Abstract
Background:
Aspirin-exacerbated respiratory disease (AERD) is characterized by eosinophilic rhinosinusitis, nasal polyposis, aspirin sensitivity, and asthma.
Materials and Methods:
Among 37 patients undergoing endoscopic sinus surgery, 28 received systemic steroids preoperatively. Nasal polyps were harvested from all 37 patients. After routine processing of paraffin sections, immunohistochemistry was performed using specific antibodies for HPGDS, eosinophil peroxidase (EPX), and DP2.
Results:
Expression of HPGDS, DP2, and EPX by eosinophils was higher and more frequent in patients with non-preoperative steroid therapy. Likewise, HPGDS and DP2 were highly expressed in activated eosinophils in the nasal polyps, but not in normal eosinophils.
Conclusion and Significance:
This study provides clear evidence that systemic steroid therapy inhibits eosinophil activation and decreases HPGDS and DP2 expression in patients with AERD, indicating a reduction in prostaglandin D2 production and hence control hyperplasia of nasal polyps.
Introduction
Aspirin-exacerbated respiratory disease (AERD) is a nonallergic hypersensitivity condition in which severe respiratory symptoms occur due to inhibition of cyclooxygenase (COX). It is characterized by the triad of aspirin sensitivity, asthma, and nasal polyps. 1,2
In AERD, inhibition of COX-1 by nonsteroidal anti-inflammatory drugs (NSAIDs) may lead to overproduction of cysteinyl leukotrienes (CysLTs) mediated by 5-lipoxygenase, 3,4 with both CysLTs and arachidonic acid metabolites being involved. Patients with AERD have high urinary levels of leukotriene E4 (LTE4), a marker of CysLT production, 4,5 and urinary LTE4 increases markedly after administration of NSAIDs including aspirin. 3,6 In addition, LTE4 is increased in the respiratory tract of patients with AERD after aspirin inhalation, 7 and the upper respiratory tract (including nasal polyps) is the main site of CysLT production in AERD. 4,8
Unlike CysLTs, the role of the COX system in AERD is unknown. Prostaglandin E2 (PGE2), a major COX metabolite in the urine and nasal polyps of patients with AERD was low levels. 3 Exacerbation of AERD may occur because endogenous PGE2 production is decreased due to reduced COX-2 expression, leading to overproduction of CysLTs and eicosanoid imbalance. 6,9 Prostaglandin D2 (PGD2), another major metabolite of the COX system, promotes allergic asthma. 9 It also accelerates eosinophil infiltration into the trachea, decreases airway hyperreactivity in PGD2 receptor–deficient mice, and exacerbates asthma in transgenic mice with increased PGD2 production. 8 However, little information is available about the COX system in AERD, and there have been reports about PGD2 synthase, 10 but few reports about the PGD2 receptor.
Hematopoietic PGD2 synthase (HPGDS) is a PGD2 synthase expressed by eosinophils. There are 2 receptors for PGD2, which are PGD2 receptors 1 and 2: DP1 and DP2 (PTGDR2, also known as chemoattractant receptor homologous molecule expressed on Th2 cells). Coupling of Gi protein with DP2 mediates PGD2 chemotaxis for lymphocytes such as Th2 cells and various inflammatory cells. 11 -13
Although steroids are effective for AERD, long-term administration is not recommended because of various adverse reactions. Effective treatment causing fewer reactions could possibly be developed by elucidating the mechanism through which steroids suppress AERD. Therefore, we investigated the role of PGD2 and the COX system in AERD by examining nasal polyps resected from patients with AERD.
Materials
The subjects comprised 37 patients with AERD and 4 patients with non-AERD who underwent endoscopic sinus surgery from July 2003 to April 2012. Both AERD and non-AERD cases were clinically diagnosed by history of asthma symptoms, respiratory reactions to NSAIDs, blood examination, nasal examination, and pulmonary function test. All patients with AERD had a history of asthma attack after NSAID administration. Among 37 patients with AERD, 28 patients were administered a steroid, whereas 9 patients were not administered a steroid before endoscopic sinus surgery. These 2 groups were not intentionally divided according to age, sex, laboratory findings, and symptoms/signs (Table 1).
Clinical Characteristics of the Patients With AERD With or Without Preoperative Steroid Therapy.a
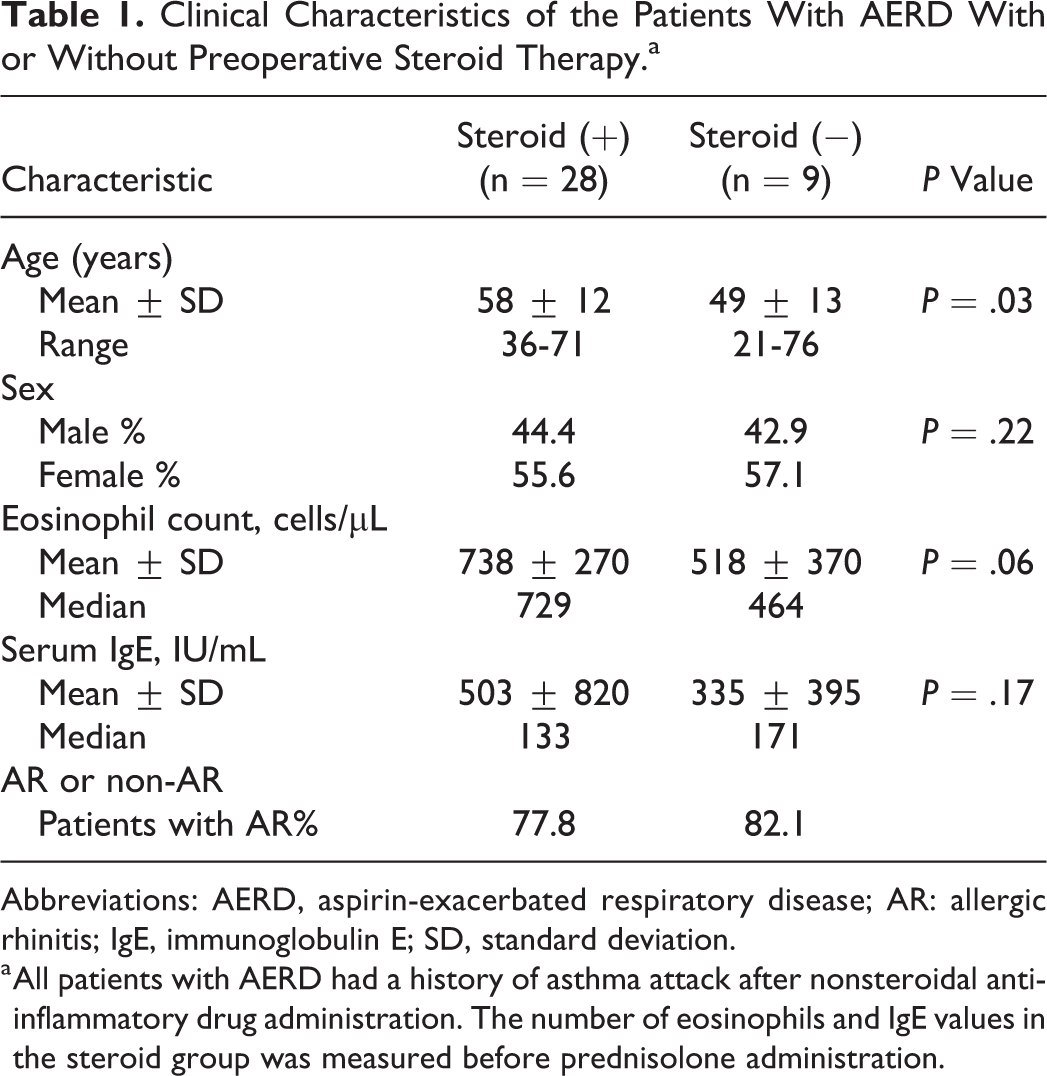
Abbreviations: AERD, aspirin-exacerbated respiratory disease; AR: allergic rhinitis; IgE, immunoglobulin E; SD, standard deviation.
a All patients with AERD had a history of asthma attack after nonsteroidal anti-inflammatory drug administration. The number of eosinophils and IgE values in the steroid group was measured before prednisolone administration.
Methods
Study Outline
We show the outline of this research method in Figure 1. The diagnosis of AERD and non-AERD according to clinical symptoms/signs was all confirmed by eosinophil infiltration and HPGDS expression in the nasal polyps of the patients. Among the 37 patients with AERD, 28 patients were administered prednisolone (5 mg/d, for 8 days) before endoscopic sinus surgery, whereas 9 patients were not administered prednisolone. Both groups of patients did not receive any steroid therapy for 3 months prior to this study. Nasal polyps were collected during the operation, and HPGDS, anti-eosinophil peroxidase (EPX), and DP2 expressions were studied along with eosinophil infiltration.
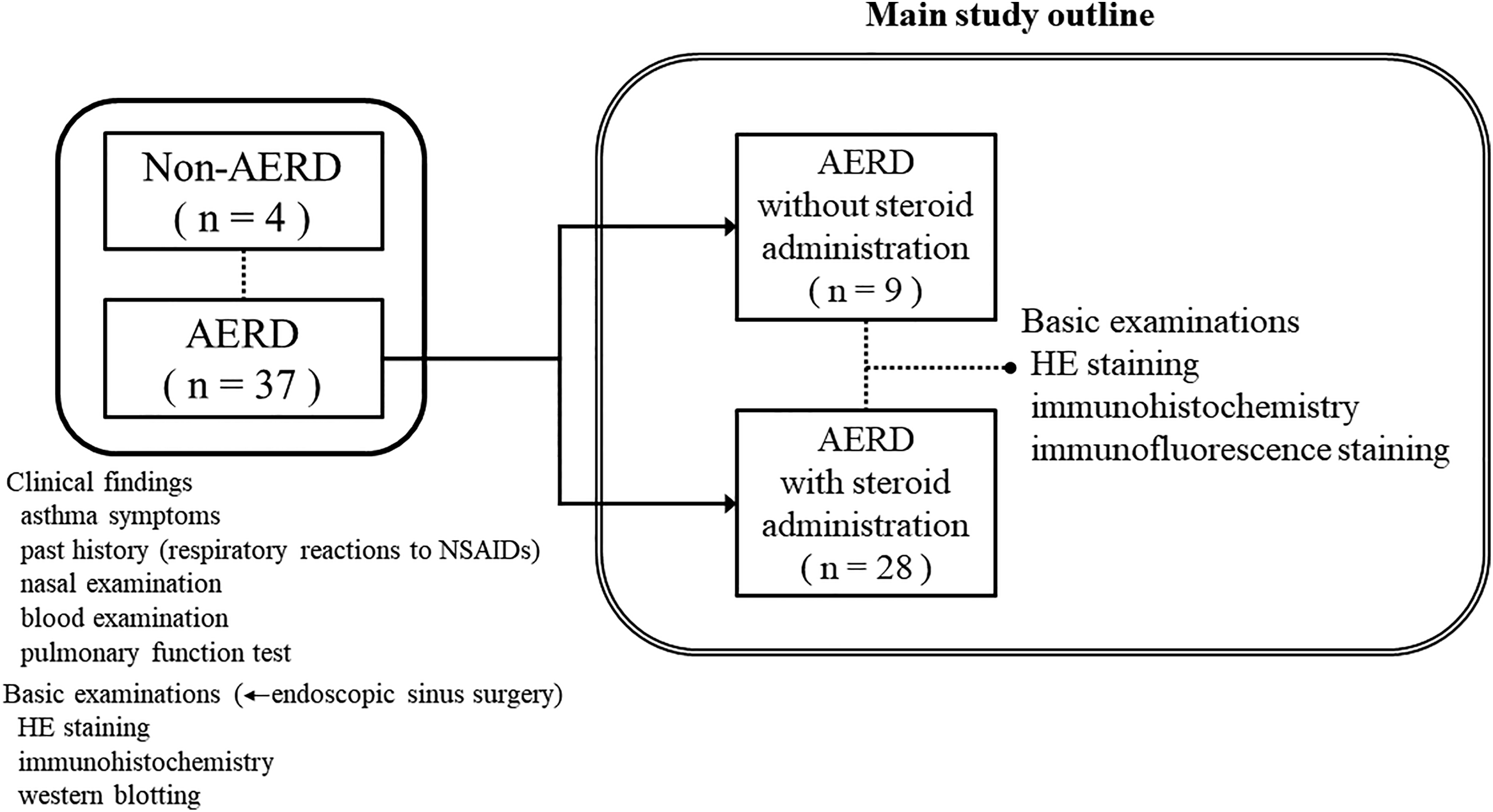
Outline of research method for this study. After blood examination, 28 patients with aspirin-exacerbated respiratory disease (AERD) among 37 were administrated prednisolone at 5 mg/d for 8 days prior to endoscopic sinus surgery.
Antibodies
A novel mouse monoclonal anti-human DP2 (anti-hDP2) was generated in Osaka Bioscience Institute, Department of Molecular Behavioral Biology (see details below). Other primary antibodies were as follows: rabbit polyclonal antibody against HPGDS and membrane-associated PGE synthase 1 (mPGES-1; Cayman Chemicals, Buffalo, New York), EPX antibody (Chemicon, Billerica, Massachusetts), rat monoclonal antibody (mAb) against CD11b, mouse mAb against CD16, and anti-HA tag antibody (Abcam, Cambridge, United Kingdom). A Dako EnVision HRP/DAB kit (Dako Japan, Tokyo, Japan) was used to stain paraffin-embedded sections.
Generation of hDP2 Antibody
A 19-mer peptide (CAASPQTGPLNRALSSTSS) at the C-terminal appendage of human DP receptor was designed, synthesized, coupled to thyroglobulin (Immuno-Biological Laboratories, Gunma, Japan), and administered subcutaneously to DP receptor KO mice (Balb/c) followed by a booster injection. Splenocytes from immunized mice were fused with P3U1 myeloma cells. Positive hybridoma clones were isolated by limiting dilution and IgG antibody was purified from the serum-free culture medium. The immunoglobulin isotype was determined with an IsoStrip Mouse Monoclonal Antibody Isotyping Kit (Roche Diagnostics, Mannheim, Germany), according to the manufacturer’s instructions.
Specificity of Generated Mouse Anti-hDP2 mAb
To investigate the specificity of the generated mouse anti-hDP2 mAb, HEK293 cells expressing HA tag receptors, hDP1, hDP2, mouse DP1 (mDP1), and mDP2 were stained consecutively with purified mouse anti-hDP2 IgG and anti-HA tag antibodies. The reactivity of the antibodies was visualized with microscope. Reactivity of the purified mouse hDP2 mAb found solely with cells expressing hDP2-HA reveals that the generated antibody is highly specific to hDP2 (Figure 1). Cells expressing hDP1, mDP1, or mDP2, though showed positive HA staining, showed no interaction with mouse hDP2 mAb (Figure 2).
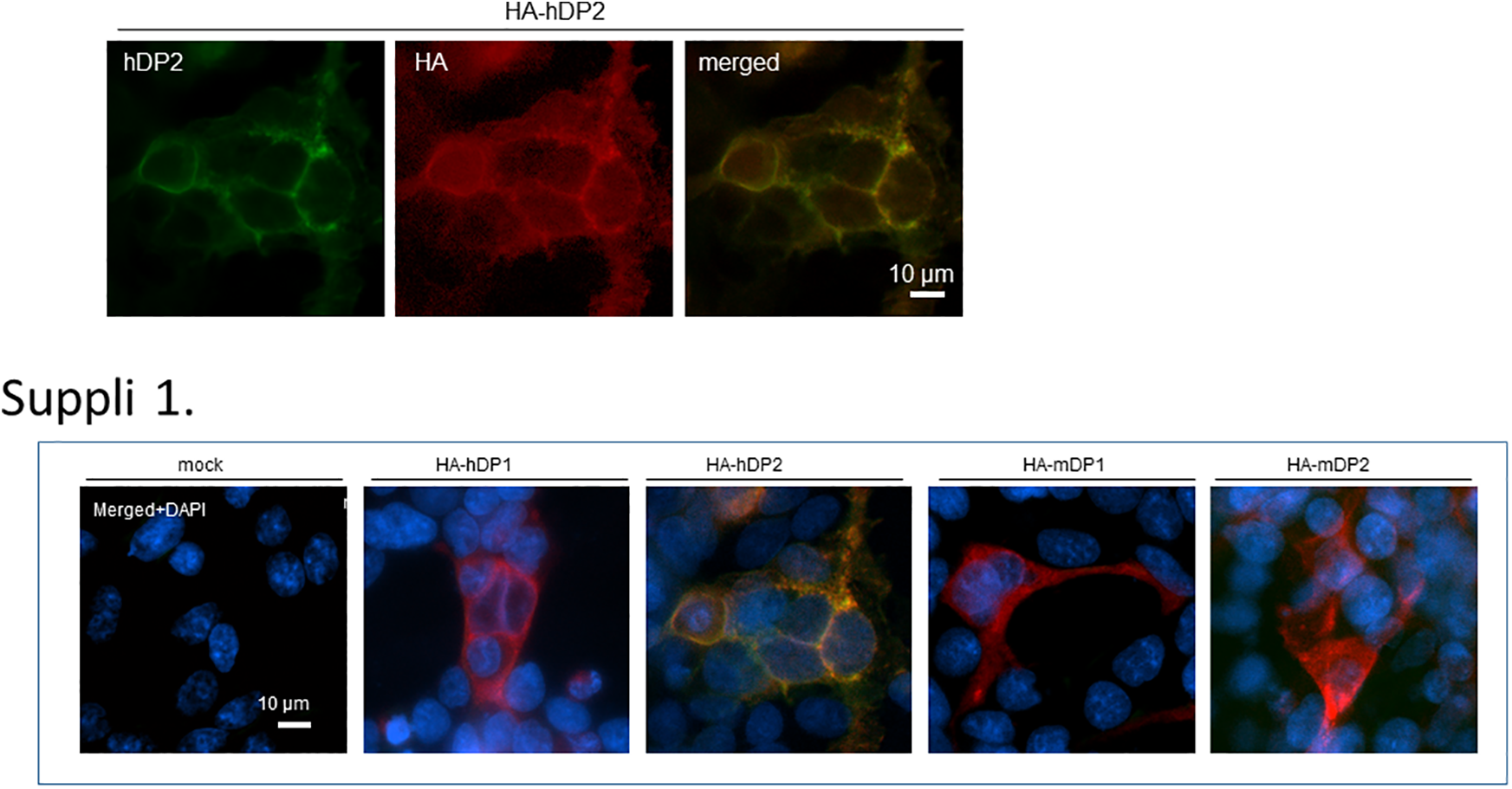
Immunofluorescent double staining of HEK293 cells with forced expression of human prostaglandin D2 (DP2) antibody with an HA tag (HA-hDP2). Staining with HA-hDP2 showed that the antibody and HA were highly expressed at the same sites in the cell wall (merged image). There was no reaction with HA-human DP1, HA-mouse DP1, or HA-mouse DP2, so the antibody reacted specifically with the human DP2 receptor.
Handling of Tissue Samples
Nasal polyps were collected during endoscopic sinus surgery. Each polyp was divided into 2 pieces, with a half snap-frozen at −80°C, whereas the other fixed in 10% neutral formaldehyde solution, embedded in paraffin, and sliced to 4-µm serial sections.
Assessment of Eosinophil Infiltration
Consecutive hematoxylin and eosin (HE)-stained sections were reviewed under an all-in-one fluorescence microscope (BZ-8000; Keyence, Itasca, Illinois). Infiltration of eosinophils in nasal polyps was compared between patients with AERD and non-AERD by taking the average from the number of eosinophils in 5 randomly selected microscopic fields (×400) per nasal polyp.
Immunohistochemistry
To investigate HPGDS and mPGES-1 expression, sections were deparaffinized with xylene and rehydrated in a graded alcohol series. After treatment in 0.3% H2O2 for 30 minutes, sections were incubated overnight at 4°C with antibodies for HPGDS (diluted 1:10 000) or mPGES-1 (diluted 1:1000) after blocking with 10% goat serum for 1.5 hours. The tissues were then incubated with EnVision+ HRP labeled polymer secondary antibody (Dako) for 1 hour and the protein expression was visualized using diaminobenzidine tetrahydrochloride. Images were obtained with a BZ-8000 microscope (Keyence).
Western Blotting
Nasal polyps in phosphate-buffered saline containing protease inhibitor cocktail (complete EDTA-free; Roche, Basel, Switzerland) were homogenized by using a handheld homogenizer (Taitec, Koshigaya, Japan). Proteins in the supernatant after centrifugation were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis on 10%:20% gradient gel and transferred to polyvinyl difluoride membrane (Immobilon; Millipore, Billerica, Massachusetts) by electrophoresis at 100 mA for 1.5 hour. The membranes were incubated with blocking buffer (5% skim milk in Tris-buffered saline [500 mM NaCl, pH 7.5] with 0.05% Tween 20) for 1 hour at 25°C before incubating overnight with HPGDS antibody (diluted 1:2000) at 4°C. The membrane was then treated with enhanced chemiluminescence detection reagent (Amersham Biosciences, Little Chalfont, Buckinghamshire, United Kingdom) and exposed to autoradiography film (Kodak XOMAT AR film; Eastman Kodak, Rochester, New York).
Immunofluorescence for Eosinophils
Eosinophils in the nasal polyps of steroid and nonsteroid groups were identified by immunofluorescent double staining with CD11b and CD16 antibodies. Expression of EPX, HPGDS, and DP2 was visualized by immunofluorescent double staining in eosinophils. Eosinophil peroxidase may be a marker of eosinophilic inflammation and is used to assess the activity of eosinophils. CD11b is a marker of monocytes, granulocytes, and natural killer cells, and eosinophils were identified as CD16-negative granulocytes with the typical eosinophil morphology. Sections were incubated with primary antibodies for HPGDS (1:5000), EPX (1:500), DP2 (1:500), CD11b (1:1000), or CD16 (1:1000), followed by incubation with respective secondary antibody (AlexaFluor488 donkey anti-mouse IgG [1:1000], AlexaFluor594 donkey anti-rabbit IgG [1:1000], or AlexaFluor594 donkey anti-rat IgG [1:1000]; Abcam). Then nuclei were stained with 4′,6′-diaminodino-2-phenylindole (ProLong Gold; Cell Signaling Technology, Tokyo, Japan), and the staining was visualized with an inverted fluorescence microscope (DMI6000AFC; Leica, Wetzlar, Hesse, Germany).
Positive cells were counted in 5 randomly selected microscopic fields (×400), and the average total number of positive cells was expressed as percentage over the total number of eosinophils per nasal polyp.
Statistical Analysis
The significance of differences between 2 groups was calculated by using the Mann-Whitney U test for unpaired data and a probability (P) value <.05 was considered statistically significant.
Ethical Considerations
This retrospective study was approved by the institutional review board of Osaka Medical College Hospital.
Results
Nasal Polyps of AERD and Non-AERD Patients
The HE-stained sections showed markedly greater eosinophil infiltration in the nasal polyps of patients with AERD as compared with non-AERD patients (Figure 3, upper). Immunostaining revealed high HPGDS expression in AERD polyps (Figure 3, lower), whereas mPGES-1 expression was too low to be assessed in both cases (data not shown). Furthermore, Western blotting confirmed that HPGDS expression was higher in AERD polyps (lanes 3 and 4) than in non-AERD polyps (lanes 1 and 2; Figure 3, lower right).
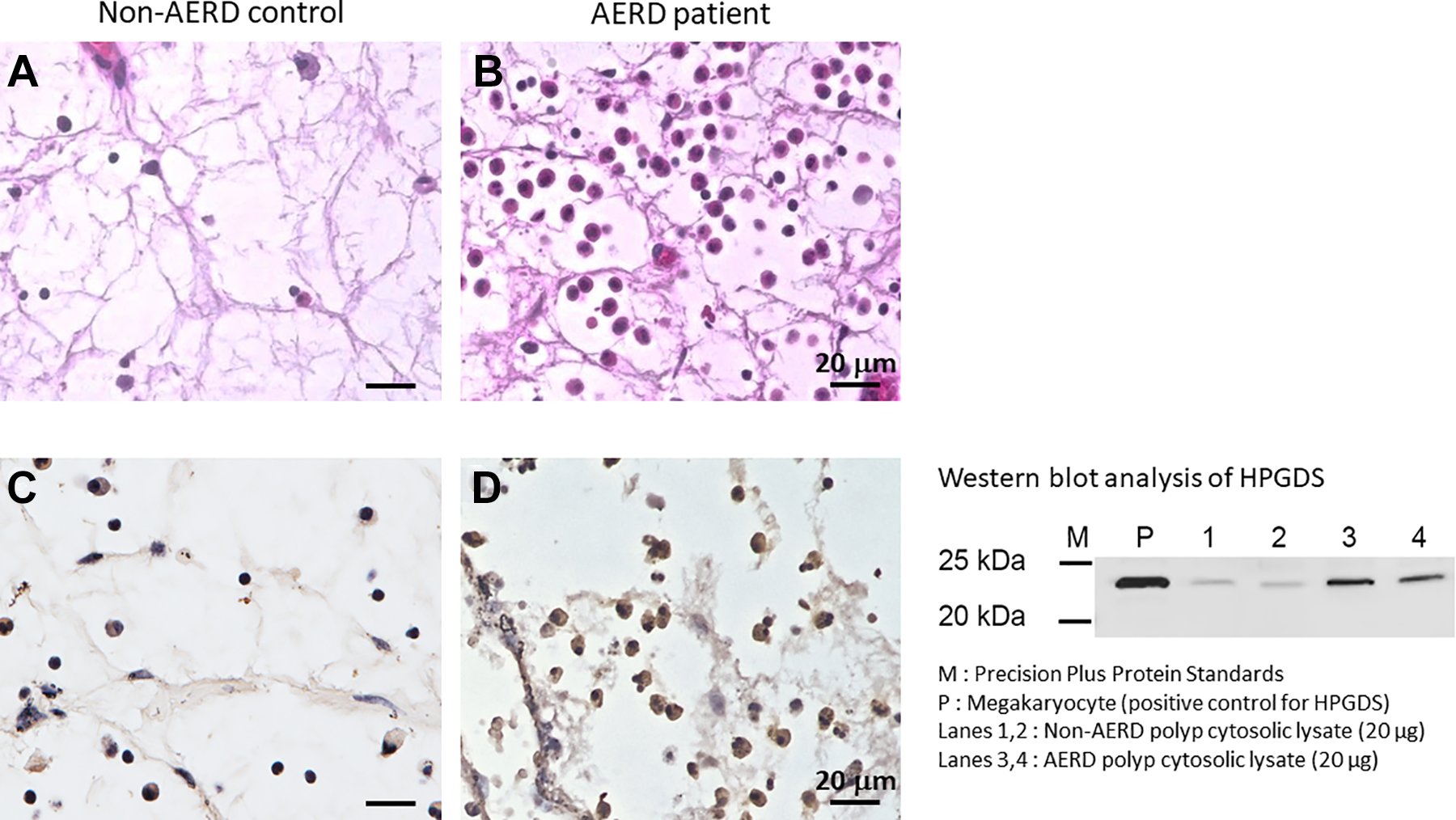
Comparison of nasal polyps from aspirin-exacerbated respiratory disease (AERD) and control patients. Marked eosinophil infiltration in a polyp from a patient with AERD compared with that from a control patient (A, B; HE staining). Hematopoietic prostaglandin D2 synthase (HPGDS) showed higher expression in the nasal polyp from a patient with AERD compared with that from a control patient (C, D; immunohistochemistry). Western blotting for HPGDS showed higher expression in AERD nasal polyps (lanes 3 and 4). P, positive control; lanes 1 and 2, nasal polyps from patients with non-AERD.
Localization of HPGDS and DP2 Expression, and Activated Eosinophils
Eosinophil peroxidase is a marker of activated eosinophils 14 in AERD nasal polyps, whereas HPGDS is an enzyme in the downstream of COX pathway. Immunofluorescent double staining with HPGDS and EPX showed that both proteins were not only highly expressed but also localized in the same cells, indicating that HPGDS was expressed in activated eosinophils (Figure 4, upper). Moreover, DP2 also colocalized with HPGDS (Figure 4, lower). These results indicated that high expression of both HPGDS and DP2 is concurrent with the occurrence of activated eosinophils infiltrating AERD nasal polyps.
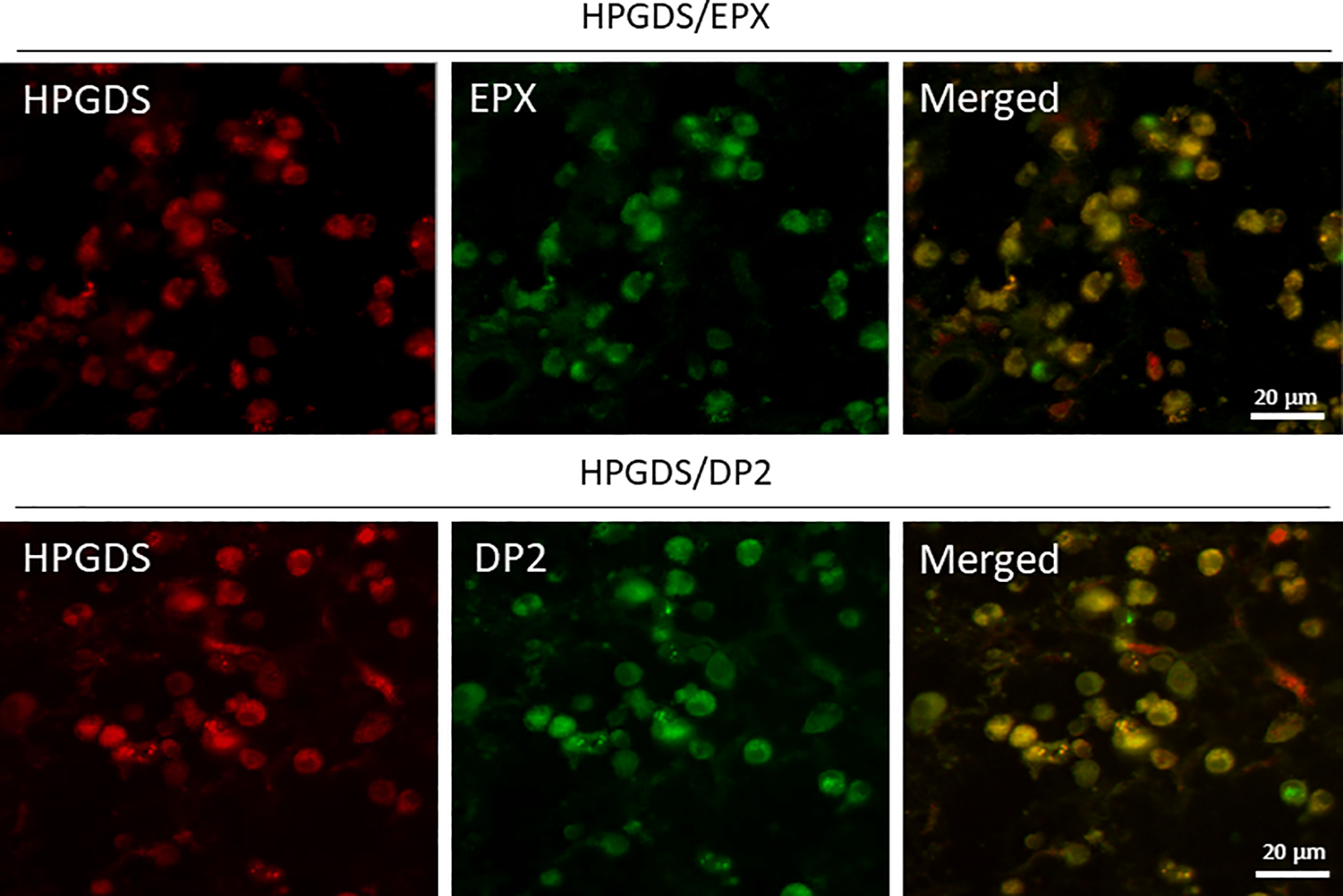
Immunofluorescent double staining of a nasal polyp from a patient with aspirin-exacerbated respiratory disease (AERD; ×400). Hematopoietic prostaglandin D2 synthase (HPGDS) and eosinophil protein X (EPX), a marker of activated eosinophils, were highly expressed and were generally localized in the same cells (upper panels). Prostaglandin D2 (DP2; the receptor for PGD2) was also highly expressed and was generally localized in the same cells (lower panels).
Effect of Steroid Therapy
Whether steroid therapy renders influence on the expression of HPGDS, DP2, and EPX was investigated in polyps from patients with AERD with or without preoperative steroid therapy. Indeed, HPGDS and EPX were markedly decreased in the steroid group (Figure 5, left), although HPGDS was expressed by residual activated (EPX-positive) eosinophils, that is, EPX-positive cells corresponded to HPGDS-positive cells and inactive eosinophils rarely expressed HPGDS (Figure 4, left). Likewise, DP2 expression was markedly inhibited by preoperative steroids, and cells without HPGDS expression corresponded to those not expressing DP2 (Figure 5, right). These results inferred that steroid therapy suppressed the expression of HPGDS and DP2 as it did in decreasing the prevalence of activated eosinophils in nasal polyps.
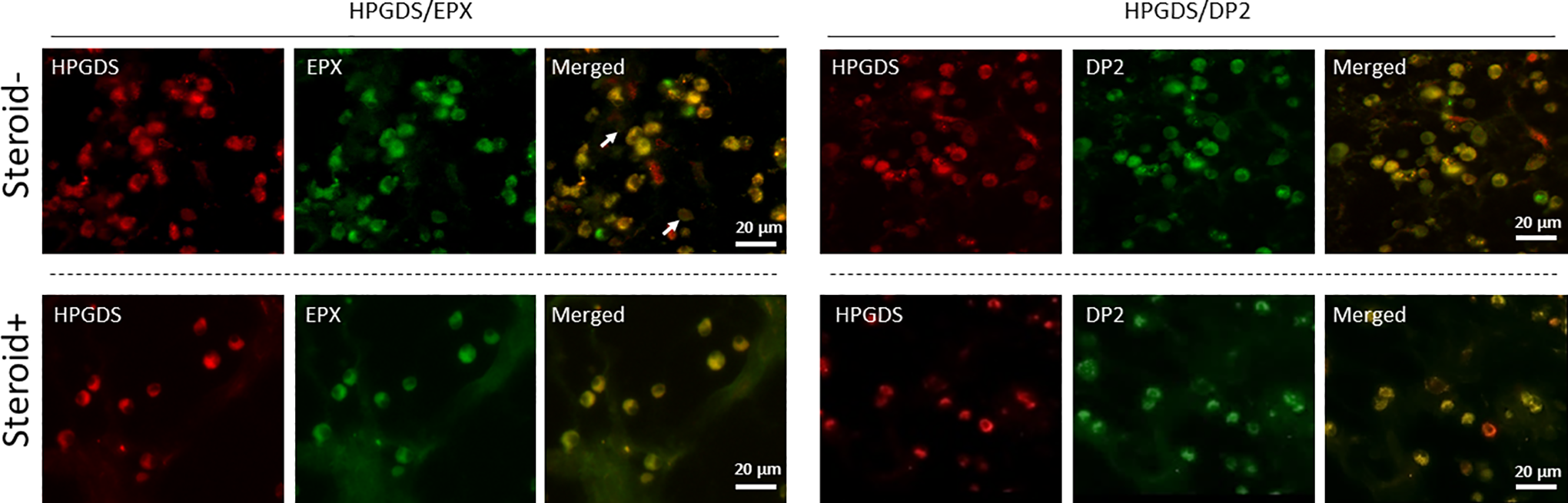
Influence of steroid therapy on hematopoietic prostaglandin D2 synthase (HPGDS), eosinophil protein X (EPX), and prostaglandin D2 expression. Left panels: Immunofluorescent double staining (×400) revealed a significant decrease in cells positive for HPGDS and EPX in nasal polyps from the steroid group compared with the nonsteroid group. Hematopoietic prostaglandin D2 synthase and EPX were generally expressed in the same cells, and HPGDS + EPX + eosinophils were decreased by steroid administration. White arrows indicate EPX-positive cells. Inactive eosinophils rarely expressed HPGDS. Right panels: Cells positive for HPGDS and DP2 were significantly decreased in nasal polyps from the steroid group compared with the nonsteroid group. Hematopoietic prostaglandin D2 synthase and DP2 were generally expressed in the same cells, and HPGDS + DP2 + eosinophils were decreased by steroid administration.
Decrease of HPGDS or DP2 Expression and Activated Eosinophils by Steroid Therapy
Taken that the above results showed preoperative steroid therapy had markedly inactivated eosinophils with subsequent marked decrease in the expression of HPGDS and DP2, we further investigated the effects of steroids by statistical analyses (Figure 6). The total eosinophil count per nasal polyp was 143.3 in the nonsteroid group and 39.6 in the steroid group, showing a 73.3% decrease. In addition, the number of activated eosinophils (EPX-positive cells) per nasal polyp was 96.6 in the nonsteroid group and 14.1 in the steroid group (85.5% decrease). Furthermore, cells expressing HPGDS decreased from 115.2 to 14.9 (87.1% decrease) and cells expressing DP2 decreased from 118.9 to 15.2 (86.2% decrease).
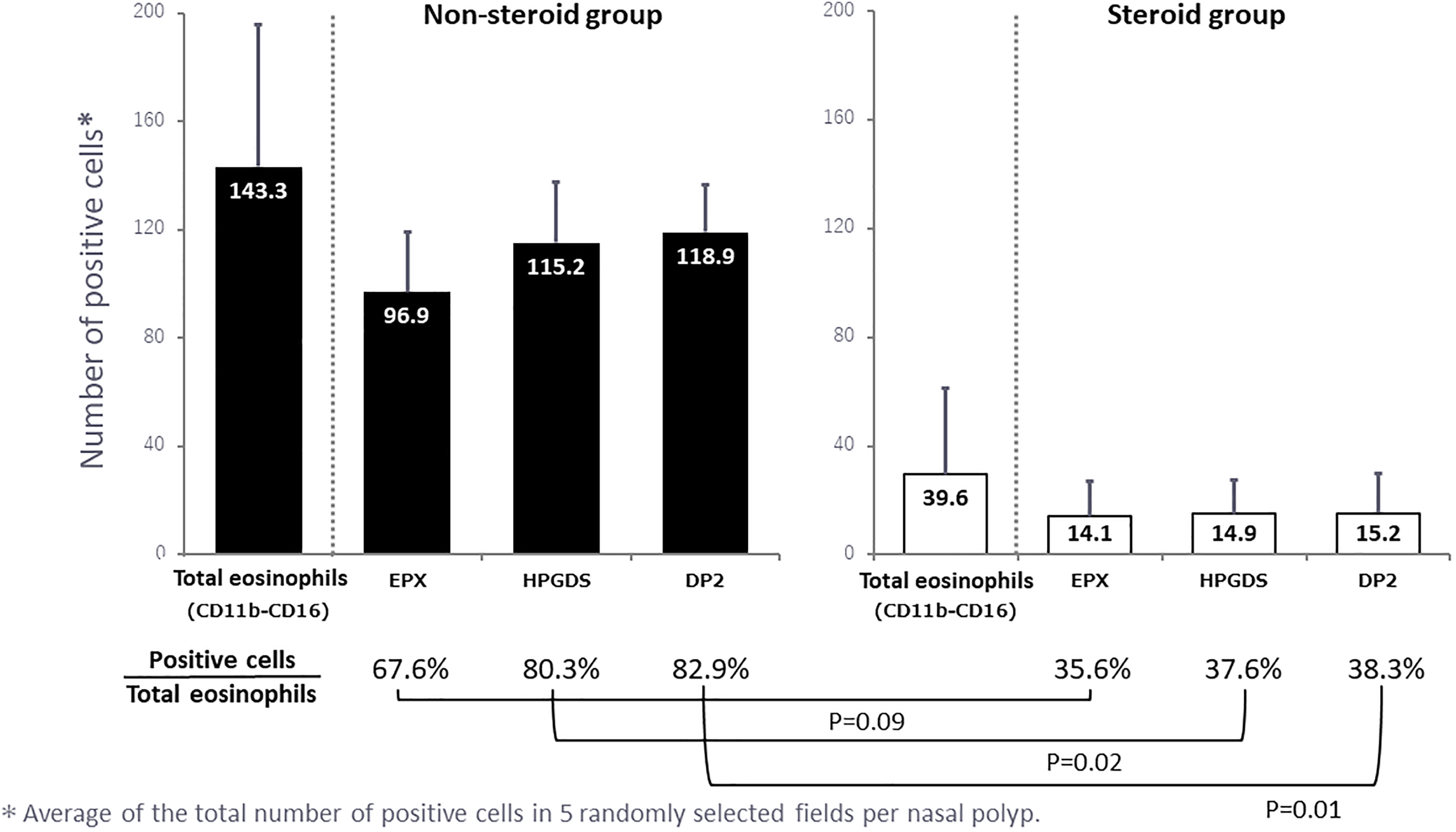
Quantitative data for the steroid and nonsteroid groups. The number of cells positive for each marker was counted in 5 randomly selected microscopic fields (×400) per polyp and the mean values were calculated. In addition, the number of positive cells was expressed as a percentage of the total eosinophil count and the results are shown below the graphs.
When the percentage of cells positive for HPGDS, EPX, and DP2 was calculated relative to the total eosinophil count, the decrease of each cell type in the steroid group was more marked than the actual decrease of the eosinophil count (Figure 6). In particular, the rates of HPGDS- and DP2-positive cells were 80.3% and 82.9% in nonsteroid group, whereas in steroid group, the rate of each positive cells was significantly low, 37.6% and 38.3%, respectively.
Discussion
The main findings of the present study were as follows: (1) HPGDS and DP2 were highly expressed in activated eosinophils in nasal polyps; (2) steroid therapy decreased eosinophil infiltration in nasal polyps, thus leading to a greater reduction in activated eosinophils over total eosinophil count; (3) steroid therapy also markedly reduced the expression of EPX, HPGDS, and DP2 in activated eosinophils in nasal polyps; and (4) the decrease in the percentage of eosinophils expressing EPX, HPGDS, and DP2, particularly the latter two, was more marked than the decline in the eosinophil count in response to steroid therapy.
Thus, steroid therapy selectively suppressed activated eosinophils and might concurrently lead to the inactivation of PGDS and DP2 involved in PGD2 metabolism, one of the downstream of the COX pathway. As for dosage of the steroid in this study, considering the effects on the whole-body and intraoperative effects (such as bleeding), administration of the steroid was set to a small dose (5 mg/d). Because the average body weight of 28 recipients was 53.6 kg, which is lighter than Westerners, the dose may increase when we calculate the dose as per body weight.
The expression of COX-2 is decreased in nasal polyps from patients with AERD, resulting in the decreased production of anti-inflammatory PGE2. Prostaglandin E2 has both anti-inflammatory and bronchodilatory effects, and it inhibits 5-lipoxygenase. 15 The decreased production of anti-inflammatory lipoxins has also been pointed out in AERD. The present study revealed a low mPGES expression in nasal polyps, thus suggesting a decrease in PGE2 production. In contrast, HPGDS was overexpressed, so production may have switched from anti-inflammatory PGE2 to pro-inflammatory PGD2. Reduced PGE2 production may lead to the overproduction of PGD2 via the COX system, and PGD2 could be important for initiating AERD along with an overproduction of CysLTs.
Both CysLTs and PGD2 were reported to cause the exacerbation of asthma. 9 Prostaglandin D2 may act via the DP receptor because allergic symptoms are alleviated by a DP receptor antagonist, 16 while asthma is aggravated in transgenic mice with increased PGD2 production. 9 Because PGD2 activates eosinophils and promotes LTC4 release, 17 it may be involved in leukotriene metabolism like PGE2. 15
Nasal polyps were resected from our patients with AERD when they had no asthma attack, suggesting that PGD2 production by COX-1 is constitutively elevated because Daham et al reported that COX-1 is the main source of PGD2 in humans. 18 Therefore, we can explain the mechanism of AERD exacerbation from the aspect of the COX-PGD2 system because NSAIDs generally inhibit both COX-1 and COX-2.
Nasal polyps of eosinophilic chronic rhinosinusitis (ECRS) are important in AERD. 19 Tissue levels of nonspecific IgE are increased in eosinophilic nasal polyps of patients with AERD. 20 In AERD, arachidonic acid metabolites are derived from nasal polyps 6 and urinary LTE4 decreases markedly after the resection of polyps. These mediators are derived from cells infiltrating nasal polyps, thus emphasizing the important role of eosinophilic polyps in AERD.
Mast cells were reported to be the major source of PGD2 in eosinophilic inflammation. 21 However, this study and previous investigations have found few mast cells in nasal polyps, unlike prominent eosinophil infiltration. In addition, activated eosinophils show high expression of HPGDS and DP2, while steroid therapy inhibits these cells, suggesting that PGD2 is involved in AERD. We previously found high expression of HPGDS in eosinophilic polyps. 22 Feng et al showed that the gene expression of eosinophils from tissue with AERD increased levels of HPGDS. 10 In this study, activated eosinophils showed high expression of both HPGDS and DP2, therefore suggesting that these cells may produce both PGD2 and its receptor in AERD.
In this study, we showed that HPGDS and DP2 are overexpressed in nasal polyps and that the HPGDS/DP2 system was inhibited by steroid therapy. Both HPGDS and DP2 were highly expressed in the same activated eosinophil. Moreover, the rates of HPGDS- and DP2-positive cells were 80.3% and 82.9% in the nonsteroid group, whereas in the steroid group, the rate of each positive cells was significantly low, 37.6% and 38.3%, respectively. Therefore, we have found that the PGD2 system plays important roles in the pathophysiology of AERD. However, it is still unknown what kind of roles the PGD2 system takes in the overview of AERD.
Conclusion
Previous investigations of ECRS have identified prominent eosinophil infiltration in nasal polyps. Activated eosinophils in nasal polyps show increased expression of HPGDS and DP2, whereas steroid therapy strongly inhibits these cells. Accordingly, new HPGDS inhibitors or more effective DP2 antagonists could possibly become additive treatments for ECRS to reduce dependence on steroids.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
