Abstract
Objective:
Previous studies suggested that hypoxia-inducible factor-1α (HIF-1α) plays an important role in the progression of inflammation and remodeling of chronic rhinosinusitis with nasal polyposis. However, the molecule mechanisms of HIF-1α activation and regulation of cytokine expressions, such as interleukin (IL) 25 and IL-17RB, in nasal polyposis are not clear.
Method:
The IL-25 and IL-17RB levels in human nasal epithelial cells after stimulation by lipopolysaccharide (LPS) were detected by enzyme-linked immunosorbent assay method, and the proteins of HIF-1α and p-Akt were detected by Western blot method. Moreover, we evaluated the cytokine levels in the nasal mucosa of a murine model of nasal polyposis.
Results:
The levels of IL-25 and IL-17RB showed dose- and time-dependent release in response to LPS stimulation. The proteins of HIF-1α and p-Akt were both increased significantly after LPS stimulation. After inhibition of PI3K/Akt pathway by PI3K inhibitor LY294002, the levels of IL-25 and IL-17RB and HIF-1α were decreased by LPS stimulation.
Conclusions:
Inhibition of PI3K or HIF-1α pathway could significantly reduce growth factor production and decrease nasal inflammation. The HIF-1α pathway could be a novel therapeutic approach for reducing nasal airway inflammation and remodeling in nasal polyposis.
Introduction
There are 2 subtypes of the chronic rhinosinusitis (CRS), with polyps (CRSwNP) and without polyps (CRSsNP). The prevalence of CRSwNP ranged from 0.5% to 4.3%, which is less common than CRSsNP in national population surveys. 1 However, there are about 38% to 69% of CRSwNP cases commonly leading to revision endoscopic sinus surgery therapy. 2 –4 Olfactory impairment is the common symptom of CRSwNP patients 5 and is also likely to endorse nasal obstruction. 6
Previous studies suggested that hypoxia-inducible factor 1α (HIF-1α) was overexpressed in the lung and nasal mucosa, which played an important role in airway inflammatory responses. 7,8 The member of interleukin (IL) 17 cytokine family, IL-25, has been overexpressed in many inflammatory animal models, such as atopic dermatitis, asthma, and pulmonary fibrosis. It is said that eosinophils or TH2 cytokines would been producted in bronchoalveolar lavage fluid and lung tissue after IL-25 intraperitoneal or intranasal administration. 9,10 Recently, IL-25 also reported to play a main role in promoting TH2-mediated inflammation. 11 However, IL-25 roles in Asian patients with CRSwNP at present have not been illuminated.
Chronic rhinosinusitis with polyps is a multifactorial disease, as a result of the persistent inflammation of nasal cavity and sinuses. The pathogenesis of CRSwNP remains unclear. In the present study, the mechanisms between the IL-25 production and HIF-1α and lipopolysaccharide (LPS) activation in vitro and in vivo were investigated. We had emphasized the role of PI3K/Akt/HIF-1α pathway in regulating IL-25 expression by LPS stimulation.
Materials and Methods
Cells Lines
Human nasal epithelial cells (HNEpCs) were purchased from American Type Culture Collection and were grown in standard culture medium (RPMI-1640 containing 10% fetal bovine serum, 2 mM
Lipopolysaccharide Stimulation
When HNEpCs reached 80% to 90% confluence, the cells were washed by phosphate-buffered saline (37°C, pH 7.4) and added fresh culture medium. The LPS (Sigma, St Louis, Missouri) was added to the cultures at the concentrations of 0, 0.1, 0.5, and 1 μg/mL and incubated at 37°C for 0 to 24 hours.
Hypoxia-Inducible Factor 1α shRNA Transfection and Inhibitor Treatment
Human nasal epithelial cells were transfected by HIF-1α short hairpin RNA (shRNA) or control shRNA 36 hours before LPS (1 μg/mL) stimulation. The used HIF-1α shRNA oligonucleotide primers were: forward 5′-AATTCHCCHHCCHCYHHAHACACAATCATATCTCGAGATATGATTGTGTCTCCACGGTTTTTTG-3′ and reverse 5′-GATCCAAAAAACCGCTGGAGACACAATCATATCTCGAGATATGATTGTGTCTCCAGCGGCCGGCG-3′. After 24-hour transfection, the cells were harvested.
The LY294002 (Sigma), PI3K-specific inhibitor, was dissolved in dimethyl sulfoxide (DMSO). Serial concentrations of LY294002 (0, 5, 10, 20 μmol/L) and DMSO vehicle (<0.1%) were administrated into the cultures 1 hour before LPS stimulation for 24 hours. There was no cytotoxicity observed by Trypan blue dye exclusion.
Enzyme-Linked Immunosorbent Assay
The IL-25 and IL-17RB expression levels in the supernatant were detected by enzyme-linked immunosorbent assay (ELISA) method according to the instruction of ELISA kits which were purchased from BioLegend (San Diego, California).
Western Blot Analysis
Proteins were extracted from whole cell lysates and separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis, then transferred to a polyvinylidene fluoride membrane. The primary antibodies against HIF-1α and p-Akt (Abcam, Cambridge, Massachusetts) and against IL-25 and IL-17RB (Santa Cruz Biotechnology, Santa Cruz, California) were used. The membranes were then incubated with secondary antibodies (Abcam). Detection was performed using the LI-COR Odyssey Scanning Infrared Fluorescence Imaging system (LI-COR Biosciences, Lincoln, Nebraska).
In Vivo Experiments
Eight-week-old healthy male BALB/c mice (26-38 g) were obtained from the Experimental Animal Center of Central South University. The mice were immunized by 100 μg/mouse LPS and 1 mg/mouse aluminum hydroxide (Sigma) by intraperitoneal injection. The sensitization was repeated at days 1, 8, and 15, then followed LPS solution by intranasal instillations on days 22 to 29. All of the procedures were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals with the approval of the Institutional Animal Care and Use Committee of Central South University.
Mice were divided into 4 groups consisting of 3 mice in each group, including (1) negative control group: saline stimulation with vehicle treatment (SAL + VEH); (2) positive control group: LPS stimulation with vehicle treatment (LPS + VEH); (3) LY294002 group: LPS stimulation with LY294002 treatment (LPS + LY294002); (4) 2ME2 group: LPS stimulation with 2-methoxyestradiol (2ME2, HIF-1α inhibitor) treatment (LPS + 2ME2). The dose of LY294002 was 7.5 mg/kg/d and the dose of 2ME2 was 30 mg/kg/d, which were administered by intranasal instillation 2 hours before stimulation.
Hematoxylin and Eosin Staining
The tissues were fixed using 10% formalin at room temperature for 24 hours and embedded in paraffin and sectioned to 3 to 5 μm. After hydrating the samples, the sections were stained in hematoxylin solution for 1 minute and were washed under running tap water at room temperature for 5 minutes. Samples were then stained in working eosin Y solution for 10 seconds. The slides were viewed under a light microscope. The number of infiltrating eosinophils for each animal was expressed as the mean value from the 5 fields.
Statistical Analysis
All statistical analysis was carried out using the SPSS version 18.0 software (SPSS, Inc, Chicago, Illinois). All values were expressed as the mean (standard deviation) from at least 3 repeated individual experiments for each group. The differences among groups were analyzed by 1-way analysis of variance followed by Bonferroni multiple comparison tests or
Results
Lipopolysaccharide Stimulation Increased IL-25 and IL-17RB Proteins in the HNEpCs
The cytokine levels were determined after LPS stimulation at different concentrations (0-1 μg/mL) and different time points (0-24 hours). The results showed that IL-25 and IL-17RB levels were determined after LPS stimulation in a dose- and time-dependent manner. The most prominent elevated levels of IL-25 and IL-17RB were 1 μg/mL and 24-hour LPS stimulation (data not shown).
Lipopolysaccharide-Induced Cytokine Expression via the HIF-1α Pathway
During LPS stimulation at 0, 0.1, 0.5, and 1 μg/mL for 24 hours, the HIF-1α and p-Akt protein were overexpressed significantly in HNEpCs using Western blotting (Figure 1A and B), and both increased significantly after stimulation with the higher concentrations of LPS. These results were consistent with the patterns of IL-25 and IL-17RB protein in HNEpCs (Figure 1C). Therefore, these findings suggested that IL-25 and IL-17RB overexpressed in response to LPS stimulation by activation of HIF-1α signaling pathway in HNEpCs.
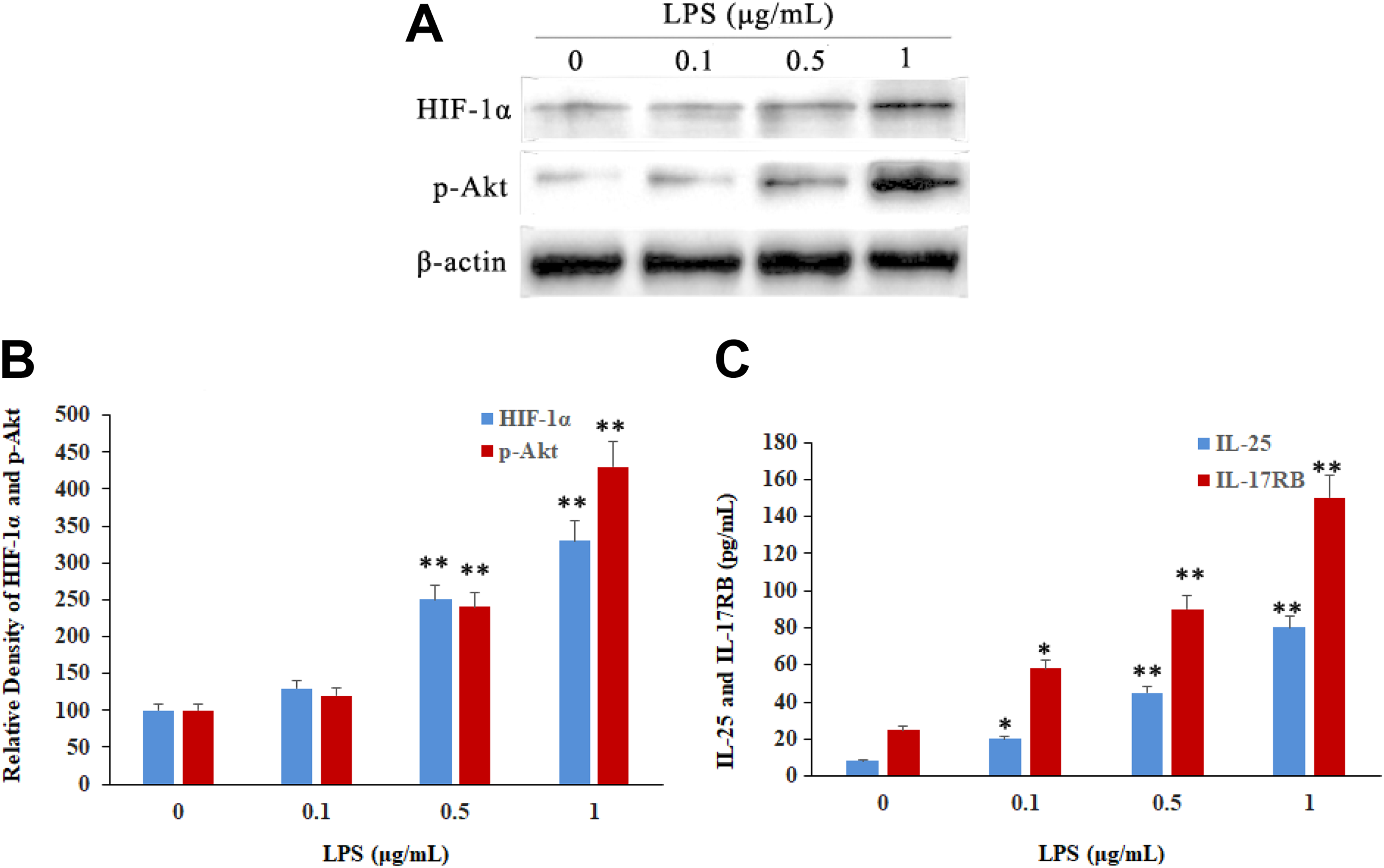
Lipopolysaccharide (LPS)-induced cytokine expression by activation of the Akt/HIF-1α pathway in human nasal epithelial cells by ELISA. Hypoxia-inducible factor 1α and p-Akt expressed by LPS stimulation for 24 hours in human nasal epithelial cells by Western blot. Lipopolysaccharide induced a dose-dependent increase in HIF-1α (A and B), p-Akt (A and C), IL-25 (D), and IL-17RB (E). Values represent the means (SD) of at least 3 independent experiments. *
To further investigate the role of HIF-1α activation of LPS stimulation by IL-25 and IL-17RB expression, the HIF-1α shRNA was transfected to HNEpCs. As shown in Figure 2A and C, HIF-1α protein was significantly upregulated after LPS (1 μg/mL) stimulation for 24 hours, then significantly downregulated after LPS stimulated cells which transfected with HIF-1α shRNA. In addition, the increased HIF-1α messenger RNA expression was also downregulated by HIF-1α shRNA transfection using reverse transcription polymerase chain reaction analyses (Figure 2B and D). Simultaneously, the increased IL-25 and IL-17RB expressions were also downregulated by HIF-1α shRNA transfection after LPS stimulation (Figure 2E and F). Therefore, the HIF-1α pathway was necessary for LPS-induced expression of IL-25 and IL-17RB.
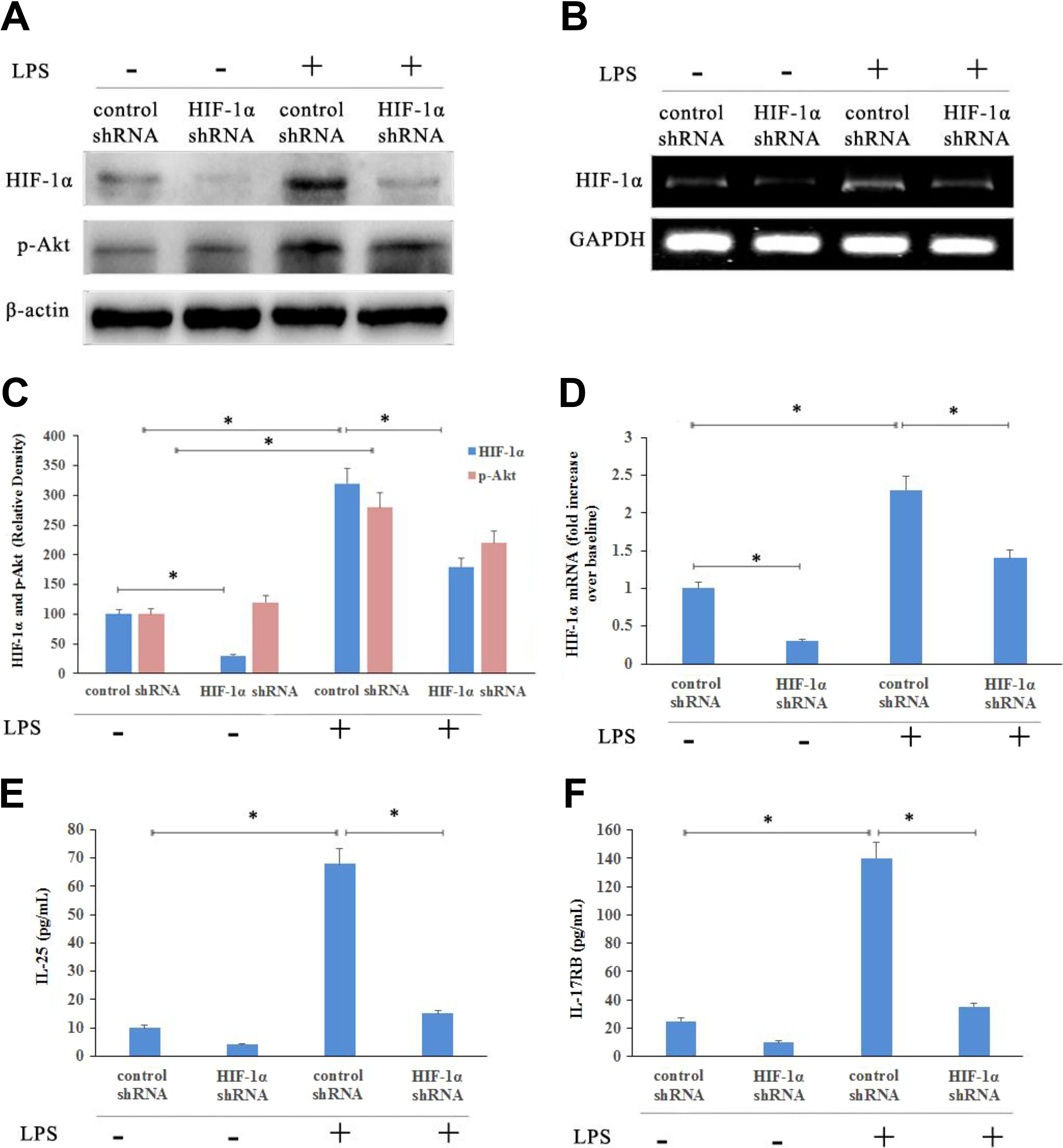
Hypoxia-inducible factor 1α knockdown reduced cytokine expression in human nasal epithelial cells. Hypoxia-inducible factor 1α and p-Akt protein (A and C) and HIF-1α mRNA (B and D) levels were determined by Western blotting and RT-PCR, respectively. The levels of IL-25 (E) and IL-17RB (F) were measured by ELISA in the supernatants of human nasal epithelial cells. Values represent the means (SD) of at least 3 independent experiments. *
Lipopolysaccharide Regulated HIF-1α Expression by PI3K/Akt Pathway
The specific PI3K inhibitor LY294002 was used to inhibit PI3K signaling activation after LPS stimulation in HNEpCs. The results showed that the p-Akt and HIF-1α were significantly decreased by LY294002 in a dose-dependent manner (Figure 3A and B). Furthermore, LY294002 also significantly inhibited IL-25 and IL-17RB levels in HNEpCs after LPS stimulation in a dose-dependent manner (Figure 3C and D). These results suggested that the LPS stimulation could induce the production of cytokine by activation of HIF-1α. However, this regulation pathway could interrupt during inhibition of PI3K/Akt pathway.
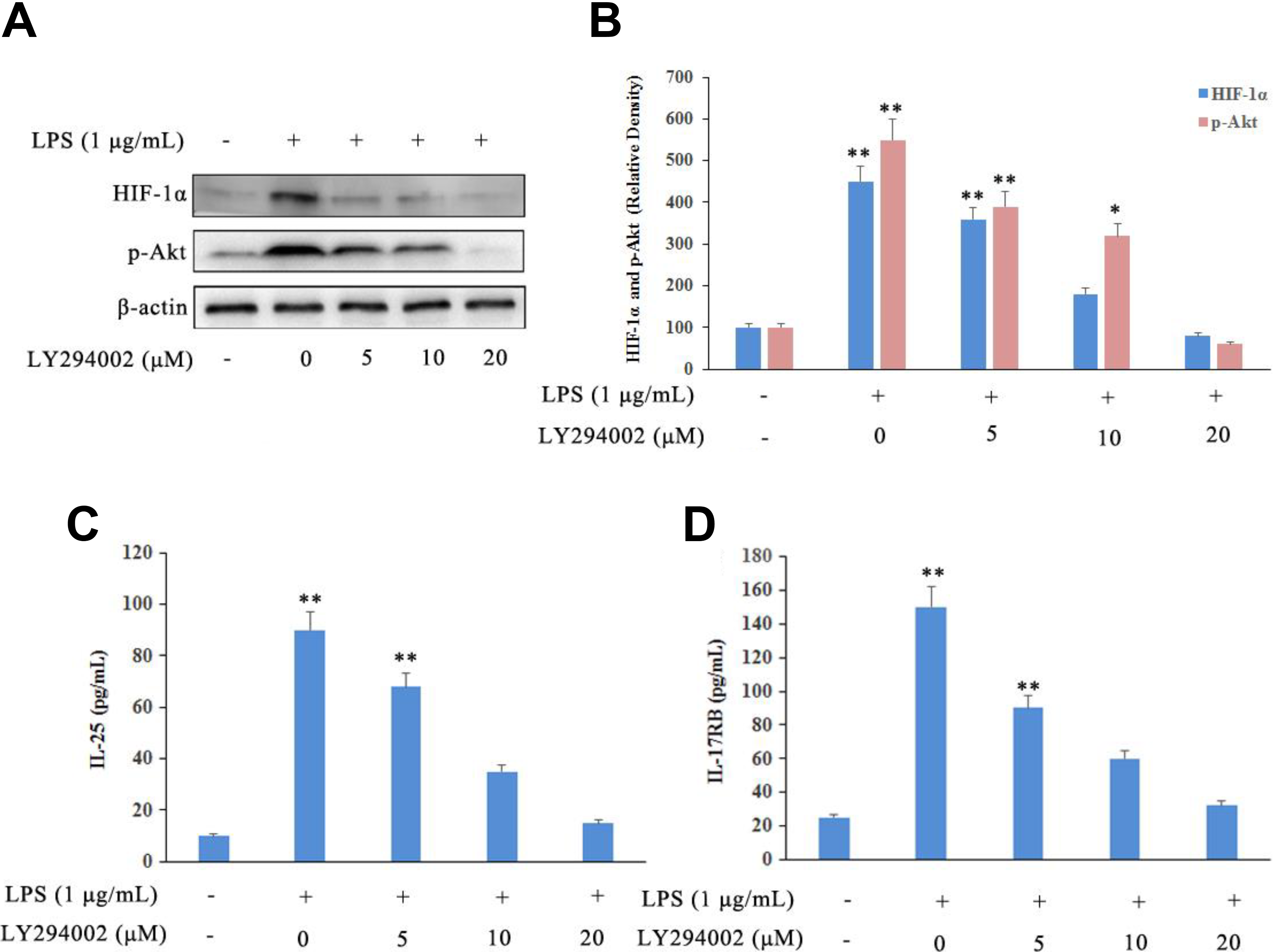
LY294002 interrupted LPS-induced PI3K signaling activation in human nasal epithelial cells. LY294002 significantly decreased the levels of HIF-1α (A), p-Akt (B) and IL-25 (C), and IL-17RB (D) production in epithelial cells in a dose-dependent manner by Western blot and ELISA analysis. Values represent means (SD) of at least 3 independent experiments. *
Regulation of Cytokine Expression by the PI3K/Akt/HIF-1α Pathway In Vivo
A CRS murine model was developed to assess the cytokine expression regulated by PI3K/Akt/HIF-1α pathway in vivo. Hematoxylin and eosin staining showed that the number of eosinophils was increased significantly after LPS stimulation with vehicle treatment, compared to the negative control group (
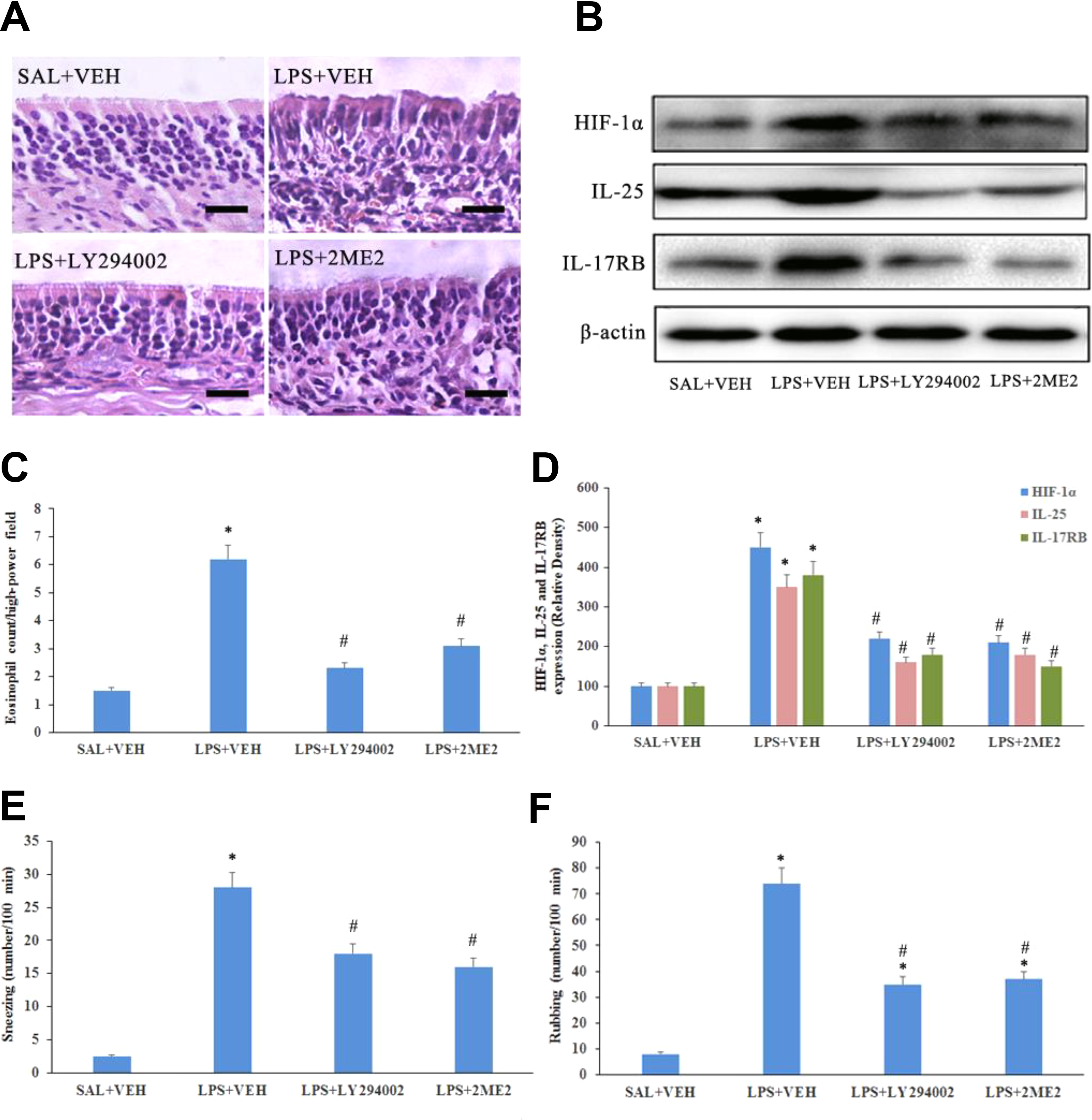
Lipopolysaccharide induced IL-25 and IL-17RB expression via the PI3K/Akt/HIF-1α pathway in vivo. Chronic rhinosinusitis mice were stimulated with LPS or saline with or without preadministration of LY294002 (PI3K inhibitor) or 2-methoxyestradiol (HIF-1α inhibitor). A, Hematoxylin and eosin (HE) staining in the nasal mucosa in each group (magnification ×400, scale bar = 20 μm). B and D, Hypoxia-inducible factor-1α, IL-25, and IL-17RB expression by Western blot. C, Eosinophil counts from HE staining were significantly increased after LPS stimulation and decreased by LY294002 or 2-methoxyestradiol (n = 3) in the nasal mucosa. Values represent the means (SD) of at least 3 independent experiments. The number of sneezes (E) and frequency of nasal rubbing (F) were counted over a 10-minute observation period immediately following LPS stimulation (n = 3).*
Discussion
During the process of respiratory movement, nasal mucosa is the first to be exposed to many microorganisms as well as their degradants, such as LPS. Respiratory diseases such as rhinosinusitis and asthma are characterized by aberrant immune function of airway epithelial cells. 12,13 Lipopolysaccharide has already been regarded as a main regulator of inflammation. High exposure level of LPS is associated with the occurrence and development of many airway diseases. 14
Interleukin-25 production by bronchial and nasal epithelial cells is regulated by transcription and protein expression, and allergen proteases can play pivotal roles in both of these biological processes. 15 Various studies have shown that IL-25 was mainly produced by mast or TH2 cells. 16,17 In the present study, IL-25 and IL-17RB were overexpressed by LPS simulation in HNEpC.
Previous studies showed that HIF-1α overexpression played a significant role in inflammatory response in the nasal cavities, not only in CRSwNP patients but also in murine models. 18 However, the biochemical mechanisms of HIF-1α activation and the relationship with cytokine release, such as IL-25 and IL-17RB in CRSwNP, are unclear. According to previous reports, we hypothesized that HIF-1α expression and cytokine release could lead to regulation of airway inflammation and remodeling.
What’s more, increasing studies have shown the role of HIF-1α in regulating allergic immune response of the PI3K/Akt pathway in allergic asthma both in vitro and in vivo. 19 However, the allergic pulmonary inflammation response regulated by PI3K/Akt pathway is not clear. Therefore, the present study investigated the HIF-1α activation and the PI3K/Akt pathway regulation and the IL-25 and IL-17RB release in response to LPS in in vitro and in vivo studies.
It is a better therapeutic strategy for understanding the precise mechanisms in the progression of allergic airway inflammation and remodeling. Some cytokines, such as IL-17A, were considered as remodeling-associated mediators in the allergic airway. 20 The present study showed that HIF-1α was a key mediator in these remodeling. Consequently, simultaneously affecting multiple cytokines (IL-25 and IL-17RB) by targeting HIF-1α may be more effective than only targeting one particular cytokine at blocking remodeling.
In conclusion, our findings indicated that LPS induced the expression of remodeling-associated mediators IL-25 and IL-17RB regulated by the PI3K/Akt/HIF-1α pathway, not only in HNEpCs but also in CRS murine model. Our results clearly demonstrated that targeting to HIF-1α could modulate allergic inflammatory responses and remodeling, which may provide the basis for developing pharmacological interventions for reducing nasal airway inflammation and remodeling in nasal polyposis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
