Abstract
Previous studies reported that positive airway pressure (PAP) treatment may improve olfaction function in patients with obstructive sleep apnea (OSA) through various mechanisms. Olfactory function before and after PAP treatment is understudied regarding patient group at issue. The aim of this study is to investigate the contribution of PAP to olfactory function in patients with OSA. The study was conducted on 26 patients with OSA (10 females and 16 males, mean age 50.1 [9.3] years) who scheduled for PAP treatment. The Connecticut Chemosensory Clinical Research Center odor test was performed before and 4 months after PAP treatment. Patients were grouped (normal, anosmia, mild hyposmia, moderate hyposmia, and severe hyposmia) with respect to olfactory function by measuring odor test parameters, including threshold determination and identification. The odor test average scores of the patients after 4-month PAP treatment compared to pretreatment scores were increased and the difference was statistically significant (P = .002). In the apnea hypopnea index groups, statistically significant difference was found in the threshold and discrimination values regarding before PAP treatment (P = .038, P = .022, respectively). This study revealed that improvement in olfactory thresholds in patients with OSA receiving PAP treatment seems to improve olfactory dysfunction. This provides minimization of OSA consequences, including progressive upper airway inflammation, cognitive impairment, and associated olfactory dysfunction. Resolving the associations between olfactory function and PAP treatment is an important area for future research.
Introduction
Obstructive sleep apnea (OSA) is classified among sleep-related breathing disorders. Recurrent apnea and hypopnea episodes induce intermittent hypoxia and oxygen desaturation with significant reduction in the brain activity in the thalamus, hippocampus, prefrontal, and posterior parietal cortices. 1 The main symptoms are daytime sleepiness, cognitive dysfunction, and decreased psychomotor performance. 2,3
Cognitive deficits in OSA are considered secondary to nocturnal hypoxemia and cholinergic neurotransmission changes. 4 Previous studies reported association of reduced cognitive ability with the olfactory functions such as smell identification and odor differentiation. 5 Olfaction has a critical role in human physiology and behavior, including nutrition, sexuality, mating selection, memory, and toxin detection. Olfactory dysfunction can lead to significant clinical problems such as depression, loss of appetite, and the disability to detect environmental hazards with subsequent reduction in quality of life. 6 Recent studies have revealed that OSA has significant negative effects on olfactory function. Also, a negative correlation between the olfactory function and the severity of OSA has been shown in the literature. 5,7
The goal in the management of OSA is to control the effects of snoring, daytime sleepiness, and neurocognitive impairment, as well as limiting morbidity and mortality associated with long-term cardiovascular disease and motor vehicle accidents. 8 Positive airway pressure (PAP) is accepted as the gold-standard treatment in OSA disease. 9 Previous studies reported that PAP treatment may improve olfaction in patients with OSA through various mechanisms. 5,10,11 The aim of this study is to investigate the contribution of PAP to olfactory function in patients with OSA.
Patients and Methods
This study was conducted between January 2017 and January 2018 in a tertiary care hospital, with the approval of the ethics committee. The ethics committee’s approval number is 1199.
Patients were evaluated in the respiratory sleep disorders and snoring outpatient clinic. Twenty-six patients with OSA (10 females and 16 males, mean age 50.1 [9.3] years) referred to PAP treatment were enrolled in the study. All patients had diagnostic workup, including oropharyngeal examination, flexible endoscopy, and polysomnography (PSG) results.
The inclusion criteria of the study were patients who have used PAP for at least 4 months following the diagnosis of OSA. Patients with acute upper or lower respiratory tract infections as well as the ones having chronic rhinosinusitis, nasal cavity disease, nasal surgery, nasopharynx tumors, oropharynx or laryngeal tumors, cranial disorders, neurodegenerative and systemic diseases associated with the olfactory dysfunction, post-traumatic olfactory disorders, and major depressive disorders were all excluded from the study.
All participants had a full night PSG with a minimum of 7 hours of recording time. According to the American Academy of Sleep Medicine (AASM) guidelines, apnea–hypopnea index (AHI) scoring was assessed as normal (0-5), mild OSA (5-14), moderate OSA (15-29), and severe OSA (>30). 12 The patients in the study were grouped as normal, mild, moderate, and severe with respect to AHI results.
Patients were informed about the treatment options, including conservative measures, upper airway and maxillofacial surgery, intraoral devices, hypoglossal nerve stimulation, and PAP. The patients with mild OSA with the symptoms of intensive daytime sleepiness, insomnia, and associated cardiovascular morbidities who are noncompliant for surgery and other treatment options as well as the patients with moderate and severe OSA ineligible for other treatment alternatives were enrolled to PAP treatment. 12
The Connecticut Chemosensory Clinical Research Centre (CCCRC) test was done before the patients’ PAP treatment and 4 months after the treatment. The CCCRC test was conducted as described previously elsewhere. 13,14
The CCCRC test is composed of n-butanol odor threshold test and odor identification test. N-butanol test was numbered 0 to 7. The bottle number 0 contained the highest concentration, at 4% n-butanol, and the remaining bottles contained one-third dilutions of the previous concentration, prepared with distilled water. N-butanol test was scored out of 7 (0: worst, 7: best olfaction), and the mean score was calculated as the odor threshold test score. 14
In healthy volunteers at a previous study identified substances known to our community for use in odor determination tests. For the remaining 7 bottles (which contained aromas of cinnamon, cocoa, coffee, soap, peanuts, baby powder, and naphthalene), the odor determination test results were evaluated over 7 parameters according to the correct and incorrect answers. The scores for each nostril were noted separately for these 7 odors according to correct and incorrect results. The n-butanol and odor tests were scored and divided by 4 to determine the average value between 0 and 7.
Patients were grouped according to the odor test results: anosmia (0-1.75), advanced hyposmia (2.00-3.75), moderate hyposmia (4.00-4.75), mild hyposmia (5.00-5.75), and normal smell (6.00-7.00). 15
Statistical Analysis
SPSS program was used for statistical analysis. Descriptive statistics were given as the number and percentage for categorical variables, mean for numerical variables, standard deviation, minimum, maximum, median, and 95% confidence interval. Numerical variables in the dependent group differences that allow normal distribution were analyzed using paired t test condition. The relation between numerical variables was analyzed using the Pearson correlation analysis since the parametric test condition was provided. The differences between the numerical variables in the groups were compared with the Kruskal-Wallis test since the parametric test condition was not provided. The ratios in groups were tested by χ2 analysis. Statistically significant level of α was accepted as P <.05.
Results
The distribution of the patients in our study was 61.5% (16) males and 38.5% (10) females. The mean of our patients was 50.1 (9.3) and the mean AHI score was 24.4 (10.1). The evaluation of AHI scores revealed 4 patients with mild OSA, 15 with moderate OSA, and 7 with severe OSA. On average, the PAP setting was 7.1 (1.4) cm H2O and the PAP use was 5.1 (2.3) h/night (Table 1).
Patient Characteristics and Polysomnographic Measures.
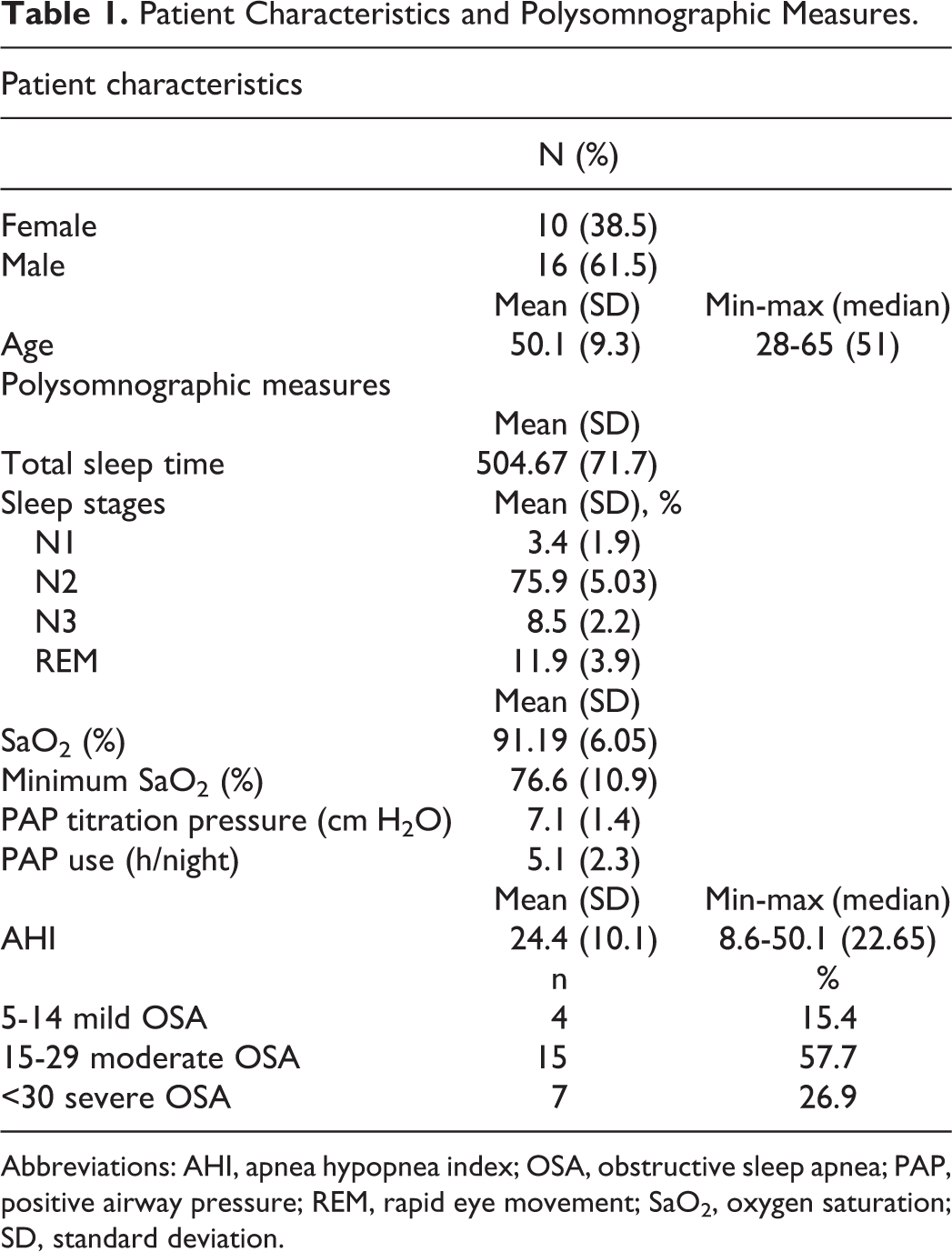
Abbreviations: AHI, apnea hypopnea index; OSA, obstructive sleep apnea; PAP, positive airway pressure; REM, rapid eye movement; SaO2, oxygen saturation; SD, standard deviation.
In the following PAP treatment, a decrease in the number of patients in anosmia and severe hyposmia groups was noted while there was an increase in the number of patients in moderate, mild hyposmia, and normal odor groups (Table 2). After 4 months of PAP treatment, the odor test averages increased. The difference between before and after PAP treatment was statistically significant (P = .002; Figure 1).
Distribution of the Odor Test Groups According to Pretreatment and Posttreatment Odor Test Results.
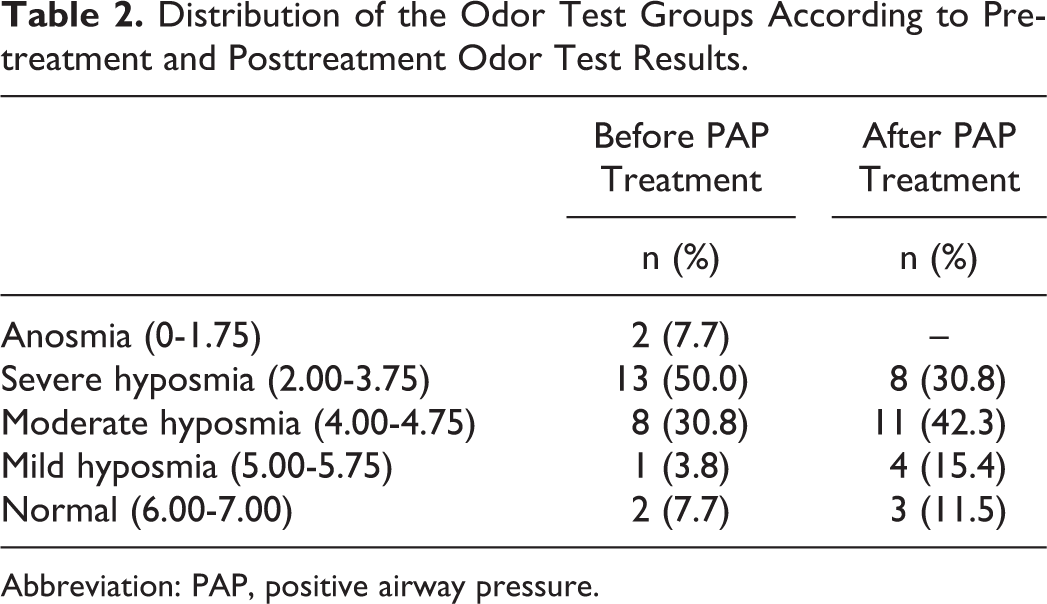
Abbreviation: PAP, positive airway pressure.
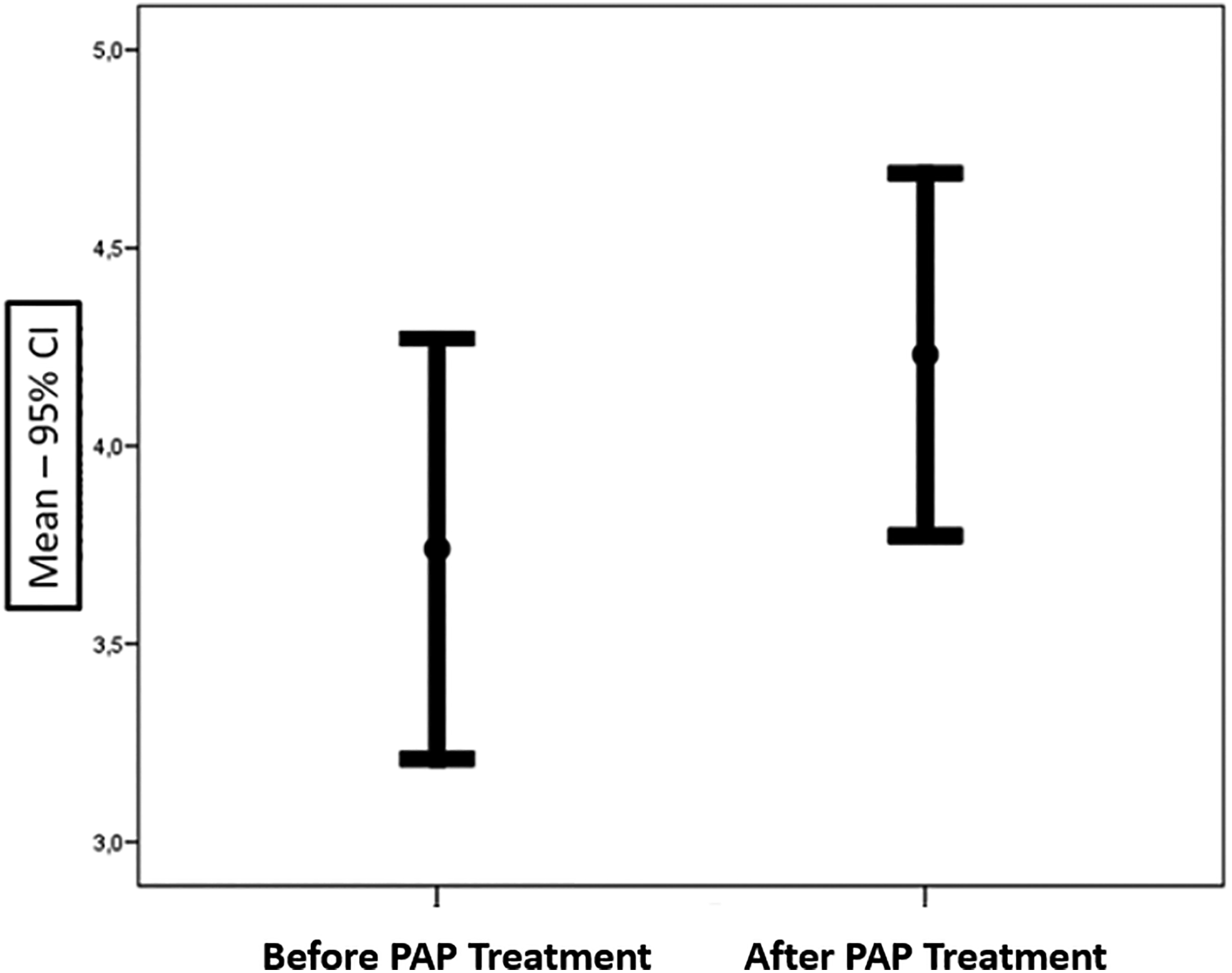
The difference between before and after positive airway pressur treatment.
Statistically significant correlation was not detected between AHI levels and olfactory test averages (P = .985, P = .508, respectively; Table 3).
Correlation Between AHI Level and Before and After PAP Treatment Odor Test.

Abbreviations: AHI, apnea hypopnea index; PAP, positive airway pressure.
In the AHI groups, statistically significant correlation was not detected in before and after PAP treatment odor test averages (P = .056, P = .512, respectively; Table 4).
Correlation of AHI Groups Before and After PAP Treatment.
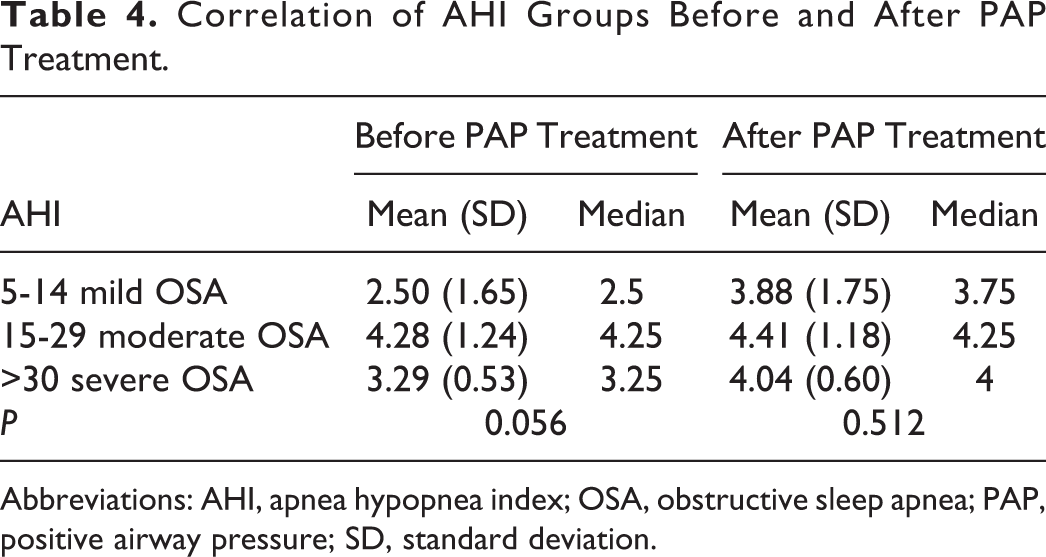
Abbreviations: AHI, apnea hypopnea index; OSA, obstructive sleep apnea; PAP, positive airway pressure; SD, standard deviation.
In the AHI groups, statistically significant difference was found in the threshold and discrimination values before PAP treatment (P = .038, P = .022, respectively; Table 5).
Distribution of the Threshold and Discrimination Values Before and After PAP Treatment in the AHI Groups.
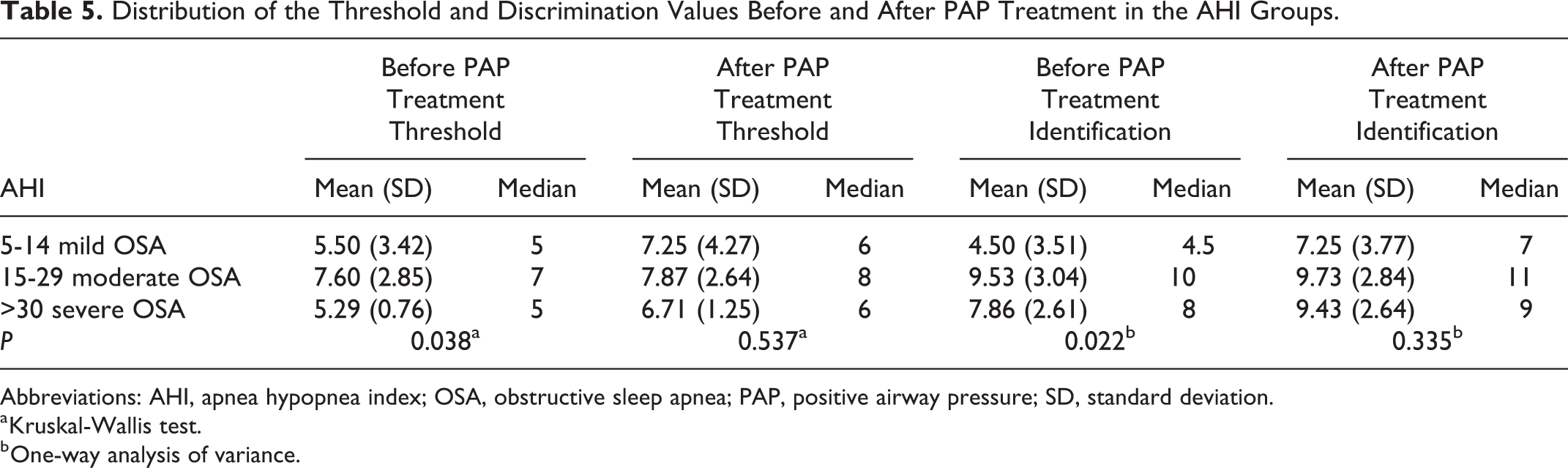
Abbreviations: AHI, apnea hypopnea index; OSA, obstructive sleep apnea; PAP, positive airway pressure; SD, standard deviation.
a Kruskal-Wallis test.
b One-way analysis of variance.
In the mild OSA group, the discriminative odor test mean values after PAP treatment were significantly higher than the test before PAP treatment (P = .010). However, in the other groups, statistically significant difference was not found between before and after PAP treatment (Table 6).
Depended Group Analysis in AHI Groups.
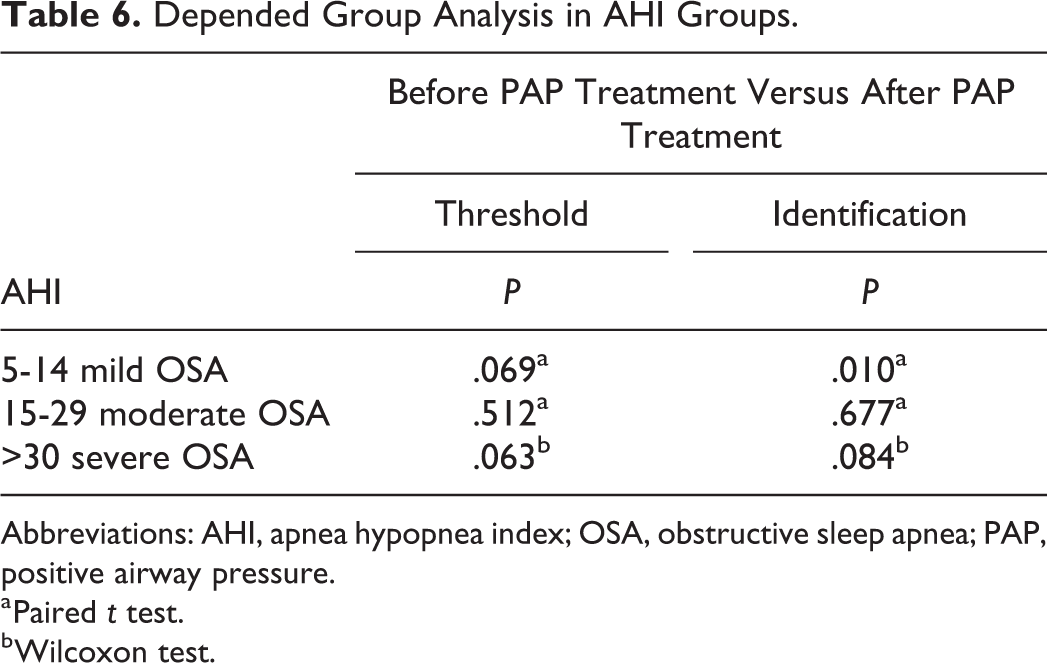
Abbreviations: AHI, apnea hypopnea index; OSA, obstructive sleep apnea; PAP, positive airway pressure.
a Paired t test.
b Wilcoxon test.
Discussion
Obstructive sleep apnea is a common disease affecting 2% to 20% of the adult population. 16 The reported prevalence is 24% and 9% in males and females, respectively. 17 In our study, a similar ratio of male (61.5%) and female (38.5%) was detected.
In the literature, the distribution of patients in the studies that investigate odor function in patients with OSA was as follows: Fu et al 6 have reported 23.7% (18) mild, 30.3% (23) moderate, and 46.1% (35) severe for 76 patients; Shin et al 7 have reported 28% (19) mild and 73% (50) moderate-severe for overall 79 patients; Boerner et al 10 have detected 9% (3) patients with moderate OSA and 91% (32) with severe OSA; Salihoglu et al 18 have revealed 33% (26) mild, 37% (29) moderate, and 29% (23) severe OSA. In our study, in terms of OSA severity, the distribution of the patients was unevenly consisting of 15.4% (4) mild, 57.7% (15) moderate, and 26.9% (7) severe. The main reason for this uneven distribution was inclusion of patients using exclusively PAP treatment as well as our rigid exclusion criteria.
According to the AASM, primary conservative treatment of OSA is PAP or mandibular advancement device. 13,14 Positive airway pressure is recommended as a first-line treatment for patients with symptomatic OSA. 17
In patients with OSA, olfactory dysfunction may be associated with various mechanisms. Several studies have reported cognitive deficits regarding patients with OSA, including memory, attention, and sensory functions as well as olfaction. Decreased (poor) neurocognitive performance has been found to be associated with olfactory dysfunction through impaired olfactory discrimination and decreased threshold. 19 On the other hand, decreased nasal airflow, intermittent hypoxia, chronic irritation, and subsequent upper airway mucosal damage may be the additional associated mechanisms responsible for olfactory dysfunction in OSA.
The clinical studies have shown that PAP treatment contributes to the respiratory and cognitive performance in OSA. 11,20 Positive airway pressure treatment may improve olfactory dysfunction in patients with OSA through diverse interactions. 5,10 The cholinergic neurotransmitter system is involved in many cognitive functions and sensitive to cerebral hypoxemia. Positive airway pressure through normalizing the synthesis of neurotransmitters may improve cognitive dysfunction in patients with OSA. 11 Increased oxygen saturation has been shown to have positive effects on neurocognitive function, including olfaction. 7,18
The threshold and identification tests measure different aspects of olfactory function. Although thresholds largely reflect the nasal mucosal and upper airway circumstances such as congestion and inflammation, identification reflects the cognitive, “central” aspect of olfactory processing. 21,22 Recent studies have reported more significant improvement in the cognitive state compared to upper airway inflammation following PAP treatment. 23 –25 In our study, we found statistically significant increase in olfactory test averages of patients with OSA after 4 months of PAP treatment (P = .002). The mechanisms having an impact on olfactory function in OSA may be associated with many factors, including oxygen saturation, duration of disease, and degree of upper airway inflammation. Several studies have reported that airway inflammation in patients with OSA having intermittent nocturnal hypoxia increases pro-inflammatory markers such as interleukin 8 and tumor necrosis factor α. 26,27 Positive airway pressure treatment may contribute olfactory function by reducing mucosal inflammation in OSA. 28,29 In our study, we found that in the mild OSA group, identification thresholds improved significantly regarding after PAP treatment (P = .010). However, in the other groups, statistically significant difference was not detected.
In the literature, Salihoglu et al 18 have reported a strong negative correlation between AHI and odor threshold scores, odor discrimination scores, and overall olfactory function (threshold-discrimination-identification) scores in 75 patients with OSA. Boerner et al 10 in a small group of patients (n = 25) could not confirm a correlation between AHI levels and odor test averages; however, they reported improvement in olfaction with PAP treatment after 3 months. In our study, statistically significant correlation was not detected between AHI levels and odor test averages (P = .985, P = .508, respectively; Table 4). We found similar results with Boerner et al, 10 but as a difference to avoid bias we excluded patients with diabetes mellitus due to reported impact on olfaction.
Our study has a few limitations, including relatively small sample size, lack of olfactory bulb volume measurement, clinical care classification findings, long-term follow-up, and a control group consisting of untreated patients with OSA due to ethical reasons.
Conclusion
This study revealed improvement in olfactory thresholds in patients with OSA receiving PAP treatment. Positive airway pressure resolving apnea episodes avoids desaturation in patients with OSA. This provides minimization of OSA consequences, including progressive upper airway inflammation, cognitive impairment, and associated olfactory dysfunction. To clarify the contribution of PAP treatment to olfactory function in patients with OSA, more comprehensive studies are required.
Footnotes
Authors’ Note
This study was carried at Sisli Hamidiye Etfal Training and Research Hospital in Istanbul.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
