Abstract
This investigation explored the outcomes of 4 standardized treatments in patients with refractory chronic rhinosinusitis (CRS), despite recent maximal medical therapy (MMT). In a prospective observational study, we compared continued nasal steroids and irrigation (cNSI), repeated MMT (rMMT), pulsed nasal steroid inhalation (PSI), and endoscopic sinus surgery (ESS). Between November 2015 and March 2016, patients with symptomatic CRS despite having received MMT during the year prior to symptom reoccurrence were offered 1 of 4 standardized treatments. Reflecting real-world conditions, patients selected their treatment option following physician counseling. Sino-Nasal Outcome Test-22 (SNOT-22) scores were obtained before treatment, at the end of treatment, and at 2 months and 1 year following treatment. The mean (± standard deviation [SD]) duration since last MMT was 144 (±36 days). Of the 130 patients, 52 selected cNSI, 16 PSI, 19 rMMT, and 43 ESS. Mean SNOT-22 scores before treatment did not significantly differ between treatments (P = .99). Overall, SNOT-22 scores decreased from 38 ± 2 before treatment to 20 ± 2 after 1 year (P < .001), with a higher reduction for patients having CRS with nasal polyps than for those without nasal polyps (35 ± 2 to 15 ± 2 vs 41 ± 3 to 25 ± 4, respectively; both P < .001). Overall, no difference between the 3 medical treatments was observed (all P > .2). Post-treatment scores following ESS (19 ± 2) were significantly lower than for each of the 3 medical treatments (cNSI 26 ± 2, P = .004; PSI 27 ± 3, P = .026; rMMT 28 ± 3, P = .008). At 1 year following ESS, 26 of 31 patients were asymptomatic and did not require additional systemic steroids, compared to 25 of 50 patients following medical treatment (P = .002). The investigated standardized treatments significantly improved SNOT-22 scores in patients with refractory CRS under real-world conditions. Both patients having CRS with and those without nasal polyps showed significant improvement in SNOT-22 scores, although a less profound effect was found among the latter group. Patients who selected ESS were less symptomatic during the first follow-up year than patients who selected medical treatment alone. Patients with refractory CRS did not benefit from an additional course of MMT in comparison to those who were treated only with cNSI.
Introduction
Chronic rhinosinusitis (CRS) is a common chronic inflammatory disease with an estimated prevalence of 4.5% to 12% in North America and Europe. 1 CRS has been reported to reduce quality of life (QoL) and lead to economic losses. 2 Previously, endoscopic sinus surgery (ESS) was considered a first-line treatment for CRS. Randomized controlled trials (RCTs), however, have not confirmed the superiority of ESS over medical treatment. 3 -5 It has thus become standard to manage CRS primarily with medical treatment, 6,7 with options including irrigation, 8 nasal steroids. 9 and maximal medical therapy (MMT). 10,11 MMT is considered appropriate for patients with CRS who do not respond to nasal steroids and irrigation. 6,7 Currently, several protocols for MMT exist. In a large review, MMT protocols were detailed in only 21% of cases, and oral antibiotics (3-6 weeks) were frequently combined with systemic corticosteroids (2-4 weeks). 12
When patients remain symptomatic despite recent MMT, their CRS is termed “refractory to MMT”, 13 -17 which has been reported in up to 60% of patients. 13 Unfortunately, definitions concerning the intensity and duration of MMT that justifies classifying patients as refractory have been inconsistent. In the European Position Paper on Sinusitis (EPOS), 6 which takes the intensity of recent medical treatment into account, the terms “partly controlled” and “uncontrolled” are proposed for refractory CRS (Table 1).
Assessment of DCS of CRS (in the Last Month) According to EPOS Recommendations. 6
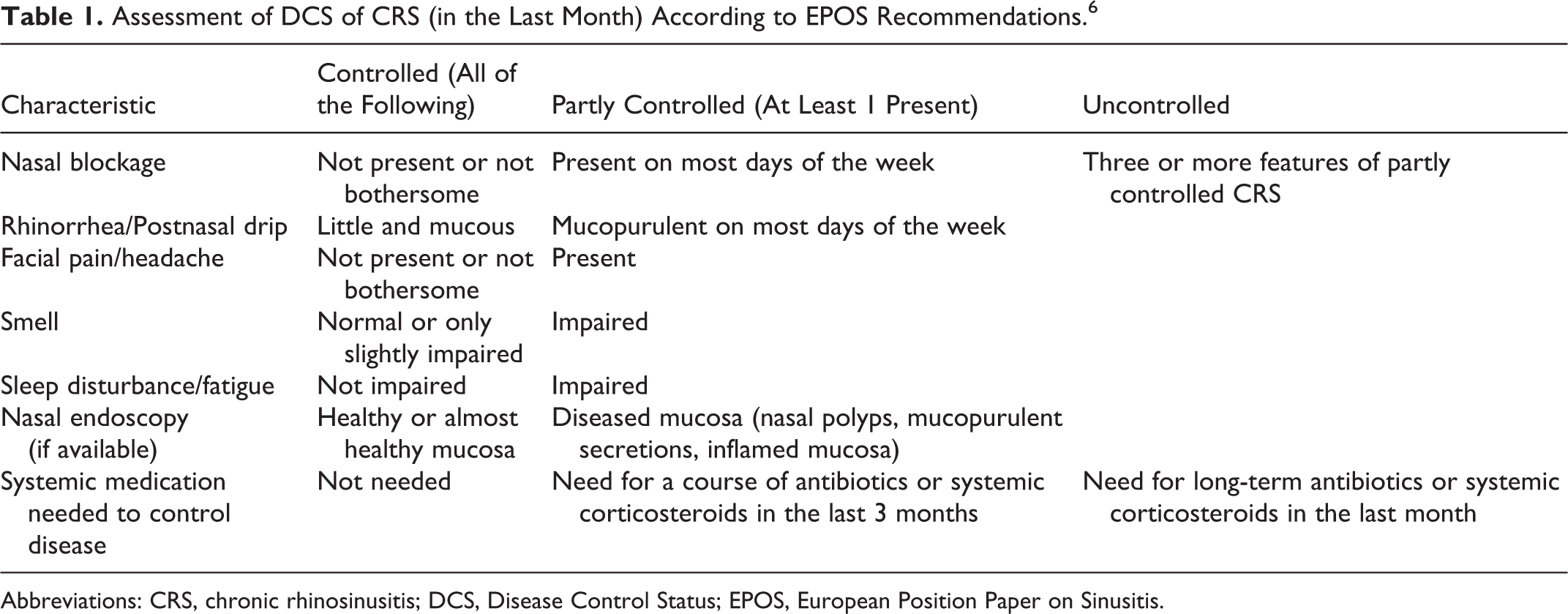
Abbreviations: CRS, chronic rhinosinusitis; DCS, Disease Control Status; EPOS, European Position Paper on Sinusitis.
For patients with CRS refractory to MMT, treatment options include continuation or intensification of nasal steroids and irrigation 18,19 or repetition of MMT. Alternatively, ESS combined with medical treatment may be offered. 6,7,14,16,17,20 Available data on the effectiveness of these treatments are sparse and have seldom been gathered under real-world conditions in which patients select treatment according to their preferences. Moreover, no explicit treatment recommendations are currently available for patients with CRS refractory to recent MMT. 16,17,21,22
In this prospective observational study, we investigated the outcomes of 4 standardized treatments (Table 2) for patients with refractory CRS, despite having received MMT during the year prior to symptom reoccurrence. Reflecting real-world conditions, patients selected their treatment themselves after receiving counseling from their physician. Sino-Nasal Outcome Test-22 (SNOT-22) scores 23 and disease control status (DCS) 6 served as outcome parameters.
Clinical Parameters and Treatment Selection of 130 Patients With Refractory CRS Following Previous MMT at T0, Grouped by Presence of Polyps.a
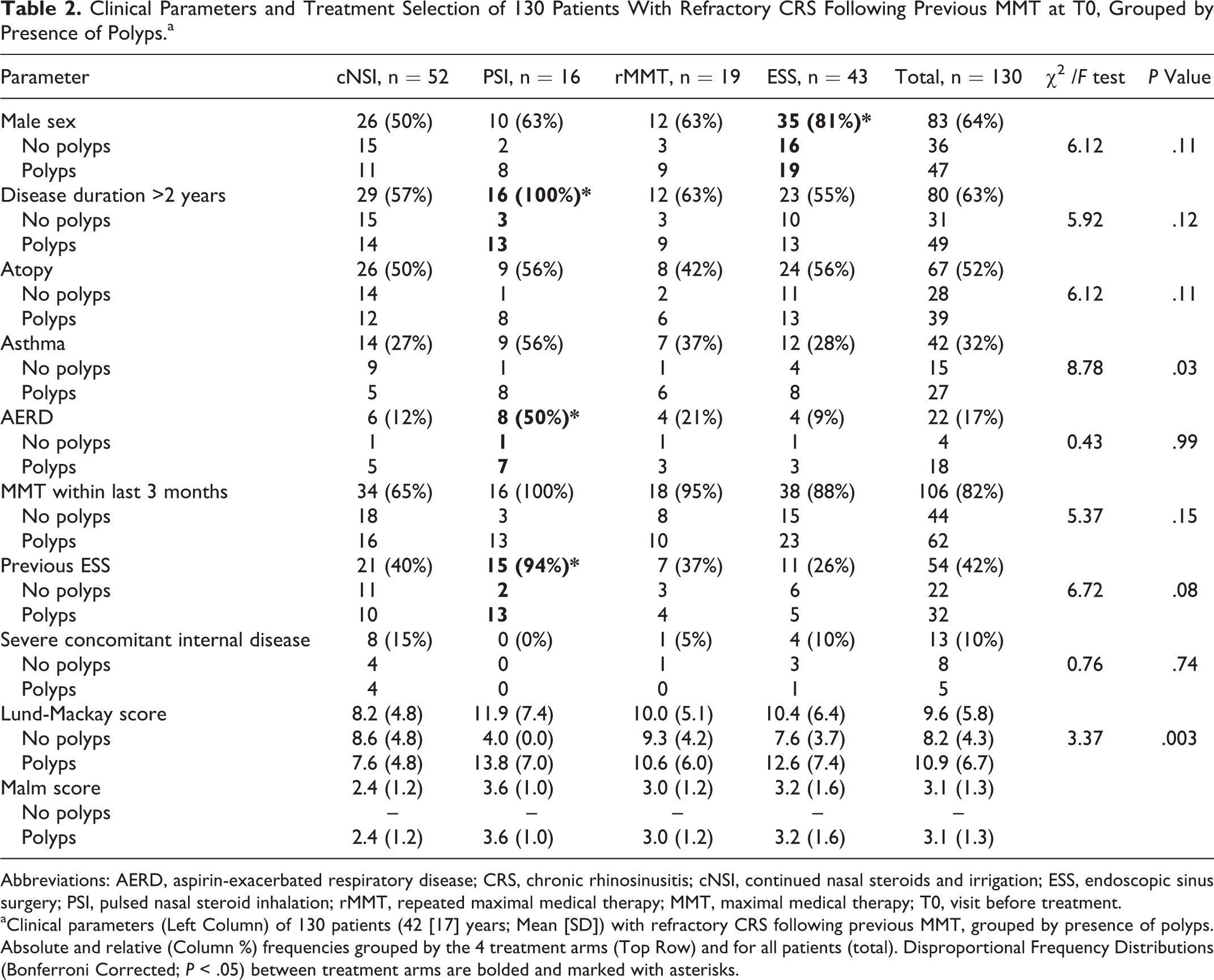
Abbreviations: AERD, aspirin-exacerbated respiratory disease; CRS, chronic rhinosinusitis; cNSI, continued nasal steroids and irrigation; ESS, endoscopic sinus surgery; PSI, pulsed nasal steroid inhalation; rMMT, repeated maximal medical therapy; MMT, maximal medical therapy; T0, visit before treatment.
aClinical parameters (Left Column) of 130 patients (42 [17] years; Mean [SD]) with refractory CRS following previous MMT, grouped by presence of polyps. Absolute and relative (Column %) frequencies grouped by the 4 treatment arms (Top Row) and for all patients (total). Disproportional Frequency Distributions (Bonferroni Corrected; P < .05) between treatment arms are bolded and marked with asterisks.
Patients and Methods
Study Overview
This prospective observational study 24,25 on treatment outcomes of 4 standardized treatments in patients with CRS refractory to MMT was conducted at the Department of Otorhinolaryngology—Head and Neck Surgery, Medical University of Innsbruck, Austria. Adult patients with symptomatic CRS with nasal polyps (CRSwNP) and without nasal polyps (CRSsNP) following at least 1 course of MMT during the year prior to inclusion date were recruited between November 2015 and March 2016 (Figure 1). The preceding MMT followed a standardized protocol requiring a tapering course of 40 mg methylprednisone once daily with a 50% dose reduction every 7 days in combination with 250 mg clarithromycin twice daily for 4 weeks in addition to nasal steroids and irrigation. CRS was diagnosed according to established criteria. 6 Patients with fungal sinusitis, dental sinusitis, ciliary dysfunction, immunodeficiency, sinus malignancy, history of substance abuse, or psychiatric illness were excluded as were pregnant or lactating women. The SNOT-22 scores and DCS 6 served as outcome parameters (Table 1). The institutional review board approved the study (AN2015-0301).
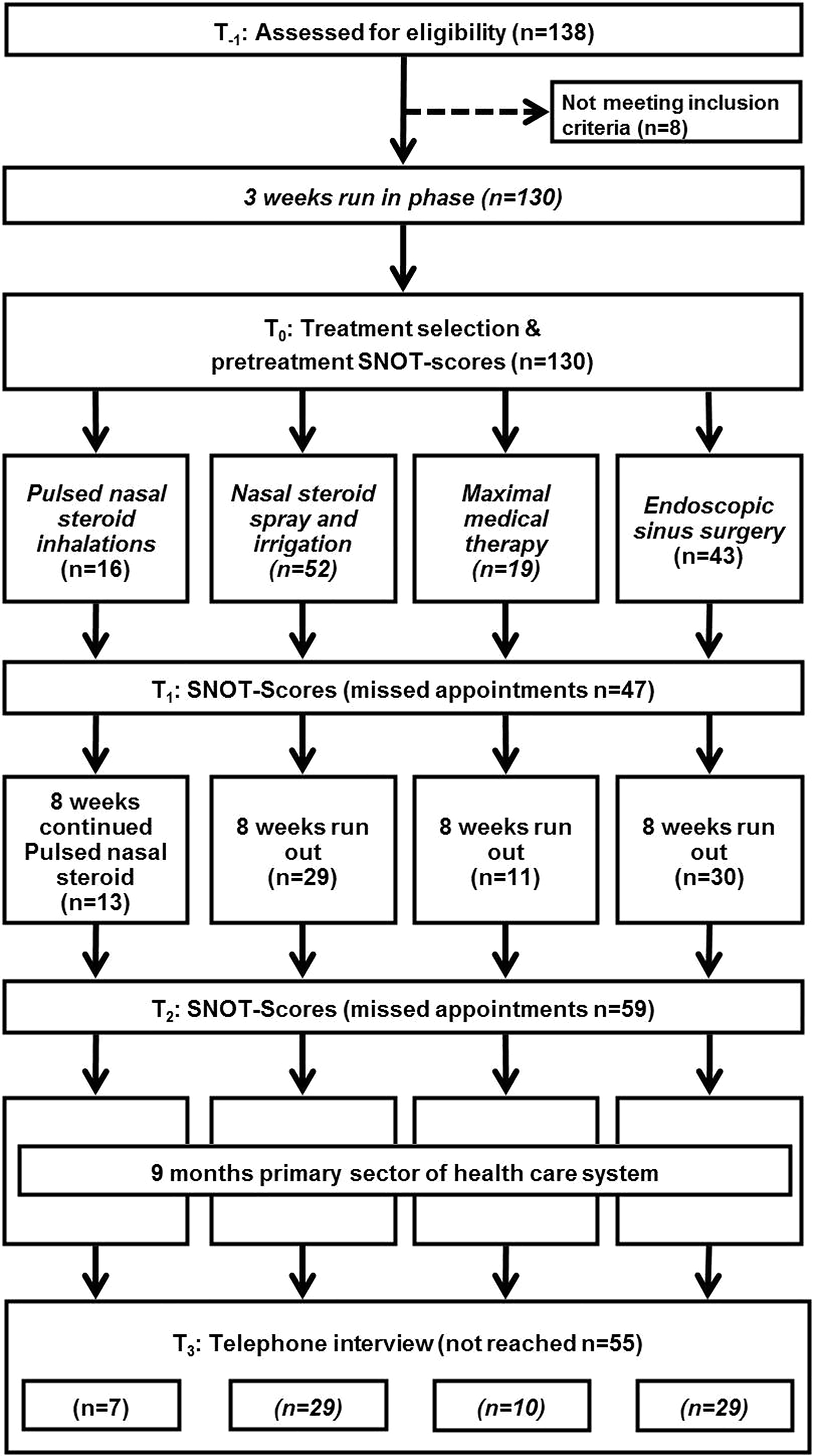
Study flow. Following inclusion, all patients underwent a 3-week run-in phase to synchronize the different treatments and account for various previous treatments before inclusion. Patients then selected 1 of the 4 treatment arms (see Table 3). The run-out phase aimed at keeping the follow-up synchronized, for example, to allow for adequate postoperative care. Following the run-out phase, treatment was continued by general practitioners and practicing otorhinolaryngologists for 9 months.
All patients received treatment for a total of 15 weeks. Week 1 to 3 was referred to as “run-in phase” established to compensate for treatment differences since the last course of MMT (Figure 1). Treatment during this phase was identical to the continued nasal steroids and irrigation (cNSI) arm (Table 3). Week 4 to 8 was referred to as “treatment phase.” In this phase, patients received 1 of 4 standardized treatments (Table 3) following physician counseling: cNSI, pulsed nasal steroid inhalation (PSI), repeated MMT (rMMT), or ESS. Patients were informed of the advantages and disadvantages of each treatment method. The main advantage communicated to patients in regard to cNSI, PSI, and rMMT was that these permitted treatment on an outpatient basis. As to disadvantages, patients were told about the low-efficacy of cNSI, the higher costs of PSI, and the frequent side effects of rMMT. The higher costs for the PSI resulted from the necessary inhalation device, which cost approximately 200€ and had to be paid by the patients themselves. The main advantage communicated to patients in regard to ESS was its high efficacy, with disadvantages being the required inpatient treatment setting for at least 1 week as well as potential surgical complications. Week 9 to 15 was referred to as “run-out phase,” established to keep the follow-up synchronized. Again, treatment during the run-out phase was identical to cNSI for all patients. Exceptions were patients with PSI; since the inhalation device had to be paid by the patient, the study concept allowed patients to continue PSI for the complete duration of the study. Consequently, all patients received cNSI at some time point during their participation with application durations ranging from 3 weeks (pNSI) to 11 weeks (rMMT, ESS) and 15 weeks (cNSI).
Four Standardized Treatment Protocols.a
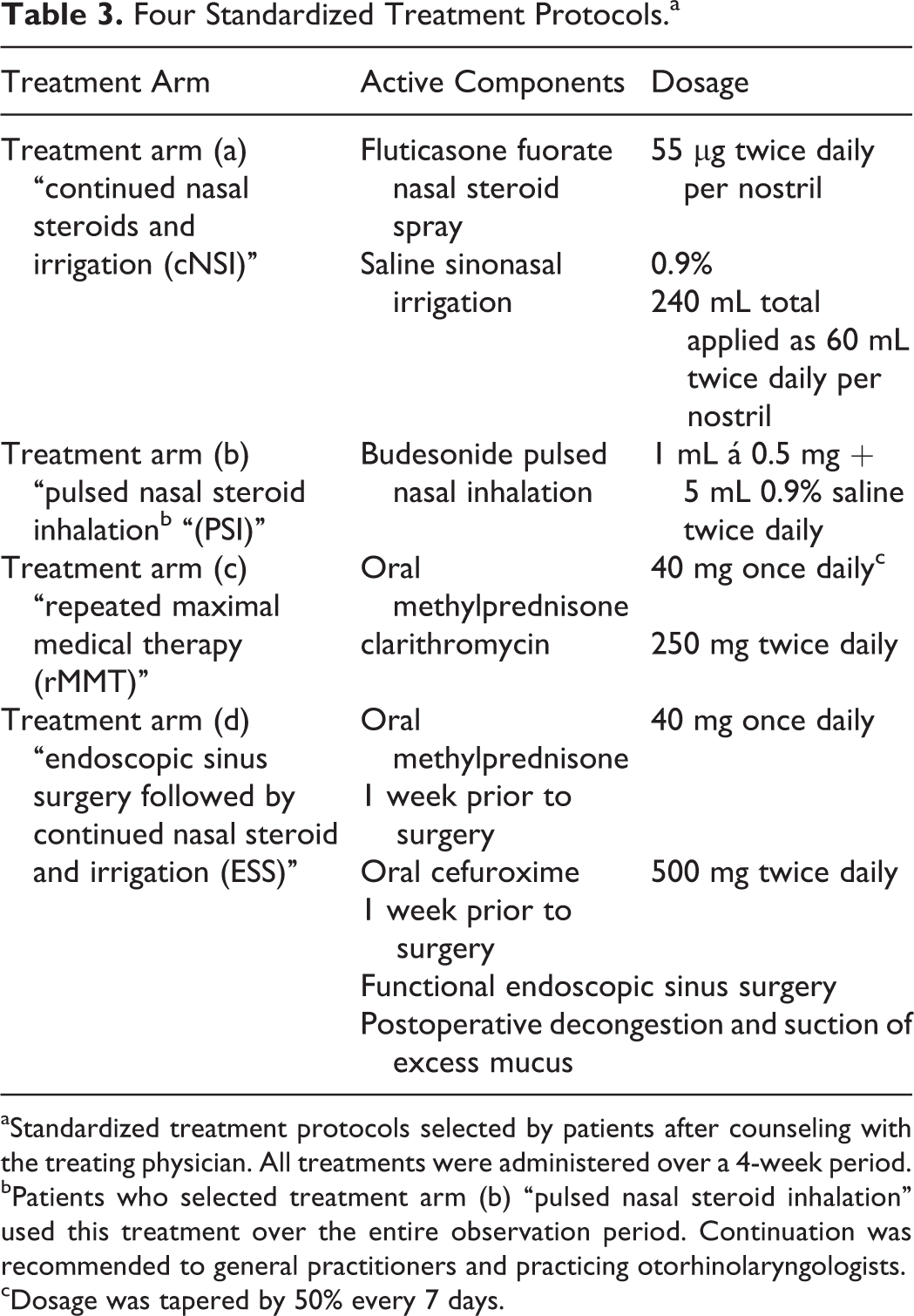
aStandardized treatment protocols selected by patients after counseling with the treating physician. All treatments were administered over a 4-week period.
bPatients who selected treatment arm (b) “pulsed nasal steroid inhalation” used this treatment over the entire observation period. Continuation was recommended to general practitioners and practicing otorhinolaryngologists.
cDosage was tapered by 50% every 7 days.
At the inclusion visit (T-1), patients’ clinical data were recorded, including possible confounders, age, sex, nasal polyps, disease duration, atopy, 26 asthma 27 or aspirin-exacerbated respiratory disease (AERD), 28 concomitant severe internal diseases, CRS treatment at inclusion, previous ESS, time since last MMT, and whether MMT had been given within the preceding 3 months (Table 2). Then, all patients entered the standardized 3-week run-in phase described above (Figure 1). Following the run-in phase at the visit before treatment (T0), baseline SNOT-22 scores 23 and DCS 6 were recorded. Thereafter, all patients began the 4-week treatment phase of their chosen treatment protocol following the physician counseling described earlier. At the visit following the treatment phase (T1), SNOT-22 scores were taken for the second time. Patients then entered the standardized 8-week run-out phase described above. Following the run-out phase (T2), SNOT-22 scores were again recorded 2 months post-treatment. After the run-out phase, patients continued to receive treatment from their primary health-care providers; recommendations for this treatment were identical to cNSI. At 1 year following inclusion (T3), SNOT-22 scores and DCS were reassessed by telephone interviews. 29
Treatment
Treatment protocols for CRS at our institution were consistent during the study period and based on standard operating procedures (Table 3). In the cNSI protocol, patients received 55 µg fluticasone fuorate spray twice daily per nostril (220 µg) in combination with a total of 240 mL high-flow 0.9% saline sinonasal irrigation per day, applied as 60-mL irrigations twice daily per nostril (Nasaline; ENTpro, Stockholm, Sweden). In the PSI protocol, patients received 1 mL á 0.5 mg budesonide in 5 mL 0.9% saline solution twice daily, applied via a pulsed nasal nebulizer (PARI Sinus; Pari GmbH, Starnberg, Germany). In the rMMT protocol, patients received a tapering course of 40 mg methylprednisone once daily, with a 50% dose reduction every 7 days in combination with 250 mg clarithromycin twice daily in addition to nasal steroids and irrigation. For ESS, we followed the principles of functional surgery. The extent of surgery was tailored to the extent of disease 20 and performed under general anesthesia. To reduce intraoperative blood loss, a 7-day oral course of 40 mg methylprednisolone once daily and 500 mg cefuroxime twice daily was administered immediately prior to surgery. 30 Postoperative care included decongestion and suction of excess mucus for 5 days postoperatively during inpatient treatment and, when requested by the patient, weekly decongestion and suction of excess mucus in an outpatient setting for another 3 weeks.
Outcome Assessment
Sino-Nasal Outcome Test-22
The SNOT-22 is a self-administered questionnaire to assess symptom severity and QoL in patients with CRS. 23 It consists of 22 items; each item is rated from 0 (“no problem at all”) to 5 (“worst possible symptom”). Total scores may range from 0 to 110, with higher scores indicating worse symptoms. Good reliability, responsiveness, and validity have been reported for the SNOT-22. 23 A minimal clinically important difference (MCID) of 8.9 score points has been reported. 23 For healthy individuals, a median score of 7 (mean 10.5; 95% CI 9-12) has been reported. 31 In the present study, the SNOT-22 was completed by the patient in the clinic at T0, T1 and T2; at T3, the SNOT-22 scores were obtained by telephone interview. 29
Assessment of disease control status
DCS 6 was assessed pretreatment (T0) and at 1 year following inclusion (T3). Patients were asked whether they had needed any systemic medication during the preceding month or during the preceding 3 months to control CRS. DCS was further assessed according to the corresponding SNOT-22 items for nasal blockage, rhinorrhea/postnasal drip, facial pain/headache, loss of sense of smell, and sleep disturbance/fatigue within the preceding month. To obtain a single value expressing DCS, redundant SNOT-22 items on nasal symptoms and sleep disturbance were aggregated, and the maximum score was used. As proposed by the EPOS, 6 disease was categorized as “controlled” when all criteria were either negative or not bothersome, indicated by a SNOT-22 score of 0 to 2, “partly controlled” when 1 or 2 of these criteria were positive, and “uncontrolled” when 3 or more criteria were positive (Table 1). The validity of the DCS concept has been explored by Snidvongs and coworkers previously. 32
Data analysis
Descriptive data are presented below for CRS and CRSsNP and CRSwNP separately. Group differences were tested using the χ2 test and analysis of variance. A linear mixed random intercept model was used to analyze the SNOT-22 time course for the 3 study visits (T0, T1, and T2) and the telephone interviews (T3). Patients served as subject variables and time as fixed effects. For the evaluation of the effect of the 4 treatments, a linear mixed random intercept model was chosen, using the post-treatment SNOT-22 scores (T1, T2, and T3) as dependent variables. To account for the correlation of post-treatment scores with pretreatment scores, pretreatment SNOT-22 scores (T0) served as covariates. Patients served as subject variables and examination times (T1, T2, and T3) as repeated variables, with a diagonal covariance structure. Time of examination (T1, T2, and T3), SNOT-22 score, and treatment were modeled as fixed effects. The intercept was modeled as a random factor, with identity as the covariance structure. The 11 possible confounders were tested as independent fixed effects. Moderator effects on the outcome of the 4 treatments were tested with the confounder × treatment interaction term. The SNOT-22 total score trajectories are presented separately for CRSsNP and CRSwNP. Effect size was evaluated using the proposed MCID for the SNOT-22 total score. 23
For calculation of DCS 6 1 year following inclusion, the 3 medical treatments were combined as a single medical treatment and compared to ESS to avoid low cell frequencies. DCS was classified as “improved” when it improved by 1 or 2 steps or as “not improved” when no change occurred or DCS deteriorated. Two-sided Fisher exact test was applied to test for independence of treatment and DCS. Equal distribution of the 11 confounders investigated among the 4 treatments was tested with the χ2 test. Column proportions were adjusted for multiple testing using Bonferroni correction. The only possible confounders with adjusted P values <.05 are reported in detail. Missing values were analyzed using the SPSS MVA procedure. All variables included in the outcome analysis were included in the missing value analysis, and Little Missing Completely at Random (MCAR) test was carried out. All statistical analyses were performed using SPSS 24 (IBM, Armonk, New York).
Results
Clinical Data
A total of 138 patients with CRS refractory to at least 1 course of MMT during the year prior to symptom reoccurrence were screened for study inclusion (T-1). Of these, 130 patients were included. Exclusion reasons were incorrect diagnosis (antral cyst; 1 patient), incorrect classification as refractory (2 patients), and nonstandardized treatment selection (5 patients). The mean age was 42 ± 17 years (range: 24-79). Mean (± standard deviation [SD]) duration since last MMT was 144 days (±36; range 132-227). Clinical parameters at inclusion (T0) of all included patients are shown in Table 2. At least 1 follow-up visit was available for 117 patients, corresponding to a dropout rate of approximately 10% (Figure 1). Missing value analysis, including SNOT-22 scores, follow-up visits, treatments, and 11 confounders revealed that missing data were random and did not follow a specific pattern (Little’s MCAR test P = .32).
Treatment Selection
Of the 130 included patients, 52 selected cNSI, 16 PSI, 19 rMMT, and 43 ESS. Patients’ treatment selection (Table 2) was significantly influenced by sex (P = .018), nasal polyps (P = .054), disease duration >2 years (P = .010), AERD (P = .001), previous ESS (P < .001), current treatment at inclusion (P < .001), and MMT within 3 months prior to inclusion (P = 0.001). Treatment selection was not influenced by age (P = .76), atopy (P = .58), asthma (P = .14), or concomitant severe internal disease (P = .28).
The SNOT-22 Scores
Time course of SNOT-22 scores
For all patients with CRS, mean SNOT-22 scores before treatment (T0) did not significantly differ between treatments (cNSI = 38 ± 2, PSI = 39 ± 1, rMMT = 37 ± 3, FESS = 38 ± 2; P = .99). For all treatments together, SNOT-22 scores significantly decreased from 38 ± 2 (estimated marginal mean [EMM] ± standard error of mean [SEM]) score points before treatment (T0) to 20 ± 2 score points 1 year following inclusion (T3) (P < .001; Figure 2). The most pronounced difference was found between before treatment (T0) and directly after the treatment phase (T1), with an improvement of 12 ± 2 score points (P < .001). No significant difference between end of treatment (T1) and 2 months post-treatment (T2) was observed (26 ± 2 vs 26 ± 3; P > .2). Between T2 and T3, SNOT-22 scores further improved by 6 ± 2 score points (P < .05). Pretreatment (T0) SNOT-22 scores were significantly correlated with post-treatment (T1, T2, T3) SNOT-22 scores (Spearman’s ρ = .65; P < .001). This correlation was observed for all treatments. For cNSI, PSI, rMMT, and ESS individually, SNOT-22 scores also significantly decreased from 38 ± 3, 38 ± 2, 37 ± 3, and 38 ± 1 score points before treatment (T0) to 23 ± 1, 22 ± 3, 24 ± 1, and 16 ± 1 score points 1 year following inclusion (T3; all P < .001; Figure 3A). Again, the most pronounced difference was found between before treatment (T0) and directly after treatment phase (T1), with an improvement of 8 ± 3, 7 ± 1, 8 ± 3, and 18 ± 2 score points, respectively (all P < .005). Additional details about the time course of SNOT-22 scores for the 4 standardized treatment protocols are provided in Table 4 and Figure 3A.
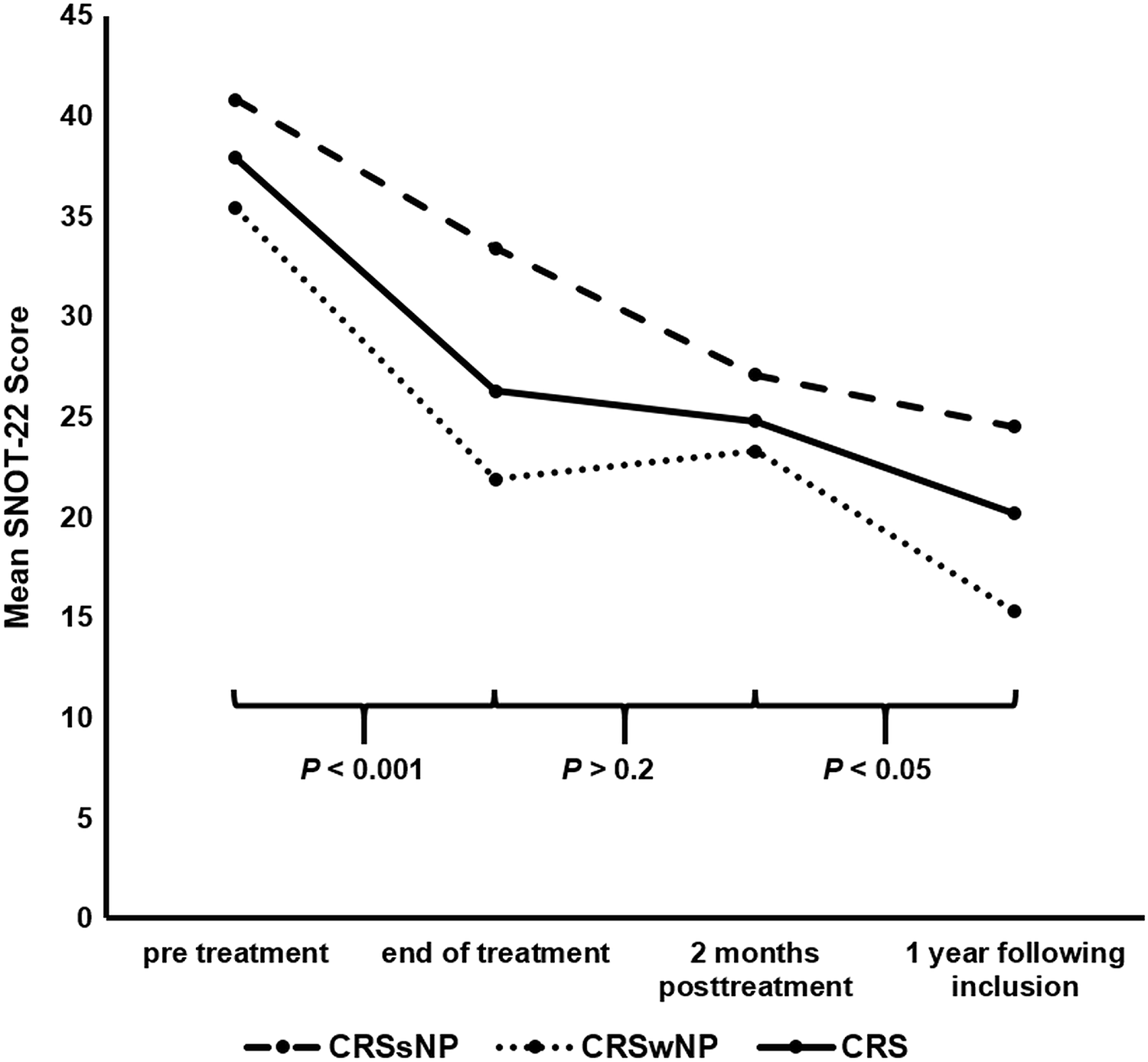
Time course of Sino-Nasal Outcome Test-22 (SNOT-22) scores (all treatment arms) in patients with refractory chronic rhinosinusitis (CRS) following previous maximal medical therapy (MMT). Line plot providing SNOT-22 scores for all treatment arms at pretreatment (T0), end of treatment (T1), 2 months post-treatment (T2), and 1 year following inclusion (T3). The continuous black line represents all patients with CRS (CRS; 37.9, 26.3, 24.8 and 20.2 mean SNOT-22 score points), the upper dashed black line represents patients without nasal polyps (CRSsNP; 40.8, 33.4, 27.1 and 24.5 mean SNOT-22 score points); the lower dotted black line represents patients with nasal polyps (CRSwNP; 35.4, 21.9, 23.3 and 15.3 mean SNOT-22 score points). The horizontal round brackets indicate the P values for patients with CRS only. All mean SNOT-22 scores for patients with CRSsNP were significantly higher than for patients with CRSwNP (all P < .05), except for 2 months post-treatment (P > .5). However, as scores for both groups of patients significantly decreased over time, mostly running in parallel, the results suggest that both phenotypes benefit from all treatment arms.
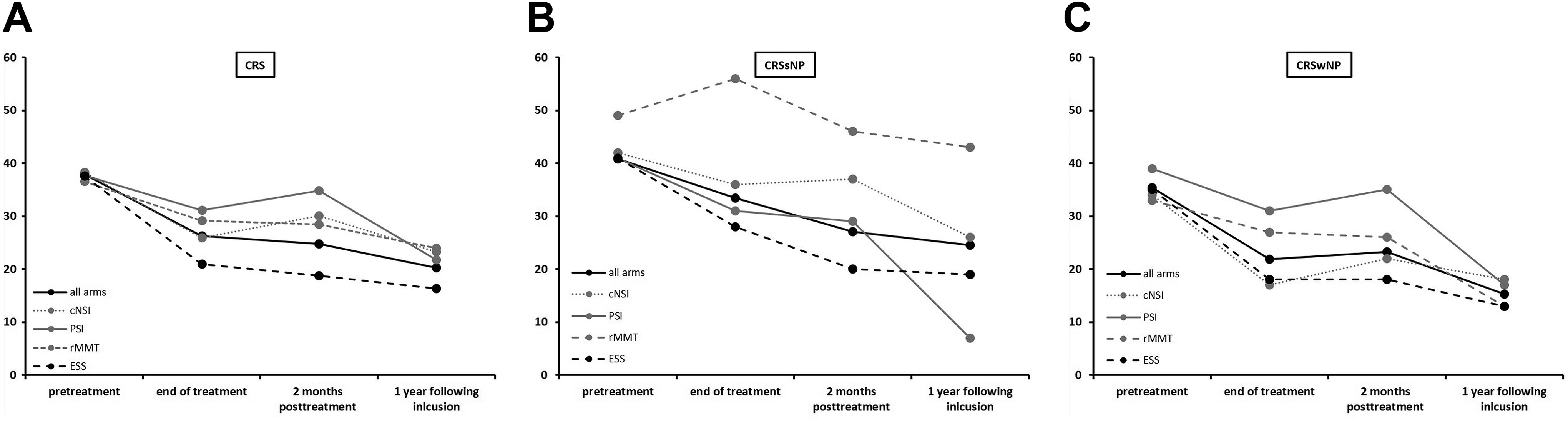
Time course of Sino-Nasal Outcome Test-22 (SNOT-22) scores (individual treatment arms) in patients with refractory chronic rhinosinusitis (CRS) following previous maximal medical therapy (MMT). Line plot providing SNOT-22 scores for all 4 treatment arms individually at pretreatment (T0), end of treatment (T1), 2 months posttreatment (T2 and 1 year following inclusion (T3) for all patients with CRS (A) and for patients with chronic rhinosinusitis without nasal polyps (CRSsNP; B) and chronic rhinosinusitis with nasal polyps (CRSwNP; c), separately. In all 3 figures, the continues black line represents all treatments together, and the gray lines represent the individual medical treatment arms (continues line: PSI, dotted line: cNSI, dashed line: rMMT). The dashed black line represents the endoscopic sinus surgery (ESS) treatment arm. Detailed mean SNOT-22 scores for each visits and treatment arm are provided in Table 4.
Mean SNOT-22 Scores for All Time Points and All Individual Treatments for Patiens With CRS, CRSsNP, and CRSwNP.a
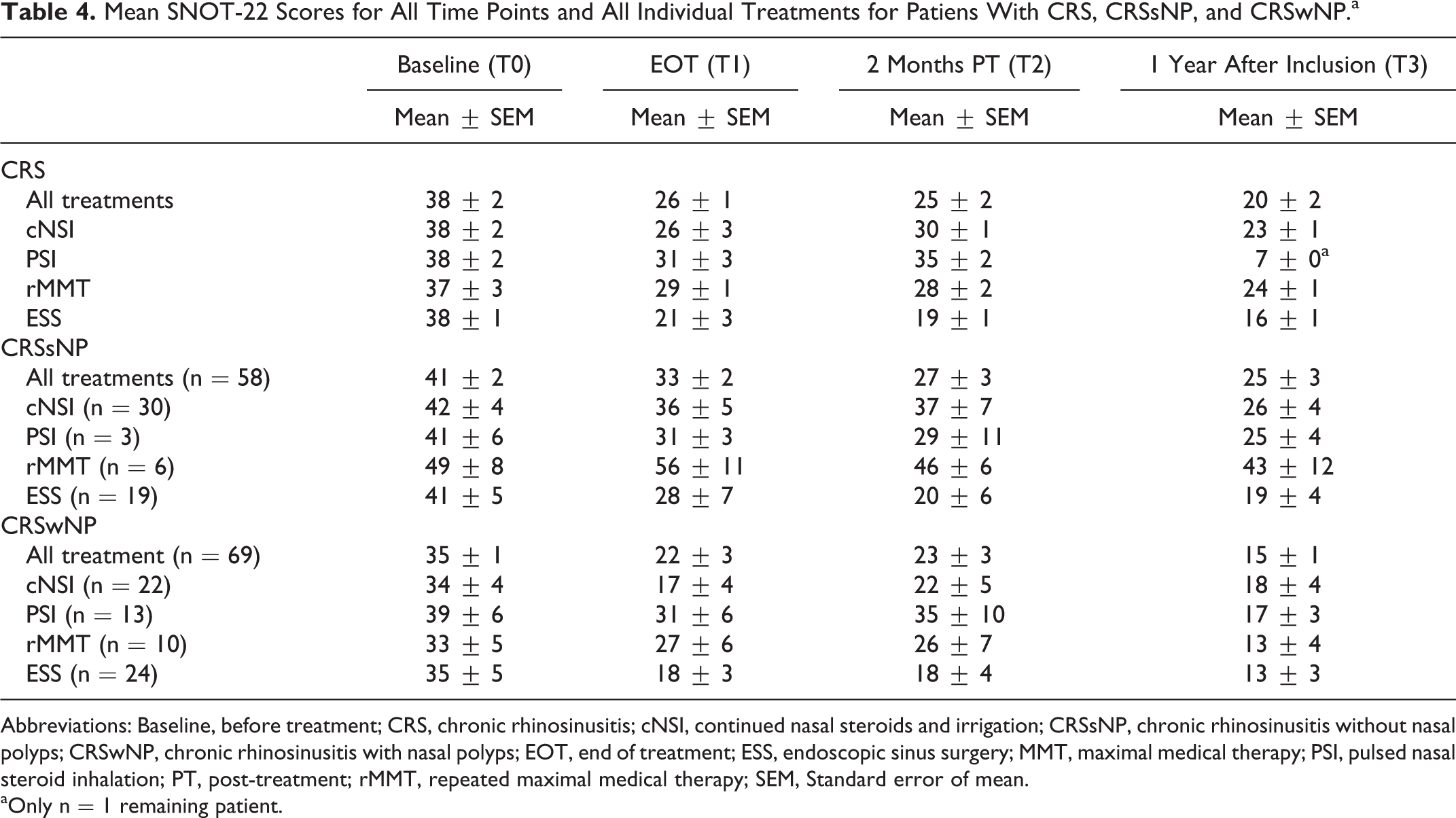
Abbreviations: Baseline, before treatment; CRS, chronic rhinosinusitis; cNSI, continued nasal steroids and irrigation; CRSsNP, chronic rhinosinusitis without nasal polyps; CRSwNP, chronic rhinosinusitis with nasal polyps; EOT, end of treatment; ESS, endoscopic sinus surgery; MMT, maximal medical therapy; PSI, pulsed nasal steroid inhalation; PT, post-treatment; rMMT, repeated maximal medical therapy; SEM, Standard error of mean.
aOnly n = 1 remaining patient.
For patients with CRSsNP, SNOT-22 scores significantly decreased from 41 ± 3 score points before treatment (T0) to 25 ± 4 score points 1 year following inclusion (T3), which is almost twice the MCID 23 (P < .001; Figure 2). The most pronounced difference was found between before treatment (T0) and directly after the treatment phase (T1), with an improvement of 8 ± 1 score points (P = .12)—although this difference was smaller than for all patients with CRS together. No significant difference between end of treatment (T1) and 2 months post-treatment (T2) or between 2 months post-treatment (T2) and 1 year following inclusion (T3) was observed (T1 vs T2: 33 ± 2 vs 27 ± 2; P = .39; T2 vs T3: 27 ± 2 vs 25 ± 4; P = .95; Figure 2). For cNSI, PSI, rMMT, and ESS individually, SNOT-22 scores also significantly decreased from 42 ± 4, 41 ± 6, 49 ± 8, and 41 ± 5 score points before treatment (T0) to 26 ± 4, 7 ± 0, 43 ± 12, and 19 ± 4 score points 1 year following inclusion (T3). The decrease for PSI has to be interpreted with caution, since only 1 participant remained at T3 (Figure 3B). Additional details about the time course of SNOT-22 scores for the 4 standardized treatment protocols are provided in Table 4 and Figure 3B.
For CRSwNP, SNOT-22 scores significantly decreased from 35 ± 2 score points before treatment (T0) to 15 ± 2 score points at 1 year following inclusion (T3), which is more than twice the MCID 23 (P < .001; Figure 2). The most pronounced difference was found between before (T0) and directly after the treatment phase (T1), with an improvement of 13 ± 3 score points (P < .001). A nonsignificant increase in mean SNOT-22 scores between end of treatment (T1) and 2 months post-treatment (T2) was observed (22 ± 4 vs 23 ± 2; P = .71). This was followed by an improvement of 8 ± 2 score points between 2 months post-treatment (T2) and end of treatment (T3), which was significant (23 ± 2 vs 15 ± 2; P = .03; Figure 2). For cNSI, PSI, rMMT, and ESS individually, SNOT-22 scores also significantly decreased from 34 ± 4, 39 ± 6, 33 ± 5, and 35 ± 5 score points before treatment (T0) to 18 ± 4, 17 ± 3, 13 ± 4, and 13 ± 3 score points 1 year following inclusion (T3). Additional details about the time course of SNOT-22 scores for the 4 standardized treatment protocols are provided in Table 4 and Figure 3C.
Post-treatment SNOT-22 scores by treatment arm
Post-treatment SNOT-22 scores at the 3 follow-up visits differed significantly between the 4 treatment methods (P = .006). The SNOT-22 scores (EMM ± SEM) were 26 ± 2 following cNSI, 27 ± 3 following PSI, 28 ± 3 following rMMT, and 19 ± 2 following ESS (P < .001; Figure 4). Post-treatment SNOT-22 scores following ESS were 8 ± 3 score points better than following cNSI (P = .004), 9 ± 4 score points better than following PSI (P = .026), and 10 ± 4 score points better than following rMMT (P = .008; Figure 4). No significant differences between post-treatment SNOT-22 scores for the 3 medical treatments were observed (all P > .2).
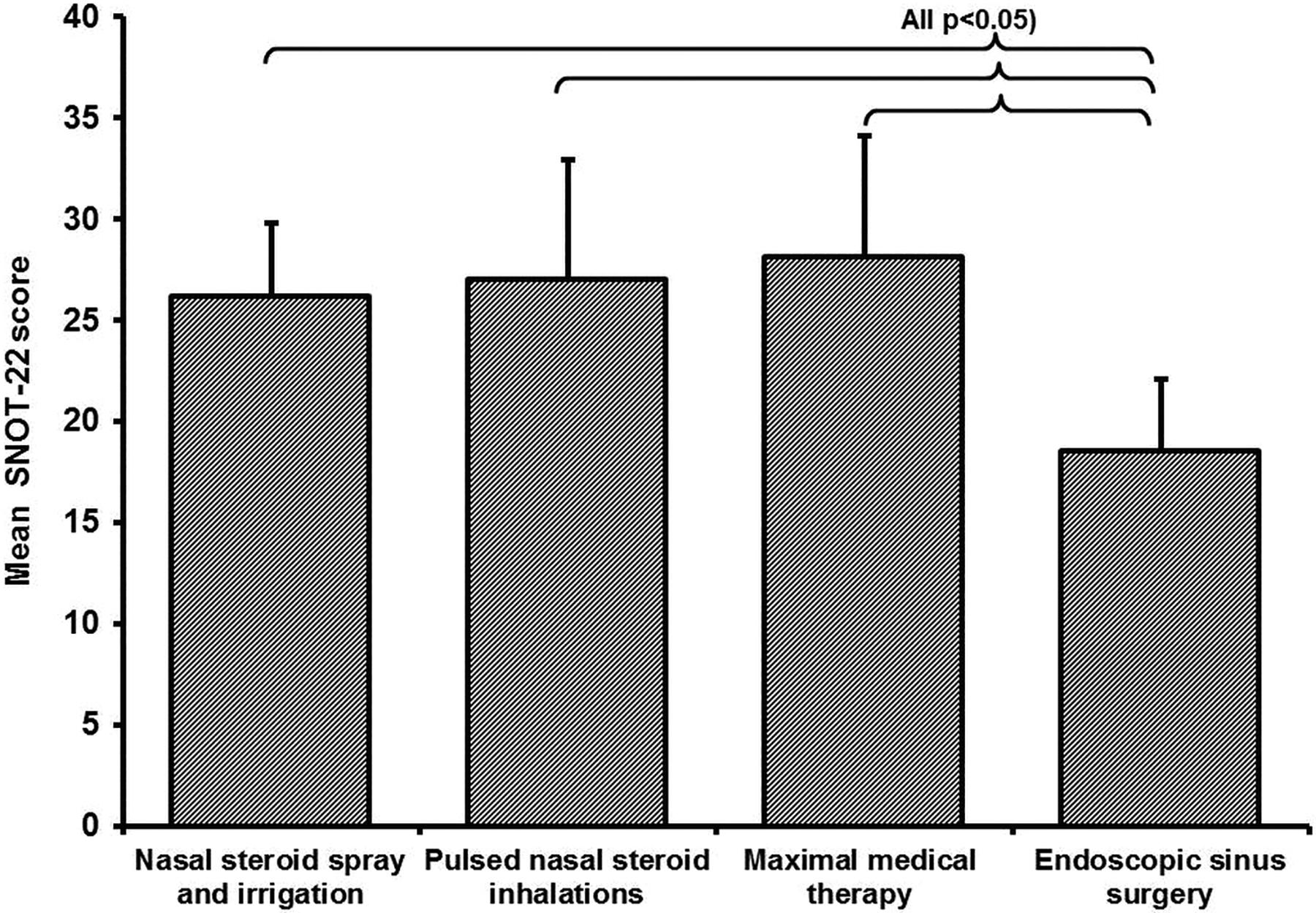
Post-treatment Sino-Nasal Outcome Test-22 (SNOT-22) scores by treatment arms in patients with refractory chronic rhinosinusitis following previous maximal medical therapy. Bar chart providing post-treatment SNOT-22 scores of the 4 treatment arms (also see Table 2). Bars represent estimated marginal means of the mixed regression model, 95% confidence intervals as whiskers. Mean post-treatment scores following endoscopic sinus surgery were lowest (best) and differed significantly from all 3 continued medical treatment arms. Mean post-treatment SNOT-22 scores did not differ significantly between the 3 medical treatment arms (all P > .2).
Effect moderation by confounders
Of the 11 investigated confounders, moderation of the observed treatment effect was found for sex (P = .07) and atopy (P = .07). Moreover, atopy (P = .04) and severe concomitant internal disease (P = .02) proved to be treatment-independent predictors of post-treatment SNOT-22 scores. All other preplanned confounders—age, nasal polyps, disease duration >2 years, asthma, AERD, previous ESS, current treatment immediately at inclusion and MMT within the 3 months prior to inclusion—were neither an independent outcome predictor nor a moderator of the treatment effect on post-treatment SNOT-22 scores (all P > .2).
Disease control status according to EPOS criteria
Data were available for 81 patients at both the pretreatment visit (T0) and at 1 year following inclusion (T3). 6 Before treatment (T0), 51 patients were uncontrolled and 30 patients partly controlled. At 1 year following inclusion (T3), 21 patients were uncontrolled, 29 patients partly controlled, and 31 patients controlled. The 3 medical treatments were combined as a single medical treatment to avoid low cell frequencies. Improvement in DCS following medical treatments was observed in 25 of 50 patients, compared to 26 of 31 patients after ESS treatment (P = .002). At T3, 3 of 31 patients were uncontrolled after ESS compared to 18 of 50 after medical treatments (P < .01).
Discussion
Refractory CRS following recent MMT is a common problem. 10,13,15 In this prospective observational study, 130 patients with CRS refractory to at least 1 course of MMT during the year preceding symptom reoccurrence were enrolled. Previous MMT had included a 4-week course of oral steroids and systemic antibiotics. The mean time since the last course of MMT was approximately 4.5 months. In a manner similar to real-world conditions, patients themselves selected 1 of 4 standardized treatments (Table 3) after physician counseling. Outcome was assessed with SNOT-22 scores 23 at regular intervals. Moreover, DCS was evaluated according to EPOS criteria. 6 The last follow-up was at 1 year following inclusion.
Patients’ selection of 1 of the 4 treatments significantly depended on sex, nasal polyps, AERD, disease duration > 2 years, previous ESS, and whether MMT had been administered within the 3 months preceding inclusion (Table 2). In the ESS treatment arm, male patients were overrepresented (35 of 43). Patients with unfavorable prognosticators frequently selected PSI: 13 of 16 patients had nasal polyps, 8 of 16 AERD, 16 of 16 disease duration >2 years, and 15 of 16 previous ESS. With the exception of sex, none of these characteristics interfered with multifactorial analysis outcomes.
Before treatment (T0), no significant difference in SNOT-22 scores between treatments was observed (P = .99), which suggests that the 3-week run-in phase successfully compensated for treatment differences since the last MMT. Overall, SNOT-22 scores for all treatments decreased from 38 score points before treatment (T0) to 20 score points at 1 year following inclusion (T3; P < .001; Figure 2). A higher reduction in SNOT-22 scores for patients with CRSwNP than for patients with CRSsNP was observed between the pretreatment visit (T0) and the visit at 1 year following inclusion (T3; 20 vs 16 score points, respectively). However, reduction in SNOT-22 scores was significant (P < .001) and almost twice the MCID of 8.9 score points, 23 suggesting that the investigated treatments are effective for both phenotypes (Figure 2). However, for all patients, mean post-treatment SNOT-22 scores remained substantially higher than in the healthy population, with an average score of 10.5. 31 Atopy (P = .043) and severe concomitant internal disease (P = .016) were treatment-independent predictors for worse post-treatment SNOT-22 scores.
Post-treatment scores for ESS were significantly lower than for each of the 3 medical treatments (Figure 4, all P < .03). The mean difference in post-treatment SNOT-22 scores between ESS and the 3 medical treatments was 8.7 score points (95% confidence interval [CI] 4.2-13.3), which is close to the MCID. 23 Posttreatment SNOT-22 scores for the 3 medical treatments did not differ significantly (all P > .2). Notably, rMMT was not superior to cNSI. This observation is especially important considering the non-negligible morbidity of MMT. 33 Sex and atopy had weak moderating effects on the effectiveness of the 4 treatments. Female patients responded particularly poorly to cNSI (P = .067). Patients with atopy performed better with cNSI and worse with the other 3 treatments in comparison to non-atopic patients (P = .065). When sex and atopy as well as their interaction term (with treatment) were included in the mixed regression model, the differential effect of the 4 treatments on post-treatment SNOT-22 scores remained unchanged (P = .008). Overall, improved DCS 6 was observed in 51 of 81 patients at 1-year follow-up. The DCS at 1 year following inclusion was observed in 26 of 31 patients after ESS, compared to 25 of 50 after medical treatments only (P = .002).
The results of this study are in line with several recent publications. 14,16 DeConde and colleagues evaluated the change of SNOT-22-derived cardinal symptoms in 342 patients with refractory CRS who selected ESS or continued medical therapy as treatment. The authors defined CRS as refractory when symptoms persisted after a 5-day trial of systemic corticosteroids. After a minimum follow-up of six months, all patients improved across all cardinal symptoms. Patients who selected ESS had significantly better outcomes than patients who selected medical treatment. 14 In a similarly designed study, Smith explored disease-specific QoL in 98 patients with refractory CRS. The authors defined CRS as refractory when symptoms persisted after 3 weeks of systemic antibiotics and nasal steroids. In patients who selected ESS, symptom improvement was significantly better than in patients who continued medical therapy. 16
Furthermore, the observed DCS (as proposed by EPOS 6 ) findings in the present study are in line with a recent publication showing that, for a total of 106 CRS patients, 10% remained uncontrolled 12 months after undergoing ESS. 32 However, reported long-term results following treatment for refractory CRS have not been as promising. Van der Veen observed high failure rates following ESS over a longer term. The CRS was considered refractory when symptoms persisted after irrigation, nasal steroids, and one or more courses of systemic corticosteroids. Of 560 patients with refractory CRS after previous medical treatment, 43.7% were uncontrolled 3 to 5 years following ESS. 17
One of the strengths of this investigation is its realistic study setting. Treatment allocation was not randomized but rather based on patient preference. While RCTs aim at determining the efficacy of a treatment under experimental conditions, observational studies are commonly used to explore treatment outcomes under real-world conditions. Almost all of patients approached for participation in the present study—all of whom were refractory despite a course of standardized MMT during the year prior to their participation—were included. By contrast, a randomized study with a surgical treatment arm can only include patients who are qualified and willing to undergo surgery. 24,25 Moreover, patients who have recently received a course of MMT or ESS are frequently excluded from the clinical trials. This selection bias was avoided by the design used in the present study. 24,25 Nevertheless, in nonrandomized studies, results are frequently biased by uneven distribution of confounders among treatment arms. 34 Indeed, several possible confounders revealed uneven distribution among treatment arms in our study. However, the results of including these factors in the multifactorial model did not suggest a severe bias. Nevertheless, this computational approach is considered much less trustworthy than RCTs. For one, there are concerns about the influence of unobserved variables in observational study settings. 25 Interestingly, a recent Cochrane Review concludes that there is little evidence for significant effect estimate differences between observational studies and RCTs. 24
A limitation of this study is that it was not randomized and therefore did not allow comparison of the effectivity of the different treatment modalities. Instead, it reports on the clinical outcomes of common treatment modalities for CRS. Moreover, patient and disease characteristics at inclusion were not homogenous. Because of the prospective observational study design, aiming at reflecting real-world conditions with patients choosing their own treatment after physician counseling, no traditional a priori sample size calculations were conducted. Nevertheless, post hoc power analysis indicated that our study was sufficiently powered (β = 0.84) to detect small-to medium effect sizes as suggested by the literature 3 -5,8 -11 within the 4 treatment groups across the 4 assessment time points (f = 0.13; α = 0.05; n = 130; r = .5). Power calculation was performed using G*Power 3.1 (Heinrich Heine University, Düsseldorf, Germany).
Furthermore, several patients had undergone MMT as much as 7 months prior to inclusion. The run-in phase might therefore appear too short to compensate for treatment differences since the last MMT. However, mean SNOT-22 scores before treatment (T0) did not significantly differ between treatment arms. This suggests that the duration of the run-in phase may be considered adequate. Additionally, the treatment phase might appear short in comparison to the run-out phase. Nevertheless, short treatment escalations in symptomatic CRS patients together with ongoing baseline treatment reflect common clinical practice. 6,7
For rMMT, oral clarithromycin and oral methylprednisone were administered for their anti-inflammatory and immunomodulatory effects. 35 While the original 2012 EPOS recommendations 6 proposed macrolides only for CRSsNP, Huang and colleagues suggest in a recent meta-analysis that adding oral clarithromycin significantly improves clinical symptoms, endoscopic, and computed tomography scores in both CRSsNP and CRSwNP over the short and medium term. 35 However, most of the studies included in that meta-analysis administered clarithromycin for a minimum of 8 weeks. 35
Taken together, patients with refractory CRS following MMT during the year preceding symptom reoccurrence were shown to benefit from all the explored standardized treatments over a 1-year observation period. For patients having CRS with and without nasal polyps, a significant improvement in SNOT-22 scores was found—less profound, however, in the group without nasal polyps. For all patients with CRS, both medical treatment alone and combined medical and surgical treatment resulted in significantly improved SNOT-22 scores and DCS. The ESS followed by cNSI was the most effective treatment protocol. Outcomes in the three medical treatment only arms did not differ. Notably, patients with refractory CRS following MMT did not benefit from an additional course of MMT in comparison to those who were only treated with cNSI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
