Abstract
Objective:
To assess the potential influence of increased time from diagnosis to treatment on survival outcomes in patients with locoregionally advanced hypopharyngeal squamous cell carcinoma (HSCC).
Methods:
Retrospective study of patients with a primary diagnosis of HSCC proposed for primary surgical treatment.
Results:
The study population included a total of 121 Caucasian patients (121 males) with HSCC. Mean age at diagnosis was 60.4 years (range: 43-83 years). All patients had cT3 or cT4 hypopharyngeal tumors. The sample presented a 5-year overall survival (OS) of 59.6% and a disease-specific survival of 74.9%. Considering univariable analysis (unadjusted), duration of adjuvant treatment (T5), pN, margins, and extracapsular spread (ECS) are factors associated with poorer survival outcomes. An increase in T5 was associated with lower OS. Results revealed that T5 higher than 43 days, pN1, pN2, pN3, no free margins, and presence of ECS were associated with lower OS. These patients have 7.465 higher hazard of death.
Conclusion:
This study suggests that duration of adjuvant therapy may be more important than other timing metrics from diagnosis to treatment. For locoregionally advanced HSCC, duration of adjuvant therapy after primary surgery higher than 6 weeks is an important feature for worse survival outcome. Preventing strategies in order to avoid radiotherapy or chemoradiotherapy breaks should be developed and optimized.
Introduction
Head and neck squamous cell carcinoma accounts for approximately 6% of all cancers worldwide, and most patients present with locally advanced diseases. 1 -3 For locally advanced resectable HSCC, organ preservation strategies using combined chemotherapy and radiotherapy (RT) as induction, concurrent, sequential, or alternating therapies have been studied in the recent decades. 4 -6 In more advanced HSCC with cartilage invasion, extralaryngeal soft tissue invasion or high-volume tumor, primary surgery with postoperative adjuvant therapy has remained the recommended therapy. 4 -7 Despite aggressive multimodality therapy using combinations of surgery, RT, and chemotherapy, oncologic outcomes remain poor. 8 As a result, there is a critical need to identify strategies to improve survival for patients with HSCC. One strategy focuses on improving the timeliness of care delivery. 8 Across the continuum of HSCC care delivery, delays are common and are a major source of preventable mortality. 9,10
To our knowledge, delays on different points from diagnosis to primary treatment and its potential influence on survival outcomes of HSCC have not been analyzed. We therefore conducted a retrospective cohort study to assess the potential influence of increased time from diagnosis to treatment on survival outcomes in patients with locoregionally advanced HSCC.
Materials and Methods
Patients
We obtained an information database containing 211 patients with HSCC diagnosed and treated in an oncological tertiary center between 2012 and 2016. Approval of the Medical Ethics Committee of Instituto Português de Oncologia do Porto was obtained (CES.155/015). This database was refined to include only patients with a primary diagnosis of hypopharynx carcinoma proposed by the Head and Neck Interdisciplinary Tumor Board for primary surgical treatment (pharyngolaringectomy and neck dissection) so that pathological staging information could be obtained.
The number of institutional affiliation, age, gender, tobacco and alcohol abuse, histopathologic tumor classification according to the WHO International Classification of Diseases for Oncology (ICD-0-3), tumor localization, clinical tumor node metastasis (cTNM), and pathological tumor node metastasis (pTMN) classification were registered. Covariates also included others significant points such as surgical margins, radiation modality, and administration of concurrent chemotherapy. The postoperative RT schedules were 60 to 66 Gy in 2-Gy fractions, 5 fractions per week over 6 to 6.6 weeks, and the postoperative radiochemotherapy was based on identical radiation treatment schedule plus concurrent cisplatin with 100 mg per square of body surface area intravenously on days 1, 22, and 43.
The database was cross-referenced to patient charts or electronic medical records to verify the integrity of the data, particularly for information involving staging, treatment, follow-up period (FU), and dates last known alive.
Intervals of Time
Time 1 (T1): Interval of time from admission in the hospital, with biopsy-proven diagnosis, to the first multidisciplinar decision (FMD). Time 2 (T2): Interval of time from FMD to primary surgery. Time 3 (T3): Interval of time from surgery to second multidisciplinar decision (SMD). Time 4 (T4): Interval of time from SMD to the beginning of adjuvant treatment. Time 5 (T5): Duration of adjuvant treatment. Total of time (T1 + T2 + T3 + T4 + T5): Interval of time from diagnosis to complete adjuvant treatment.
Survival Outcome
During the FU, patients were considered as being alive with and without oncologic disease; dead with local, regional, or distant disease; dead without oncologic disease; and finally, lost to FU. Local recurrence was defined as an invasive carcinoma developing 6 months after the conclusion of the curative treatment. Regional and distal recurrences were defined as the presence of lesions with the same histological type in regional lymph nodes, neck soft tissues, or distant localizations after the completion of the initial treatment.
The cutoff point for statistical analysis was August 2018, encompassing a minimum FU of 24 months. For overall survival (OS), follow time was considered as the time between diagnosis and death with or without disease (event of interest). Patients alive with disease, patients alive without disease, and patients lost in follow-up were censored.
For disease-specific survival (DSS), follow time was considered as the time between diagnosis and death with disease (event of interest). Patients alive with disease, patients alive without disease, patients who died without disease, and patients lost in follow-up were censored.
Statistical Analysis
A descriptive analysis of patient’s characteristics was performed considering absolute and relative frequencies (for categorical variables) and mean and standard deviation (for continuous variables). Both OS and DSS curves were calculated using Kaplan-Meier method, and statistical significance was determined by log-rank test. Univariable and multivariable Cox regression model were performed to identify potential predictive factors for OS and DSS. All analyses were performed in the software SPSS version 24, and P values below .05 were considered statistically significant.
Results
Demographic, Clinicopathologic, and Treatment Characteristics
The study population included a total of 121 Caucasian patients (121 males) with HSCC. Mean age at diagnosis was 60.4 years (range: 43-83 years; Table 1). All patients had cT3 or cT4 hypopharyngeal tumors.
Sample Characteristics.

Abbreviation: ECS, extracapsular spread.
Histopathological evaluation of the margins in primary specimens showed free margins in 63 (52.0%) patients. A total of 58 (48.0%) patients presented extraorgan invasion of adjacent structures, mostly to the larynx.
Adjuvant RT or chemoradiotherapy (CRT), depending on the patient’s age, general conditions, and comorbidities, was performed in all patients with positive or close surgical margins and in patients with other adverse features, such as primary pT4, N2 or N3 nodal disease, perineural invasion, or vascular embolism.
Overall Treatment Times
The median time from diagnosis (biopsy proven diagnosis) to the first multidisciplinary decision, where the Head and Neck Interdisciplinary Tumor Board propose primary surgical treatment, was 24.9 days (Table 2). Then, up to pharyngolaringectomy and neck dissection, the interval of time was almost 27 days (Table 2).
Time Intervals From Diagnosis to Treatment.
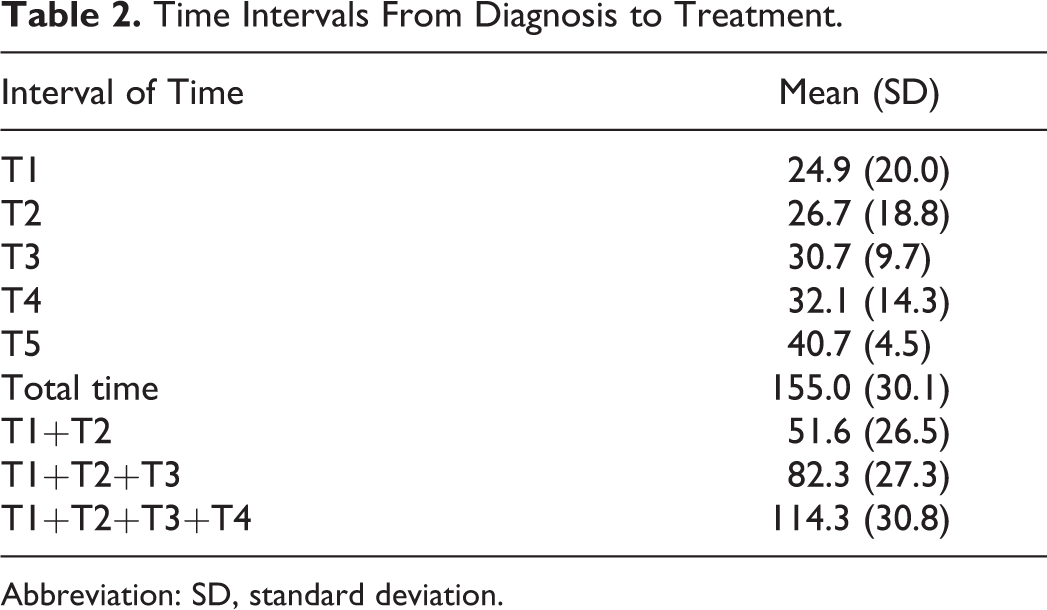
Abbreviation: SD, standard deviation.
After histopathological analysis was completed, the patient was discussed on a second multidisciplinary tumor board, usually, 1 month after surgery (Table 2). In this patient’s group, adjuvant therapy has begun 32.1 days after and during approximately 6 weeks (Table 2).
Survival Outcomes and Its Predictive Factors
The sample presented a 5-year OS of 59.6% (Figure 1) and a DSS of 74.9% (Figure 2). Considering univariable analysis (unadjusted), duration of adjuvant treatment (T5), pN, margins, and extracellular space (ECS) are factors associated with poorer survival outcomes.
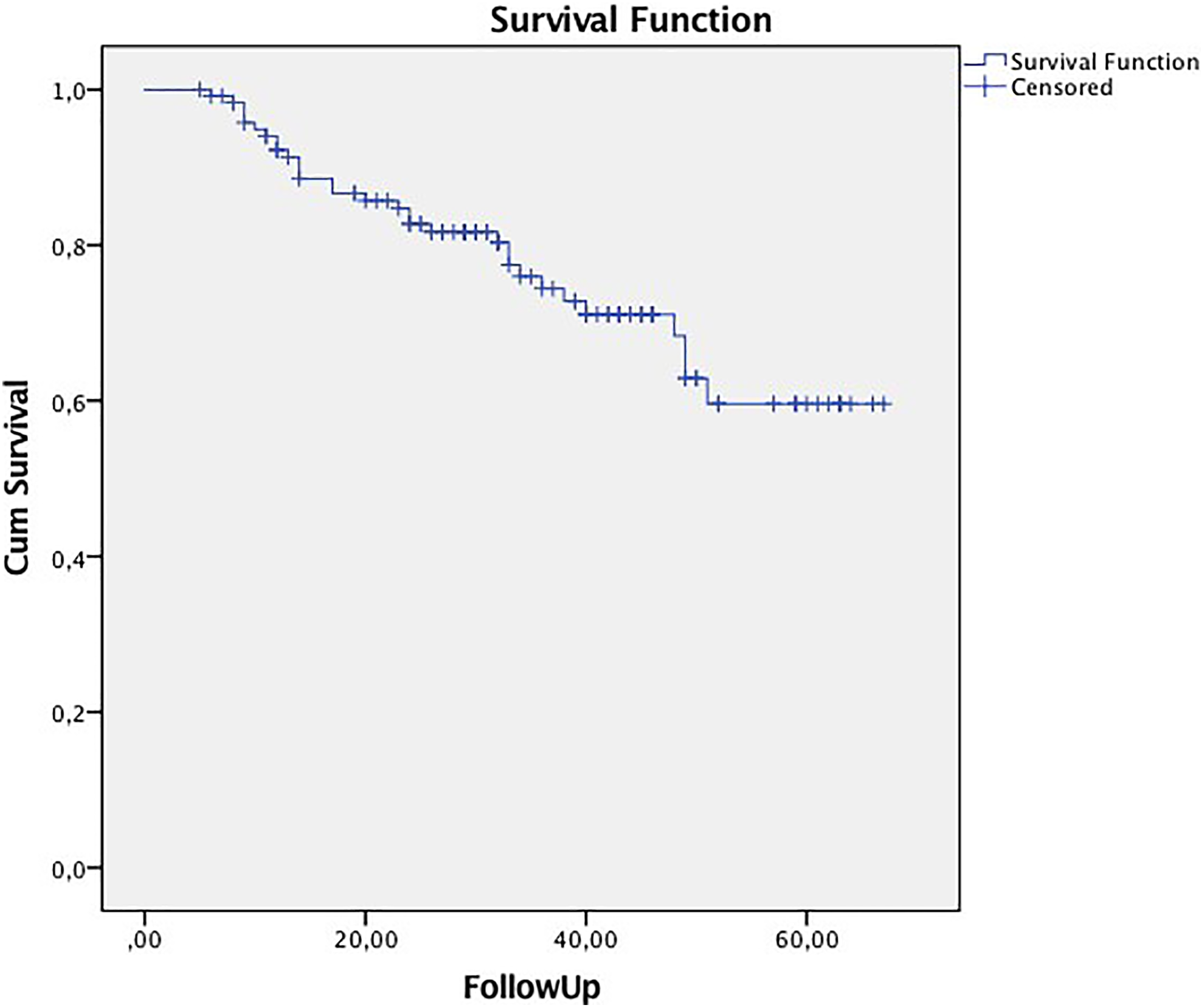
Overall survival Kaplan-Meier curves (log-rank P = .042).
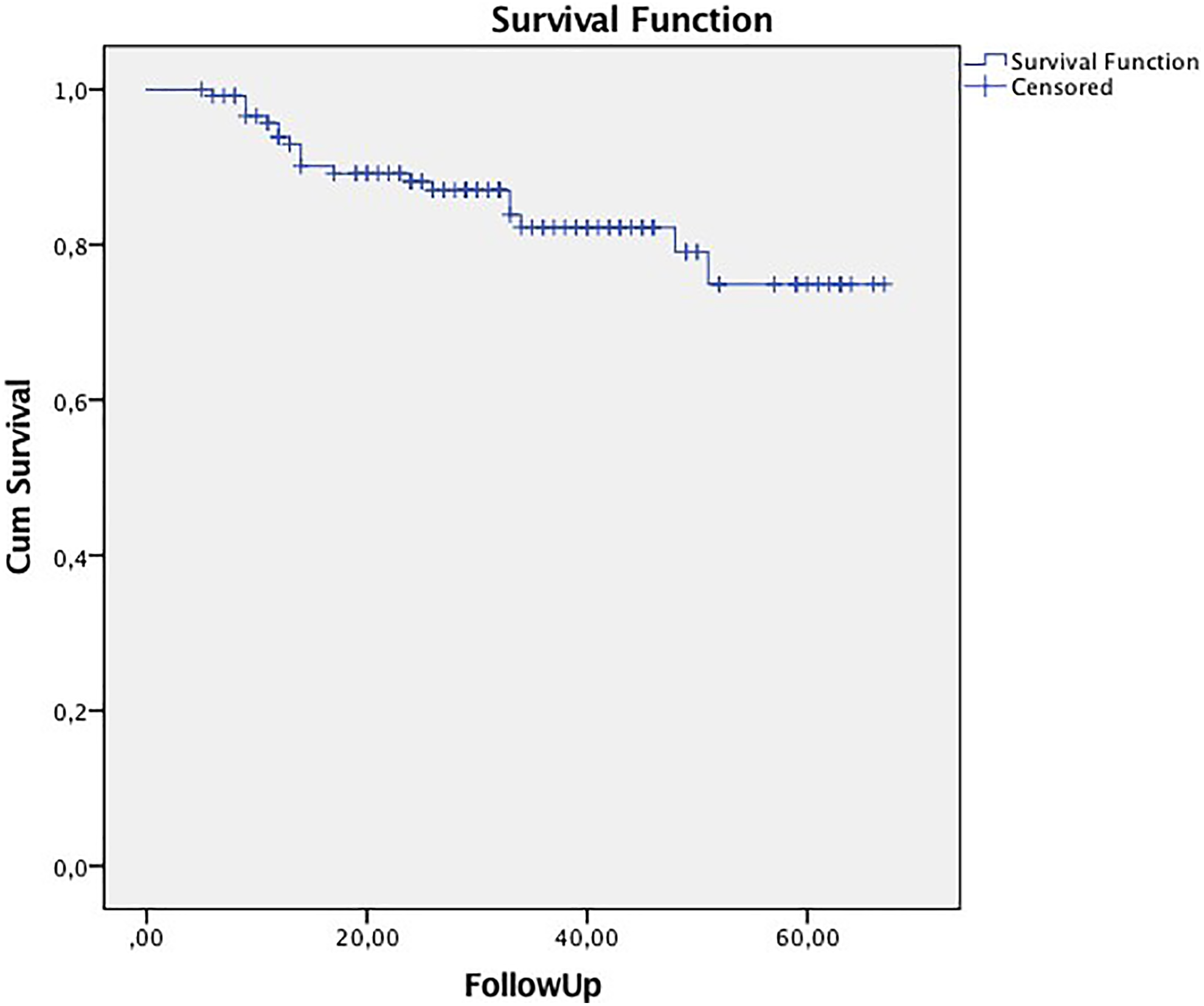
Disease-specific survival Kaplan-Meier curves (log-rank P = .042).
An increase of T5 was associated with lower OS. Groups pN2 and pN3 were associated with lower OS and DSS than pN0. Histopathological features such as positive margins or ECS were associated with lower OS and DSS than free margins or no ECS.
Duration of Adjuvant Treatment
Considering that T5 was a significant predictive factor of OS and using the Youden’s J statistics to identify the optimal cutoff provided by the receiver operating characteristic curve (T5 as variable and overall mortality as indicator), the optimal cutoff obtained was 43.5 days. Performing univariable Cox regression model, this variable is a factor associated with OS, as expected (hazard ratio: 2.244, 95% confidence interval: 1.090-4.621, P = .028).
Patients with positive margins are the group of patients who had the highest duration of adjuvant therapy (odds ratio = 2.387, P = .026; Table 3). Results revealed that T5 higher than 43 days, pN1, pN2, pN3, no free margins, and presence of ECS were associated with lower OS (Table 4). These patients have 7.465 higher hazard of death.
Predictive Factors for Duration of Adjuvant Therapy >43 Days.
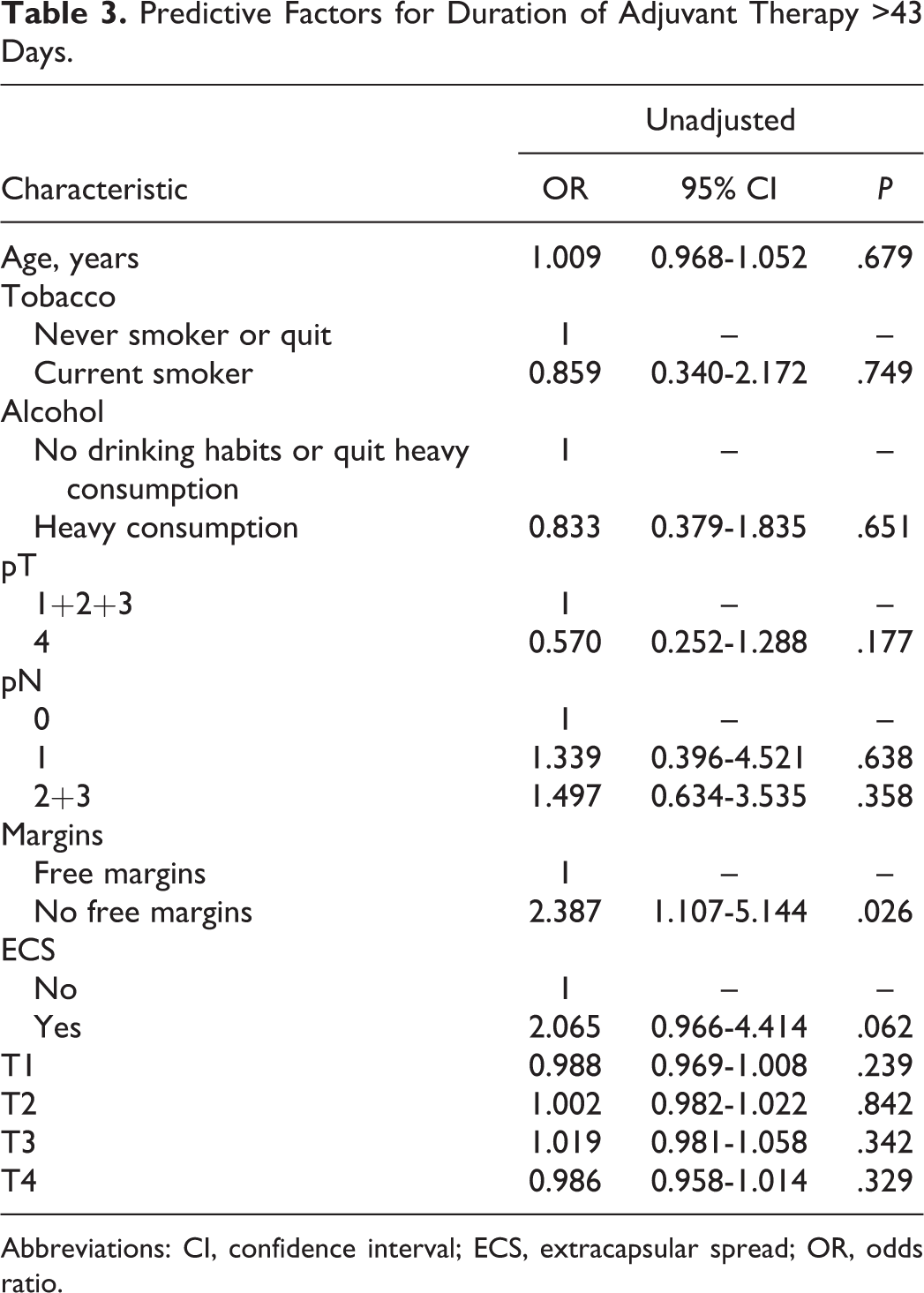
Abbreviations: CI, confidence interval; ECS, extracapsular spread; OR, odds ratio.
Predictive Factors for Survival Outcome.
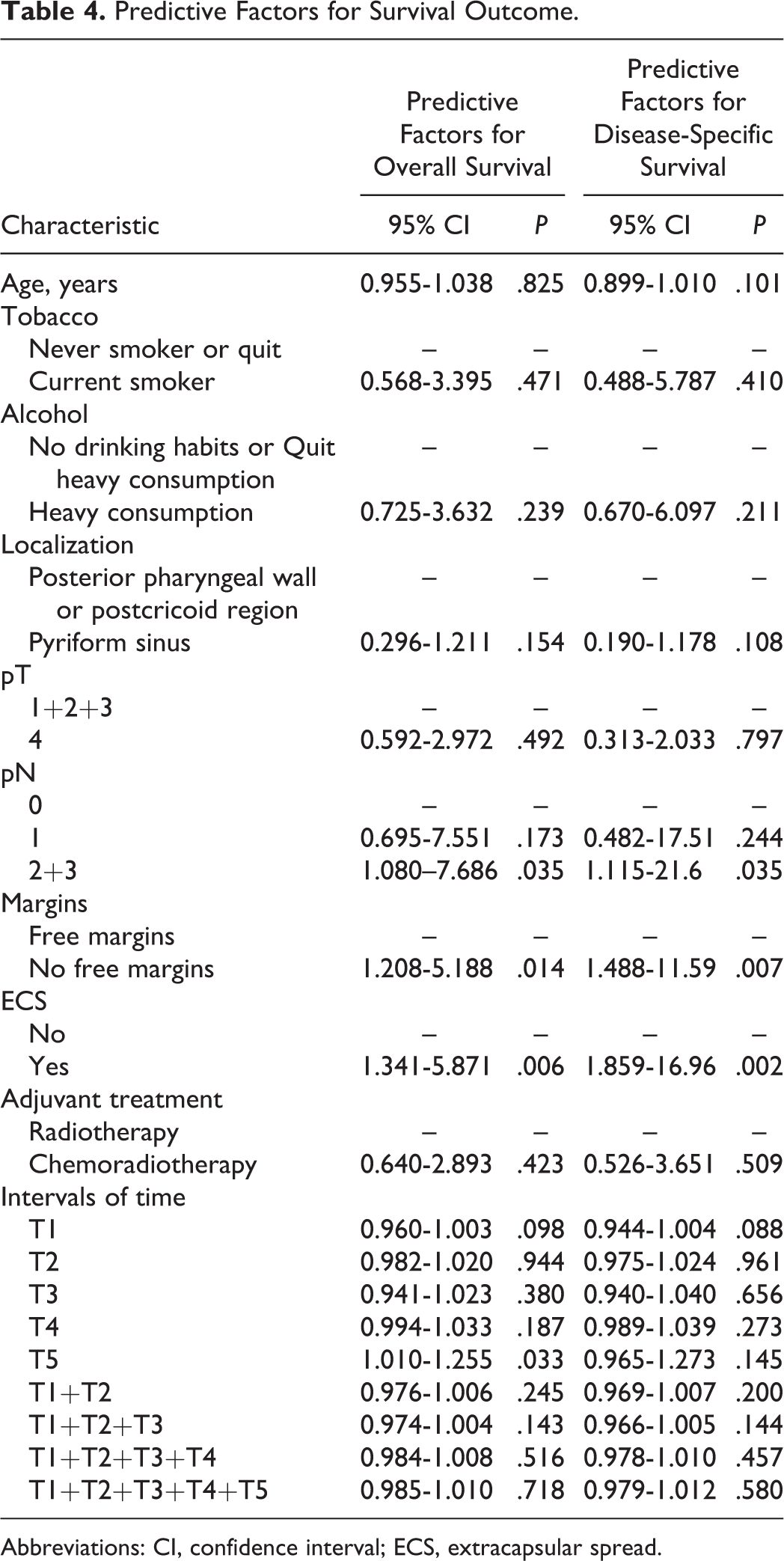
Abbreviations: CI, confidence interval; ECS, extracapsular spread.
Discussion
Clinical and demographic characteristics of the present study correlate with other series regarding the age of presentation, male predominance, and high tobacco and alcohol consumption. 11 The first aim of this study was to evaluate wait times from diagnosis to treatment of locoregionally advanced HSCC in a tertiary public oncological center.
Median time from diagnosis to surgery was 51.6 days (±26.5). During this time, patients were presented and discussed in a multidisciplinary tumor board that happens once a week. Patients had to attend this consultation, and cases were only discussed if all the studies required for an appropriate staging were available. This fact might explain why this interval of time is so high. However, in this particular cohort, this time delay did not appear to negatively affect survival outcomes, and it can be considered that advanced HSCC less than 2 months from diagnosis to primary surgery may not influence global survival outcomes.
Median time from surgery to the beginning of adjuvant therapy was 62.8 days. Ideally, patients will start treatment ≤6 weeks to allow for unplanned treatment breaks due to treatment-related toxicity. 12 The oncologic effect of Nathional comprehensive cancer network (NCCN) guideline-adherent care for timely adjuvant therapy remains uncertain, and no consensus exists. 13,14
Harris et al suggested that adjuvant RT should be started before 50 days of surgery. 14 Prior studies have shown inconsistent effects on locoregional recurrence and survival, with some finding a benefit and others finding no influence. 13 It has been argued that recent improvements in radiation technology, such as intensity-modulated radiation therapy, altered fractionation, and concurrent chemotherapy may mitigate the risk associated with delays in initiating adjuvant treatment. 15 Controversy about the ideal time for starting adjuvant therapy exists, and most of the studies published refer to head and neck cancer, not specifying subsites. Our study is focused specifically on HSCC, and the mean value obtained was slightly higher than 50 days. Besides this, it did not affect survival outcomes.
As concerns duration of adjuvant therapy, the mean time was 40.7 days, and it was found that higher duration of adjuvant therapy (>6 weeks) is associated with poorer survival outcomes. The importance of adjuvant therapy’s duration may be due to biological significance of tumor cell proliferation during CRT treatments. 16 Repopulation can be overcome with strategies such as accelerated fractionation or dose intensification, although when the dose is increased, care must be taken to account for the eventual additional delay. It is not known yet how accelerated fractionation patterns will affect postoperative wound healing and surgical site complications. 16 It would be interesting in the future to attempt which are the most important features: position of the gap during the adjuvant therapy, total number of gaps, or the number of consecutive treatments days missed.
The NCCN guidelines specify time to initiation of postoperative radiation therapy but do not address overall treatment package time. The package time is defined as the time from surgery to the completion of postoperative adjuvant therapy. There is growing evidence that package time may be more relevant than simply the time to initiation of adjuvant therapy, in part because it also helps to capture other important factors such as planned and unplanned treatment breaks. 16 Our findings corroborate this idea and suggest that within package time, eventually, it is the duration of adjuvant therapy which is the most important interval. This highlights the importance of taking supportive measures to prevent unplanned CRT breaks.
The secondary purpose of this article was to determine other potential factors that might influence survival outcomes. It was found with multivariate regression, pN1, pN2, pN3, positive margins, and presence of ECS were associated with lower OS. This is in accordance with other studies, which documented that not only the presence of regional disease but also the existence of nodal rupture are determinant factors in prognosis. 17 In the present study, the 5-year OS and DSS is similar to other series, supporting the idea that HSCC continues to have an unsatisfactory prognosis that has not changed appreciably over the past decades. 17
Our work is novel because it suggests that duration of adjuvant therapy may be more important than other timing metrics. Additional studies evaluating the reasons for RT or CRT breaks may be helpful in further understanding our practice patterns. Research is also needed in determining how to balance the need for adequate wound healing prior to initiation of radiation therapy and the importance in minimizing total package time.
Although our study did not show a significant effect of shorter time from diagnosis to treatment on clinical outcomes, we still believe that shorter wait times have a positive effect with respect to patient anxiety, mental well-being, satisfaction, physical functioning, quality of life, and cost of care. Psychological stress during the investigation period is considerable.
This study has some limitations. First of all, it is a retrospective study, and some patients exhibited a small FU that had conditioned the calculation of 5-year survival rates. Second, histopathological analysis was performed not always by the same pathologist. Third, the conclusions of this study might change if the study population size was expanded. Our data did not allow us to evaluate whether patients presenting with a less advanced HSCC were more affected by longer waiting times. Time to treatment might become a more important factor for patients with early-stage disease in order to increase the number of patients with resectable disease and to avoid tumor growth and upstaging. Nevertheless, to our knowledge, this is the first explorative study that access for advanced HSCC the potential influence of waiting timings from diagnosis to complete adjuvant treatments. Our data were collected from 1 single institution with uniform processing and reporting protocols, which highlight the reliability of our results.
This study is important because it demonstrates that the duration of adjuvant therapy is important for patients with HSCC. Notably, this measure may hold a greater impact on survival than the current NCCN metric of time from surgery to initiation of adjuvant therapy. We will continue to evaluate survival outcomes and potential influencing factors by increasing the sample size to increase the power of findings in this study.
Conclusion
This study suggests that duration of adjuvant therapy may be more important than other timing metrics from diagnosis to treatment. For locoregionally advanced HSCC, duration of adjuvant therapy after primary surgery higher than 6 weeks is an important feature for worse survival outcome. Preventing strategies in order to avoid RT or CRT breaks should be developed and optimized.
Footnotes
Authors’ Note
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
