Abstract
To evaluate the use of commercially available allogenic dural graft materials made of fetal bovine collagen, we present an analysis of our case series with use of autologous and allogenic graft materials. Patients who underwent surgical repair of a tegmen tympani defect associated with ipsilateral conductive hearing loss and cerebrospinal fluid (CSF) otorrhea using a middle cranial fossa (MCF) approach from 2004 to 2018 at Loyola University Medical Center were included. Resolution of CSF otorrhea, audiologic outcomes, facial nerve preservation, and surgical complications was analyzed. Thirty-three patients with an average age of 55.3 years (range: 21-78, standard deviation [SD]: 12.9) and body mass index of 34.4 (range: 22-51, SD: 7.4) underwent an MCF repair of a tegmen and dural defect. All patients presented with CSF otorrhea and conductive hearing loss ipsilateral to the defect. Repairs were made with combinations of allograft and autograft in 17 cases, allograft only in 15 cases, and autograft only in 5 cases. Improvement in hearing was noted in 33 cases, and resolution of CSF otorrhea was noted in 36 cases; one patient required repeat surgery which resolved CSF otorrhea. Three patients had minor complications; all these were in the autograft group. The MCF approach coupled with the use of fetal bovine collagen grafts is a safe and viable method to repair tegmen tympani and associated dural defects with salutary outcomes and low morbidity.
Keywords
Introduction
The tegmen tympani is the thin shelf of bone that forms the roof of the tympanic cavity. Defects in the tegmen can be associated with erosion of the overlying dura and serve as a path of leakage of cerebrospinal fluid (CSF) into the tympanic cavity. At times, herniation of brain tissue into the middle ear cavity may be seen in the form of an encephalocele (Figure 1). 1 The etiology of tegmen tympani defects is not clear in many cases; they may be related to congenital bone defects, 2 postinfectious erosion of the bone, prior trauma, or secondary to increased intracranial pressure. 1,3 Patients usually present with ipsilateral hearing loss, tinnitus, and aural fullness. Treatment is warranted as CSF otorrhea risks infections such as meningitis or encephalitis with high morbidity and potential mortality. 4
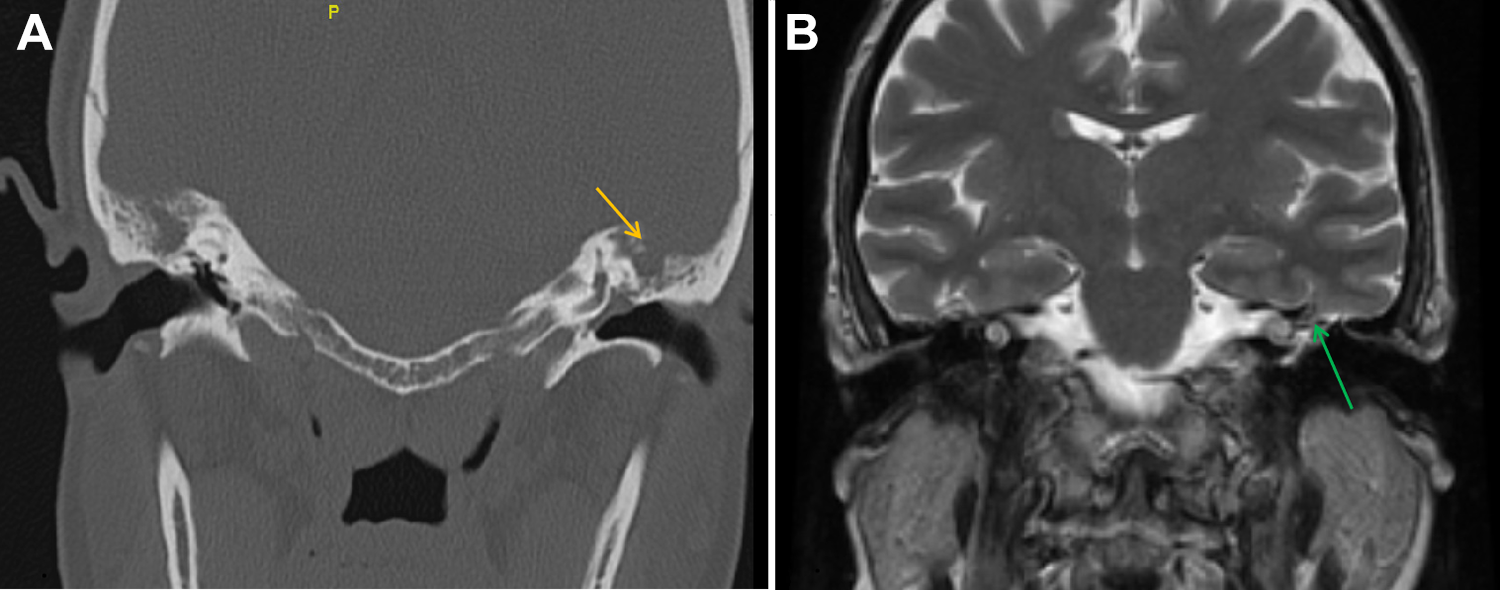
A, Coronal computed tomography cut over the area of interest. Arrow notes absence of bone thus defining a tegmen defect. In addition, note fluid signal in the middle ear cavity consistent with a middle ear effusion. B, Magnetic resonance image in coronal view in T2 sequence. Arrow points to the location of a small dural sac with fluid and brain parenchyma (encephalocele).
Tegmen defects and encephaloceles of the middle cranial fossa (MCF) are repaired surgically via a transmastoid approach or MCF approach. The transmastoid approach does not require temporal lobe retraction or cranial opening and has shorter hospital stay—however, it offers a less favorable approach for repair of the defect since the ossicles of hearing may be compromised. 5 On the other hand, the MCF approach allows direct visualization of the tegmen and dura defects, preservation of hearing, and the ability to perform a direct multilayered repair. 6
The principles of surgical repair via the middle fossa approach include identification of the bony defect, removal of herniated brain tissue (encephaloceles), and various repair strategies that aim to reconstruct the roof of the tympanic cavity (bony) and obliterate any dural tears or fistulae (dural repair). For repair of the bony defect, titanium mesh or autologous split thickness bone graft has also been used with varying results. The use of temporalis fascia has been described, in addition to multiple commercially available dural substitutes to repair the dural defect. The purpose of this study was to determine whether allogenic materials that are commercially available provided results comparable to autologous grafts and whether there was an increased risk of infection with the use of non-native repair materials.
Patients and Methods
This investigation was approved by the institutional review board of Loyola University Medical Center. A retrospective chart review of all patients surgically treated for tegmen defects or encephaloceles at our institution using the MCF approach with a minimum of 3 months of follow-up over a period of 14 years (2004-2018) was conducted. Demographic data included age, gender, body mass index (BMI), and comorbid conditions. Pre- and postoperative data included indications for surgery, methodology of repair, surgical and postoperative complications, facial, and cochlear nerve function before and after surgery, resolution of CSF otorrhea, length of stay, number of readmissions, and length of follow-up.
Data were collected and inputted into Microsoft Excel 2016 (Microsoft, Redmond, WA). Demographic factors were recorded descriptively.
Surgical Procedure
For the MCF approach, the patient is positioned supine with the ipsilateral shoulder gently elevated with a shoulder roll. Prophylactic antibiotics, 0.5 g/kg intravenous mannitol, and antiepileptics coupled with monitoring of the facial nerve are standard procedure. A C-shaped incision is made centered above the pinna with anterior limb about 1 cm anterior to the tragus. A temporalis fascia flap is harvested and set aside in saline. 7,8 A standard temporal craniotomy is carried out, with additional drilling to ensure direct access into the middle fossa floor. Standard extradural dissection is carried out with care to avoid injury to the greater superficial petrosal nerve. The tegmen defect is then encountered and plans are made for repair.
Prior to dural repair, any encephalocele is resected by a combination of bipolar electrocautery and microscissors. Electrocautery is used to gently shrink the under surface of the dura and to provide adequate visualization of the size of the dural defect. Dural repair material (autograft or allograft) is then fashioned to cover the dural defect and laid over the defect. The choice of the material used was made by the primary surgeon. These included either autograft (temporalis fascia or split thickness bone graft) or allograft (Durerepair, Medtronic, Kalamazoo, MI; DuraGen, Integra, Plainsboro, NJ); and DuraMatrix, Stryker, Kalamazoo, MI).
Repair of the tegmen bony defect then ensues with split thickness autologous bone graft harvest during the same procedure. The craniotomy flap is resecured using titanium plates and the temporalis muscle reattached. A multilayered closure of the scalp is performed.
Results
Demographics and Presentation
In all, 33 patients, 18 male and 15 female, with an average age of 55.3 years (range: 21-78, standard deviation [SD]: 12.9) underwent surgical repair of a tegmen defect associated with ipsilateral CSF otorrhea and hearing loss over a 14-year period at our institution. Four patients had bilateral interventions, and these were counted separately. In total, 37 cases were completed, with 18 left-sided repairs and 19 right-sided repairs. The average BMI was 34.4 (range: 22-51, SD: 7.4). The most common presenting symptom and sign was hearing loss and CSF otorrhea (37 cases), meningitis (2 cases), and CSF leaks in the nasal and throat cavities (2 cases). In 31 of the 37 cases, the hearing loss and CSF otorrhea occurred spontaneously in an insidious manner without any obvious precipitating event. Of the remaining 6 cases, 4 experienced a traumatic event while 2 had iatrogenic etiologies due to prior surgeries that may have contributed to the leak. Intraoperatively, 7 of the 37 cases did not have evidence of encephalocele. Encephalocele were noted in 30 of the cases, of which 7 cases had multiple encephaloceles.
A total of 7 patients underwent lumbar punctures of which 6 results were reported in the record with an average opening pressure of 31 mm Hg (range: 22-37, SD: 5). All patients had preoperative audiograms and all had some degree of hearing loss ipsilateral to the side of the CSF otorrhea (18 left sided, 19 right sided). Of these 37 cases, 17 had mixed loss, 18 from conductive loss, and 2 from sensorineural loss. Our follow-up ranged from 1 to 46 months with a mean follow-up period of 9.2 months (SD: 10). Twelve cases included the placement of a lumbar drain in the perioperative period to provide CSF diversion. The decision to place a lumbar drain was surgeon-dependent and varied based on intraoperative impressions.
Surgical Outcomes
The average length of intensive care unit stay was 2 days (range: 1-7, SD: 1.7); the average hospital stay length was 4 days (range: 1-8, SD: 1.6).
Facial nerve monitoring was standard in all cases and no patient experienced intraoperative facial nerve compromise measured as a change in electrophysiological parameters of the standard electroencephalogram monitoring. Postoperative resolution of CSF otorrhea was noted in 36 of 37 cases following the initial surgery. One patient required a second operation, which did result in resolution of the CSF leak.
Postoperative hearing improvement was noted in 33 (89%) cases of the 37 with subjective and objective (improved audiogram) findings. The 4 remaining cases did not report hearing improvement and their audiograms (3/4 patients [note 1]) showed no changes from preoperative findings.
Results Based on Repair Method
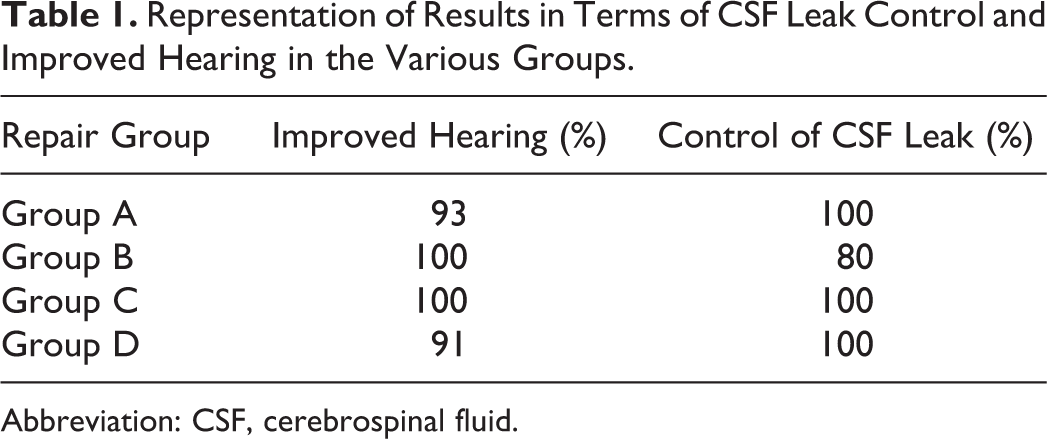
As this was a retrospective evaluation, we analyzed the data based on the type of repair material used. We have retrospectively divided the treatment groups here into 4 (Table 1): Group A. Allograft alone (n = 15) Group B. Autograft alone (n = 5) Group C. Allograft with 1 autograft (n = 5) Group D. Allograft with 2 autografts (n = 12)
Representation of Results in Terms of CSF Leak Control and Improved Hearing in the Various Groups.
Abbreviation: CSF, cerebrospinal fluid.
Group A: Patient who underwent repair with allograft alone all experienced resolution of CSF leak (15/15). In this group, all but one experienced improved hearing (14/15). This was the largest subgroup in our analysis.
Group B: This group consisted of 3 patients treated with temporalis fascia and bone, and 2 patients treated with temporalis fascia alone. Four patients in this group, experienced resolution of CSF leak (4/5), with one patient who required a reoperation. All 5 of 5 patients in this group experienced improved hearing following the procedure.
Group C: This group was treated with allograft with one type of autograft. Of all, 4 of 5 patients here were treated with allograft combined with bone, and one was treated with allograft combined with temporalis fascia. In this group, all 5 of 5 patients experienced resolution of CSF leak and improvement in hearing.
Group D: A total of 12 patients were treated with allograft, temporalis fascia, and bone graft. In this group, all experienced resolution of CSF leak. Of all, 11 of 12 patients had improved hearing.
Complications
No intraoperative complications were reported and no graft site morbidity was noted in patients who underwent autograft harvesting. However, postoperative complications were encountered in 3 patients who required readmission; one (group B) had a delayed wound infection and meningitis, which resolved with intravenous antibiotics alone. The second (group D) experienced a temporal wound dehiscence that was surgically revised and subsequently healed.
The one patient in this study who had a persistence of CSF leak was in group B and had repair completed with temporalis fascia only. The patient was taken for revision surgery and was noted to have a second MCF floor defect that was not appreciated at the time of the initial surgery. The CSF leak was controlled following the second operation.
Additionally, 1 patient (group A) experienced a left-sided facial nerve palsy, House-Brackmann 4 that began 3 days following surgery, which improved 2 months later with continued acyclovir therapy. Electromyographic monitoring before, during, and immediately after surgery, however, showed no compromise of facial nerve function on either side. Thus, this infection was likely viral in nature and unrelated to the surgery or graft material employed. No patients in this study cohort experienced mortality.
Discussion
The optimal surgical approach for repair of tegmen tympani defects is debated but our group and others have validated the effective role of an MCF approach for the reduction of the encephalocele and repair of the dural defect. The optimal repair material is also debated. In general, with the provision of newer allografts, the reliance on autografts has diminished, minimizing graft site morbidity. We sought to compare the use of fetal bovine collagen allografts with temporalis fascia autografts and bone in terms of effectiveness in resolution of CSF otorrhea and hearing improvement; an additional consideration was preservation of facial nerve integrity and avoidance of operative complications. The availability of various allograft materials was evident. We hence decided to compare all allografts derived from fetal bovine collagen with autograft material that was generally temporalis fascia or slit thickness bone graft. Numerous surgical teams participated in this process. An analysis of these graft types and their relation to outcomes, namely hearing improvement, cessation of CSF otorrhea, preservation of facial nerve function, avoidance of operative complications, indicated that the fetal bovine collagen allografts, regardless of the manufacturer, provided similar outcomes to the use of autologous materials; the use of bone augmentation also made no difference in outcome.
In terms of hearing improvement, all patients treated using dura allograft alone reported improvement. Three patients who did not have improved hearing had temporalis fascia autograft alone, temporalis fascia with bone, or temporalis fascia, bone, and dura allograft used together; this would suggest a better outcome with the use of allograft alone.
The ability of these allografts to produce outcomes similar to autografts may in part be due to their structural composition. The allografts used in these cases (Durepair, DuraMatrix, and DuraGen) are all made up of a combination of type I and type III fetal bovine collagen. This collagen has excellent tensile strength and is resilient and easily malleable. In addition to its strength, it is generally engineered to maintain a porosity through which tissue growth and vascularization can take place, aiding in the healing process. They are easy to use and can be cut and conformed to the dural defect; we lay an additional layer on the MCF floor over the tegmen tympani defect thereby bolstering the repair and avoiding the need for bone augmentation. This has obviated the use of any metal fixation devices and is a gentle buttress on the MCF floor providing minimal risk to the GSPN or other critical structures. There is also no donor site morbidity, and the closure of the temporalis fascia and subsequent scalp layers can be effected with ease. It is also likely that the cessation of CSF egress into the tympanic cavity is critical in restoring hearing obviating the need for bone or other materials. The natural thickness and malleability of the allografts and the ability to layer them provides a key advantage in this regard.
Our series reported here carries some limitations. Although all of our patients had clinical follow-up, we note that the range was wide from 1 month to 46 months. Four patients had follow-up less than 3 months in this 37 case series. Appropriate long-term follow-up in all patients would be important for this pathology, as clinical failure can occur early or late in the disease process. In addition, the study design did not allow for proper comparison between the treatment groups and limits the overall generalizability of this report. As this was a retrospective study, we evaluated the treatment groups based on the various repair materials used, but the decision to use any particular combination of repairs material was surgeon-dependent, overall limiting our ability to study comparison groups. Further, 12 patients were treated with the use of a lumbar drain for temporary CSF diversion. The decision to place a lumbar drain was made largely based on the surgeon’s intraoperative impression and was not part a preoperative protocol, thus further limiting our ability to draw conclusions from its use.
Conclusion
Defects in the tegmen tympani can pose a risk of conductive hearing loss, CSF otorrhea, or seizures, if left untreated. Surgical intervention to repair the defect is essential; both the dura and tegmen defect must be identified and addressed and any herniating parenchymal tissue reduced. Our experience suggests that the use of the MCF approach coupled with the use of fetal bovine collagen can be a safe and viable method to addressing this condition. Future studies are needed to shed light on the efficacy of the various repairs materials and to address the need for adjunctive treatment methods such as temporary CSF diversion in the perioperative period.
Footnotes
Acknowledgments
We would like to acknowledge our business administrator of digestive health & neurosciences, Danielle Young (
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Note
1. One patient had complete hearing loss preoperatively and did not have any subjective improvement. An audiogram was not repeated postoperatively.
