Abstract
Antrochoanal polyps (ACPs) are benign lesions of the paranasal sinuses. Similarities between ACPs and nasal polyps have been previously investigated. However, oxidative stress has been implicated but not investigated in terms of the etiology of ACPs. Thus, the aim of this study was to investigate oxidative stress in the etiology of ACPs. The study population comprised 93 patients divided into 3 groups. Group 1 consisted of 30 patients with ACPs. Group 2 consisted of 33 patients with nasal septal deviation. Group 3 comprised 30 healthy individuals. Venous blood was taken from all participants, and total thiol, native thiol, disulfide, and ischemic modified albumin (IMA) levels were evaluated. When the values of native thiol (P = .097), disulfide (P = .221), total thiol (P = .140), and IMA (P = .091) were compared, no significant differences were found among the 3 groups. While many studies have been conducted related to nasal pathologies and the role of oxidative stress in their etiologies, our study concluded that oxidative stress plays no role in ACP etiology.
Keywords
Antrochoanal polyps (ACPs) are benign lesions that typically arise in the mucosa of the maxillary sinus and extend to the choana. Antrochoanal polyps have 2 components: the cystic and solid parts. 1 -3 Antrochoanal polyps, which most commonly occur in children and young adults, are generally unilateral, but bilateral ACPs have been reported in the literature. 4,5
Common symptoms are nasal obstruction and unilateral nasal drainage. Nasal endoscopy, computed tomography scans, and magnetic resonance scans are important for the diagnosis. Treatment is surgery and the endoscopic endonasal resection is the most recommended technique. 6 Differential diagnosis should include the different causes of unilateral nasal obstruction and ipsilateral nasal masses. 7
The etiopathogenesis of ACPs has not been clarified, 8 -10 and further investigation is needed to discover the etiology of ACPs, particularly as it relates to the role of inflammatory mediators, oxidative stress, and tumor marker expression. Moreover, some research studies have investigated the similarities and differences between ACPs and nasal polyps (NPs). 11,12 However, while oxidative stress has been implicated in the etiology of NPs, it has not been investigated in terms of ACP etiology. 13,14
Thiols (total antioxidant capacity) are organic compounds capable of reacting with free radicals to provide protection against tissue and cellular injury caused by reactive oxygen products that contain a sulfhydryl (−SH) group bound to a carbon atom. In oxidative stress, protein sulfhydryl groups are converted into disulfides (total oxidant capacity). Disulfide bridges subsequently convert back to thiols, and this cycle continues in a balanced manner. The thiol/disulfide balance plays a critical role in oxidative stress, apoptosis, detoxification, antioxidant protection, cellular signal transmission, and enzymatic activities. Serum thiol/disulfide levels were previously analyzed indirectly until direct measurement became possible with the full automatic colorimetric method described by Erel and Neselioglu in 2014. 15
Due to the influence of free radicals, oxidative stress also causes damage to the albumin molecules. In such a case, the albumin is referred to as ischemic modified albumin (IMA). Ischemic modified albumin can be used as a sensitive biochemical marker of ischemia and oxidative stress originating as a consequence of tissue hypoxia. 16,17
The purpose of this study was to evaluate the relationship between ACPs and thiol/disulfide balance, which was used as a marker of oxidative stress. To the best of our knowledge, this study was the first to involve this novel biomarker, which has been used in the investigation of different diseases, 18,19 in patients with ACPs. It was also the first study to investigate IMA levels in ACPs.
The study group comprised 93 patients and 3 groups. Approval for the study was granted by our hospital’s ethical committee. Signed consent forms were received from all 3 group patients.
Group 1 consisted of 30 patients: only antrochonal patients who attended our ENT tertiary center clinic. Group 2 consisted of 33 patients: only nasal septal deviation (NSD) patients. Group 3 consisted of 30 patients: healthy individuals.
Patients with diabetes mellitus, obesity, cardiovascular disease, cerebrovascular disease, acute and chronic kidney disease, liver disease, acute or chronic systemic diseases such as malignancy, using antioxidants, lipid-lowering drugs or vitamins, with acute–chronic infectious diseases, other nasal pathologies except group characteristic, and using cigarettes or alcohol were excluded from the study.
Patients who were diagnosed on the basis of anterior rhinoscopy, endoscopic examination, and paranasal sinus tomography and who needed magnetic resonance scan were included in study. Endoscopic examination was performed using a 4-mm rigid 0° to 30°(Karl-Storz VR GmbH & Co, Tuttlingen, Germany) endoscope.
Laboratory Procedures
Blood specimens were collected after 12-hour overnight fasting. These were then immediately centrifuged for 10 minutes at 1500 rpm for plasma and serum separation. Serum specimens were stored at 80°C until all samples had been collected. Thiol/disulfide hemostasis parameters were subsequently studied in the specimens. All parameters were studied from the same specimen. Thiol/disulfide levels were measured using the method described by Erel. Ischemic modified albumin was measured from the same blood specimens. The data obtained were determined as total thiol, native thiol, disulfide, and IMA levels.
Thiol/Disulfide Measurement
The serum levels of total oxidant status were measured using the TOS Kit (Rel Assay Diagnostics, Ankara, Turkey). The serum levels of total antioxidant status were measured using the TAS Kit (Rel Assay Diagnostics). For oxidative stress index (OSI) calculation, the unit for TAS (millimole of Trolox equivalent per liter) was converted to micromole of Trolox equivalent per liter, and the OSI value was calculated as OSI = (TOS, lmol/L)/(TAC, lmol Trolox equivalent/L/100). 15
Ischemic Modified Albumin Measurement
Bar-Or et al’s method was used for IMA measurement. The albumin cobalt binding test is based on the fact that ischemia-induced structural change in albumin is measured with spectrophotometric measurement of the unbound cobalt after addition of cobalt. The principle of the method is briefly as follows: Serum IMA concentration is measured by spectrophotometric measurement of the unbound cobalt at 470 nm using dithiothreitol after addition of a known amount of cobalt to the serum sample. Since IMA concentrations are not standard, they were given as absorbance unit (ABSU). 17
Statistical Analysis
Data were analyzed using the SPSS version 21.0 software program (Statistical Package for Social Sciences v.21, IBM, Chicago, Illinois). Pearson χ2 test was used to investigate the association between categorical variables. The one-way analysis of variance was used to compare continuous numerical variables between groups. As descriptive statistics, the mean and standard deviation values were given. P < .05 were considered statistically significant.
Results
There was no significant difference in sex comparing the groups. But there was a significant difference between the ages comparing the groups. Group 3 has significant difference in age (P = .001). The mean age of group 1 was 31.3, group 2 was 32.4, and group 3 was 42.
Group 1, group 2, and group 3 native thiol levels were 519.92, 521.56, and 486.23 µmol/L, respectively. Disulfide levels were 17.305, 17.442, and 25.031 µmol/L, respectively; total thiol levels were 554.53, 555.27, and 523.28 µmol/L, respectively; IMA levels were 74.060, 79.572, and 74.586 ABSU, respectively.
Comparing the values of native thiol (P = .097), disulfide (P = .221), total thiol (P = .140), and IMA (P = .091) between the 3 groups, there was no significant difference (Table 1)
Characteristics of the Study Population.
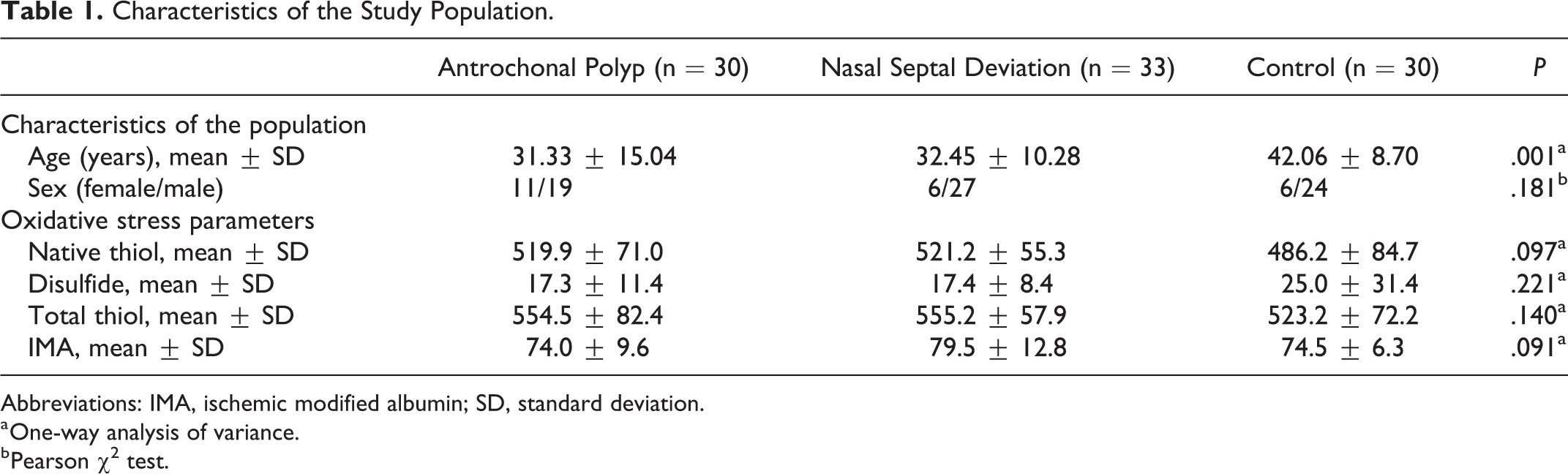
Abbreviations: IMA, ischemic modified albumin; SD, standard deviation.
a One-way analysis of variance.
b Pearson χ2 test.
Discussion
An ACP is a benign sinonasal lesion that is defined as a maxillary sinus polyp originating from the maxillary sinus mucosa that passes through the accessory ostium and extends into the choana. In general, 4% of 6% of NPs are ACPs that epidemiologically affect younger patients. 20 Antrochoanal polyps are generally unilateral, but cases of bilateral ACPs have been published. 4,5 While ACPs and NPs bear some similarities and some studies have investigated the molecular and cellular bases of NPs, because of their low incidence, little is known about ACPs. 2,7,20,21
Nasal polyps are a chronic inflammatory disease of the nasal mucosa. However, their pathophysiology remains unclear. There are no etiological factors that explain the pathogenesis of NPs, but inflammation is thought to be one of the main factors. Oxidative stress parameters are a reliable sign of inflammation.
Studies have explained the relationship between nasal polyposis and oxidative stress. A study conducted in 2016 revealed a mechanism involving oxidative stress operating in the development of NPs. 14 Another study in 2013 demonstrated that oxidative stress is at an increased level in patients with nasal polyposis. 22 In addition, oxidative stress markers increase in chronic sinusitis. 23
Nasal septal deviation is a deformity of the nasal septum from the midline that results in nasal obstruction, which may lead to hypoxia. This hypoxia may alter the cellular metabolism. Some cellular compartments may disappear, and the release of free oxygen radicals from the lysosomes may increase. In the related literature, studies have reported increased production of free oxygen radicals in NSD patients. 24 Thus, in our study, we compared NSD patients with ACP patients. As in NSD, ACPs lead to nasal obstruction, which leads to hypoxia and a potential increase in free oxygen radicals. However, to understand the role of free radicals in ACP etiology, we need to ignore nasal hypoxia.
There was a significant difference between the ages of the groups (P < .05) because group 1 included 6 patients who were younger than 18 years. Like NSD, which typically occurs in the young, ACP can be seen in childhood. The mean ages of the groups were 31.3 (group 1), 32.4 (group 2), and 42 (group 3), and we made the assumption that the age differences were not clinically significant.
There was no significant difference among the 3 groups in terms of the comparison of oxidative stress markers. With the help of these findings, we can conclude that oxidative stress does not play a role in ACP etiology.
Many studies have been conducted about nasal pathologies and oxidative stress, including septal deviation, NPs, and chronic sinusitis, 13,14,22 -24 which concluded that oxidative stress played a role in their etiologies. In our novel study, we concluded that oxidative stress has no role in the etiology of ACPs. This study revealed that ACPs have a different pathophysiology than other nasal pathologies, which requires further investigation.
Footnotes
Authors’ Note
Research involve human participants and ethical approval was obtained from the ethical committee. Informed consent form was obtained from all participants. This study has been presented at the 14th Turkish Rhinology Congress, 2018, Antalya, Turkey.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
