Abstract
Objective:
Nearly half of children who undergo tympanostomy tube (TT) insertion may experience otorrhea following surgery. We sought to review the evidence for the role of bacterial biofilms in post-tympanostomy tube otorrhea (PTTO) and the accumulated experience regarding the preventive measures for biofilm formation/adhesion on TTs.
Methods:
English literature search for relevant MeSH keywords was conducted in the following databases: MEDLINE (via PubMed), Ovid Medline, Google Scholar, and Clinical Evidence (BMJ Publishing) between January 1, 1995, and December 31, 2019. Subsequently, articles were reviewed and included if biofilm was evident in PTTO.
Results:
There is an increased evidence supporting the role of biofilms in PTTO. Studies on TT design and material suggest that nitinol and/or silicone TTs had a lower risk for PTTO and that biofilms appeared in specific areas, such as the perpendicular junction of the T-tubes and the round rims of the Paparella-type tubes. Biofilm-component DNAB-II protein family was present in half of children with PTTO, and targeting this protein may lead to biofilm collapse and serve as a potential strategy for PTTO treatment. Novel approaches for the prevention of biofilm-associated PTTO include changing the inherent tube composition; tube coating with antibiotics, polymers, plant extracts, or other biofilm-resistant materials; impregnation with antimicrobial compounds; and surface alterations by ion-bombardment or surface ionization, which are still under laboratory investigation.
Conclusions:
Currently, there is no type of TT on which bacteria will not adhere. The challenges of treating PTTO indicate the need for further research in optimization of TT design, composition, and coating.
Introduction
Treatment of otitis media (OM) consists of systemic antibiotics in cases of acute otitis media (AOM) episodes in certain scenarios, 1 and for those presenting with recurrent AOM episodes and/or prolonged otitis media with effusion (OME), which is associated with hearing loss and/or speech and learning difficulties, surgical placement of tympanostomy tubes (TTs) is frequently performed. 2 Other nonsurgical options, such as steroids, decongestants, antihistamines, or other treatments were shown to be ineffective. 3 In a study analyzing US multihospital network claims database between 2004 and 2010, it was reported that 19% of children aged 0 to 3 required at least 1 set of TTs, and 4.6% required 2 or more TT sets, making TT insertion one of the most common surgical procedures in children. 4 Complications associated with TT insertion include tympanic membrane perforation, early or late extrusion, myringosclerosis, and most importantly post-tympanostomy tube otorrhea (PTTO), which is the most common complication. 5,6
The reported incidence of PTTO ranges from 3.4% to 74% as reported in a big meta-analysis, which reflects a big variability in the definition of PTTO and rate calculations of PTTO in the different studies and the calculation of PTTO incidence either per patient or per ear studied. 7 Persistent PTTO is classified as either acute/early, which lasts <8 weeks and occurs at an incidence of 7.4% from all operated cases, or chronic/prolonged, which lasts >8 weeks and occurs at an incidence of 3.8% from all operated cases. 5 The positive culture rate from PTTO cases varies. This variability is explained by the differences in study design and time from beginning of symptoms to time of swabbing and method of culture processing (standard cultures, polymerase chain reaction or DNA pyrosequencing analysis). 8 Studies using conventional microbiological cultures showed that Haemophilus influenzae, Staphylococcus aureus, and Pseudomonas aeruginosa were the most common microorganisms isolated in untreated, uncomplicated PTTO cases. Because S aureus and P aeruginosa are normal inhabitants of external auditory canal, their detection in samples from this site does not imply a pathogenic role. However, when a prosthesis, such as TT is in place, the pathogenicity of these 2 bacterial species is enhanced. A sharp decrease in the prevalence of Streptococcus pneumoniae isolations was evident after the introduction of pneumococcal conjugate vaccines. 9
Treatment of PTTO is either topical, combining antibiotics and steroids or antibiotics only, or with oral antibiotics. 10 In a recent meta-analysis, it was reported that both topical antibiotic–glucocorticoid and antibiotic-only drops were superior to watchful waiting for the treatment of PTTO. The odds of clinical cure were 12 (95% credible interval [CrI], 1.9-82) times higher (number needed to treat [NTT]: 2.2, assuming a reference rate of 0.45) for antibiotic–glucocorticoid drops and 7.3 (95% CrI, 1.2-51) times higher (NNT: 2.5, assuming a reference rate of 0.45) for topical antibiotic drops. 6 At times, treatment failure of PTTO necessitates the removal and/or changing of the offending TT. 4,11 The bacterial world is divided into planktonic and sessile bacteria. 12 In the first case, bacterial cells are free, mobile, in clusters (suspended in the microenvironment), and susceptible to the antibiotics, antibodies, the phagocytosis, and the action of the complement. Planktonic bacteria are the vegetative form isolated in the laboratory and responsible of acute infections. Sessile bacteria grow in forming biofilms embedded in a matrix of self-produced extracellular polymeric substances, adherent to the surfaces (notably prosthesis, such as TT) or cells of the microenvironment, protected from chemical, physical, and cellular aggression. It is probably the form most represented in the nature, in the microbiote, and during subacute and chronic infections. For otitis, it is believed that the main mechanism for AOM is the release from the biofilm of the nasopharyngeal microbiota of bacteria that pass from the sessile stage to the planktonic stage. 13 By contrast, in OME and chronic OM, sessile bacteria and biofilms are almost always found. 14
In this manuscript, we sought to review the current evidence for the presence of biofilms in PTTO and the laboratory methods used both in vitro and in vivo to reduce biofilm load on TTs.
Methods
For this review, we searched for the following keywords: [‘tympanostomy tube’ or ‘ventilating tube’ or ‘grommet’ or ‘pressure equalizer’] AND [‘otorrhea’ or ‘discharge’ or ‘pus’] AND [‘biofilm’] in various electronic databases: MEDLINE (via PubMed), Ovid Medline, Google Scholar, and Clinical Evidence (BMJ Publishing). These databases were searched from January 1, 1995, and December 31, 2019, for English language manuscripts. The authors reviewed the retrieved manuscripts and chose to present the relevant data. We also added some demonstrative figures from our ex vivo biofilm on TT project (not published elsewhere). The theme of this review is narrative, a meta-analysis was not performed due to the heterogeneity of the methodology in those reports and small numbers of patients.
Results
Biofilm
Bacterial biofilms are complex, organized microbial communities, in which bacteria coexist in an extracellular polysaccharide matrix which permits communication and diffusion of nutrients, leading to increased antimicrobial and host defense resistance. 15 It has been demonstrated that bacterial biofilms have been involved in the pathogenesis of AOM, and to a greater extent in OME 16,17 and in long-standing PTTO. 18,19
The complexity and dynamics of biofilms can be observed by biofilm imaging optical technology, including scanning electronic microscope (SEM), fluorescent in situ hybridization, and confocal laser scanning microscopy (CLSM). These techniques are used to visualize 3D structures and assess the existence of biofilms. An illustrative SEM image showing a P aeruginosa biofilm is shown in Figure 1.

Scanning electron microscope image of Pseudomonas aeruginosa biofilms, ×10 000 magnification.
Evidence for Biofilm Involvement
Evidence for biofilms in OM
Following the publications that demonstrated the existence of adherent mucosal biofilms in the middle ear mucosa in experimental H influenzae-induced OM in a chinchilla models, 20 it has been hypothesized that chronic OM in humans is a mucosal biofilm infection as well. The first evidence came from middle ear mucosa biopsy specimens obtained from children during TT insertion for the treatment of OME/recurrent AOM. The CLSM images were obtained from mucosal specimens and were evaluated for biofilm morphology using generic stains and species-specific probes for H influenzae, S pneumoniae, and Moraxella catarrhalis. Mucosal biofilms were visualized in 92% of the specimens. This direct detection of biofilms on middle ear mucosa specimens from children with OME and recurrent OM, but not from mucosal specimens of the control group (children who had cochlear implant surgery), supported the hypothesis that chronic middle ear diseases are biofilm-related. 21 Since then, many studies have reported the existence of biofilm in many OM and chronic adenoiditis cases, especially in children, which is the suggested source for the development of recurrent or chronic middle ear diseases, such as recurrent AOM, persistent OME, and chronic OM. 22
In the molecular level, an essential component of the biofilm structure in OM is the DNABII, a small family of bacterial DNA-binding proteins, which play a critical role extracellularly in the formation and maturation of bacterial biofilms. 23
Bedside detection of biofilms
A new emerging noninvasive technique for bedside middle ear imaging relies on optical coherence tomography (OCT). A portable, handheld OCT system otoscope was used in a study by Monroy et al. 24 Patients with chronic OME just before TT insertion were in vivo imaged before and after myringotomy and TT placement, and the aspirated middle ear effusion (MEE) was ex vivo imaged too. Biofilms were recorded in the MEE before it was suctioned. Subsequent reports from this group using this technology clearly demonstrated the content of the middle ear before surgery in different OM states, and free-floating biofilm colonies were demonstrated several times. 25,26 Figure 2 shows 2 illustrative cases of biofilms as observed with the OCT technology.
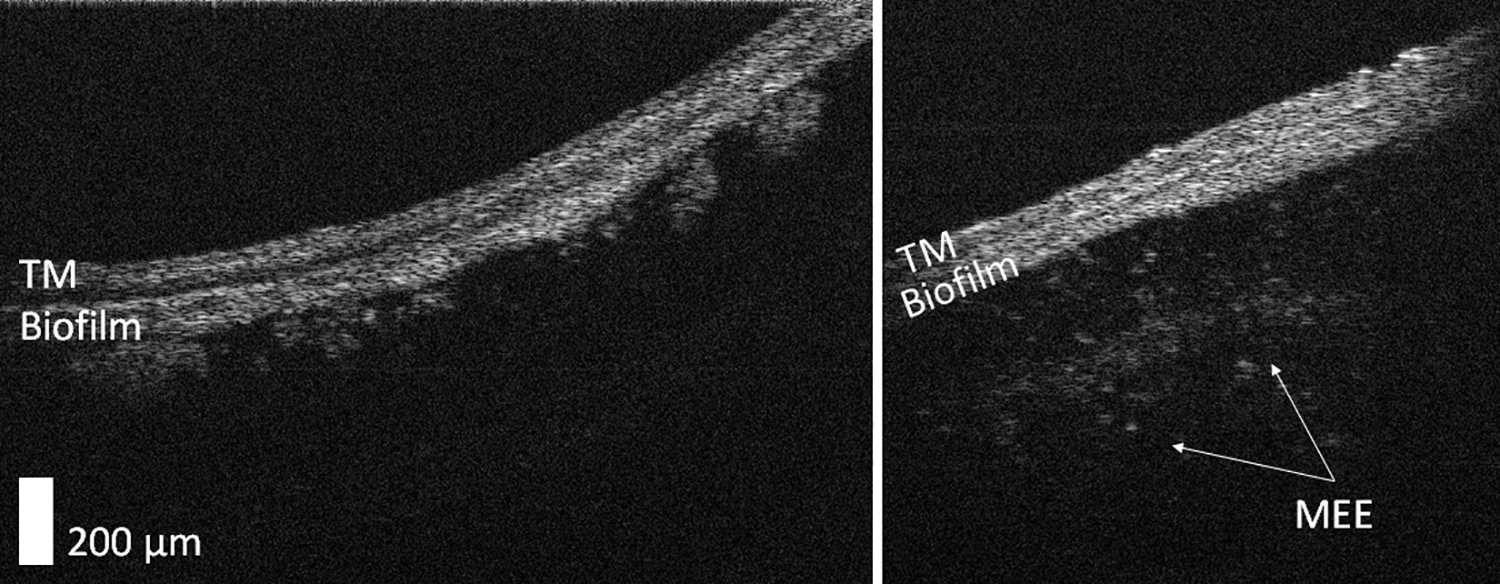
Optical coherent tomography images are from 2 different pediatric patients (6 and 5 years) who were diagnosed with bilateral otitis media with effusion. Both images are on the same scale. Right, Before myringotomy and tympanostomy tube insertion. A more film-like biofilm is present, along with a middle ear effusion containing scattering particles. Left, Before myringotomy and tympanostomy tube insertion. Note the presence of a biofilm that has “mushroom”-like extensions. TM indicates tympanic membrane. Contributed by Jungeun Won and Stephen Boppart, Biophotonics Imaging Laboratory, University of Illinois at Urbana–Champaign, Urbana–Champaign, Illinois.
Biofilms in PTTO
More than 20 years ago, several authors came to the understanding that TTs could be coated with antibiofilm materials. 27,28 Even so, we still find ourselves today without the “perfect solution” for the TT biofilm problem. Biofilm was found on TTs that were removed due to P aeruginosa persistent PTTO resistant to ciprofloxacin otic drops. In all cases, ciprofloxacin-resistant P aeruginosa (CRPA) was the only organism grown. Scanning electronic microscope imaging showed that the surface of the silicone TTs contained undulations or microfissures and that the thick biofilms presented on most TT surfaces were densities with no intervening spaces, consistent with biofilms. 29 Figure 3 shows in an illustrative case of ex vivo image of biofilm on TT removed from a child with persistent PTTO that eventually needed to be removed.

Scanning electron microscopy image showing biofilm colonies in the internal lumen of a tympanostomy tube removed from a child with persistent otorrhea, ×142 magnification.
In a recent study, samples from pediatric patients with PTTO were sent for both microbial culture and biofilm analysis. 30 For biofilm assessment, frozen samples were sectioned and then labeled using a rabbit anti-integration host factor (IHF), a bacterial DNA-binding protein. Of 15 pediatric otorrhea samples analyzed, 9 (60%) contained solids that were positive for labeling of IHF and 75% yielded positive bacterial cultures that included H influenzae, methicillin-resistant S aureus (MRSA), S pneumoniae, M catarrhalis, and P aeruginosa. The positive labeling of otorrhea solids for extracellular DNA and IHF, in combination with microbiological culture results, indicated that biofilms likely played a key role in chronic PTTO. Exposure of bacterial biofilms to antibodies against DNABII proteins destabilized the extracellular matrix and led to the collapse of the biofilm structure in OM animal models. 31 These findings also suggested that DNABII proteins in association with environmental DNA may serve as an important therapeutic target in PTTO. Lack of response to topical/systemic treatment of PTTO treatment has also been suggested to be associated with biofilms. 32
Antibiofilm Agents
Even before it was established that PTTO is in essence a biofilm-mediated disease, multiple authors have tried to find solutions to prevent biofilm formation/adhesion on TTs. 18 These attempts can be divided into several categories: (1) structural/material studies attempting to find the right material and/or the structure which will minimize/inhibit biofilm adherence on TT, (2) TT surface alterations studies, and (3) studies examining coating materials aimed to prevent or reduce biofilm adhesion on TTs.
Structural/material modifications
In 2016, Ovnat Tamir et al showed that the main distribution of experimental P aeruginosa biofilms on TTs was mainly located in the perpendicular junction between the body and the flanges as well as around rims of the TTs. 33 These zones, termed “prone zones,” were considered as potential future target areas for changes in the design or antibiofilm coating materials which may prevent PTTO. Figure 4 shows the distribution of biofilms on the right-angle bifurcations of an Armstrong-type TT, which was in vitro inoculated with an experimental biofilm-forming P aeruginosa.

Scanning electron microscope images of Pseudomonas aeruginosa biofilms on an Armstrong ventilating tube (T-tube). A, ×39 magnification of the flanges bifurcation area. B, ×100 magnification of the white inset in (A), showing numerous biofilms in the neck–flange junction and the main lumen of the tube.
In 2018, Knutsson et al published follow-up data of their 2016 randomized control trial of TTs composed of different materials and shapes: silicone-long, silicone-short, fluoroplastic-long, fluoroplastic-short. 34 They randomized 378 children per ear, so that most children had received different types of TT in each ear. Time to first PTTO episode was significantly longer for silicone tubes; however, the hazard ratio did not change when adjusted for tube shape.
In an in vitro study, Joe and Seo used a Nitinol (nickel–titanium) TTs that were smooth with minimized surface area, which were coated with titanium oxide (TiO2) that formed an oxide layer on the metallic target. 35 Titanium oxide exhibited antibacterial activity and inhibited biofilm formation via a mechanism involving the production of reactive oxygen species. Such TTs, which combined TiO2 coating and minimized surface area of the Nitinol stent, inhibited P aeruginosa biofilm formation. These studies may indicate that both material and geometry play a crucial role in PTTO prevention.
Surface modifications
Multiple experiments have been performed on TT surface alterations, in order to prevent biofilm adhesion. The 2 leading methods are (1) surface ionization, thought to increase the hydrophilicity of the TT surface, resulting in a decrease in bacterial biofilm adherence, and (2) ion bombardment, thought to smooth the surface of the TT, resulting in a less rugged surface for biofilm adhesion. In 1998, Biedlingmaier et al studied 4 different kinds of TTs: (1) Armstrong-style silicone, (2) fluoroplastic, (3) ionized treated, modified silicone tube, and (4) silver oxide–coated silicone Armstrong tube. 36 These tubes were immersed in vitro in different broths enriched with biofilm-producing bacteria, including P aeruginosa, S aureus, and Staphylococcus epidermis. Only the ionized treated silicone TTs remained free of apparent bacterial contamination. The authors demonstrated that the fluoroplastic tube appeared quite resistant to biofilm formation. In 1999, Saidi et al studied 5 different kinds of TTs, some were coated/surface altered and some were not. 27 The different TT types were (1) silicone, (2) silver oxide–impregnated silicone, (3) fluoroplastic, (4) silver oxide–impregnated, and (5) ion-bombarded silicone. They demonstrated that in their in vivo model, based on the inoculation of the middle ear of guinea pigs by S aureus treated with TTs, heavy bacterial biofilm loads developed on all TTs, except the ion-bombarded silicone tubes. The ion-bombarded silicone tubes showed only rare isolated clusters of individual bacteria. In contrast, other authors have shown that ion-bombarded silicone TTs were inefficient in the inhibition of CRPA biofilm formation. 37 In 2006, Tatar et al studied a prospective cohort of 30 untreated silastic TTs and 16 ionized, processed TTs. They demonstrated that the ionized TTs were more robust than their controls to inhibit bacterial biofilm growth. 38
Cell-to-cell communication, also known as quorum sensing, plays a critical role in biofilm development. Furanone is a known quorum sensing inhibitor, which has been studied for P aeruginosa biofilm inhibition. In a report by Kim et al, TTs with P aeruginosa biofilms were plated with furanone alone, ciprofloxacin alone, and furanone with ciprofloxacin. 39 While the cultures with furanone or ciprofloxacin alone had similar colony counts and biofilm growth as the saline control, the furanone and ciprofloxacin culture was successful in the inhibition of biofilm formation.
Coating materials
Experimental TT coatings with antibiofilm material may be divided into different groups causing both inhibition of bacterial growth/adhesion: antibiotics, long-term antimicrobial release, plant extracts, polymers, or other materials.
Antibiotics/antimicrobials
Different antibiotics have been used in order to prevent biofilm adhesion with different degrees of success. In vitro studies included piperacillin–tazobactam-coated TTs that were shown to inhibit CRPA biofilm formation, 40 while vancomycin-coated TTs were resistant to MRSA biofilm formation. 41
Other more complex long-term release polymers were studied in vitro. One study showed a potential benefit for ciprofloxacin and Elvax (solidified polymer melt), which presented a 2-stage antibiotic release. 42 Ofloxacin eluded degradable TTs, fabricated from L-lactide-co-e-caprolactone (PLA-PLC), have been studied in vivo and demonstrated a decrease in P aeruginosa biofilm formation. 43 N-acetylcysteine (NAC) is a thiol-containing antioxidant and a mucolytic agent presenting with antimicrobial and antibiofilm properties against some pathogens. 44 It has been used in vitro with TT immersed in MRSA and Quinolone-resistant Pseudomonas aeruginosa (QRPA) broths. Various NAC concentrations were added to each TT, in order to study the adhesion and formation of biofilm formation. N-acetylcysteine significantly decreased the adhesion of MRSA and QRPA in a concentration-dependent manner and significantly inhibited the rate of biofilm formation by MRSA and QRPA strains at all concentrations. The rate of biofilm formation by MRSA and QRPA strains decreased equally at each concentration of NAC. 45
Chitosan is a natural amino polysaccharide that provides antimicrobial efficiency due to its small size. 46 In an in vitro study, chitosan nanoparticles were prepared by ionic gelation (in ionic gelation method, chitosan polysaccharide is dissolved in aqueous acidic solution to get the cation of chitosan) and were used to encapsulate a co-amoxiclav drug mixture and their consequent adsorption onto O2 plasma-activated silicone TTs. 47 The activity was tested against S aureus test strain and was proven that these coated TTs had excellent antimicrobial and biofilm inhibition properties.
In an in vitro study by Banerjee et al, Next-Science (NS), an antimicrobial/antibiofilm agent, was used to destroy the extra-polysaccharide matrix of the bacterial biofilm and kill the bacteria within the biofilm. The authors showed the good effectiveness of NS in inhibiting the development of P aeruginosa and S aureus biofilms on the inner and outer surface of TTs. 48
Polymers
Different polymers changing surface hydrophobicity can alter the ability of bacterial surface adherence. These polymers aim to maximize the difference in hydrophobicity between the bacteria and the surface of the device, thus minimizing the chance of adherence. An in vitro study indicated that fluoroplastic TTs coated with a hydrophilic polymer, phosphorylcholine, were resistant to P aeruginosa and S aureus biofilm formation. 28 However, a different in vivo study in 70 children with ipsilateral phosphorylcholine-coated fluoroplastic TT and a contralateral uncoated fluoroplastic TT failed to show significant difference in biofilm-associated PTTO between these 2 types of TTs. 49
Another hydrophilic polymer is polyvinylpyrrolidone (PVP), which has been studied both as a single agent and in combination with silver oxide coating and thought to inhibit integral bacterial metabolic processes, such as iron hemostasis and disulfide bond formation. 50 In an in vitro study, it was demonstrated that PVP and silver coating reduced P aeruginosa biofilm formation, and PVP was superior to silver oxide. The combination of PVP and silver oxide was associated with a greater biofilm adhesion inhibition. 51
Plant extracts
Various plant extracts have been shown to exhibit antibiofilm properties. In vitro study of tea tree oil–coated silicone TTs prevented biofilm formation against MRSA, and antimicrobial activity was illustrated with zones of inhibition in comparison to vancomycin. 52 Further studies are still required in order to fully assess the potential benefit of this substance and its effect on the middle and inner ear.
Other substances
Human serum albumin (HAS)-coated titanium TT were studied in vitro, 53 demonstrating a decrease in S aureus biofilm formation, while having a nonsignificant effect of P aeruginosa biofilm formation. 54 In an in vivo study of the same TT type, no significant differences were shown between HAS-coated and uncoated TTs.
A randomized double-blind controlled study of 125 children which received a silver oxide–coated TT in one ear and a silastic TT in the other ear demonstrated that the silver oxide silastic tubes diminished the incidence of long-term PTTO, probably due to the disruption of key biochemical and metabolic bacterial processes, including disulfide bond formation and iron homeostasis. 55
Organoselenium (OSe) has been shown to have antibiofilm properties. In an in vitro model, OSe-coated silicon TTs have been shown to inhibit S aureus, H influenzae, and M catarrhalis biofilm formation. 56
Antibodies Against DNABII Proteins
It was shown that at each junction of crossed strands of bacterial extracellular DNA, there is the presence of the DNABII proteins. This protein family is immunolabeled by antibodies directed against IHF, and their localization indicates that this family of proteins may play a critical role in the structural stability of the biofilm. Recently, one study suggests that antibodies against the DNABII family proteins inhibited sinus implant biofilms. 57
Conclusion
Currently, there is not one type of TT on which bacteria will not adhere. The challenges of treating PTTO indicate the need for further research in optimization of TT design in addition to development of novel therapies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Marom is a medical advisor for Novus Therapeutics Inc, Irvine, California.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
