Abstract
Chronic rhinosinusitis with nasal polyps is a multifactorial disease of the nasal and paranasal sinus mucosa and it includes, as comorbidities, anatomic and morphologic alterations, allergic rhinitis, and immunologic diseases. We investigated matrix metalloproteinases (MMP-2, MMP-7, and MMP-9) and their tissue inhibitors (TIMP-1 and TIMP-2) concentration in different etiopathogenetical groups of patients with nasal polyposis (NP) in relation to recurrence after sinonasal surgery. The study group consisted of 45 patients with NP (those with allergic rhinitis, nonallergic rhinitis and asthma or nonallergic rhinitis, and obstruction of osteomeatal complex [OMC]) who underwent endonasal sinus surgery. We also collected 10 patients who underwent septoplasty as control. Immunohistochemistry of nasal mucosa fragments, Western blotting, and polymerase chain reaction analysis showed increased MMPs levels (MMP-9 more than MMP-2 and MMP-7) and decreased tissue inhibitors of MMPs levels (TIMP-1 less than TIMP-2), in patients with chronic rhinosinusitis with nasal polyps compared with control group, in particular in patients with nonallergic rhinitis and asthma compared to those with allergic rhinitis and nonallergic rhinitis and obstruction of OMC. We observed a higher risk of recurrence in patients with nonallergic rhinitis and asthma than in those with allergic rhinitis and nonallergic rhinitis and obstruction of OMC after 36 months from surgery. In this research, we evaluated pathogenesis of NP related to MMPs and their inhibitors concentrations in polypoid tissue.
Keywords
Introduction
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a multifactorial disease of the sinonasal mucosa characterized by the infiltration of inflammatory cells (eg, eosinophils, lymphocytes, and plasma cells), 1 modifications of epithelial cell differentiation, and tissue remodeling including basement membrane thickening, gland modifications, edema, and extracellular matrix (ECM) accumulation. 2
Chronic rhinosinusitis with nasal remains one of the most difficult challenges in otorhinolaryngology as its etiology and pathophysiology are still unclear, its medical treatment is unsatisfactory, and its frequent recurrences lead to repeated surgical interventions. 3 Different comorbidities, such as osteomeatal complex (OMC) obstruction, allergic rhinitis, and other diseases (ie, Widal syndrome, Churg-Strauss syndrome, etc), are linked to CRSwNP. 3 -7 Multifactorial etiology is also observed in polyposis or multiple adenoma of another district like bowel. 8
In CRSwNP, the biomorphological and molecular conditions that induce polyps formation are today objects of study, but recently some authors have suggested that ECM degradation and synthesis, basement membrane thickening, and rupture are the major factors that determine the shift from chronic inflammation to polyposis. 9 -12
The most important components of the proteolytic system involved in the physiologic remodeling of ECM are matrix metalloproteinases (MMPs) and their tissue inhibitors (TIMPs). Extracellular matrix integrity is ensured by a constant and dynamic balance between synthesis and degradation of its components. 13 Earlier studies have investigated the possible role that these proteins play in the etiopathogenesis of sinonasal polyposis. 14
However, no studies to date have investigated the mechanisms of degradation and synthesis of ECM and the concentrations of MMPs and TIMPs in the different etiopathogenetical forms of CRSwNP in relation to recurrence. Therefore, we investigated (1) MMPs and TIMPs expression in polyps of patients with CRSwNP; (2) MMPs and TIMPs tissue expression in the different etiopathogenetical groups of patients with CRSwNP; and (3) the relationship between the recurrences of CRSwNP after sinonasal surgery and the expression of MMPs and TIMPs.
Methods
Patients were enrolled in the Head and Neck Surgery Unit, between March 2013 and April 2018. The study group consisted of 45 patients (30 males and 15 females, range age of 22-67, median age 40.2 years) affected by CRSwNP, who underwent endonasal sinus surgery (ESS). Middle turbinate mucosae from 10 patients (5 males and 5 females) undergoing septoplasty were collected as controls in patients affected by CRSsNP. All the patients of the study group were evaluated with nasal fiberoptic endoscopy (2.7 mm in 0°, 30°, 45°, and 70°), high-resolution computed tomography (CT) scans; allergen-specific responses were characterized by paper radioimmunosorbent test (PRIST) and radio allergosorbent test (RAST), and skin-prick tests.
Patients and controls were assigned to 4 study groups as follows: Fifteen patients with CRSwNP and allergic rhinitis (group A), 11 polysensitive and 4 monosensitive; Twelve patients with CRSwNP and nonallergic rhinitis with asthma (group B), 7 with aspirin-exacerbated respiratory disease (AERD) or Widal disease or Samter Triad (nasal polyposis [NP], aspirin hypersensitivity, and bronchial asthma), 5 with Churg-Strauss disease (granulomatous angiitis, eosinophilia, NP, and asthma); Eighteen patients with CRSwNP and nonallergic rhinitis and OMC obstruction (group C), such as septum deviation and concha bullosa of the middle turbinate (12 patients), paradoxical curve of the middle turbinate (3 patients), turbinal–septal contact with OMC obstruction (3 patients, 2 of whom with associated paradoxical curve of the middle turbinate); Ten patients with nasal septum deviation and inferior turbinate hypertrophy and concha bullosa of middle turbinate (3 patients) with CRSsNP (control group).
Patients with AERD and Churg-Strauss were included in the same group (B) based on the common pathophysiological mechanism, the endotype underplayed by Th2-type cellular response.
All the patients of the study group had clinical signs of chronic sinusitis (congestion, rhinorrhea, and, in some cases, facial pain) for more than 3 months. These signs had not improved after more than 2 cycles of medical treatment including systemic antibiotics and topical steroids for at least 14 days.
Nasal polyps from all patients were collected during surgery of the ethmoid and/or the maxillary sinus; tissue specimens from control were obtained from the head of the middle turbinate mucosa. All samples were collected and stored in accordance with the requirements of the Hospital and University Ethics Committee and in conformity with the principles stated in the Declaration of Helsinki. Specimens were divided into 3 parts and used for paraffin embedding, molecular, and cellular biology, respectively. Each experiment was performed in triplicate.
After 1 month from surgery, the patients of each group underwent a specific therapy: Group A (patients with allergic rhinitis) underwent a topic medical treatment with fluticasone furoate, 2 puffs (50 mg) each nostril in the morning, for 40 days every 2 months, and oral antihistamines (rupatadine 10 mg 1 tablet die) for 40 days every 2 months; Group B (patients affected by nonallergic with asthma) was treated with fluticasone furoate, 2 puffs (50 mg) each nostril in the morning, for 40 days every 2 months, and antileukotrienes (montelukast 10 mg 1 tablet), each day for 40 days, every 2 months; Group C (patients with nonallergic rhinitis and OMC obstruction) underwent only isotonic nasal saline douches, every day for 15 g every month.
Follow-up included clinical evaluation at 1, 3, 6, 9, and 12 months after surgery and then every 6 months for 3 years; CT scan control was performed every 12 months. Every patient underwent clinical control with nasal endoscopy during each follow-up session. Lund-Kennedy Endoscopic score and Lund-Mackay CT score were collected at 12, 24, and 36 months after surgery in order to evaluate recurrence rate.
The expression of the MMP-9, MMP-7, MMP-2, TIMP-1, and TIMP-2 was investigated, in each sample by immunohistochemistry, Western blot, and reverse transcription polymerase chain reaction (RT-PCR) at the Morphology Unit of the Department of Medicine and Health Sciences “Vincenzo Tiberio” of the University of Molise (Campobasso, Italy) as detailed below.
Immunohistochemistry
Fragments obtained from the sinonasal mucosa were fixed in buffered 10% formalin, embedded in paraffin, and sectioned. Serial 4-μm-thick sections of these specimens were deparaffinized and treated for immunoperoxidase staining. 15 Sections were covered with primary monoclonal antibodies against MMP-2 (1:500), MMP-7 (1:250), and MMP-9 (1:200) (Sigma, St Louis, Missouri), and polyclonal antibodies against TIMP-1 (1:500) (Sigma) and TIMP-2 (1:200; Santa Cruz Biotechnology, Santa Cruz, California). After several washes in phosphate-buffered saline (PBS), sections were covered with peroxidase-conjugated immunoglobulin (Dako, Carpentaria, California) and incubated in a moist chamber for 45 minutes at 37°C, then washed again in PBS and incubated with avidin–biotin peroxidase (ABC/HRP; Vector Laboratories, Burlingame, California) for 30 minutes. The dye reaction was obtained by incubating for 10 minutes in a solution of diaminobenzidine tetrahydrochloride containing 0.03% of hydrogen peroxide to neutralize endogenous peroxidases. Slides were finally washed in water, counterstained with hematoxylin, rewashed in water, and then mounted in aqueous glycerol solution. Samples were observed with a Leica DMLB (Leica Microsystem, Bannockburn, Illinois, USA) connected to a PC by means of a digital Leica LC200 photo camera, by 3 pathologists who scored immunopositivity as (−) for absence and (+/++/+++) for the presence of staining.
Western Blot Analysis
Specimens were washed in PBS for 30 minutes at 4°C with lysate buffer (1% Triton X-100 in PBS), containing the following protease inhibitors: EDTA 10 mL, phenylmethylsulfonyl fluoride 10 mL, trypsin/chymotrypsin 10 mL, leupeptin 4.6 mL, E-64 3.6 mL, and aprotinin 10 mL. Lysates were centrifuged at 10 000 rpm for 30 minutes at 4°C. Supernatants were harvested and protein concentrations in cell lysates were determined by a colorimetric assay (Bio-Rad, Richmond, Colorado). For each sample, 100 µg of total proteins were incubated in Laemmli sample buffer and separated by electrophoresis in 30% sodium dodecyl sulfate-polyacrylamide gels. Gels were electroblotted on polyvinylidene difluoride filters (Millipore, Bedford, Massachusetts); membranes were colored with Ponceau. 16 After several washes in PBS-Tween 20 to remove exceeding coloring agent, membranes were incubated in Petri capsules and blocked for 1 hour in fat-free milk. After 3 passages in washing solution, membranes were incubated overnight at 4°C with the specific primary monoclonal antibodies against MMP-2, MMP-7, MMP-9, TIMP-1, and TIMP-2. After addition of a radish peroxidase-conjugated anti-mouse goat immunoglobulin G secondary antibody (Bio-Rad), diluted 1:3000 in PBS (Bio-Rad), membranes were incubated for 45 minutes at room temperature. After several washes, membranes were stained with an enhanced chemiluminescence (ECL) detection kit (Amersham Biosciences, Little Chalfont, Buckinghamshire, United Kingdom) for MMP-2, MMP-7, MMP-9, and TIMP-1 detection and ECL plus (Amersham Biosciences) for TIMP-2. A quantitative analysis was performed using scanner densitometry (SnapScan 1212, AGFA). Images were analyzed using the ImageQuant software (Amersham Biosciences).
Semiquantitative RT-PCR
Specimens (150-300 mg) were cryopreserved at −80°C and then lysated with Triazol (Life Technologies, Rockville, Maryland) to obtain total RNA. Total RNA was isolated, precipitated, and measured with an Eppendorf Bio Photometer (Eppendorf AG, Germany) with absorbance parameters from 260 to 280 nm. The estimated ideal absorbance ratio at 260 nm (DNA) versus 280 nm (proteins) was 1.8. The quality of RNA in all specimens was confirmed with a 2% agarose gel, which allows visualizing the 28S and 18S bands as markers of RNA integrity. Each total RNA sample was retrotranscripted to complementary DNA (cDNA) using First-Strand cDNA Synthesis (Amersham Pharmacia Biotech, Arlington Heights, Illinois). Each cDNA sample (3 µL) was amplified by PCR using target gene primers and primers for the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) for internal control. 17 Primer sequences for RT-PCR were designed using the software Primer 3 (available online at http://simgene.com/Primer3; Figure 1). The PCR mixes were run for end point amplification in a Techgene Thermal Cycler (Kraler Scientific Inc, Waltham, Massachusetts, USA) with the following parameters: denaturation at 95°C for 5 minutes; 25 cycles at 95°C for 1 minute, 55°C for 1 minute (annealing temperature), and 72°C for 1 minute; and 72°C for 10 minutes. The reaction total volume was 50 µL: 3 µL cDNA, 10 to 20 pmol each primer, 200 mM each deoxynucleoside triphosphate, 1.5 mM MgCl2, 20 mM reaction buffer, and RNAse- and DNAse-free H2O. The amplified products (12 µL of each sample) were analyzed by electrophoresis in a 2% agarose gel containing ethidium bromide, followed by photography under ultraviolet illumination. The levels of target messenger RNA were estimated by densitometric scanning and normalized against GAPDH-loading controls. Densitometric analyses of the PCR products were performed using the ImageJ v1.49q software (available online at http://imagej.net). Each PCR product was purified with QIAquick PCR purification kit (Qiagen, California) and the sequence was verified using the dideoxy method with Applied Biosystem Model 373A (Applied Biosystem, Foster City, California).
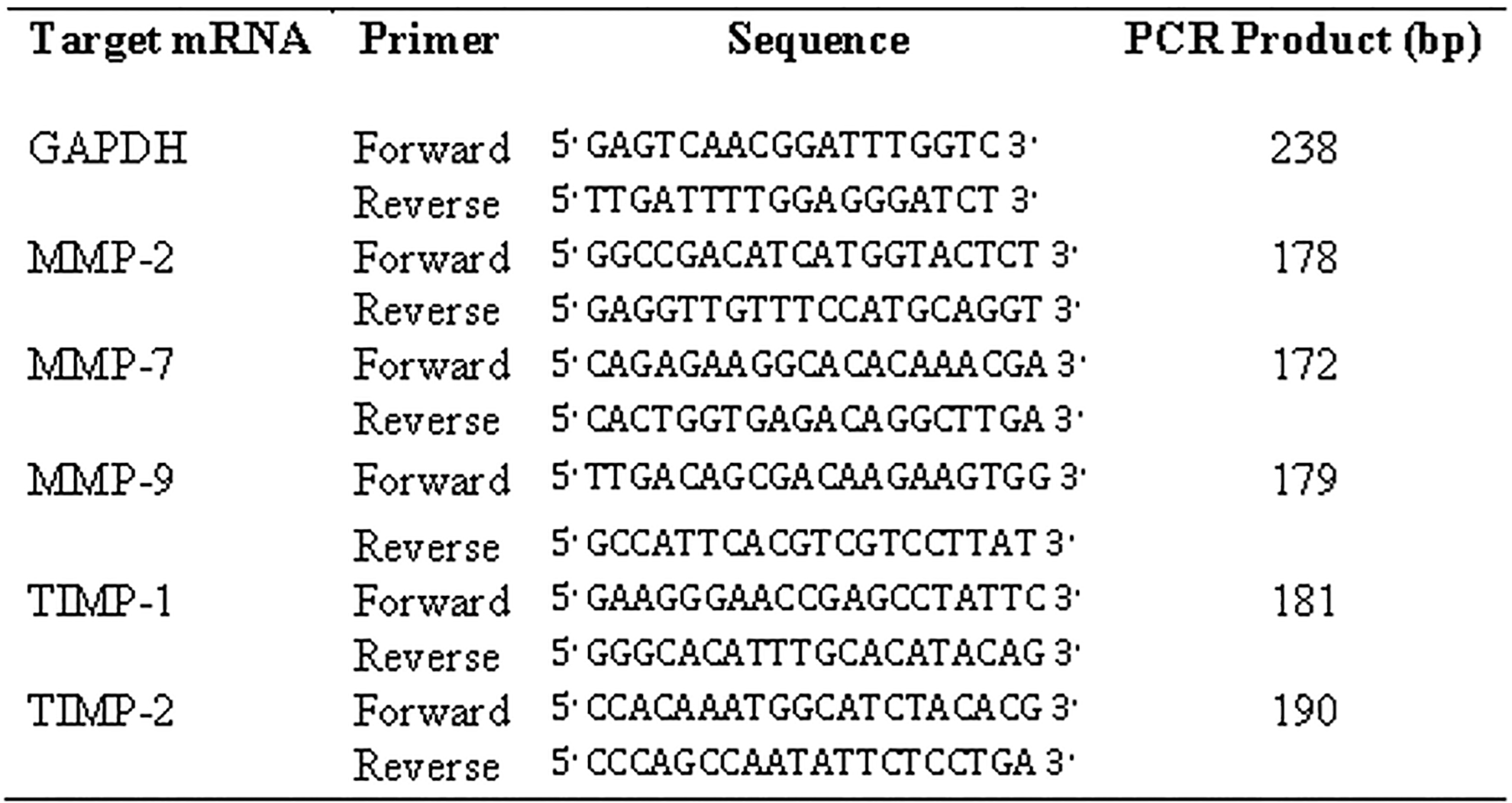
Primer sequences for reverse transcription polymerase chain reaction.
Statistical Analysis
The data were analyzed using the SPSS software version 24.0 (SPSS Inc, Chicago, Illinois). Data are expressed as the mean ± standard deviation. Rhinofiberendoscopy and CT data at follow-up were analyzed using the χ2 test. Statistical analysis for immunohistochemical data, Western blot, and RT-PCR was performed using the Mann-Whitney U test, and differences having a P value <.05 were considered significant.
Results
Tissue concentrations of individual MMPs and TIMPs were assessed separately in 3 individual groups of patients (A, B, and C) having CRSwNP (see “Methods” section) and in a control group and these results are summarized in Table 1. These results show an increase in concentrations of MMP-9, MMP-7, and MMP-2 as well as a reduction in the concentrations of TIMP-1 and TIMP-2 in samples from patients with CRSwNP compared to controls (P < .001). Concentrations of MMPs appear to be greater in the group B (patients with nonallergic rhinitis and asthma) compared to group A (patients with allergic rhinitis) and in group C (patients with nonallergic rhinitis and OMC obstruction; P < .001). On the other hand, the concentrations of TIMPs were reduced in patients assigned to group B compared to patients assigned to groups A and C (P < .001). It is interesting to note that MMP-9 appears to be expressed at higher concentrations than the other 2 MMPs studied (MMP-2 and MMP-7), while TIMP-1 is expressed at lower levels than TIMP-2. The concentrations of MMPs and TIMPs in these 3 groups (A, B, and C) of patients with CRSwNP and in the control group are shown in Figure 2 for RT-PCR analysis and in Figure 3 for Western blotting analysis.
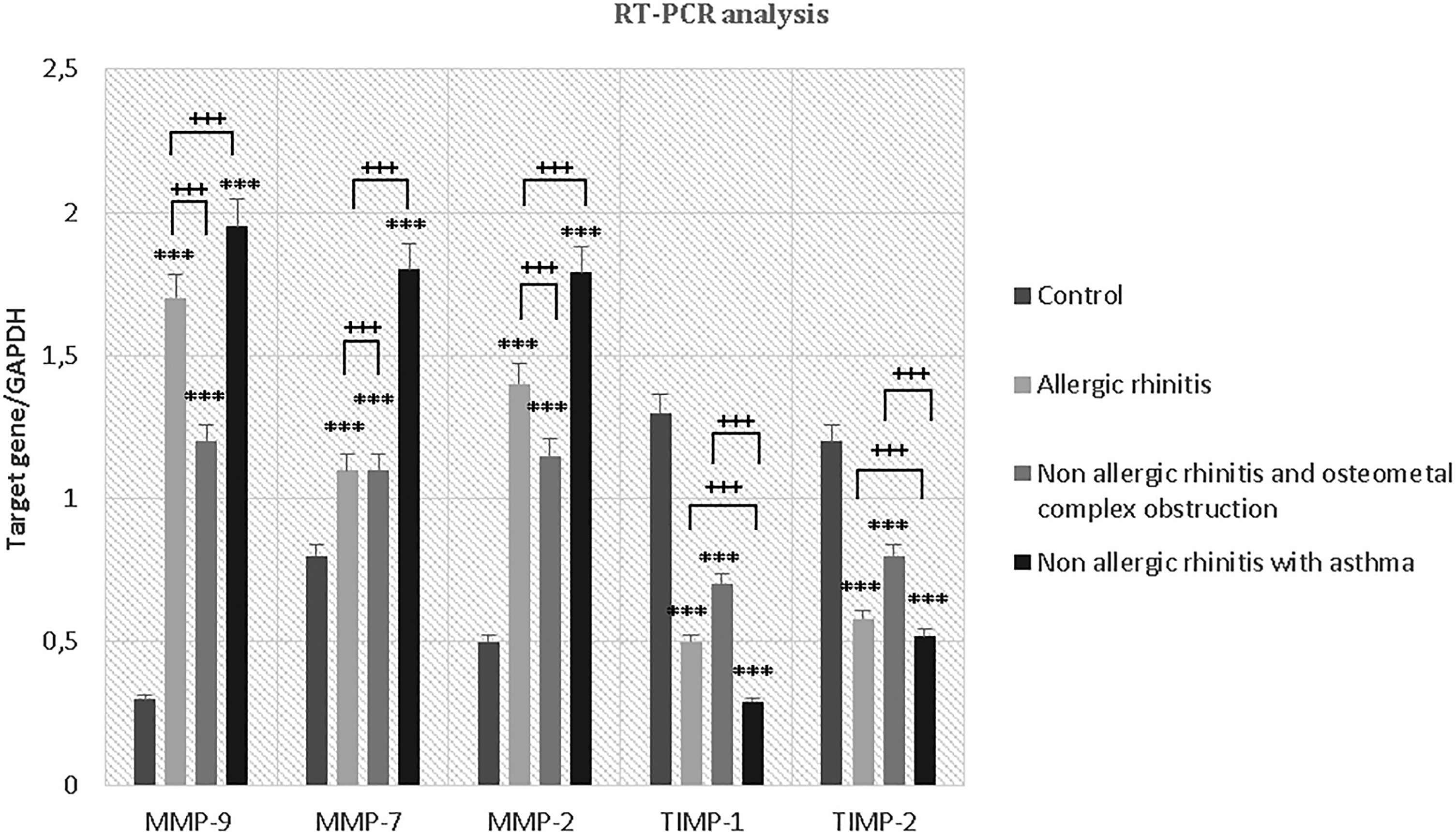
Reverse transcription polymerase chain reaction analysis. Expression of the matrix metalloproteinases (MMP-9, MMP-7, and MMP-2) and their tissue inhibitors (TIMP-1 and TIMP-2). An increase in MMP-9, MMP-7, and MMP-2 is observed in all groups compared to control group (P < .001* vs control), as well as a reduction in TIMP-1 and TIMP-2 (P < .001* vs control). Analyzing the levels of expression between groups, instead, we see that concentrations of MMPs appear to be greater in the patients nonallergic rhinitis and asthma compared to those with allergic rhinitis and patients with nonallergic rhinitis and osteomeatal complex obstruction (P < .001+). On the other hand, the concentrations of TIMPs were reduced in patients with nonallergic rhinitis and asthma compared to those with allergic rhinitis and those with nonallergic rhinitis and osteomeatal complex obstruction (P < .001+). MMPs indicates matrix metalloproteinases; TIMP, tissue inhibitor of metalloproteinase.
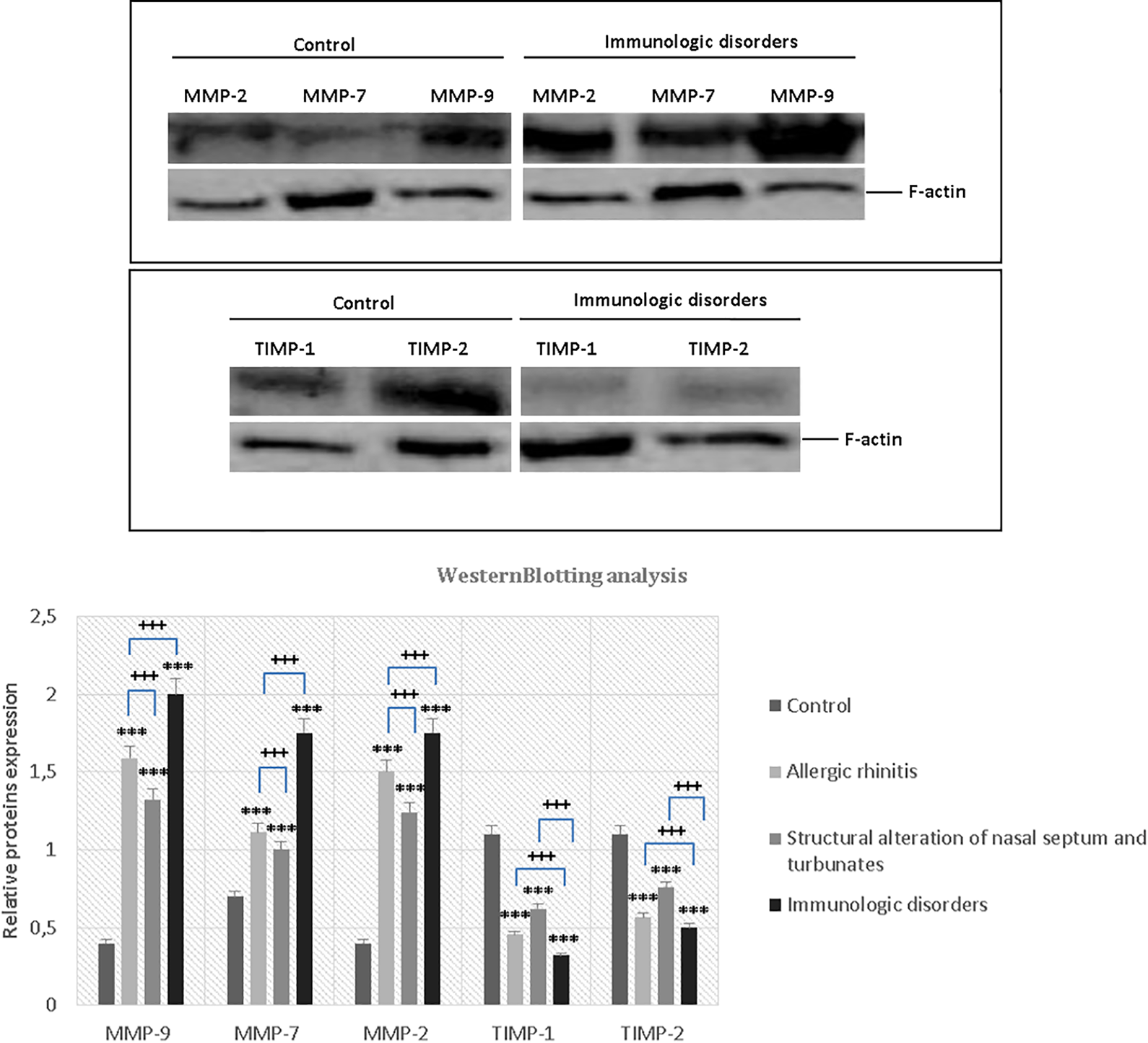
Western blot analysis. The expression of the matrix metalloproteinases (MMP-9, MMP-7, MMP-2) and their tissue inhibitors (TIMP-1 and TIMP-2) was investigated. An increase in MMP-9, MMP-7, and MMP-2 is observed in all groups compared to control group (P < .001* vs control), as well as a reduction in TIMP-1 and TIMP-2 (P < .001* vs control). Analyzing the levels of expression between groups, instead, we see that concentrations of MMPs appear to be greater in the patients with nonallergic rhinitis and asthma compared to those with allergic rhinitis and in patients with nonallergic rhinitis and osteomeatal complex obstruction (P < .001+). On the other hand, the concentrations of TIMPs were reduced in patients with nonallergic rhinitis and asthma compared to patients with allergic rhinitis and those with nonallergic rhinitis and osteomeatal complex obstruction (P < .001+). MMPs indicates matrix metalloproteinases; TIMP, tissue inhibitor of metalloproteinase.
Western Blotting Results of MMPs and TIMPS Tissue Concentrations in Patients With CRSwNP (Groups A, B, and C and Control Group).a
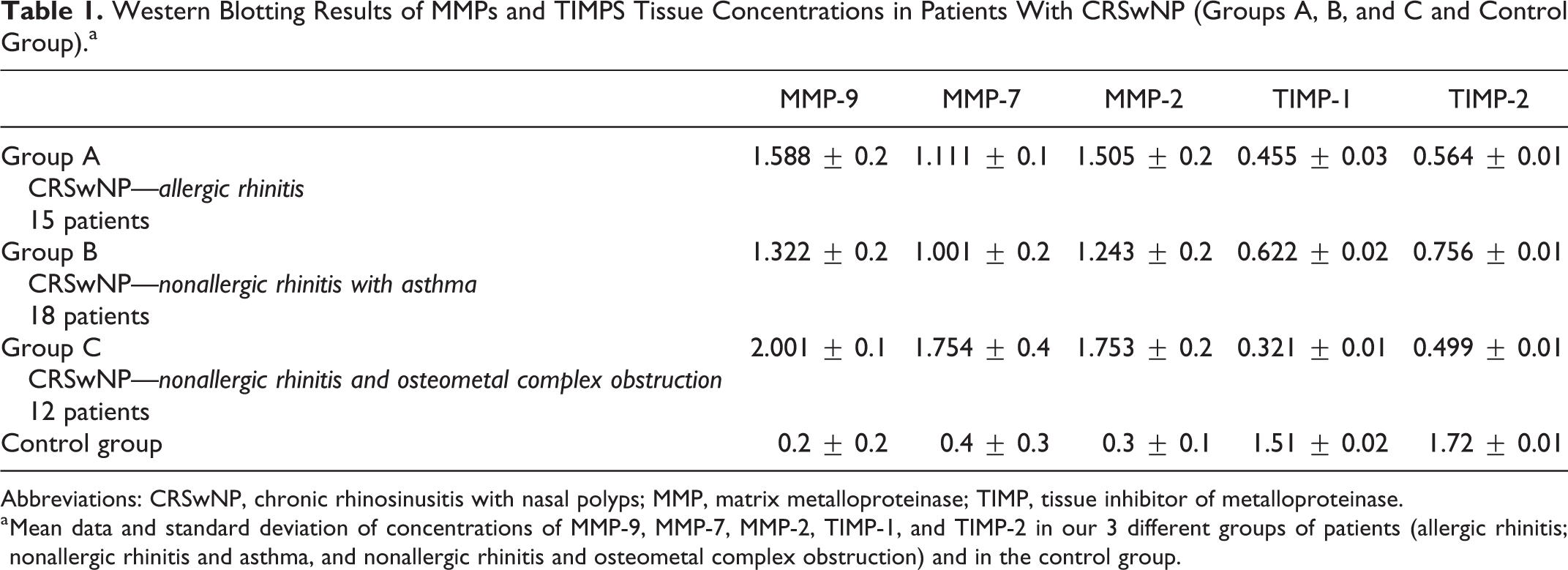
Abbreviations: CRSwNP, chronic rhinosinusitis with nasal polyps; MMP, matrix metalloproteinase; TIMP, tissue inhibitor of metalloproteinase.
a Mean data and standard deviation of concentrations of MMP-9, MMP-7, MMP-2, TIMP-1, and TIMP-2 in our 3 different groups of patients (allergic rhinitis; nonallergic rhinitis and asthma, and nonallergic rhinitis and osteometal complex obstruction) and in the control group.
The results of the immunohistochemical staining of sinonasal tissue from the patients examined are summarized in Figure 3. The samples collected were analyzed using avidin-biotin peroxidase (ABC/HRP), showing that both MMPs and TIMPs were concentrated in subepithelial mucous glands regions (Figure 3).
Endoscopic evaluations (Lund-Kennedy scores) and CT scan evaluations (Lund-Mackay scores) in follow-up showed recurrences at 36 months after surgery: in 6 (40%) of 15 patients of group A, 9 (75%) of 12 of patients of group B, and in only 2 (11.1%) patients of group C. Follow-up at 12 and 24 months after surgery did not show any recurrences in all the patients. The mean scores at Lund-Kennedy and Lund-Mackay evaluations, for each group of patients, were indicated in Table 2. Differences between groups of these scores were statistically significant at unpaired t test (P < .05).
Endoscopic and CT Scan Evaluation Scores at 36 Months After Surgery Follow-Up.
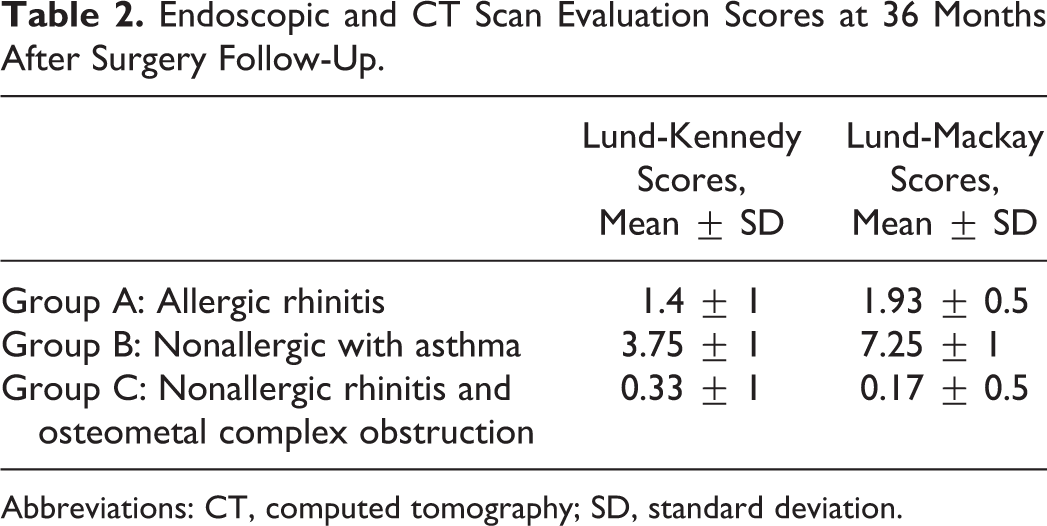
Abbreviations: CT, computed tomography; SD, standard deviation.
The incidence of recurrence in patients with nonallergic rhinitis and OMC (group C) was significantly lower compared to patients with nonallergic rhinitis and asthma (group B) and to allergic patients (group A). Differences between MMPs and TIMPs expressions in recurrent and nonrecurrent patients were statistically significant at Mann-Whitney U test (P < .05); MMPs increased, while TIMPs decreased in recurrent patients compared to nonrecurrent ones.
Discussion
Chronic rhinosinusitis with nasal polyps is an outgrowth of nasal mucosa, usually due to prolonged irritation. Nasal mucosa is a very humid tissue layer that helps protect the inside of the nose and sinuses and is involved in a lot of functions. Several factors such as sex hormones and/or neuropeptides are involved in the regulations of nasal mucosa activities. 18 -20
Bachert et al 21 in 2003 observed plasma retention and albumin within polyps and suggested that albumin favors the growth of nasal polyps, which is downregulated by steroids. Watelet et al in 2004 analyzed the biomorphological alterations in CRSwNP and in chronic rhinosinusitis without nasal polyps (CRSsNP) and then compared the results to look for abnormalities of sinonasal mucosa that could lead to polyps’ formations. In both CRSwNP and in CRSsNP, they found edema, inflammatory cells infiltration, fibrosis, and epithelial degradation; but in CRSwNP, they observed pseudocysts in the submucosal region. Pseudocystic formations are edematous cavities inside the connective tissue due to abnormal interstitial fluid accumulation. 22
In the past year, Kanai et al studied fibroblasts and their stimulation in nasal polyps formation induced by vascular endothelial growth factor, while Wu et al analyzed Th17/Treg cells ratio and its imbalance in inducing nasal polyps formation due to alterations in mucosal remodeling. 23 A higher expression of angiogenic factors and metalloproteinases was observed in other tissues-like ectopic endometrial layer in patients affected by endometriosis. 22,23
Bachert et al, Can et al, and Takabayashi et al investigated the roles of MMPs and TIMPs in CRSwNP and observed increased levels of MMPs, in particular MMP-7 and MMP-9, and decreased levels of TIMPs, leading to an increased MMP to TIMP ratio. These authors also studied MMPs and TIMPs levels in CRSsNP; in these studies, the MMP/TIMP ratios were decreased or, in some cases, similar to those found in control patients. 11,12,14,21
Chronic rhinosinusitis with nasal polyps for its predisposition to recurrence remains one of the most challenging diseases in rhinology. Its etiology is still unknown, but several studies in literature established that both MMPs and TIMPs play an important role in the balance between production and degradation of the individual components of the ECM and, therefore, in the pathogenesis of CRSwNP. 11,12,14,21 Recently some reports have documented that the MMPs that are most involved in NP are MMP-9 and MMP-7, while the role of MMP-2 is still unclear. 24 -26
However, overexpression of MMP-9, MMP-7, and MMP-2 cannot be the only explanation for tissue remodeling in CRSwNP, since high concentrations of MMP-9 were found also in CRSsNP. 24 -26 For this reason, additional parameters are likely to explain the differences in tissue remodeling, such as the expression of TIMPs, in particular TIMP-1. 24 -26 In literature, we found increased expression of TIMP-1 and MMP-9 in CRS, which did not alter their physiological ratio; in contrast, the expression of TIMP-1 in NP is not significantly higher and is, in some cases, lower compared to controls, resulting in considerably increased MMP-9 to TIMP-1 ratio. 11,24 -28 The reduced expression of TIMP-1 in these patients could, therefore, result in an unopposed function of MMP-9 and contribute to tissue remodeling, which characterizes CRSwNP. 28 -31 Likewise, a recent study analyzing the expression of another tissue inhibitor, TIMP-2, found significantly greater levels in patients with CRSsNP relative to those having CRSwNP. 32
Our study compared the concentrations of MMP-2, MMP-7, and MMP-9, and their tissue inhibitors, TIMP-1 and TIMP-2, by immunohistochemistry, Western blot analysis, and RT-PCR of samples tissue from patients with different forms of CRSwNP (allergic rhinitis, nonallergic rhinitis and asthma, and nonallergic rhinitis and OMC obstruction) and controls. In addition, we studied recurrence at 36 months of follow-up after ESS in relation to the expression of MMPs (MMP-2, MMP-7, and MMP-9) and TIMPs (TIMP-1 and TIMP-2) in recurrent CRSwNP at 36 months. Wang et al 32 in 2013 analyzed this correlation after 6 months of follow-up but did not study recurrence in different etiopathogenetical forms of CRSwNP and MMPs and TIMPs.
Our findings show increased concentrations of MMP-9, MMP-7, and MMP-2 and reduced concentrations of TIMP-1 and TIMP-2 in all patients with CRSwNP compared to controls, in agreement with other studies (Table 1). 26 However, when comparing the concentrations of these proteins in 3 different etiopathogenetic series of patients with CRSwNP, we appreciated the existence of important differences in these study groups. Specifically, the increase in MMPs concentrations and the concomitant decrease in the levels of TIMP-1 and TIMP-2 appear significantly more pronounced in patients having a syndromic disease relative to those with allergic diathesis or with morphostructural alterations of the sinonasal district. The MMP to TIMP ratio appears, therefore, significantly increased in nasal polypoid tissue, compared to controls, and significantly more in syndromic patients. Notably, an increased MMP to TIMP ratio in patients with syndromic disease was also associated with significantly higher incidence of recurrence (75%) during follow-up (at 36 months) relative to patients with other etiopathogenetic forms of polyposis. Nasal fiberoptic endoscopy during the follow-up showed a recurrence in 6 (40%) of 15 patients of group A, 9 (75%) of 12 of patients of group B, and in only 2 (11.1%) patients of group C. The incidence of recurrence in patients with morphostructural changes (group C) was significantly lower than those with immunologic alterations (group B) and to allergic patients (group A), with evidence of statistical significance (χ2 = 80.97, P < .001 and χ2 = 20.63, P = .03, respectively). This indicates the existence of a possible causative connection between the balance of MMPs and their inhibitors, and the tendency to recurrence after surgical treatment.
Results from the present study confirmed increased levels of MMPs and decreased levels of TIMPs in patients with CRSwNP compared with control group. 11-12,21 -23,25,26 The MMP-9 was more expressed than MMP-2 and MMP-7 and TIMP-1 was less expressed than TIMP-2 (P < .001); patients with nonallergic rhinitis and asthma presented higher incidence of recurrence than those with allergic rhinitis or those with morphologic alteration of nasal septum and turbinates (P < .001). Our study may, therefore, lay the basis for the development of new pharmacological or genetic protocols aimed at controlling the disease recurrence. In particular, the use of molecules able to reduce the MMPs to TIMPs ratio, such as synthetic inhibitors of MMPs (peptidomimetics of collagen, eg, Batimastat and Marimastat; nonpeptidomimetics, eg, BAY 12-9566, AG3340, CGS-27 023, and BMS-275291; derivatives of tetracyclines; and bisphosphonates), 30 could prove a viable therapeutic strategy in the prevention of recurrence in patients with NP. The weaknesses of this research are the sample size and the maximum 3-year follow-up.
In this research, CRSwNP was analyzed considering phenotypes and endotypes: MMP-2, MMP-7, MMP-9, TIMP-1, and TIMP-2 presented different concentrations in polyps of patients of the different groups considered. Although this is an original article in literature, it needs to be investigated further. The next step will be to evaluate the correlation between different cytokine patterns and analyzed phenotypes.
Footnotes
Authors’ Note
G. Guerra and D. Testa share the first authorship of this article and should be considered as co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
