Abstract
Sphenoid wing meningiomas are tumors that typically present with vision deterioration and neurological changes due to their proximity to the sella, cavernous sinus, and other vital structures. Some unusual symptoms have also been described in the literature, such as cognitive dysfunction, parkinsonism, and intracerebral hemorrhage. In this report, we detail another unusual case of sphenoid wing meningioma in a 63-year-old female who presented with left sudden sensorineural hearing loss. A brief review of the literature is also included.
Keywords
Introduction
Meningiomas are the most common benign intracranial tumors arising from arachnoid epithelium, accounting for nearly a third of all primary brain tumors reported in the United States. The overall incidence of meningiomas is higher among women and increases with age in both men and women. 1 Meningiomas can be classified into various subtypes based on their location and histological grade according to the World Health Organization (WHO) classification of brain tumors. The vast majority (90%) of these tumors are WHO grade 1 (benign), with less than 10% being grade 2 (atypical) and grade 3 (malignant) tumors. 2
The diagnosis of meningioma is made with clinical examination findings and neuroimaging using head computed tomography (CT) or magnetic resonance imaging (MRI) scans. Stereotactic or endoscopic transsphenoidal biopsy can aid in the diagnosis of meningiomas and allow for pathological classification of the tumor. Depending on the tumor size and location of symptomatic cases, the treatment is often microscopic resection or radiosurgery with radiation to reduce recurrence rates. 3,4 Asymptomatic patients diagnosed by imaging are followed with observation and do not usually require surgical intervention. 5
About 20% of all meningiomas are sphenoid wing meningiomas. While most meningioma subtypes commonly present with headache, altered mental status, and paresis, the frequently reported symptoms of sphenoid wing meningiomas include proptosis and progressive visual deterioration. The manifestations classically seen in patients with sphenoid wing meningiomas correspond to the anatomic involvement of the primary tumor. Sphenoid wing meningiomas commonly arise from or extend into the orbital or temporal fossa, producing the symptoms of proptosis, impaired extraocular movements, and loss of facial sensation, while those involving the optic nerve result in vision deficits. 6 Due to the slow-growing nature of sphenoid wing meningiomas, these classic symptoms, when present, typically have a mild onset. Thus far, only a handful atypical presentations have been reported in the literature, including cognitive impairment, intracerebral hemorrhage, and parkinsonian features. 7 -9 In this report, we describe another unique manifestation of sphenoid wing meningioma: sudden sensorineural hearing loss (SSNHL) and ear pain.
Case Report
Our patient is a 63-year-old female with no past medical history, who was in a normal state of health until 5 days prior to her presentation to the clinic. She reported having worsening bilateral frontal headaches, associated with episodic left inner ear pain, and progressive left hearing loss over this period of time. The appearance of her symptoms was abrupt in nature, and she denied experiencing similar symptoms in the past. She denied ear ringing or fullness, vision changes, seizures, facial or trigeminal nerve deficits, weakness, anosmia, psychomotor changes, weight loss, night sweats, or fevers.
Upon arrival to our institution, intravenous (IV) dexamethasone was ordered and stereotactic MRI of the brain was recommended (Figure 1). The MRI revealed an enhancing 2.4 × 3.6 × 4.7 cm left parasellar mass with nodular margins that narrowed the left cavernous carotid artery, extended medially into the suprasellar cistern, posteriorly into the left ambient cistern and circumferentially around the basilar artery. Anteriorly, the mass abutted the left orbital apex and inferiorly, the left foramen ovale, with meningeal enhancement. There was also encasement of the left cranial nerve 8 (CN VIII) and narrowing of the left internal carotid artery. Mass effect on the brain stem and left temporal lobe was also noted. The patient underwent a whole-body CT scan that showed no additional lesions or masses. At this point, we recommended audiogram, thin slice CT scan of the paranasal sinuses and skull base, and a transsphenoidal approach (TSA) for tissue sampling of the skull base mass.
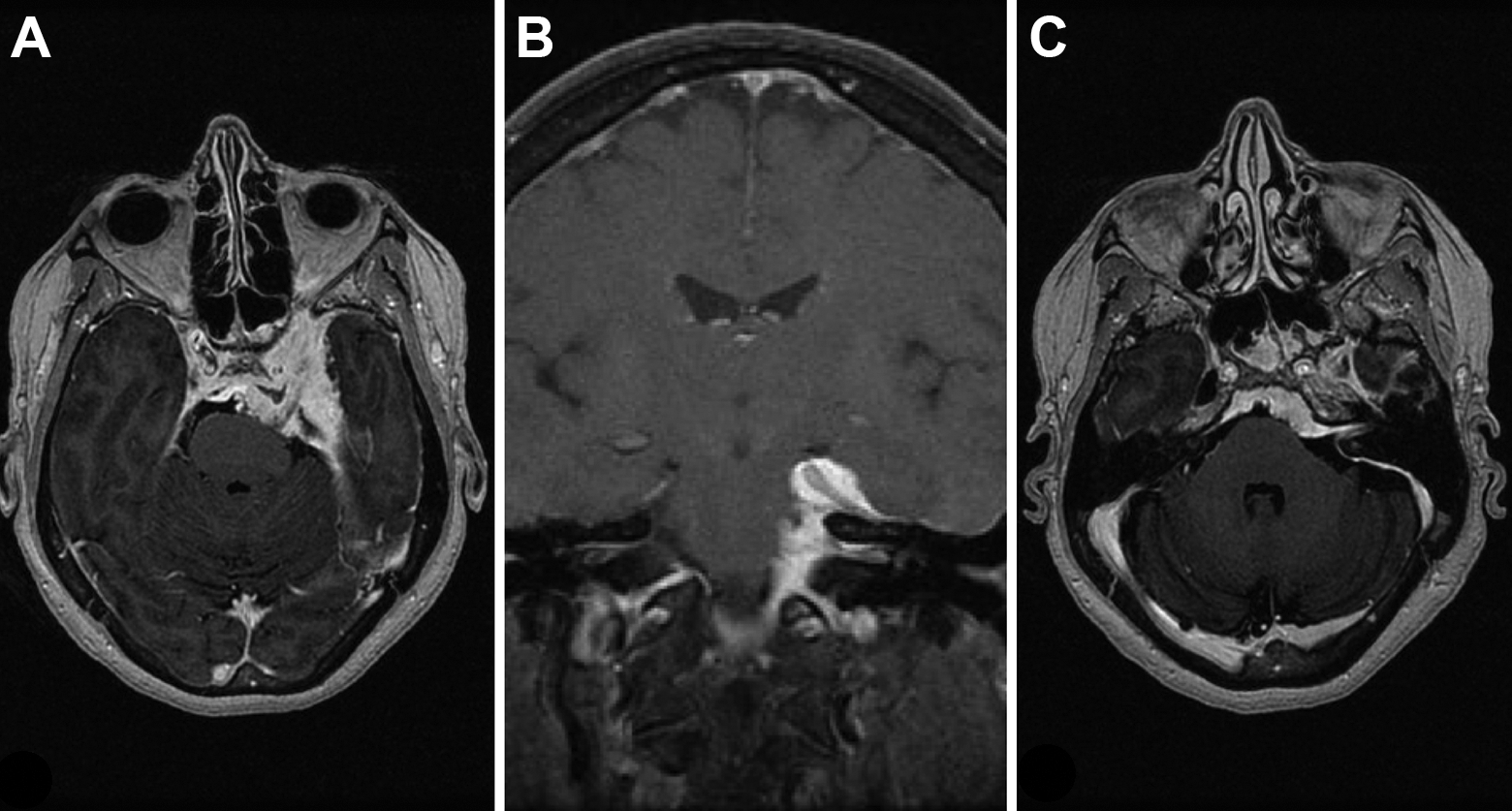
Stereotactic MRI of the brain. A, An axial cut depicting the size of the sphenoid wing meningioma and its mass effect on the brain stem. B, A coronal cut demonstrating the sphenoid wing meningioma extending into the internal auditory canal. C, An axial cut demonstrating the sphenoid wing meningioma extending into the internal auditory canal. MRI indicates magnetic resonance imaging.
The patient was then followed up in the outpatient setting. The TSA specimen sample revealed a final diagnosis of a WHO grade 1 meningothelial meningioma. The audiogram revealed severe left sensorineural hearing loss (SNHL) with a speech recognition threshold (SRT) of 85 dB and a word recognition score (WRS) of 0% at 95 dB. After discussing treatment options for the SNHL, including use of intratympanic steroids, the patient refused further medical or surgical intervention. Repeat audiogram 2 weeks after having received IV dexamethasone during her hospital admission showed no interval change in hearing improvement or worsening. Improved audiogram was noted at 4 weeks, revealing severe to moderate SNHL with SRT of 50 dB and WRS of 60% at 70 dB (Figure 2).
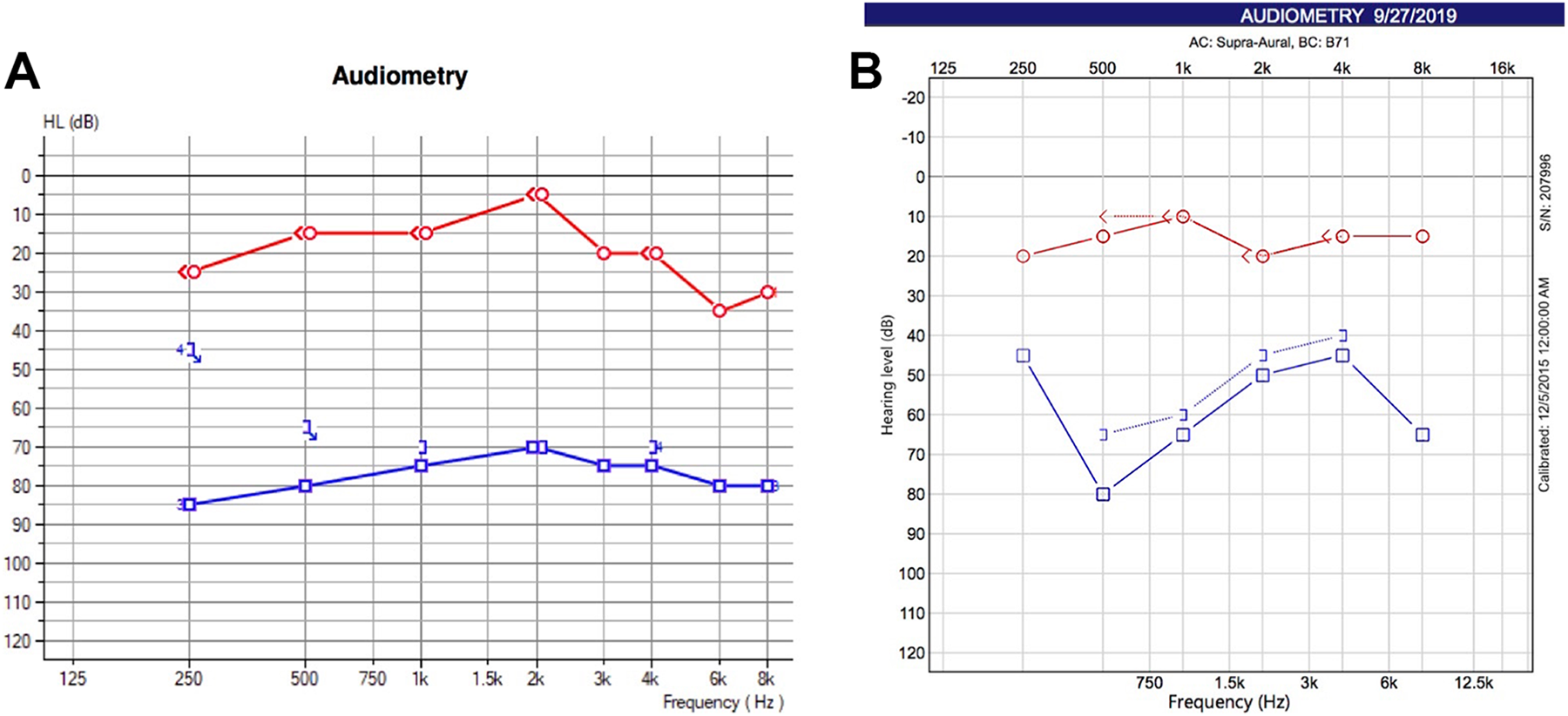
A, Initial audiogram. AD: mild high-frequency SNHL; SRT 15 dB, WRS 100% at 50 dB. AS: severe SNHL; SRT 85 dB, WRS 0% at 95 dB. B, Third audiogram after 1 month. Status: improved. AD: normal hearing; SRT 20 dB, WRS 100% at 40 dB. AS: severe to moderate SNHL; SRT 50 dB, WRS 60% at 70 dB. SNHL indicates sensorineural hearing loss; SRT, speech recognition threshold; WRS, word recognition score.
Discussion
Sphenoid wing meningiomas may present in multiple ways due to their potential involvement of vital structures in the brain including the cavernous sinus, cranial nerves, and internal carotid artery. 10 Proximity to these structures is the primary reason why these tumors typically manifest as neurological deficits, and most commonly, visual changes. Nonetheless, atypical symptoms have been described in the literature, including cognitive impairment, intracranial hemorrhage, and parkinsonian features. 7 -9,11 To our knowledge, this is the first report to describe SSNHL as the initial presentation of a patient with sphenoid wing meningioma.
Cognitive impairment as a presenting feature was first described by Saha and his colleagues and was determined to result from mass effect of the tumor on the frontal lobe in a patient with sphenoid wing meningioma. On examination, the patient demonstrated impaired reasoning, problem solving, and judgment. 7 Intracranial hemorrhage has also been reported in the setting of sphenoid wing meningiomas due to various proposed mechanisms including tumor infarction and secondary bleeding, tumor invasion into an artery, stretching and disruption of bridging veins, histamine-induced vasodilation, and venous hypertension from occlusion of venous sinuses. 8 In another patient, hemi-parkinsonism was reported as the presenting feature due to compression of the basal ganglia by a sphenoid wing meningioma. The patient was a 54-year-old female who developed resting tremor, cogwheel rigidity, bradykinesia, generalized seizures, decreased arm swing with gait, and bilateral papilledema. Her tumor was resected using a right frontotemporal craniotomy approach and her symptoms resolved in the postoperative period. 11
The patient in our report was found to have a large tumor centered in the left sphenoid wing with some evidence of extension into the internal auditory canal and encasement of the left CN VIII. Audiometry revealed left severe SNHL. For patients presenting with SNHL, regardless of the etiology, there is evidence to support the use of oral, IV, and intratympanic steroids for treatment of symptoms. 12 Particularly in patients with intracranial masses as the etiology, steroids may have the additional benefit of reducing tumor vasogenic edema that may be causing SNHL. Of note, intratympanic steroids are currently the standard of treatment since they can achieve higher concentrations of steroid in the cochlea, while maintaining lower levels in the systemic circulation and thus reducing the risk of secondary effects. 13,14 Clinicians may also opt for the use of hyperbaric oxygen as initial or salvage therapy. 13 The guidelines for the management of SSNHL were recently updated and described by Chandrasekhar et al. 13 Our patient received IV dexamethasone during her hospitalization and experienced some subjective and objective improvement in her hearing at 1 month interval following steroid administration. It remains unclear whether other patients with SNHL secondary to meningioma may achieve benefits from such therapy.
The close proximity of sphenoid wing meningiomas to critical neurovascular structures may pose a challenge for surgical resection. Various surgical approaches such as frontotemporal craniotomy and the cranio-orbitozygomatic approach for achieving complete resection of such tumors without compromising vital structures have been described in the literature. 15 In patients with tumors deemed unresectable, other management options including stereotactic radiosurgery and fractionated radiation therapy may also be considered. 16,17 More recently, clinical reports have also demonstrated achievement of complete resection with optic canal unroofing and clinoidectomy. 16
Conclusion
While sphenoid wing meningiomas most commonly present with visual symptoms, it is important for clinicians to remain cognizant of less frequent, but possible manifestations of these slow-growing tumors. Thus far, only a small number of cases have reported atypical presentations of sphenoid wing meningioma, including intracranial hemorrhage, cognitive deficits, and hemiparkinsonism. 7 -9,13,18
Footnotes
Authors’ Note
As per university standard, the patient’s written consent has been obtained.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
