Abstract
Objective
The present study investigates risk factors and clinical outcomes of postoperative pneumolabyrinth following the middle cranial fossa approach for superior semicircular canal dehiscence repair, a complication that has not been documented previously.
Study Design
Retrospective cohort study.
Setting
Tertiary/quaternary care referral center.
Methods
We conducted a retrospective review of 332 middle cranial fossa procedures from 2014 to 2020 at a tertiary/quaternary care institution. Upon identifying pneumolabyrinth cases from postoperative computed tomography temporal bone scans, we conducted multivariable logistic regression analysis to explore demographic and clinical factors that were independently linked to this complication. We also compared the rates of postoperative symptoms among patients with different grades of pneumolabyrinth.
Results
We identified 41 (12.3%) pneumolabyrinth cases. Patients with older age, higher preoperative pure tone average, and frank dehiscence were at higher risk for pneumolabyrinth. Furthermore, patients with pneumolabyrinth reported significantly higher rates of postoperative dizziness and exhibited significantly greater pure tone average and air-bone gap postoperatively as compared with patients without pneumolabyrinth. Finally, higher-grade pneumolabyrinth was associated with increased rates of postoperative hearing loss, and grade III pneumolabyrinth was associated with higher rates of postoperative tinnitus vs grade I and II cases.
Conclusion
Pneumolabyrinth following the middle cranial fossa approach was associated with poor clinical outcomes, including dizziness and auditory impairment. Old age, high preoperative pure tone average, and frank dehiscence were risk factors for this complication. The highlighted parameters may be important to note when considering this approach as a treatment option and while monitoring postoperative recovery.
Keywords
Superior semicircular canal dehiscence (SSCD) syndrome is a rare inner ear disorder caused by the absence or thinning of the bone overlying the superior semicircular canal SSC, which can elicit interferences with perilymphatic flow from the cochlea to the labyrinth. 1 As a result, audiologic and vestibular functions can be impaired, causing symptoms such as autophony, aural fullness, tinnitus, and vertigo (Tullio phenomenon and Hennebert sign). 2 If conservative regimens fail, SSCD is typically treated surgically with either transmastoid repair or the middle cranial fossa approach (MCF). 3 Both approaches have been found to offer symptom resolution with minimal risk.4-7
At our institution, patients with SSCD are treated with the MCF performed collaboratively by a neurotologist and a neurosurgeon. 8 Upon exposure of the temporal fossa, the dehiscence site is sealed off with bone wax embedded with a titanium plate or autologous bone chip. The repair is then stabilized with collagen sponges and fibrin glue.8,9 Over the years, pneumolabyrinth—which is characterized by the presence of air bubbles in the otic labyrinth that results in tinnitus, hearing loss, and dizziness—was occasionally identified in some patients after MCF surgery.10-13
Since pneumolabyrinth is an uncommon condition that is often preceded by trauma, instances of post-MCF pneumolabyrinth have not been documented in the literature.11-14 In this study, we fill this paucity of literature with a retrospective review of MCF surgery conducted at our institution over the past seven years. We hope to identify risk factors and symptomatology of post-MCF pneumolabyrinth that can aid clinical management and current understanding of this condition.
Methods
Data Collection
Study approval was obtained from the UCLA Institutional Review Board (#21-001908). This is a retrospective review of 332 consecutive MCF surgical procedures at a tertiary/quaternary referral center from January 2014 to December 2020 performed by the same head and neck surgeon (Q.S.G.) and neurosurgeon (I.Y.) for SSCD repair. Selection criteria for the surgery were patient history, symptoms, and preoperative computed tomography (CT) temporal bone findings that were consistent with SSCD (eg, autophony and pulsatile tinnitus with clear dehiscence on CT). Vestibular evoked myogenic potential testing was obtained for controversial cases with unclear evidence from CT findings and clinical manifestation. However, since vestibular evoked myogenic potential study was not routinely obtained, it was omitted from this review.
Variable Characterization
Pneumolabyrinth cases were identified by reviewing the CT temporal bone scans obtained immediately after the MCF (within 24 hours). Postoperative CT images were routinely ordered for every patient undergoing the MCF to assess for markers and early signs of complications. The pneumolabyrinth identification was done independently by radiology as well as a single researcher to confirm the presence of air and ensure a consistent standard across cases. Both raters were blinded to patients’ postoperative symptoms at the time of review. In very few instances when the raters disagreed, the researcher rereviewed the cases with radiologist input and reached a consensus.
Confirmed pneumolabyrinth cases were classified into three grades according to the volume and region of air present within the labyrinth. Cases with small air bubbles (diameter <1.5 mm on coronal image) located only in the SSC were classified as grade I. Cases with large air bubbles (diameter >1.5 mm) but located only within the SSC were classified as grade II. Finally, cases with large air bubbles extending into the vestibule or regions of the labyrinth other than the SSC were classified as grade III. Diameter was defined as the largest distance between two boundaries of an air bubble. An example of a pneumolabyrinth case from each grade is presented in Figure 1 .
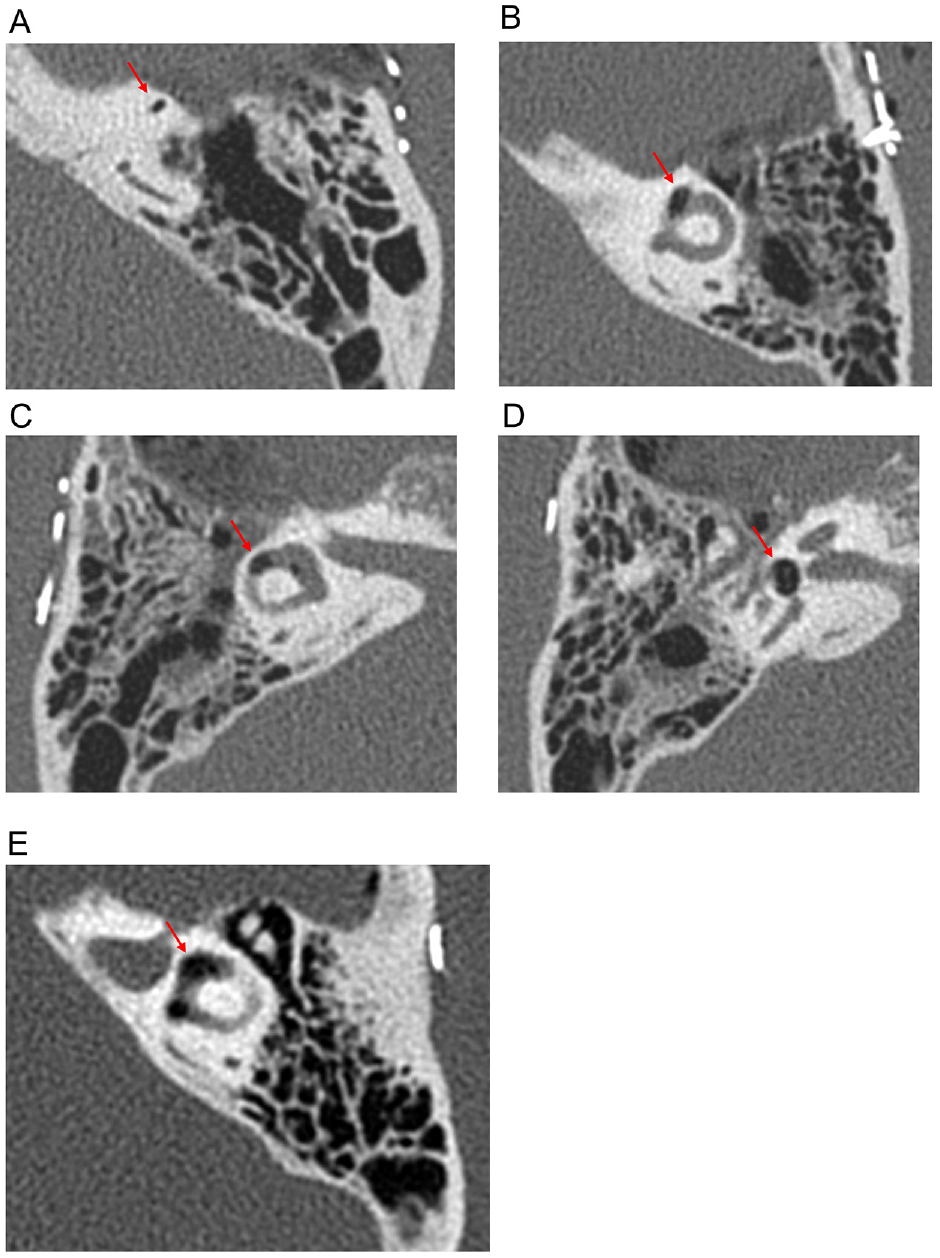
Examples of pneumolabyrinth: (A) grade I, (B) grade II, and (C-E) grade III. Panels C and D are different slice views of the same ear.
Other variables of interest were recorded: age, sex, type of dehiscence (frank or near dehiscence), prior ear trauma, prior ear surgery, laterality of SSCD, intraoperative cerebrospinal fluid leak, and symptoms reported by patients on a symptom questionnaire postoperatively. Symptoms assessed by the questionnaire were autophony, amplification of other sounds, aural fullness, tinnitus, hyperacusis, hearing loss, vertigo, dizziness, disequilibrium/imbalance, oscillopsia, and headache. Postoperative symptoms were assessed at follow-up visits post-MCF. The latest follow-up data available were used for analysis. SSCD is divided into two types of dehiscence: frank dehiscence (the bone overlying the SSC is absent) and near dehiscence (the bone overlying the SSC is very thin). 15 Prior ear trauma was defined as any pressure-induced or concussive injury that can exacerbate SSCD symptoms. Examples include barotrauma such as scuba diving injuries or concussive blows to the head. Prior ear surgery consisted of tympanoplasty, stapedectomy, mastoidectomy, myringotomy tube placement, cochlear implantation, round or oval window occlusion, and perilymphatic fistula repair.
Pre- and postoperative audiometry data were evaluated. From each audiometry study, pure tone average (PTA), air-bone gap (ABG), and word recognition score were calculated according to guidelines from the Hearing Committee of the American Academy of Otolaryngology–Head and Neck Surgery.16,17 PTA was calculated as the averaged air conduction threshold at 500, 1000, 2000, and 3000 Hz. If conduction data were unavailable at 3000 Hz for any particular audiogram, the averaged threshold between 2000 and 4000 Hz was taken. ABG was calculated as the averaged difference between pure tone air and bone conduction at 500, 1000, 2000, and 3000 Hz. The word recognition score was calculated as the percentage of single-syllable words presented at a standardized level that was correctly identified by the patient. Pre- and postoperative hearing scattergrams were also presented as recommended by the American Academy of Otolaryngology–Head and Neck Surgery. 16 In this study, separate scattergrams were created for nonpneumolabyrinth and pneumolabyrinth cases to allow for meaningful comparison.
Statistical Analysis
A multivariable logistic regression model was constructed with pneumolabyrinth status (yes/no) as the outcome variable. Covariates were age (continuous), sex, prior ear trauma, prior ear surgery, SSCD laterality, intraoperative cerebrospinal fluid leak, and postoperative symptoms assessed by the questionnaire (excluding variables exhibiting a high degree of collinearity with many other symptoms). Follow-up duration (months) was controlled for in the model.
Type of dehiscence was not included in the multivariable model due to the low case count of near dehiscence among pneumolabyrinth cases (n = 1). Instead, a Fisher’s exact test was employed to assess the relationship between dehiscence type and pneumolabyrinth status. Audiogram findings were omitted from the multivariable model due to the high degree of collinearity between audiogram metrics and a wide range of audiologic-related symptoms. Instead, a series of independent samples t tests were employed comparing pre- and postoperative PTA, ABG, and word recognition score between patients with and without pneumolabyrinth. Finally, the association between pneumolabyrinth grade and rates of postoperative symptoms was assessed via Kendall’s tau-c or Cramér’s V.
Due to the multiple comparisons performed, risks of alpha inflation were reduced with P value adjustments within hypotheses per the Benjamini-Hochberg procedure. 18 Subset analysis assessing rates of symptoms among grades of pneumolabyrinth were conducted only among pneumolabyrinth cases. These analyses were more limited in statistical power due to low case counts (n = 41). Therefore, P value adjustments were not performed, and interpretations did not rely on P values but instead focused primarily on effect sizes (Cramer V and Kendall tau-c) to assess for clinical relevance. Statistical significance was assessed at a two-tailed α = 0.05. All statistical analyses were conducted with SPSS version 27.0.1.0 (IBM).
Results
Table 1 presents a summary of patient population characteristics and study variables. Our sample had a female predominance at 63%, and the average age was 50 years. The mean follow-up period was 5.36 months (range, 0.03-35.6) for the overall sample, 5.27 months (range, 0.03-30.5) for patients without pneumolabyrinth, and 5.81 months (range, 0.03-35.6) for patients with pneumolabyrinth.
Demographic Profile and Clinical Characteristics of SSCD Cases, 2014-2020. a
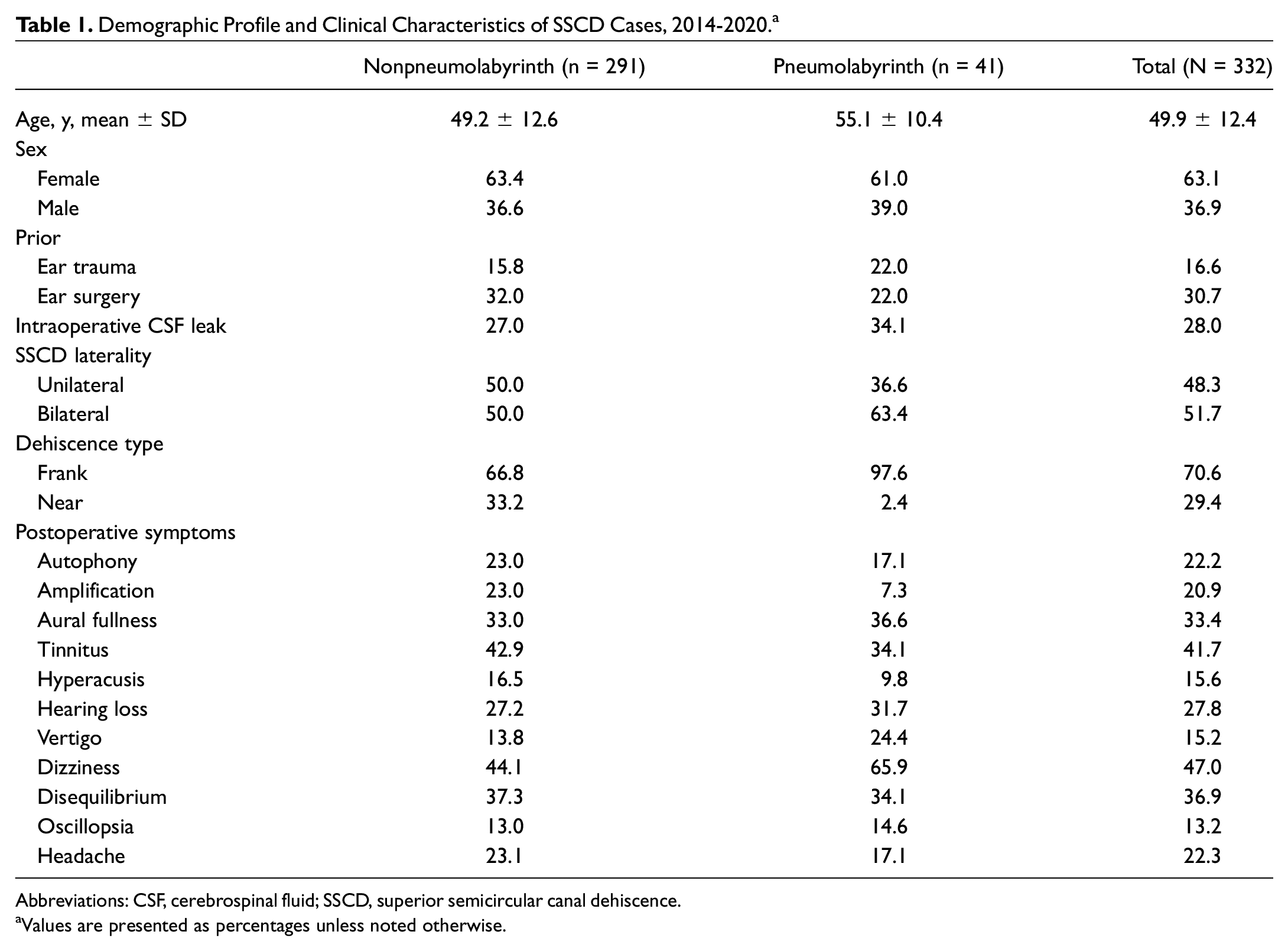
Abbreviations: CSF, cerebrospinal fluid; SSCD, superior semicircular canal dehiscence.
Values are presented as percentages unless noted otherwise.
After reviewing 332 postoperative CT scans, we identified 41 (12.3%) cases of pneumolabyrinth: 12 grade I, 19 grade II, and 10 grade III. Most frequently reported symptoms postoperatively were dizziness (65.9%), aural fullness (36.6%), and tinnitus (34.1%) among patients with pneumolabyrinth, as compared with dizziness (44.1%), tinnitus (42.9%), and disequilibrium (37.3%) among patients without pneumolabyrinth.
Among pneumolabyrinth cases, 97.6% were frank dehiscence, and only one case was near dehiscence prior to SSCD repair. However, 66.8% of nonpneumolabyrinth cases were frank dehiscence and 33.2% were near dehiscence prior to SSCD repair. The Fisher’s exact test revealed that postoperative pneumolabyrinth was significantly more likely to occur in frank dehiscence cases vs near dehiscence cases (P < .001, phi = 0.22).
Results from multivariable logistic regression analysis are presented in Table 2 . After controlling for follow-up duration and important demographic, surgical, and clinical factors, we found that age and postoperative dizziness were independent predictors of postoperative pneumolabyrinth. Specifically, higher age was independently associated with increased risks of pneumolabyrinth (adjusted odds ratio, 1.05 [95% CI, 1.01-1.09]), and patients who reported dizziness postoperatively were more likely to have pneumolabyrinth than patients who did not (adjusted odds ratio, 3.56 [95% CI, 1.52-8.33]).
Predictors of Pneumolabyrinth: Multivariable Logistic Regression Model. a
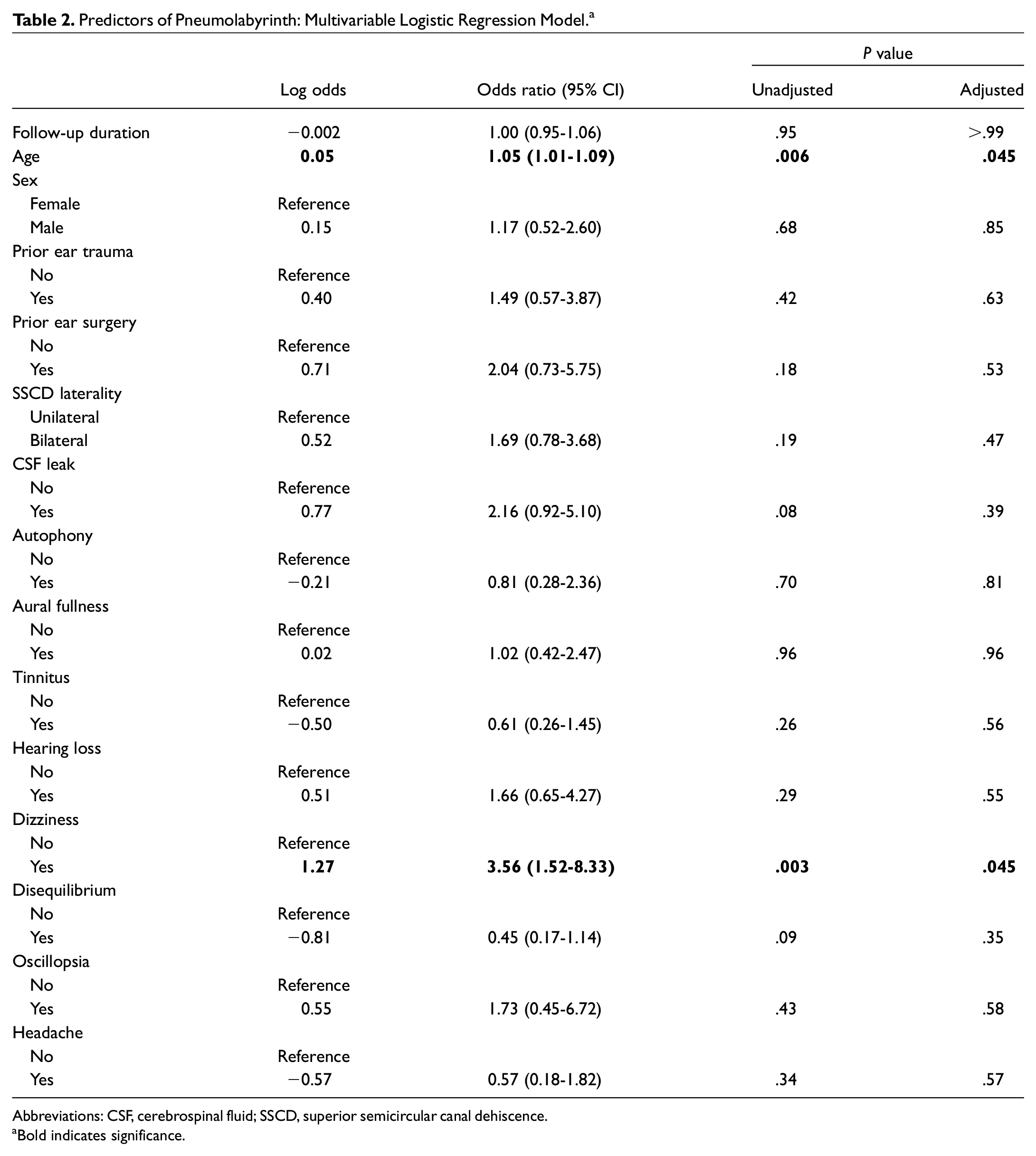
Abbreviations: CSF, cerebrospinal fluid; SSCD, superior semicircular canal dehiscence.
Bold indicates significance.
Preoperative and pre- and post-MCF hearing scattergrams among nonpneumolabyrinth and pneumolabyrinth cases are illustrated in Figures 2 and 3 . Audiometry metrics and results from independent samples t tests are presented in Table 3 . Results indicated that pneumolabyrinth cases exhibited significantly higher preoperative PTA (adjusted P = .024, Cohen’s d = 0.50), postoperative PTA (adjusted P = .039, Cohen’s d = 0.51), and postoperative ABG (adjusted P = .038, Cohen’s d = 0.50) as compared with nonpneumolabyrinth cases.
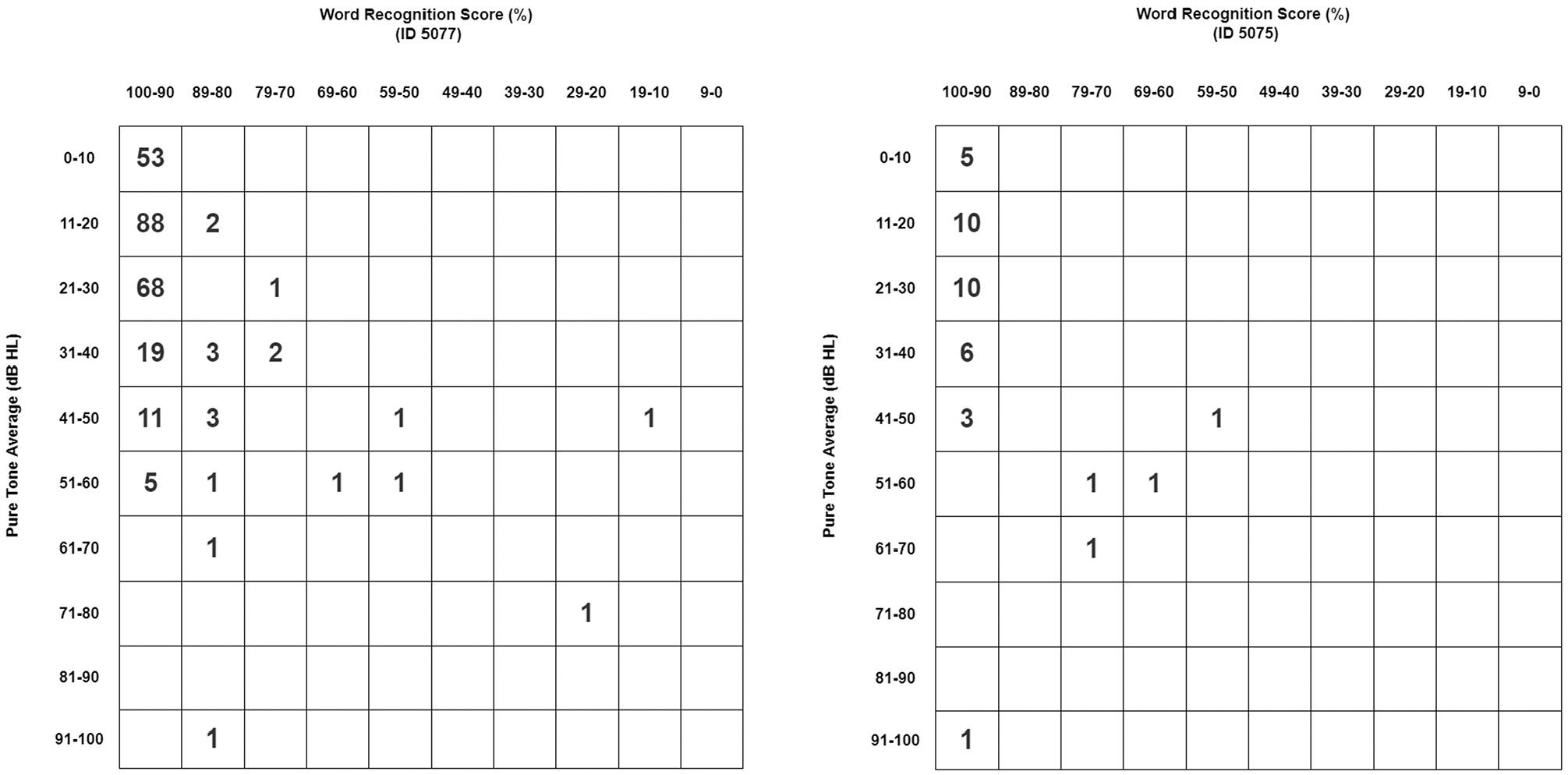
Preoperative hearing scattergrams among nonpneumolabyrinth (left) and pneumolabyrinth (right) cases.
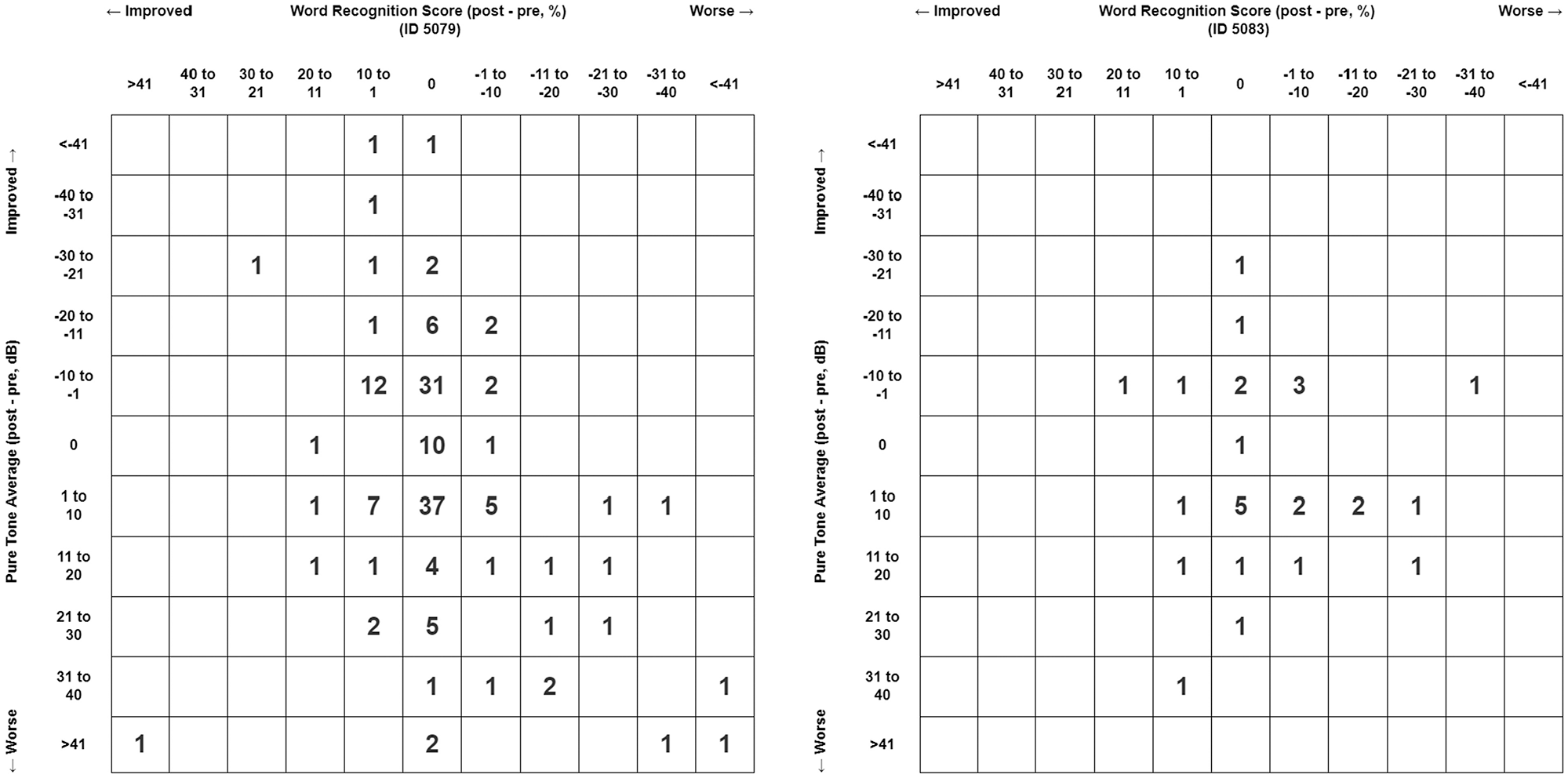
Pre- and postprocedural hearing scattergrams among nonpneumolabyrinth (left) and pneumolabyrinth (right) cases.
Audiometry Findings for Nonpneumolabyrinth vs Pneumolabyrinth Cases. a
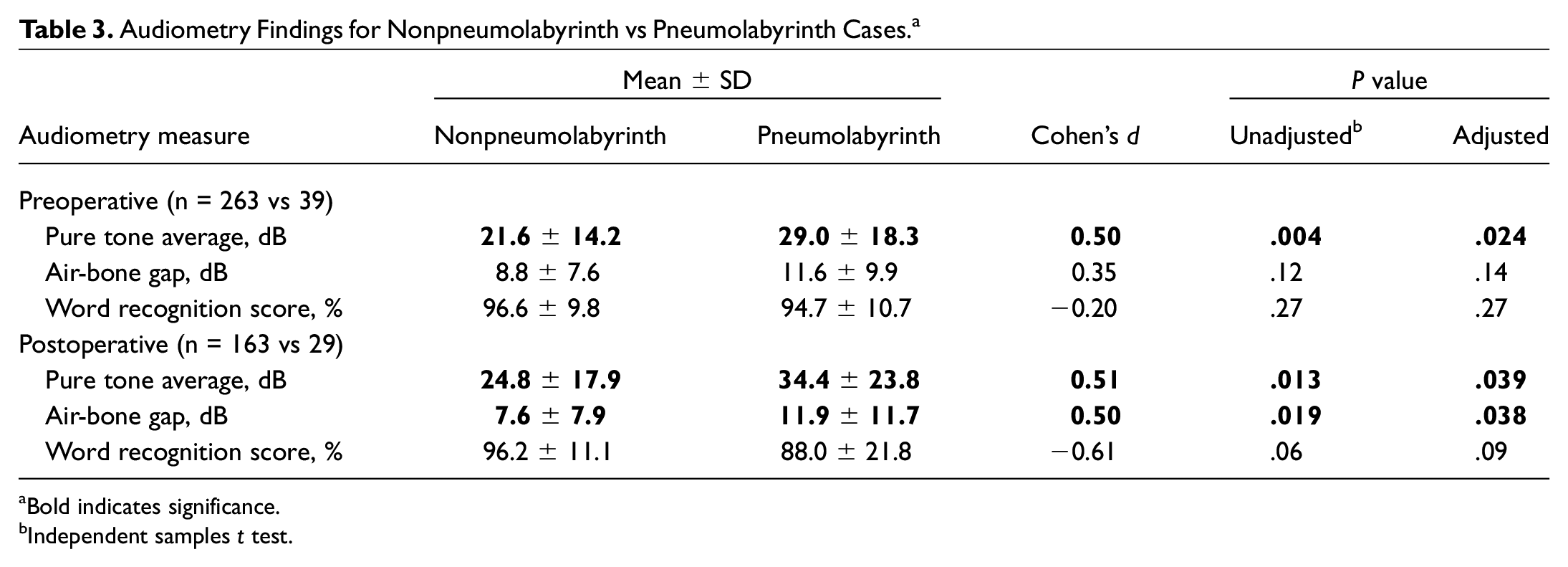
Bold indicates significance.
Independent samples t test.
Rates of symptoms among patients with grade I, II, and II pneumolabyrinth are presented in Figure 4 . Bivariate statistics reveled a medium to strong positive association between pneumolabyrinth grade and rates of postoperative hearing loss (Kendall’s tau-c = 0.31, P = .024). Furthermore, grade III pneumolabyrinth was associated with significantly higher rates of postoperative tinnitus vs grade I and II (Cramér’s V = 0.43, P = .021). Pneumolabyrinth grade was not strongly associated with rates of autophony (Kendall’s tau-c = 0.16), aural fullness (Kendall’s tau-c = −0.12), dizziness (Kendall’s tau-c = −0.03), disequilibrium (Kendall’s tau-c = 0.10), oscillopsia (Kendall’s tau-c = −0.06), and headache (Kendall’s tau-c = −0.13).
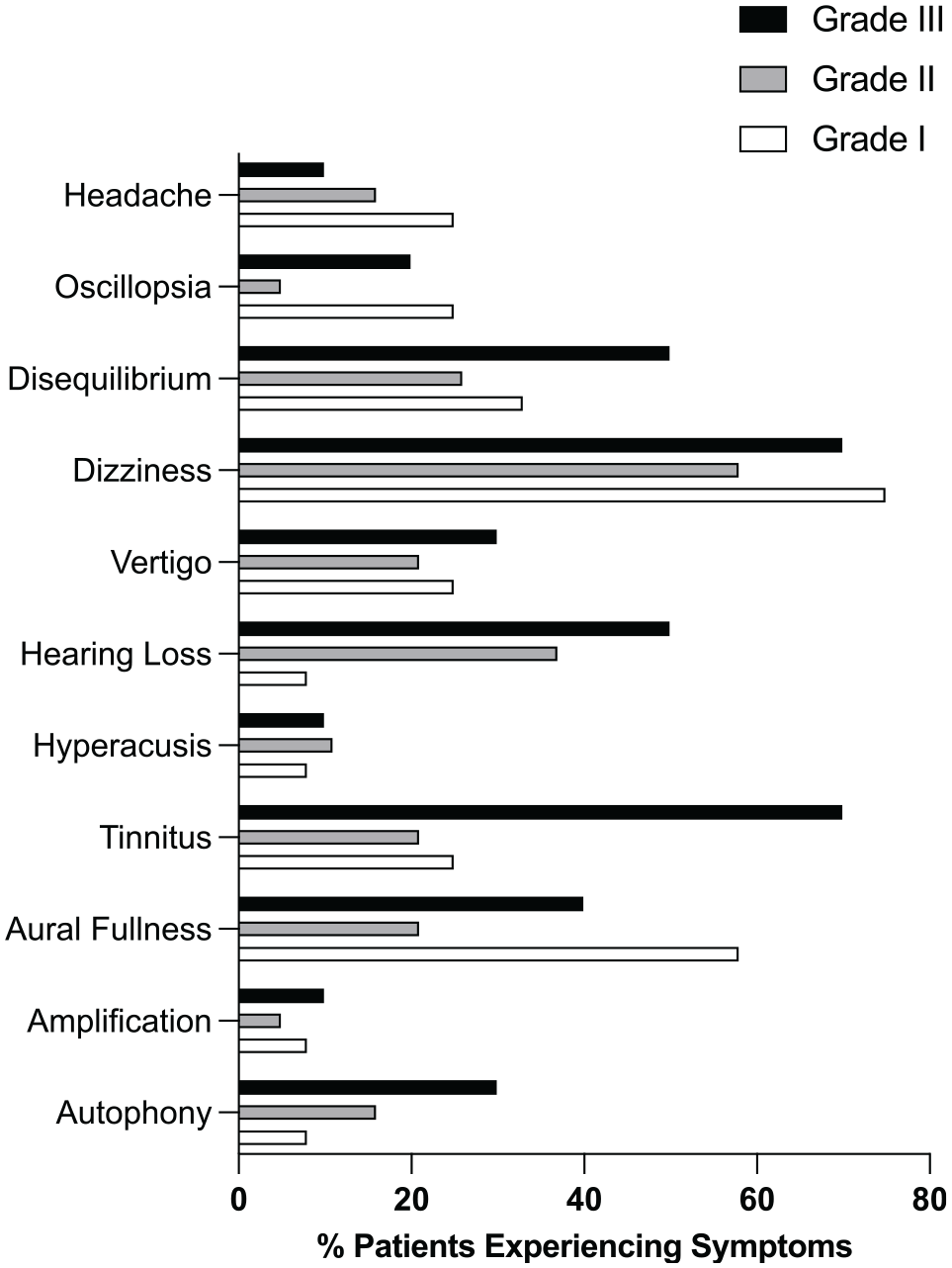
Rates of postoperatively reported symptoms among pneumolabyrinth cases (grades I-III).
Discussion
While post-MCF pneumolabyrinth has not been documented previously, other iatrogenic etiologies of pneumolabyrinth, such as stapes surgery and cochlear implant, have been reported.14,19,20 For the case of cochlear implant, opening of the perilymphatic space during surgery was suggested as the likely mechanism of air entry. 19 We add to the literature, reporting that pneumolabyrinth can occur after the MCF, which is likely due to air exposure of the SSC during surgery. The optimal management of pneumolabyrinth is unclear. Conservative treatments such as antibiotics or corticosteroid have been adopted to date. 14
Our analysis suggests that patients with pneumolabyrinth were significantly more likely to report dizziness after the MCF than patients without, even after controlling for demographic and important clinical factors. Similarly, a recent study found that pneumolabyrinth after cochlear implantation was significantly associated with dizziness. 21 Two hypotheses were proposed to explain this link: first, pneumolabyrinth can cause disturbances to perilymphatic homeostasis, and thus elicit dizziness; second, pneumolabyrinth can increase the weight of cupula and result in its higher sensitivity to head movements. 21 While these speculations are compelling, additional investigations are warranted. Nevertheless, our finding supports that post-MCF dizziness can be highly suggestive of pneumolabyrinth, and should therefore be closely monitored.
We found that patients with higher-grade pneumolabyrinth, which is indicative of increased air bubble volume and extent of labyrinth occupancy, reported hearing loss at significantly higher rates. Furthermore, patients with grade III pneumolabyrinth experienced postoperative tinnitus at significantly higher rates than patients with grade I and II pneumolabyrinth. Hearing loss and tinnitus are known symptoms of pneumolabyrinth.10-13 Our finding adds to this knowledge, suggesting that larger air bubbles in the labyrinth can exacerbate patients’ experiences with hearing loss and tinnitus. This is expected, as perilymph is essential for audiologic function, and increased perturbance of acoustic communication can result when a higher proportion of the labyrinth is occupied by air. In summary, our findings suggest that patients with post-MCF pneumolabyrinth are likely to experience dizziness, and dizziness is usually accompanied by hearing loss and/or tinnitus among patients with large pneumolabyrinth.
Moreover, we found that patients with pneumolabyrinth exhibited significantly higher postoperative ABG and PTA than patients without. Prior research has indicated that pneumolabyrinth-induced perilymphatic fistula can manifest in the form of conductive hearing loss. 22 As a high ABG is typically indicative of conductive hearing loss, our finding may be explained by the highlighted knowledge in current literature. Furthermore, a high PTA is typically suggestive of poor auditory function. Since pneumolabyrinth has been linked to poor hearing thresholds, it may be responsible for the elevated postoperative PTA among patients with this complication. 14 However, our results demonstrated that patients with pneumolabyrinth had higher PTA prior to the MCF. As such, it is possible that high PTA is a risk factor, rather than a manifestation, of post-MCF pneumolabyrinth. Hearing impairment (high PTA) could be caused by factors related to perturbance of the structural integrity or function of the inner ear. The same factors may predispose these patients to higher risk of pneumolabyrinth after undergoing an invasive procedure such as the MCF. Our findings suggest that post-MCF pneumolabyrinth is associated with high PTA and ABG. Therefore, these metrics should be routinely evaluated pre- and postoperatively.
Our analysis revealed that older patients were at higher risk for pneumolabyrinth, even after adjusting for a variety of clinical parameters. Risk for SSCD was previously shown to increase with age. 23 Specifically, it was argued that the SSC roof would wear with age as a result of the chronic pressure imposed by cerebrospinal fluid and temporal bones. 24 Recent microanatomic analysis of the inner ear with three-dimensional imaging found that the physiologic aging process was associated with contortion of the vestibular membranous labyrinth. 25 Hence, our finding may be explained by the more deformed and delicate membranous labyrinth in older patients, which is likely more prone to issues upon exposure that introduces air. Patients’ age may be important to consider before performing the MCF.
Air entry to the labyrinth during the MCF can transpire through several potential mechanisms. First, inappropriate suctioning of the canal, either with a suctioning tube or during dehiscence plugging, can lead to air perforation into the labyrinth. 26 However, both surgeons are aware of this phenomenon and actively avoid such a procedure during MCF surgery. Second, the dura may adhere to the lining of the membranous labyrinth. As such, when the dura is elevated, the membranous labyrinth may be violated. This hypothesis is consistent with our finding that most cases of pneumolabyrinth occurred in patients with frank dehiscence as opposed to near dehiscence. While near dehiscence is sometimes treated by drilling the thin bone to create an opening for plugging, we do not adapt such practice. 27 Instead, we reinforce the thin bone with plugging of bone wax. Therefore, if the membranous labyrinth was violated when the dura was being elevated, patients with near dehiscence would theoretically still have a thin layer of protection against air entry into the labyrinth, whereas patients with frank dehiscence would have direct exposure of the canal, and thus be at higher risk for pneumolabyrinth.
Several practices may prevent or minimize the severity of pneumolabyrinth. Attempts to avoid suctioning during the MCF and perform dehiscence plugging with a high level of caution can prevent trauma to the membranous labyrinth. If elevation of the dura in fact leads to violation of the membranous labyrinth, minimizing the time between dehiscence exposure and sealing could ensure that the volume of air entered, if any, is low.
The underwater transmastoid approach for SSCD repair was recently developed to prevent intraoperative perilymph loss. 28 Surgery was performed with the mastoid immersed in a balanced salt solution and yielded favorable outcome with minimal hearing loss complications. A similar underwater approach was adapted for cochlear implantations, which showed that such an approach was effective in minimizing significant variations in cochlear pressure and permitted nontraumatic insertion of the electrode. 29 Since pneumolabyrinth formation is expected to arise from exposure of the perilymph to air, risks of pneumolabyrinth may be reduced if a similar underwater approach is adapted for the MCF. This underwater approach would eliminate suctioning during surgery, thereby reducing risks of pneumolabyrinth caused by inappropriate suctioning. 30 However, the underwater approach has been shown to be feasible for only the transmastoid approach. The MCF is expected to be technically challenging when the surgical field and endoscope are both submerged in water, although efforts are warranted to assess its feasibility. Therefore, the appropriateness of this approach may vary depending on patient circumstances and surgeon preferences. Additional investigations are needed on potential surgical maneuvers that can minimize the risk of post-MCF pneumolabyrinth.
Limitations
Several limitations should be noted. First, symptoms were reported by patients at postoperative visits, and recall bias can be at play. Furthermore, standards for identifying pneumolabyrinth from CT scans could be inconsistent across radiologists. However, this risk was minimized as a single researcher reviewed all scans, employing a consistent standard. Finally, due to inconsistent protocols across external audiometry studies, a portion of our sample did not have adequate data for calculations of audiometry metrics, especially for postoperative audiograms. Nonetheless, there remained a sizable number of patients with sufficient audiogram parameters that allowed for meaningful analysis. Bivariate statistics revealed that population characteristics were not significantly different between those included in the analysis and those excluded due to insufficient audiometry data ( Table 4 ). As such, this limitation likely did not significantly affect our findings. Despite these potential issues, this study, to our knowledge, is the first to present and characterize post-MCF pneumolabyrinth. With a large multiyear sample, we provided several risk factors of this complication, which can guide clinical practice. Therefore, this study can be an important contribution to the literature.
Population Characteristics of Cases With Sufficient vs Insufficient Postoperative Audiogram Data.

Abbreviations: CSF, cerebrospinal fluid; SSCD, superior semicircular canal dehiscence.
Conclusions
Among 332 MCF surgical procedures, 41 cases of postoperative pneumolabyrinth were identified. Old age, high preoperative PTA, and frank dehiscence were risk factors for this complication. Patients with pneumolabyrinth reported higher rates of postoperative dizziness and exhibited higher PTA and ABG postoperatively as compared with patients without pneumolabyrinth. Finally, higher-grade pneumolabyrinth was associated with increased rates of postoperative hearing loss, and grade III pneumolabyrinth was associated with higher rates of postoperative tinnitus vs grade I and II pneumolabyrinth. These factors may be important to note when considering the MCF as a potential treatment option.
