Abstract
Background:
Aspirin-exacerbated respiratory disease (AERD) is characterized by asthma, chronic rhinosinusitis with polyps, and sensitivity to aspirin (ASA). Optimal treatment requires coordinated medical and surgical management, with prior studies showing that a single-center approach to AERD management yields improved sinonasal patient outcomes. Here, we sought to evaluate whether institutional experience enhances pulmonary and sinonasal outcomes in patients with AERD undergoing functional endoscopic sinus surgery (FESS) followed by ASA desensitization (AD).
Methods:
Single-center, retrospective cohort study evaluating patients undergoing FESS and AD from 2016 to 2024. Cohorts were defined as “early” (2016-2019) and “late” (2020-2024). Demographics, clinical history, and quality of life metrics [eg, 22-Item SinoNasal Outcome Test (SNOT-22) score] were assessed. Cohorts were compared with statistical significance defined as P < .05.
Results:
Two hundred sixty-two patients (n = 145 early, 117 late) met inclusion criteria. The late cohort was younger (mean 49.6 vs 56.4 years, P = .042) with fewer prior surgeries (1.33 vs 2.64, P ≤ .001) at the time of referral. Results showed a reduced need for inhaled corticosteroids and beta-agonists in the late cohort and improved SNOT-22 rhinologic sub-scores in the early post-treatment period (post-FESS, pre-AD; 2-3 months post-treatment; 4-6 months post-treatment), with a reduced need for revision surgery (0% vs 6.9%, P = .010).
Conclusions:
Greater institutional experience in AERD management yields significantly improved pulmonary outcomes and more rapid improvement in sinonasal outcomes. This was felt to be due to enhanced communication and coordination of care between the allergist and rhinologist, with implications for healthcare expenditures.
Plain Language Summary
Greater institutional experience in the management of aspirin-exacerbated respiratory disease results in a reduced need for inhaled asthma therapies and improved early sinonasal quality of life, with a reduced need for revision sinus surgery.
Keywords
Introduction
Aspirin-exacerbated respiratory disease (AERD) is a chronic inflammatory disease characterized by chronic rhinosinusitis with nasal polyps, asthma, and sensitivity to aspirin (ASA) and other non-steroidal anti-inflammatory drugs. AERD affects more than 7% of adults with asthma but is often underdiagnosed due to its complex symptomatology.1,2 Symptoms typically begin with sinonasal symptoms followed by lower respiratory disease, with variable timing of onset of ASA sensitivity onset.3,4 Due to the severity of the disease, overall quality of life (QOL) is significantly impaired in patients with AERD.4,5 Furthermore, definitive diagnosis may take time due to the asynchronous nature of symptom onset, making increased awareness, early diagnosis, and timely evidence-based intervention crucial to improved outcomes in AERD management. 3
Treatment of AERD requires close collaboration between the treating rhinologist and allergist to coordinate therapy, 6 which includes complete functional endoscopic sinus surgery (FESS) followed by ASA desensitization (AD)7-9 as the standard of care at our institution.10-12 The University of Pennsylvania AERD Center, founded in 2016, offers a multidisciplinary single-center approach to the management of this complex patient population and is housed in the Department of Otorhinolaryngology – Head & Neck Surgery, Division of Rhinology. Patients undergo medical and surgical work-up and treatment within the same clinic by a single allergist (J.V.B.) and a small group of rhinologists (J.N.P., N.D.A., M.A.K., and J.E.D.).
Our prior study demonstrated improved sinonasal patient outcomes following the development of this multidisciplinary, single-center treatment model. 13 Herein, we sought to better understand whether patient outcomes may be similarly enhanced with greater institutional and provider experience. It was hypothesized that both pulmonary and sinonasal patient outcomes would correlate with institutional experience, emphasizing the importance of close collaboration between allergist and rhinologist in the care of patients with AERD.
Materials and Methods
A single-center retrospective cohort study reviewing all patients undergoing FESS followed by AD at the University of Pennsylvania AERD Center between 2016 and 2024 was performed. Subjects were excluded if their immediate pre-desensitization surgery was not performed at the University of Pennsylvania. Patients were divided into 2 cohorts based on the date of surgery. The “early” cohort was defined as those undergoing treatment from 2016 to 2019, and the “late” cohort was defined as those undergoing treatment from 2020 to 2024. Patient demographics, preoperative and postoperative medication use, operative reports, need for revision surgery, and results of the 22-Item SinoNasal Outcome Test (SNOT-22), 14 pre- and serially post-treatment, were reviewed. The date of surgery was used as the date of the index event.
Differences in continuous variables for the cohorts were analyzed utilizing 2 sample t-tests, and categorical variables were analyzed utilizing chi-square tests of independence. The extent of frontal surgery was compared using a 2-proportion z-test. SNOT scores were compared using Wilcoxon rank sum tests. Statistical significance was defined as P < .05. All statistical analyses were performed using R (version 4.1.2, R Foundation for Statistical Computing, Vienna, Austria). Institutional review board approval was obtained from the University of Pennsylvania with a waiver of informed consent due to the retrospective nature of the study.
Results
A total of 262 patients (early cohort: n = 145, late cohort: n = 117) were identified. Patients undergoing treatment in the late cohort were on average younger (49.6 vs 56.4 years of age, P = .042) compared with the early cohort. There were no significant differences in patient sex, pre-operative biologic use, Asthma Control Test (ACT) score, and total SNOT-22 and rhinologic subscores between cohorts. The number of prior sinus surgeries in the late cohort was on average lower (1.33 vs 2.64, P ≤ .001) compared to the early cohort. Analysis of operative reports revealed no significant difference in the extent of frontal sinus surgery (Draf IIb vs Draf III) between cohorts. Notably, the time to AD in the early cohort was longer compared with the late cohort (17.8 vs 10.1 weeks, P = .004; Table 1).
Patient Characteristics.
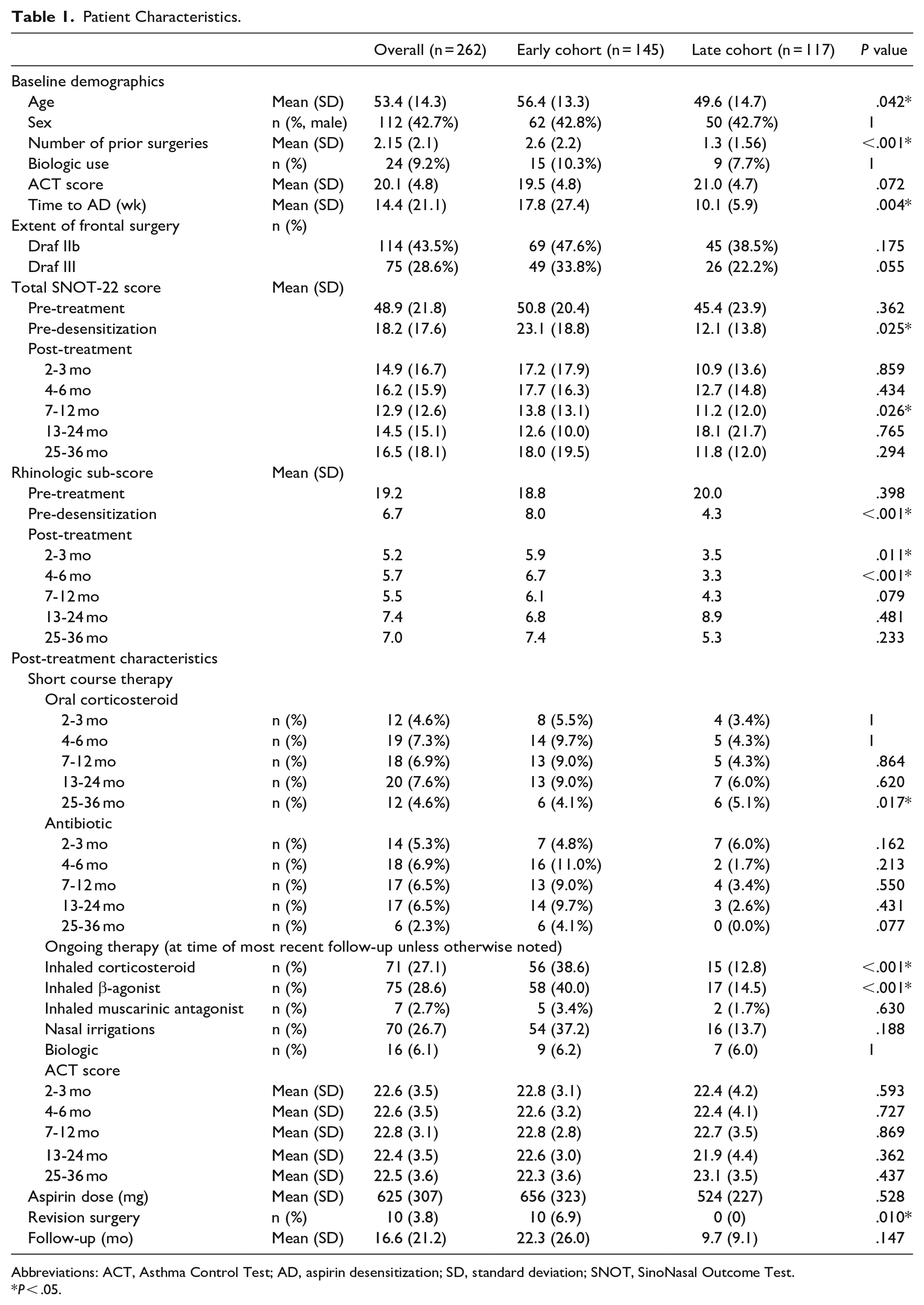
Abbreviations: ACT, Asthma Control Test; AD, aspirin desensitization; SD, standard deviation; SNOT, SinoNasal Outcome Test.
P < .05.
Analysis of post-treatment medication use (Table 1) revealed no significant difference in short-course antibiotic or oral corticosteroid therapy, apart from at the 25 to 36 month time point when a slightly increased rate of steroid burst use was found in the late cohort. There were significant reductions in the use of inhaled corticosteroids and beta-agonists for asthma control in the late cohort relative to the early cohort (corticosteroids: 12.8 vs 38.6%, P < .001; beta-agonists: 14.5 vs 40.0%, P < .001) at the time of most recent follow-up. Of the 24 patients on a biologic pre-treatment, 10 were able to discontinue therapy post-treatment. Sixteen patients started biologics following treatment. This was done for improved asthma control in 50% of patients and improved sinonasal control in the remainder of patients. There were no significant differences between cohorts.
The SNOT-22 rhinologic sub-score was significantly reduced (ie, improved) in the late cohort at 3 time-points: post-FESS/pre-AD (Δ-3.76, P ≤ .001), 2 to 3 months post-treatment (Δ-2.4, P = .011), and 4 to 6 months post-treatment (Δ-3.37, P < .001). There was no difference in rhinologic sub-score at 7 to 12, 13 to 24, and 25 to 36 months post-treatment. This was reflected in the total SNOT-22 scores, which were significantly reduced in the late cohort at 2 time points: post-FESS/pre-AD (Δ-10.9, p < .001) and 2 to 3 months post-treatment (Δ-6.9, p < .001). There was no significant difference at 4 to 6, 13 to 24, or 25 to 36 months post-treatment (Table 1 and Figure 1). While both cohorts exhibited a statistically improved post-treatment ACT score compared with pre-treatment, the degree of improvement was not significantly different between cohorts. There was a notable and significant reduction in the need for revision surgery in the late cohort (0% vs 6.9%, P = .010). The mean daily ASA dose at last follow-up was not significantly different between the cohorts (524 vs 656 mg, P = .528). The average follow-up duration was 16.6 months.
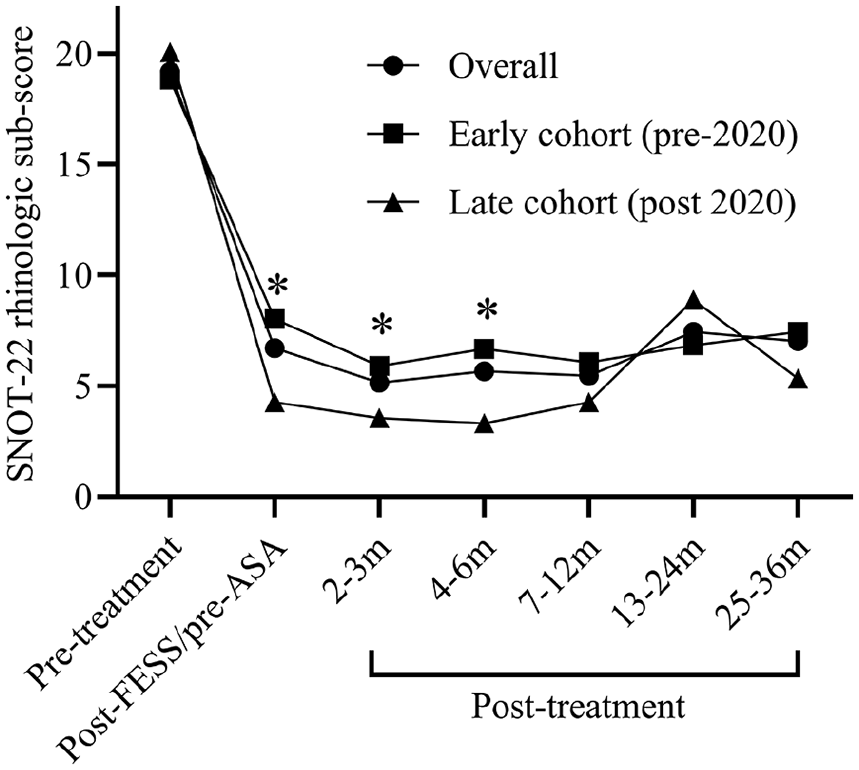
Pre- and post-treatment SNOT-22 rhinologic sub-scores.
Discussion
We previously showed that a multidisciplinary, single-center approach to the management of AERD results in improved sinonasal patient outcomes. 13 Herein we demonstrate that within our single-center model, greater institutional experience results in significantly improved pulmonary outcomes post-treatment (as measured by a reduced need for both corticosteroid and beta-agonist inhalers) and a more rapid improvement in sinonasal QOL (as measured by the SNOT-22 score). Notably, the enhanced pulmonary metrics occurred in the setting of no significant differences in ACT score, as both cohorts exhibited well-controlled asthma pre- and post-treatment. Improved sinonasal outcomes were largely driven by the rhinologic sub-score, and were nearly 3 times the minimally clinically important difference for the test of 8 points, indicating profoundly improved QOL. Importantly, patients are now being referred to our institution at an earlier age, allowing for increased quality-adjusted life years post-treatment. The time to AD is also reduced in the late cohort and exhibits lower variability, suggesting more streamlined transitions of care. A significantly reduced rate of revision surgery also correlated with greater institutional experience. Given that revision surgery is a significant driver of increased costs in ambulatory sinus surgery, 15 this study has important implications for healthcare expenditures.
The etiology of this improvement in pulmonary and sinonasal outcomes with institutional experience is likely multi-factorial and may reflect optimized clinical workflow. Importantly, the reduced time to AD could be contributing. Early AD may capitalize on reduced sinonasal and systemic inflammation in the early post-operative period to yield quicker and more durable improvements post-treatment. At our center, providers utilize a shared clinic space, facilitating synchronous visits, streamlined communication, and real-time decision-making. While this was true in both cohorts, the learning curve of multi-disciplinary collaboration is well-established,16,17 and may improve patient compliance with treatment and follow-up. In addition, clinical support staff gain familiarity with treatment protocols. While our study did not directly evaluate this question, patient feedback regarding the multidisciplinary team approach was overwhelmingly positive.
Limitations of the study include the trend toward shorter follow-up duration in the late cohort, which could impact our ability to detect a difference in need for revision surgery or biologic therapy. However, this did not meet statistical significance. In addition, the single-center nature of the study limits generalizability; however, given the significant volume of patients treated at the center, this study provides important data as to mechanisms for improving the care of patients with AERD.
In summary, increased institutional experience and a single-center model for the management of AERD yield favorable pulmonary and sinonasal outcomes and may have the potential to reduce overall healthcare expenditures. Our experience suggests that a longitudinal relationship between treating allergist and rhinologist is of utmost importance to enhance communication and coordination of care for this complex patient population.
Footnotes
Author Note
The project was presented at the 2024 American Rhinologic Society at the Combined Otolaryngology Spring Meetings.
Ethical Considerations
This study received ethical approval from the University of Pennsylvania Institutional Review Board (protocol #854217) on August 22, 2023.
Consent to Participate
Written informed consent was waived by the IRB.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Thomas B. McCabe and Jeannette E. Laws McCabe Fund at the University of Pennsylvania.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the study are available from the corresponding author on reasonable request.
