Abstract
Background:
Evaluate the impact of patent tympanostomy tubes (TT) on otoacoustic emissions (OAE) in patients with normal pure tone audiometry (PTA).
Methods:
Retrospective review of patients who underwent TT placement with paired postoperative audiogram and OAEs at 1, 2, 4, and 8 kHz were included. Frequencies with PTA >20 dB or nonpatent TTs were excluded. OAEs were graded by signal-to-noise ratio and distortion product as “present,” “reduced,” or “absent.”
Results:
In total, 124 patients (217 ears) were included with a mean age (SD) of 3.9 (3.2) years and OAE testing occurring 4.5 (4.7) months postoperatively. OAE absence rates were 85% at 1 kHz (n = 165), 57% at 2 kHz (n = 203), 56% at 4 kHz (n = 206), and 83% at 8 kHz (n = 135).
Conclusion:
Absent OAEs were the most common result at all frequencies, especially 1 and 8 kHz. Overall, 78.8% of patients failed at least one tested frequency, despite normal PTA and patent TTs, suggesting a potential direct impact of TTs on OAE outcomes. Clinicians may need to exercise caution when interpreting OAEs in patients with patent TTs, and further research may warrant a reevaluation of OAE utility in this context.
Keywords
Introduction
The outer hair cells of the cochlea generate otoacoustic emissions (OAEs) in response to auditory stimulation, providing an objective assessment of cochlear function. 1 OAEs are widely used for hearing screening, particularly in pediatric populations, due to their noninvasive nature and sensitivity. 2 Among the several types of OAEs, transient evoked OAEs (TEOAEs) are often preferred for initial screenings due to their ability to assess a broad frequency range of the cochlea. 3 Distortion product otoacoustic emissions (DPOAEs) are especially valuable in evaluating higher frequencies, such as in pediatric patients, due to their frequency-specificity 3 ; however, their diagnostic reliability may be influenced by middle ear conditions. 4
Otitis media (OM) is a common cause of transient hearing loss in pediatric populations. OM complicated by middle ear fluid is further classified into otitis media with effusion (OME) or acute otitis media (AOM), with AOM being associated with signs and symptoms of infection.5,6 OME is the most common cause of acquired pediatric hearing impairment and is considered chronic OME (cOME) if effusions persist for over 3 months.7-9 Recurrent AOM (RAOM) is defined as either 3 or more episodes of AOM within a 6 month period or 4 or more episodes within a year. 5 Treatment for cOME or RAOM may include medical treatment (ie, antibiotics) or surgical placement of tympanostomy tubes (TT), which is the most-frequently-performed pediatric surgical procedure in the United States.9-11 Placement of TTs ventilates and improves middle ear function; however, their presence alters the anatomy and acoustics of the middle ear, and may influence the reliability and outcomes of OAEs. 12
Prior studies evaluated the feasibility of OAEs in pediatric patients with varied methodology and inconsistent findings.4,12-15 The current literature mostly suggests that the presence of TTs does not significantly impact diagnostic reliability and outcomes of OAEs, and a negative impact of TTs on OAE outcomes is scarcely reported. 16 This lack of consensus may arise from differences in subject inclusion criteria, such as tube patency assessment, the threshold for OAE absence, or the presence of residual underlying middle ear dysfunction.
Herein, we aimed to address a gap in the knowledge and build upon previous research by strictly controlling for normal audiometric thresholds and tube patency to assess whether the presence of TTs influences outcomes of OAE testing in a pediatric population. This study aimed to isolate the direct effect of TTs on OAE outcomes by employing rigorous inclusion criteria based on tympanometry and audiogram data.
Methods
This retrospective chart review was performed in a single-specialty private practice on pediatric patients seen by 4 fellowship-trained pediatric otolaryngologists in Southeast Florida. Institutional review board exemption was granted (IRB # 2021-171). Electronic Health Records (EHR) were queried for Current Procedural Terminology codes for TT placement between October 2018 and March 2021.
This initial cohort was narrowed to include patients who underwent unilateral or bilateral TT placement and had postoperative pure tone audiometry (PTA) and OAE data available for review in the EHR. Further inclusion criteria required OAE data to be limited to ears with patent TTs and normal hearing thresholds at corresponding PTA frequencies. On PTA, normal hearing was defined as an air conduction threshold ≤20 dB, based on the calibration of the audiometer. TT patency was determined via tympanometry, either by large ear canal volume or inability to form a seal. OAE frequency data were excluded if it corresponded to missing or abnormal audiometric thresholds or if tympanometry indicated a nonpatent tube (types A, B, C, or small canal volume). In addition to excluding abnormal PTA results, we also excluded tests deemed unreliable, as their results could not be confidently interpreted. Care was taken by the audiologist to assess the reliability of PTA responses, including having patients repeat responses at each frequency, and patients with unreliable testing were excluded.
In this study, DPOAEs were performed exclusively. The possible OAE frequencies that correspond to audiograms were 1, 2, 4, and 8 kHz. Results of OAE testing were categorized based on signal-to-noise ratio (SNR) and distortion product (DP) as “present” (SNR ≥6 + DP ≥0), “reduced” (SNR ≥6 + DP <0), or “absent” (SNR <6 dB + any DP value). 17
Statistical analysis included chi-squared analysis for nonparametric variables and simple logistic regression for parametric variables, in the GraphPad Prism 10.0 software (La Jolla, CA, USA). The significance level was set at .05.
Results
A total of 124 patients, consisting of data from 217 ears (OAEs), met inclusion criteria. The cohort had a mean (SD) age of 3.9 years (3.2), ranging from 0.9 to 16.7 years, and were predominantly male (61.3%). Indications for TT placement were relatively balanced between RAOM (53.5%) and cOME (46.5%). The mean interval from TT placement to OAE testing was 4.5 months (4.7). Demographics and other study characteristics are outlined in Table 1.
Demographic Variables and Clinical Characteristics of Participants.
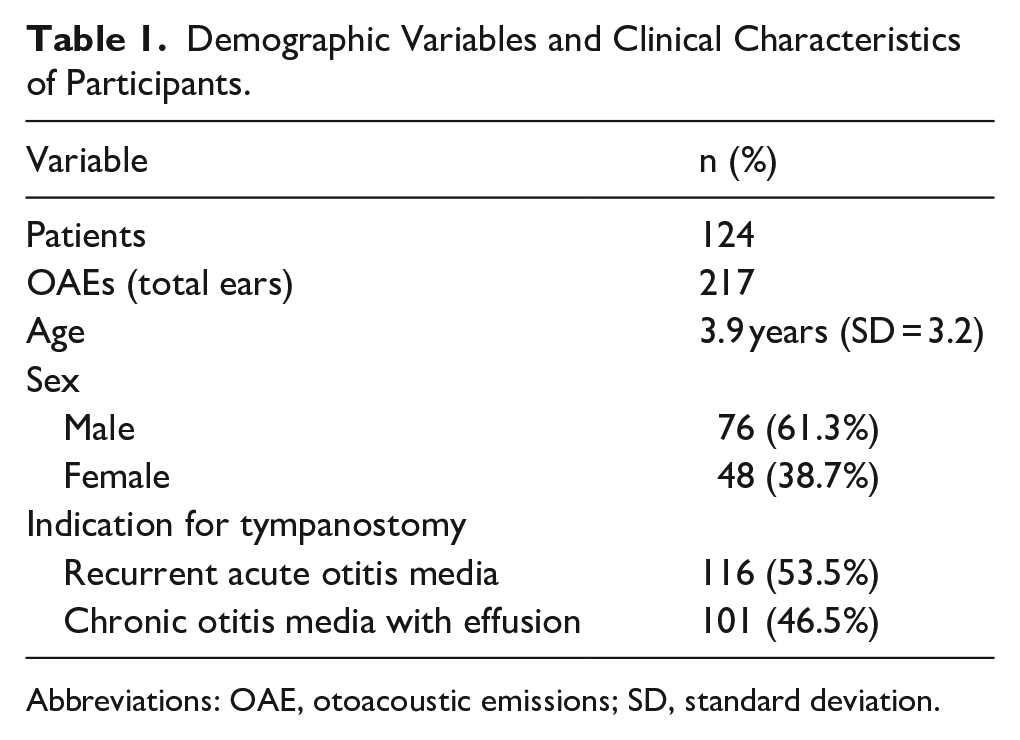
Abbreviations: OAE, otoacoustic emissions; SD, standard deviation.
Table 2 describes the prevalence of “absent” OAE responses per ear tested, in the presence of normal PTA data across the 4 frequencies considered. Overall, 21.2% of patients had zero “absent” OAE responses, 17.1% had 1 “absent” response, 12.0% had 2 “absent” responses, 18.0% had 3 “absent” responses, and 31.8% of patients had 4 “absent” responses.
Number of Frequencies With “Absent” OAE Responses Despite Normal PTA Hearing.

Abbreviations: No., number; OAE, otoacoustic emissions; PTA, pure tone audiometry.
At the individual frequency level, “absent” OAEs were most common at the lowest (1 kHz) and highest (8 kHz) frequencies. At 1 kHz (n = 165), 85% of OAEs were absent, 2% were reduced, and 13% were present. At 2 kHz (n = 203), 57% were absent, 2% were reduced, and 41% were present. At 4 kHz (n = 206), 56% were absent, 1% were reduced, and 43% were present. At 8 kHz OAEs (n = 135), 83% were absent, 10% were reduced, and 7% were present. The relative distribution of responses per frequency is displayed in Figure 1.
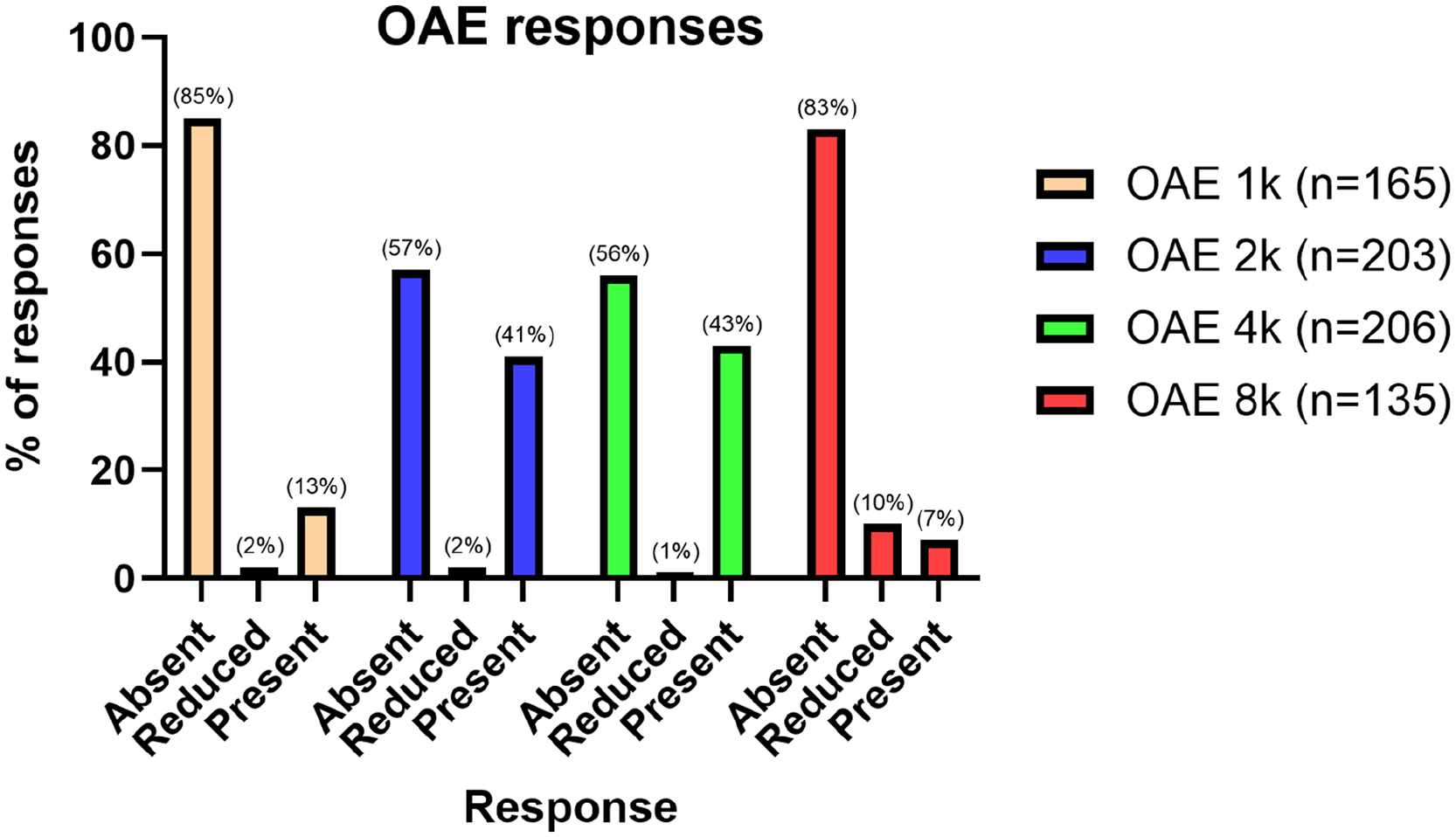
Otoacoustic emission responses across tested frequencies. Responses are expressed in relative percentage per frequency tested.
OAE results were analyzed on a per-patient basis to assess the degree of concordance between ears. Among patients with bilateral TTs and OAE data (n = 92), overall concordance across all tested frequencies was 81.4%. The percentage of consistent bilateral responses per frequency was as follows: 1 kHz (86.4%), 2 kHz (75.0%), 4 kHz (82.1%), and 8 kHz (84.2%).
Subgroup analyses were also performed to evaluate other factors that may influence OAE outcomes. The chi-squared analysis revealed no significant differences in the presence or absence of OAEs based on TT placement indication (Figure 2) or gender. Similarly, simple logistic regression did not demonstrate any significant relationship between age and OAE response.
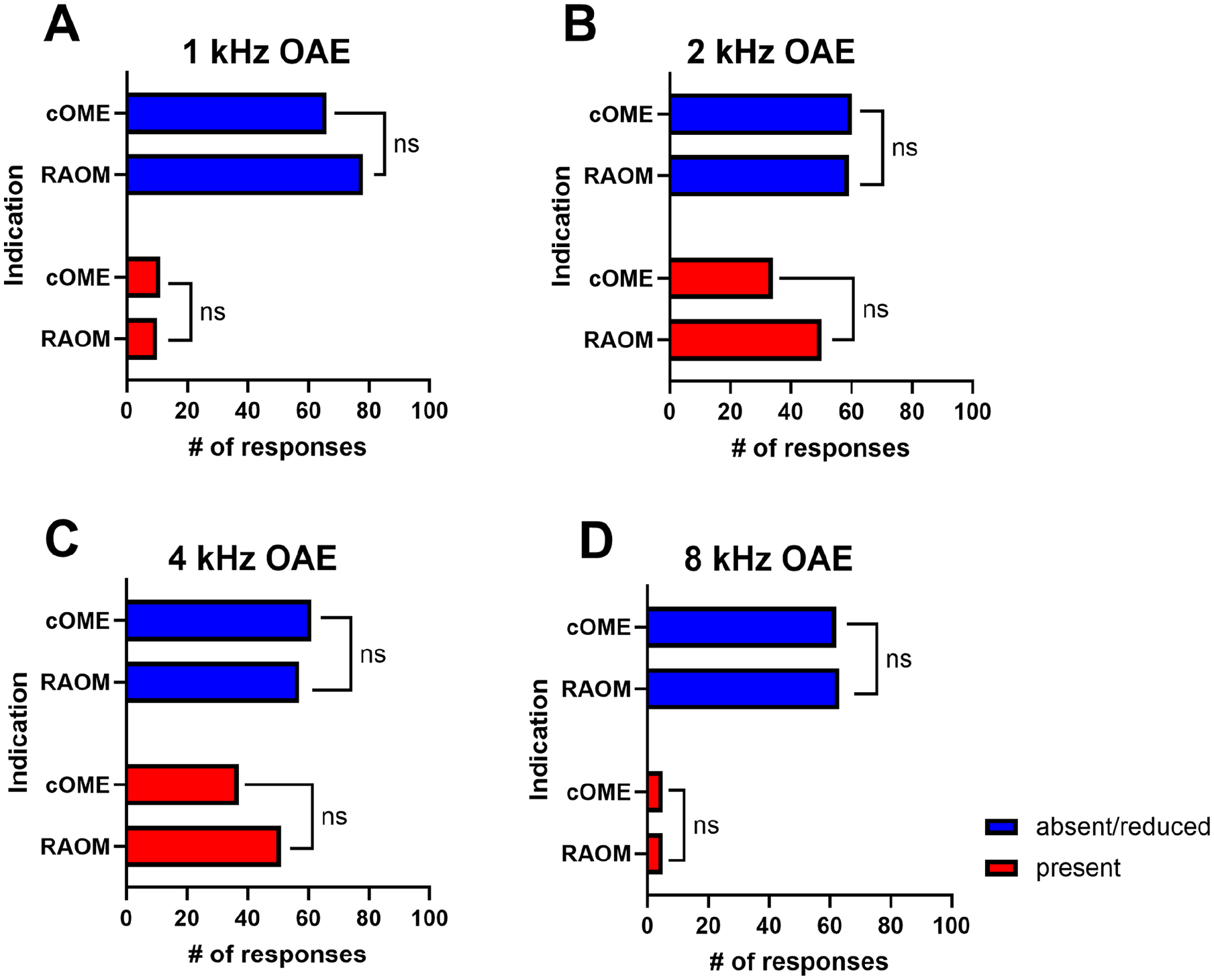
Otoacoustic emission responses stratified by tympanostomy tube indication, per frequency tested. cOME, chronic otitis media with effusion; RAOM, recurrent acute otitis media; OAE, otoacoustic emissions; ns, nonsignificant.
Discussion
OAE testing relies on middle ear function, and its interpretation can be complicated by middle ear disease, as conditions like OM and effusion are known to reduce OAE responses.18,19 However, the impact of TTs on OAEs, upon the resolution of middle ear dysfunction, has been insufficiently explored. Considering the sensitivity of OAE testing, “present” OAE responses are expected to coincide with normal PTA at equivocal frequencies in most cases. Therefore, we aimed to isolate the potential direct effects of patent TTs on individual OAE frequency responses by strictly controlling for normal PTA thresholds and TT patency. In this study, 78.8% of patients demonstrated at least one absent OAE response across the 4 tested frequencies. While this was particularly true at frequency extremes, there was still >50% incidence of “absent” OAE responses at 2 and 4 kHz. No significant relationship was found between OAE response and diagnostic indication, age, or gender.
Our findings appear to contradict with most prior pediatric studies suggesting the feasibility and accuracy of OAE testing in pediatric patients with TTs.4,12,14,15 Fritsch et al reported a high rate (81%) of “passing” TEOAEs with normal hearing and patent TTs, using a methodologic design that resembles the present study, although the use of TEOAEs may contribute to the observed discrepancies in the findings between our studies. 4 Similarly, Topolska and Baczek found “valid” DPOAEs in almost all ears at 1 month postoperatively compared to a control group, though decreased response amplitudes suggested a potential impact of the TT. 14 Notably, their distribution of OAE responses echoed our findings, with absent responses being most common at 1 and 8 kHz, albeit with less failures than our cohort. Saleem et al reported 100% present TEOAEs in patients with normal postoperative PTA, and Patel et al observed significantly-greater rates of present OAE responses postoperatively than preoperatively, but did not detail the OAE type or response grading criteria.12,15 Investigations by Charlier and Debruyne 20 and Gordts et al 21 also concluded that OAE testing was overall feasible and accurate among patients with TTs.
These studies emphasize the potential utility of OAEs in patients with TTs, but inconsistencies in methodologies and stated criteria complicate direct comparisons with our results. Regardless, our findings suggest that TTs may directly influence OAE responses, particularly at frequency extremes. Consequently, clinicians may need to cautiously interpret OAEs in pediatric patients with patent TTs, as absent responses may reflect a TT-related effect as opposed to cochlear dysfunction. Accordingly, conventional audiometry may be necessary in these cases; however, further studies are needed to clarify this notion.
When considering the findings of this study being particularly pronounced at frequency extremes (1 and 8 kHz), there are multiple theories as to why this may be the case. Low frequencies are known to be affected in particular by middle ear effusion, which could be the case if any of our patient cohort had residual middle ear fluid, although it would not have been enough to cause a conductive hearing loss on their audiogram.22,23 Tympanic membrane stiffness and mass, both of which could be theoretically increased by the presence of a TT, may reduce OAE responses at both low and high frequencies, respectively. 24 Another consideration is whether our findings would be reproducible using TEOAEs with the same inclusion criteria. Given that prior studies have employed both DPOAEs and TEOAEs with generally-consistent results, we would not expect to see significant differences.
Beyond inherent TT effects, other considerations may influence OAE responses in our study cohort. Norowitz et al. retrospectively found that the number of historical AOM episodes correlated with higher rates of failed OAE screenings later in life, suggesting potential long-term effects of AOM on OAE responses. 25 Similarly, in a case-control study, Sanfins et al. reported absent and diminished OAE amplitudes in children with a history of RAOM and TTs compared with those in controls, despite not finding a difference in audiometric thresholds between groups. 26 These findings introduce the possibility of altered OAE responses in our study cohort due to permanent changes related to a history of RAOM or cOME, as opposed to direct effects of the TT. It is unclear whether this corresponds to clinical hearing loss, but potential mechanisms could be related to pathogen-specific effects like eustachian tube ciliary damage secondary to negative middle ear pressure or general inflammation associated with OM directly transmitted to the cochlea.27,28 If the inflammatory response is similar in both ears, this notion would be consistent with the 81.4% overall agreement observed between ears in our cohort of patients with bilateral TTs.
Another factor that could affect OAE results, independent of the tube, is the previously-reported age-related decline in OAE responses.29,30 However, we examined the potential impact of age on OAE outcomes in our patient cohort, and our logistic regression analysis found no significant associations at any OAE frequency.
Given the findings of the present study, alternatives to OAE testing in patients with TTs may be considered. Chiefly, particularly in newborns and young children, automated auditory brainstem response (ABR) may be considered. Although ABR generally takes longer to perform than OAEs and may require sedation in some cases, it can provide more robust results than OAEs. Previous investigations have reported a negative impact of TTs on intraoperative ABR responses immediately after tube placement, partially mediated by the effusion type (worse with mucoid).31,32 Huang et al compared ABR responses pre-TT placement to 1 day postoperatively, in comparison with healthy controls, and found that ABR responses improved after TT placement but did not reach the normal levels of the control group. 33
The strengths of our study include strict inclusion criteria and a relatively-large sample size compared with prior studies, although our study is not without limitations. A retrospective study design introduces a sampling bias toward patients with complete audiometric and OAE data in the EHR. Additionally, effusion type was not accounted for, and frequency analysis was restricted to the available overlapping frequencies between PTA and OAE (ie, 1, 2, 4, and 8 kHz), despite there being a wider range of available testable frequencies on OAE’s. Potentially, including additional or more finite OAE frequencies may reveal different patterns. Previously-investigated factors such as a history of AOM or cOME can also negatively impact OAE responses, which may be a confounder in our evaluation of the effects of a TT. Future investigations would benefit from an increased sample size, prospective design, a comparison of DPOAE versus TEOAE findings, and a wider frequency range to better characterize the impact of TTs on OAEs.
Conclusion
Absent OAEs were the most common response at all frequencies, especially 1 and 8 kHz. Overall, 78.8% of patients failed at least one tested frequency, despite normal PTA thresholds. These findings suggest that TTs may directly impact OAE responses, potentially limiting their reliability as a measure of cochlear function in this population. Clinicians may need to exercise caution when interpreting OAE findings in patients with patent TTs, as results may reflect the presence of the tube itself. Further studies are needed to explore this notion and potentially inform a reassessment of the utility of OAEs as a diagnostic tool in patients with TT.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon request.
