Abstract
Primary atrophic rhinitis is a disease of the nose and of paranasalsinuses characterized by a progressive loss of function of nasal and paranasal mucosa caused by a gradual destruction of ciliary mucosalepithelium with atrophy of serous–mucous glands and loss of bonestructures.The aim of this study was to evaluate the therapeutic effects of topic α-tochopherol acetate (vitamin E) in patients with primary atrophicrhinitis based on subjective and objective data.We analyzed 44 patients with dry nose sensation and endoscopic evidence of atrophic nasal mucosa. We analyzed endoscopic mucosascore, anterior rhinomanometry, and nasal mucociliary clearance before and after 6 months of topic treatment with α-tochopherol acetate. For statistical analysis, we used paired samples t test (95% confidence interval [CI], P < .05) for rhinomanometric and muciliary transit time evaluations and analysis of variance 1-way test (95% CI, P < .05) for endoscopic evaluation. All patients showed an improvement in “dry nose” sensation and inperception of nasal airflow. Rhinomanometric examination showed increase of nasal airflow at follow-up (P < .05); nasal mucociliaryclearance showed a reduction in mean transit time (P < .05); and endoscopic evaluation showed significative improvement of hydration of nasalmucosa and significative decreasing nasal crusts and mucusaccumulation (P < .05). Medical treatment for primary atrophic rhinitis is not clearly documented in the literature; in this research, it was demonstrated that α-ochopherol acetate could be a possible treatment for atrophic rhinitis.
Keywords
Introduction
The definition of dry nose involves several clinical conditions such as the anterior dry rhinitis, primary atrophic rhinitis (PAR) and secondary atrophic rhinitis (SAR), and their complications like ozena and empty nose syndrome. 1 Atrophic rhinitis (AR) is a disease of the nose and paranasal sinuses of considerable clinical interest in otolaryngology. Fraenkel described it for the first time in 1876, but its etiopathogenesis is still debated nowadays. 1-2 According to its etiology, it is classified into PAR and SAR and it is characterized by a progressive loss of function of nasal and paranasal mucosa caused by the gradual destruction of the ciliary mucosal epithelium or respiratory epithelium, by the atrophy of exocrine serous-mucous glands, and by the loss of underlying bone structures. 1 Moreover, the disease involves the metaplastic replacement of the squamous epithelium and subsequent loss of mucociliary clearance. 1 This morphostructural damage of nasal mucosa leads to clinical manifestations such as nasal congestion and paradoxal nasal respiratory obstruction, despite an increase in nasal spaces (paradox stuffy nose) and persistence of secretions; these conditions are mainly determined by the loss of the nasal nerve sensitivity due to submucosal atrophy. 1-2
The diagnosis is clinical based on the subjective and objective findings: increased mucociliary clearance transit time, alteration in rhinomanometric values, nasal symptoms (congestion, dry nose, nasal respiratory obstruction, nasal crusts, mucus secretion, and hypo/anosmia), and epistaxis. 1-2
Several authors have investigated the role of α-tocopherol acetate for its anti-inflammatory, immune, and antioxidant functions have been widely documented in the literature with restoration of epithelium in skin, in oral and vulvovaginal mucosa, and in gastric mucosa and nasal mucosa. 3 -14
Vitamin E acts as a cofactor for the binding of different enzymes for oxidative cascade reaction: it prevents oxidation and destruction of membrane lipids; it interacts with different cellular proteins that regulate the transcription and the expression of genes that code for cytokines and chemokines. 3,9,13
The aim of this study was to evaluate the effects of the therapeutic protocol with α-tocopherol acetate in patients with PAR without infection, based on the subjective and objective measures.
Patients and Methods
From October 2017 to September 2018, we enrolled 44 patients (29 female and 15 male) aged between 34 and 70 years, mean age 57.2 years old, with clinical history and objective findings of PAR. Most of the patients referred hyposmia/anosmia associated with the sensation of dry nose. Informed consent was obtained from all individual participants included in the study. The research protocol was approved by University Control Group; this study was conducted according to the World Medical Association Declaration of Helsinki. This is a retrospective observational research.
The inclusion criteria were clinical evidence of paradox nasal stuffiness sensation, hyposmia/anosmia, sensation of “dry nose,” and endoscopic and computed tomography (CT) scan evidence of mucosal epithelium atrophy with abnormal expansion of paranasal sinuses and nasal spaces. Patients with the history of previous nasal surgery, allergic chronic vasomotor rhinitis, chronic granulomatous disease, use of topical nasal drugs, diagnosis of Sjogren syndrome, prior radiotherapy of the head and neck, and complications of the disease such as septal perforation and crusts infections were excluded. Patients underwent, after general ear, nose and throat examination, a CT scan of the nose and paranasal sinuses, endoscopic rhinologic evaluation, rhinomanometry, and nasal mucociliary clearance (NMC) test with charcoal and saccharine powder.
At endoscopic rhinologic evaluation, using a quantitative scale, we analyzed: – State of hydration of nasal mucosa (0, dry mucosa; 1, partially wet mucosa; and 2, wet mucosa); – presence of nasal crusts (1, 2, and 3 for poor, moderate, and severe crusting; 0 for absence of crusting); – presence of mucus accumulation (1, 2, and 3 for poor, moderate, and severe mucus accumulation; 0 for absence of mucus accumulation).
The endoscopic evaluation was performed always by the same specialist since this score was a subjective data.
Bilateral anterior rhinomanometry (evaluated at 150 Pascal drop pressure) was conducted in basal condition and 5 minutes after nasal decongestion with naphazoline 0.1% nasal spray (1 puff each nostril) in order to analyze nasal flow rates (cm3/s) and nasal resistances (Pa/cm3/s) using ATMOS® Rhino 31 (anterior measurements using olive measuring probe). We obtained 8 groups of results before and after topic treatment with α-tocopherol acetate: – Nasal airflow basal before topic treatment (AFbasalT0); – nasal airflow basal after topic treatment (AFbasalT1); – nasal resistance basal before topic treatment (RbasalT0); – nasal resistance basal after topic treatment (RbasalT1); – nasal airflow after decongestant before topic treatment (AFdecongT0); – nasal airflow after decongestant after topic treatment (AFdecongT1); – nasal resistance after decongestant before topic treatment (RdecongT0); – nasal resistance after decongestant after topic treatment (RdecongT1).
The NMC test was performed 2 hours before rhinomanometry using a mixture of charcoal and 3% of saccharine powder, at 1 to 3
The treatment scheme used for this study was the nasal administration of pure α-tocopherol acetate 2 puffs in each nostril, 3 times a day, for 6 months, so the follow-up was performed at the end of medical treatment.
For statistical analysis, we used descriptive data, means, and standard deviations of each group of results at both rhinomanometric and NMC transit time values before and after topic treatment; and then we compared means between different groups by means of paired samples t test (95% confidence interval (95% CI), P < .05). For endoscopic scores, we used analysis of variance 1-way test (95% CI, P < .05).
Results
In all patients selected for treatment, we analyzed endoscopic evaluation, nasal airflow, and nasal resistances rates at basal and after decongestant rhinomanometry and NMC transit time (Tables 1 -3).
Hydration of Nasal Mucosa, Nasal Crusts and Mucus Accumulation Mean Scores at Endoscopic Evaluation Before and After Topic Treatment With α-Tocopherol Acetate.a
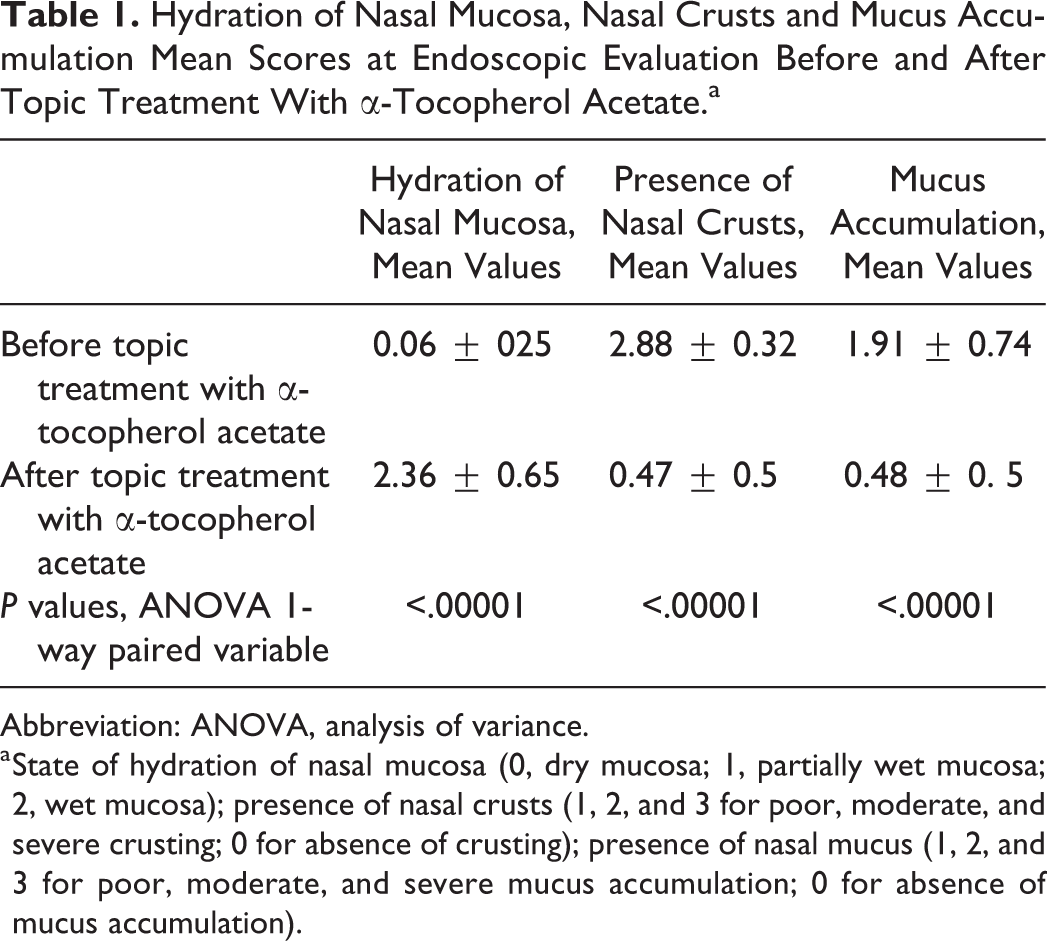
Abbreviation: ANOVA, analysis of variance.
a State of hydration of nasal mucosa (0, dry mucosa; 1, partially wet mucosa; 2, wet mucosa); presence of nasal crusts (1, 2, and 3 for poor, moderate, and severe crusting; 0 for absence of crusting); presence of nasal mucus (1, 2, and 3 for poor, moderate, and severe mucus accumulation; 0 for absence of mucus accumulation).
Rhinomanometric Medical Treatment.a
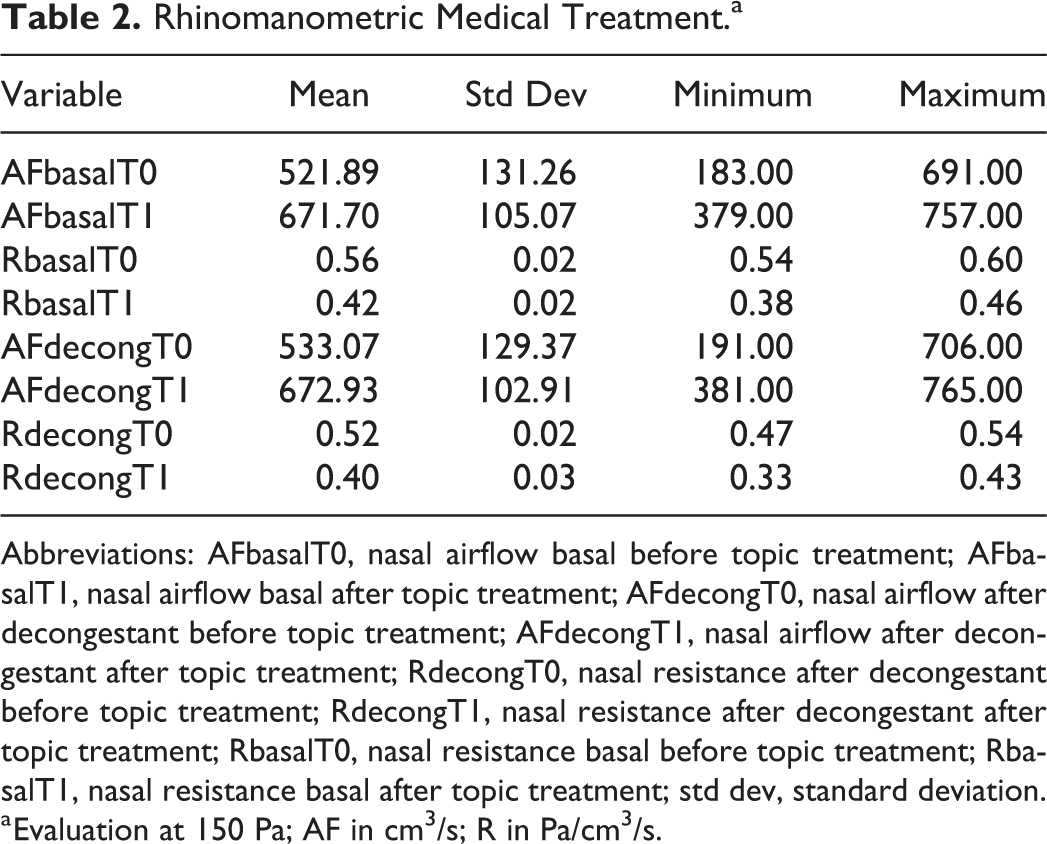
Abbreviations: AFbasalT0, nasal airflow basal before topic treatment; AFbasalT1, nasal airflow basal after topic treatment; AFdecongT0, nasal airflow after decongestant before topic treatment; AFdecongT1, nasal airflow after decongestant after topic treatment; RdecongT0, nasal resistance after decongestant before topic treatment; RdecongT1, nasal resistance after decongestant after topic treatment; RbasalT0, nasal resistance basal before topic treatment; RbasalT1, nasal resistance basal after topic treatment; std dev, standard deviation.
a Evaluation at 150 Pa; AF in cm3/s; R in Pa/cm3/s.
NMC (Transit Time) Results Expressed in Minutes.a
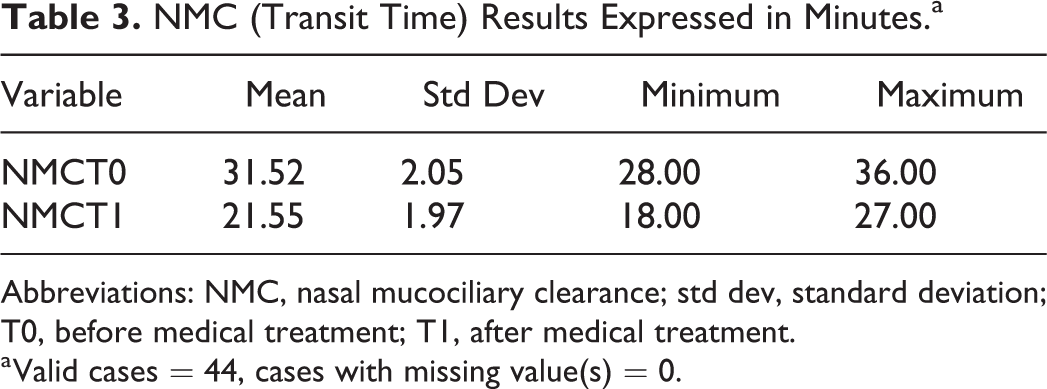
Abbreviations: NMC, nasal mucociliary clearance; std dev, standard deviation; T0, before medical treatment; T1, after medical treatment.
a Valid cases = 44, cases with missing value(s) = 0.
We observed at endoscopic examination a greater hydration of the nasal mucosa than before topic treatment and a decrease of crusts and mucus accumulation (P < .05; Table 1). Before topic treatment with α-tocopherol acetate, we observed dry mucosa with severe crusting and moderate mucus accumulation; after topic treatment, mucosa was wet with poor of absent crusting and mucus accumulation (Figures 1 and 2).
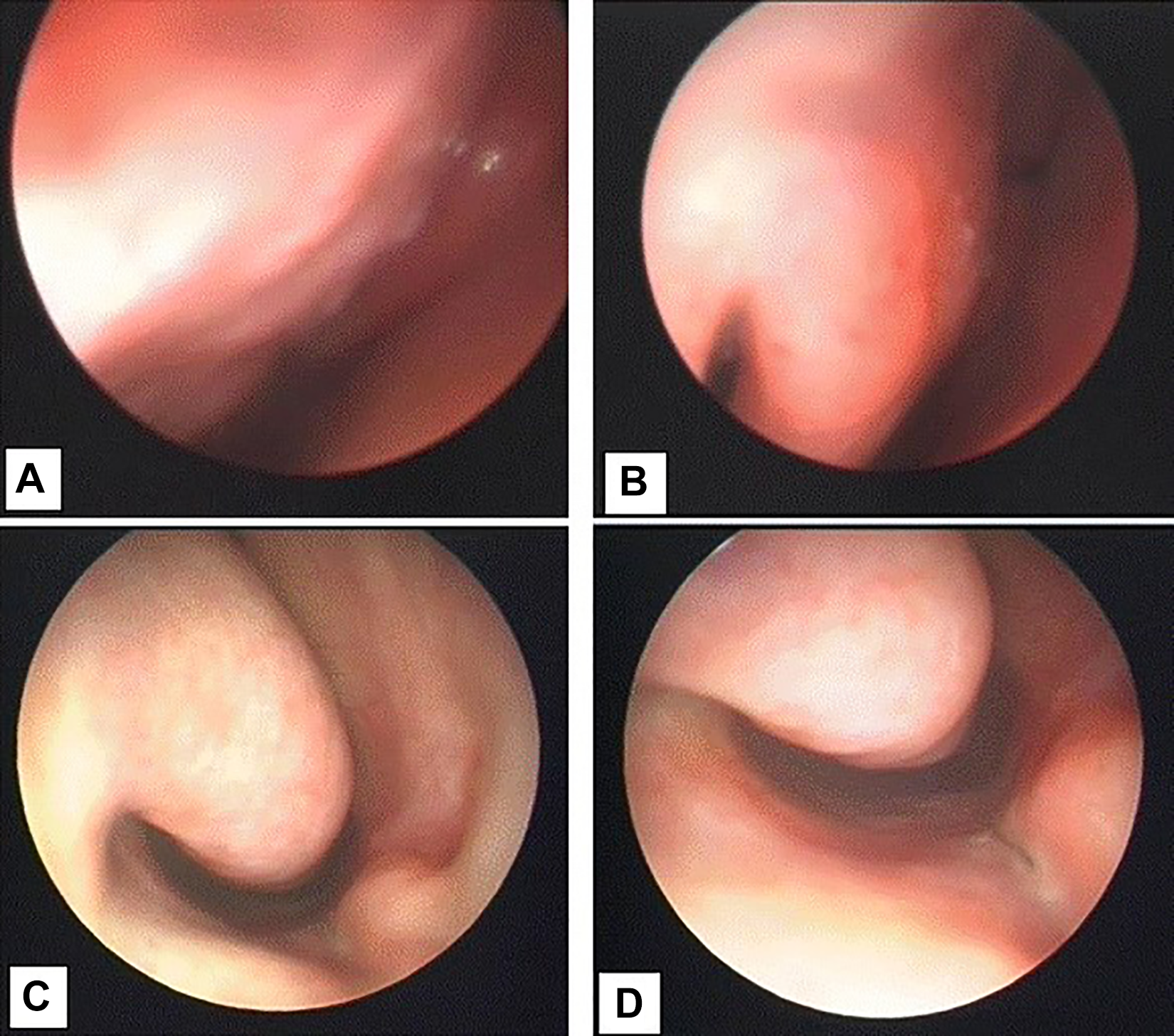
Endoscopic view of nasal mucosa (inferior turbinate) in primary atrophic rhinitis before medical treatment: (A) medial face; (B-D) head.
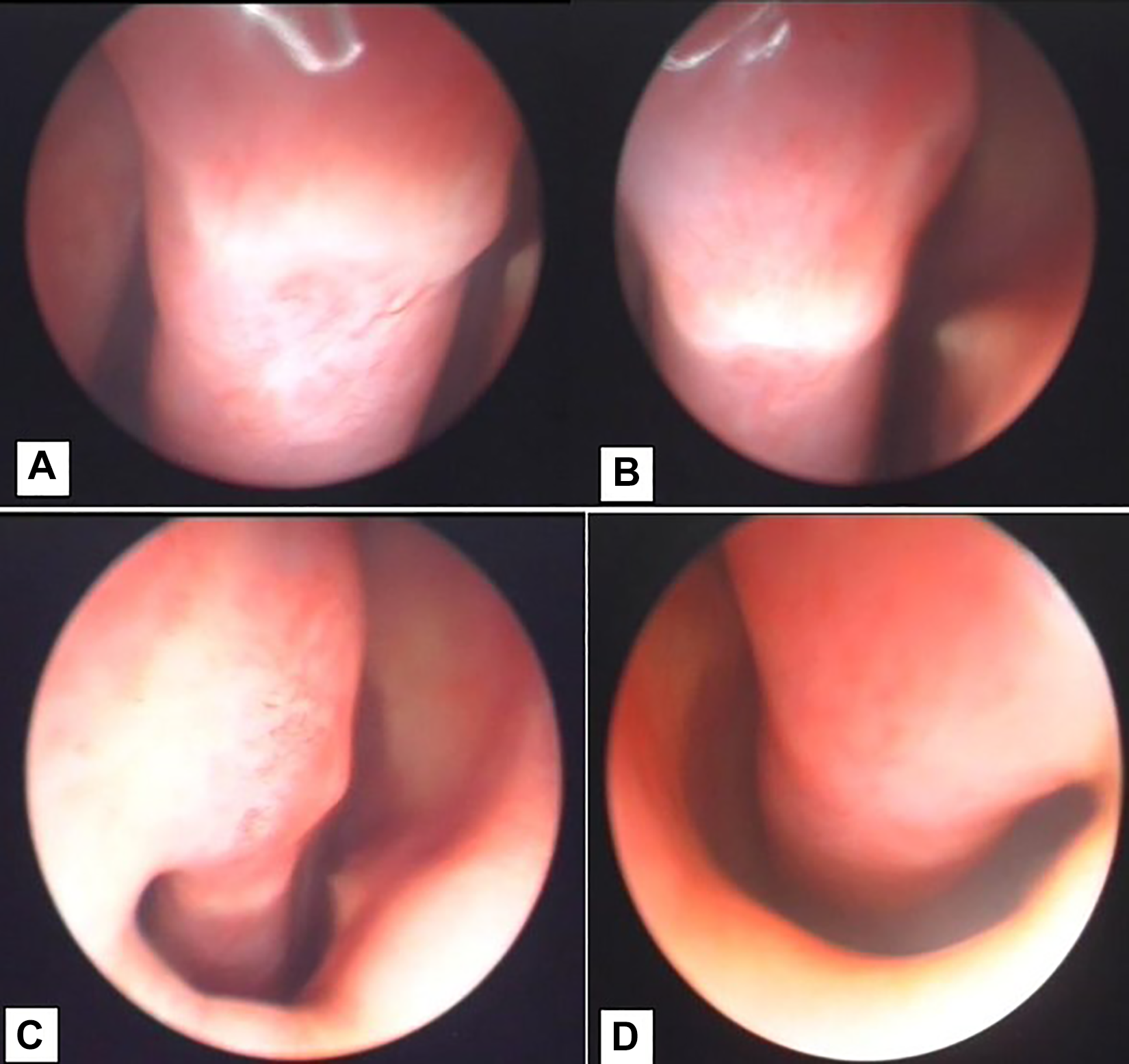
Endoscopic view of nasal mucosa (inferior turbinate) in primary atrophic rhinitis after medical treatment: (A) inferior face; (B-D) head.
At rhinomanometric examination, the analysis of nasal airflows before and after medical treatment, both at basal and after decongestant evaluation, demonstrated increased airflows at follow-up with statistical significance (P < .05); while nasal resistances did not have significative differences before and after topic treatment with α-tocopherol acetate (P > .05; Table 4).
Paired Samples t Test For Rhinomanometric Values.a
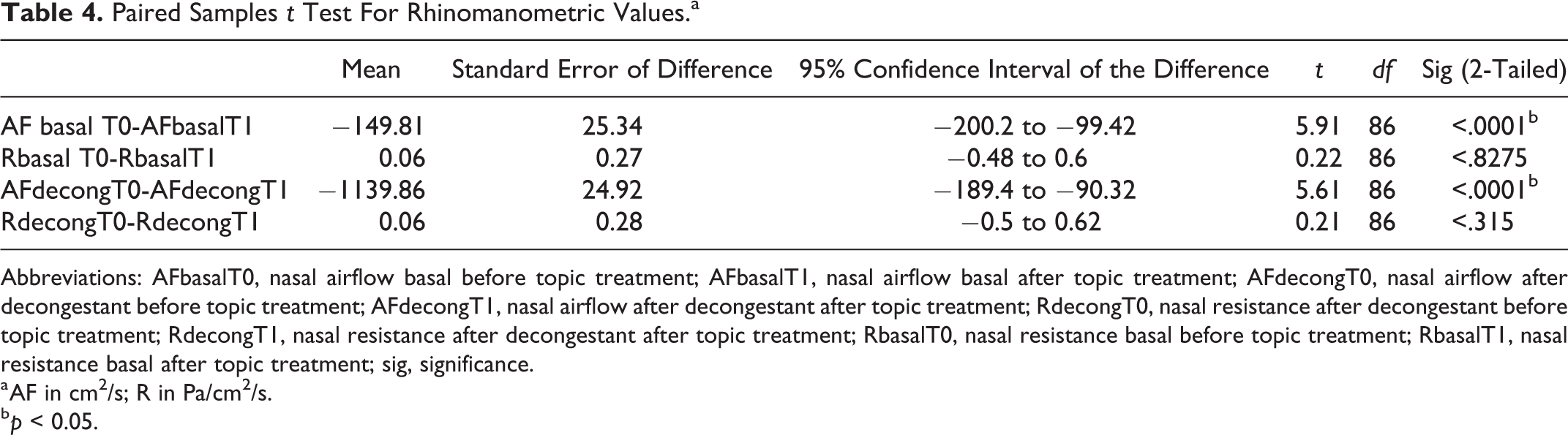
Abbreviations: AFbasalT0, nasal airflow basal before topic treatment; AFbasalT1, nasal airflow basal after topic treatment; AFdecongT0, nasal airflow after decongestant before topic treatment; AFdecongT1, nasal airflow after decongestant after topic treatment; RdecongT0, nasal resistance after decongestant before topic treatment; RdecongT1, nasal resistance after decongestant after topic treatment; RbasalT0, nasal resistance basal before topic treatment; RbasalT1, nasal resistance basal after topic treatment; sig, significance.
a AF in cm2/s; R in Pa/cm2/s.
b p < 0.05.
The NMC test before topic nasal treatment showed a severe prolonged time (mean 31.52 minutes) instead after medical topic treatment with α-tocopherol acetate, the mean transit time was 21.55 minutes (prolonged transit time) with statistical significance (P < .05; Table 5).
Paired Samples t-Test for Nasal Mucociliary Clearance Transit Time Values.
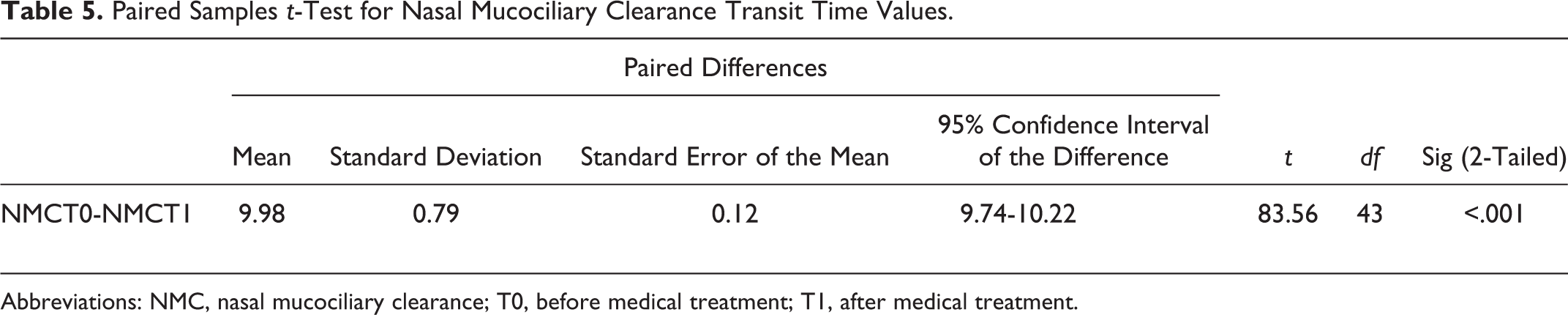
Abbreviations: NMC, nasal mucociliary clearance; T0, before medical treatment; T1, after medical treatment.
All patients showed an improvement in “dry nose” sensation and in the perception of nasal airflow (apparent remission of paradoxical stuffy nose sensation) with an improvement of hyposmia/anosmia.
Discussion
Nasal-sinusoidal walls are lined by nasal mucosa providing to several functions like heating up, humidifying and purifying inspired air, and nonspecific and specific acting against environmental pathogens and others. Nasal mucosa functions are regulated by several factors like nervous system and sex hormones acting in different manner during life. 16
Dysregulation or dysfunction of these mechanisms leads to several clinical conditions characterized by dry nose such as PAR, SAR, and their complications. 1,2
Primary chronic AR is a clinical condition with a higher prevalence in women after puberty, associated with hereditary factors, endocrine imbalances, racial factors, nutritional deficiencies such as lack of vitamin A or D, iron, and autoimmune disorders. 16
The diagnosis was clinical while CT scan was indicated when signs of chronic rhinosinusitis are found or to obtain adjunctive evidence of PAR. 1,2
Treatment for PAR is not well defined, and it is often empirical. 17-18 The saline washes must be considered as the first choice for several authors to promote the cleaning of the nasal cavity and to remove secretions and crusts, which could provoke secondary infections. 17-18 Other therapeutic approaches propose the use of bicarbonate antiseptic solutions in which the diborated sodium acts as an antiseptic and antibacterial substance; bicarbonate sodium helps to dissolve the crusts; and the chloride sodium makes the solution isotonic. 17-18 Glycerin drops or spray associated with glucose can be used because they allow the lubrification of the nasal mucosa. 17-18 The glucose fermentation acidifies the pH and hinders bacterial growth. 17-18
Bacterial superinfections are treated with specific antibiotics such as rifampicin 600 mg daily for 12 weeks; ciprofloxacin 500 to 750 mg for 8 weeks. 19
Surgical treatments, instead, provides for the partial or complete closure of the nostrils with autologous or synthetic implants. 19 Other alternative treatments described in the literature propose the use of liposucked with autologous platelet-reached plasma, subcutaneous fat, cancellous bone, autologous bone marrow grafts, grafts of placenta, or adipose tissue. 20
In our previous study, we found a decreased healing time in elderly patients affected by chronic rhinosinusitis after endoscopic sinus surgery treated with topic nasal α-tocopherol acetate for 3 months. 14
In consideration of our previous research, 14 the treatment scheme used for the present study was the nasal administration of α-tocopherol acetate 2 puffs in each nostril 3 times a day, for 6 months. The follow-up was performed after 6 months of medical treatment and consists of the endoscopic rhinologic examination, basal and after decongestant rhinomanometry, and NMC transit time test.
All patients showed an improvement in nasal respiratory function for increased nasal airflow; the patients had a better response to rhinomanometric tests after treatment (increased nasal airflow). Nasal resistances, according to the literature, did not have significative differences before and after medical treatment proposed in this research, neither after decongestant: maybe due to a vascular depletion in AR and to the duration of treatment. 18
Furthermore, the NMC test, after treatment, showed a reduction of the mean transit time near the normal transit time (up to 20 minutes but less than 31 minutes). The duration of NMC in normal individuals is up to 20 minutes, it is prolonged if it is 21 to 31 minutes; it is considered severely or grossly prolonged if it is 31 to 60 minutes or up to 60 minutes. 21
The results obtained suggest the use of α-tocopherol acetate in PAR; this study investigated an aspect scientifically poorly explored: What is the most correct strategy in the pharmacological treatment of PAR? Moreover, we also want to give an impulse for scientific studies on the effect of vitamin E, and specifically of α-tocopherol acetate, in the nasal tropism.
PAR and ozena are often used to indicate the same clinical condition, even if it is important to distinguish them because of in the second one, we find bacterial infection. Medical treatment for PAR is not clearly documented in the literature in terms of follow-up and clinical evaluation with subjective (symptoms) and objective methods (endoscopic evaluation, rhinomanometry, and NMC transit time) and medical treatment. In the present study, we showed relevant results in nasal functions after medical topic treatment with α-tocopherol acetate (vitamin E), and these results lay the foundation for further application of this molecule in sinonasal pathology.
Footnotes
Authors’ Note
Giuseppina Marcuccio and Domenico Testa contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All participants in peer-review and publishing have no conflicts of interest since they have no financial or personal relationship that might bias or be seen bias their work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
