is rarely indicated or useful. Quinolone drops have concentration-dependent bactericidal activity, which enhances efficacy when the local concentration exceeds the MIC.
Recommendations for action
When “resistant” MRSA is encountered on a culture, topical therapy should be continued and oral antibiotics reserved for treatment failures. Caregivers should also be instructed on proper administration of ototopical drops to maximize entry into the ear canal, tube, and middle ear space. Aural toilet before administering drops is not essential for efficacy, 35 but when otorrhea is visibly obstructing the ear canal, it can be cleaned most effectively using “tissue spears,” created by tightly twisting the corner of a facial tissue into a point (spear), tearing off the distal 1 in, and then inserting the torn edge into the canal to absorb drainage. 36 This is repeated until the canal is clear, then drops are administered.
What Is the Best Way to Manage a Child With Recurrent or Persistent TTO?
Background and context
Recurrent acute TTO and persistent (chronic) TTO affect 7.4% and 3.8% of children with tubes, respectively, but can be extremely frustrating for the child, family, and clinician.
Current best evidence
During an episode of acute TTO managed with topical antibiotic drops, about 50% of children recover within 3 to 4 days, 90% within 7 days, and a plateau of 95% after 14 days. 34 When otorrhea persists for 2 weeks or longer with a nonintact tympanic membrane (tube or perforation), additional topical antibiotics, with or without concurrent oral antibiotic, appear effective, but the quality and consistency of supporting evidence is low. 37
Expert insight and opinion
Effective management of recurrent or persistent TTO begins with identifying the likely cause, based on the laterality and temporal pattern (Table 5). 2 Unilateral TTO is more likely to involve local, ear-specific, factors, and bilateral TTO is more likely to reflect a systemic cause. Dry period(s), even if only last a few days, imply reversible disease with a recurring (but time limited) insult, suggesting a good prognosis. In contrast, lack of a dry period (even if brief) suggests a chronic underlying condition and potential need for intervention.
Causes of Recurrent and Persistent Tympanostomy Tube Otorrhea.
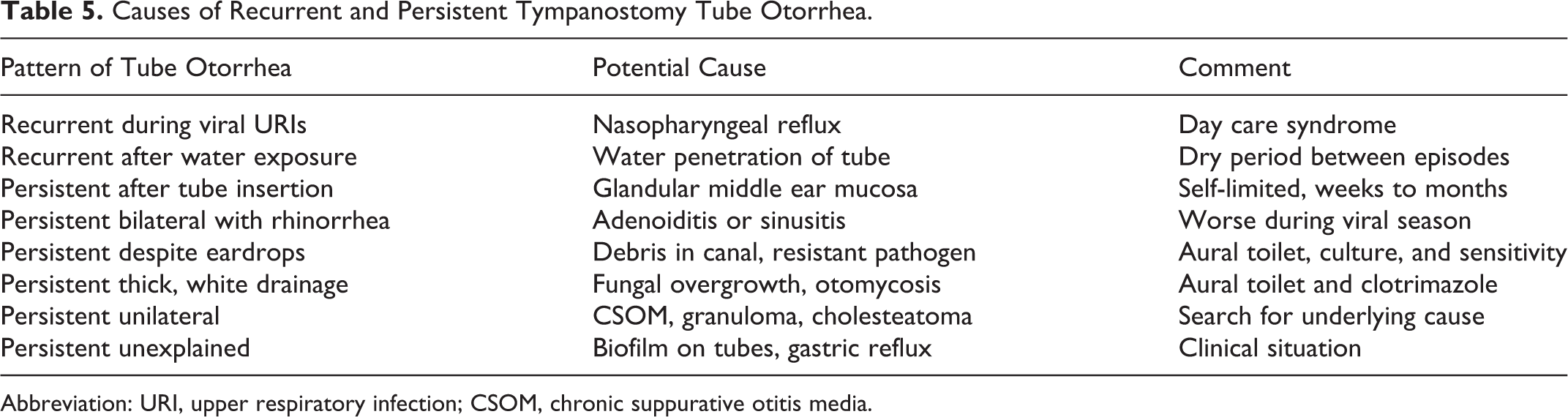
Abbreviation: URI, upper respiratory infection; CSOM, chronic suppurative otitis media.
Recommendations for action
Clinicians should strive to identify the underlying cause for recurrent or persistent TTO using the information in Table 5, recognizing that the cause may not be found with certainty. A particularly frustrating situation for caregivers is frequent and repeated TTO corresponding with upper respiratory infections (row 1 in Table 5), which may be impossible to prevent because of a short, floppy, and patulous Eustachian tube that facilitates reflux of nasopharyngeal secretions into the middle ear and out the tube. Explaining to caregivers that this is the “runny ear” equivalent of a “runny nose” can relieve some concern.
Conclusions
Tympanostomy tubes are a safe, effective, and time-tested intervention that can improve quality of life and reduce the burden of MEE and recurrent AOM in appropriately selected children. 4 Tubes can also help optimize the listening and learning environment for children with developmental delays, but in the author’s experience, tube insertion is often not recommended in a timely fashion for at-risk children with persistent OME (Table 3). Clinicians should follow evidence-based guidelines for tube insertion (Table 2), although the procedure is nearly always elective and there is substantial opportunity for shared decision-making with caregivers.
Despite a substantial, and constantly growing, volume of high-level evidence on managing children with tympanostomy tubes, there will always be gaps, uncertainties, and controversies that benefit from clinician experience and expert opinion. In that regard, the issues discussed in this review article (Table 1) will hopefully aid clinicians in everyday, pragmatic decisions. With regard to the future, I anticipate that in-office tube insertion will become more popular because of caregiver demand, regardless of the final verdict regarding the impact of general anesthesia on child brain development. The role of automated tube insertion is uncertain because of difficulties in recovering the expense of a single-use device and current uncertainties about properties of the proprietary tubes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
