Abstract
The management and prognosis of oral squamous cell carcinoma (OSCC) depend on tumor stage and lymph node (LN) metastasis status. Early-stage (T1/T2 N0M0) OSCC comprises a heterogeneous group. We evaluated the role of histological parameters including worst pattern of invasion (WPOI) and tumor budding to determine the risk of LN metastasis in cases of OSCC and to determine the risk of recurrence and death in early-stage OSCC in north Indian patients. All cases of buccal mucosa and tongue SCC which underwent excision over 4 and half years were reviewed for histological parameters including histologic grade, WPOI, tumor budding, lymphovascular emboli (LVE), perineural invasion (PNI), depth of invasion (DOI), host lymphocyte response, and stromal response and compared to LN metastasis. Clinical follow-up of early-stage tumor was obtained and compared. A total of 126 cases of OSCC were included, of which 48 showed LN metastasis. Histological grade, WPOI, tumor budding (≥3/×40 field), LVE, and PNI were significantly associated with risk of LN metastasis. On multivariate analysis, WPOI and tumor budding were 2 most significant factors. Among the early-stage tumors with available follow up (n = 48), DOI, WPOI, tumor budding, and LVE were associated with a shorter overall survival, although it was not statistically significant. To conclude, WPOI and tumor budding are important risk factors for predicting LN metastasis in all stages of OSCC and associated with a poorer outcome in early-stage tumors. These are easy and reliable prognostic factors and should be included in the histopathological reporting guidelines.
Keywords
Introduction
Oral cavity cancer is one of the most common malignancies worldwide. It causes substantial mortality and morbidity. Oral squamous cell carcinoma (OSCC) is of significant public health problem in India, and it accounts for approximately 30% of all cancers in the country. 1 There is a worrying rise in cancer of upper aerodigestive tract among both sexes in India. According to the projected risk, cumulative oral cavity cancer will become the leading malignancy in the country by 2020. 2
Prognosis of OSCC depends on multiple factors. Tumor stage and lymph node (LN) status are the 2 most important factors that determine the treatment and outcome. For staging purpose, the entire oral cavity cancers including the anterior two-thirds of tongue (oral tongue) are staged together as they have similar biological behavior. 3 Lymph node metastasis is best determined by histological examination of the resected LNs. Clinical examination and radiological investigations show a sensitivity of 60% to 70% to detect nodal metastasis. 4 It is generally agreed upon that T1/2, N0, M0 tumors show a favorable outcome and are called early-stage cancers. 5 However, prognosis of the patients with early-stage OSCC is heterogeneous. While most of the patients have a prolonged disease-free survival, some patients of early cancer show early relapse and death due to disease. 6,7 Researchers have evaluated different clinical and histological parameters for predicting the risk of LN metastasis in oral cavity cancers and risk of recurrence in early-stage tumors. Various histological parameters are associated with a higher incidence of nodal metastasis in oral cavity cancer. Lymphovascular emboli (LVE) and perineural invasion (PNI) are well-known histological risk factors for predicting nodal metastasis. 8 However, LVE and PNI are difficult to evaluate, especially in small biopsies. Other histological parameters that have been evaluated previously include degree of tumor histological differentiation, host lymphocyte response (HLR), stromal response, and invasive tumor front. 9
Invasive tumor front is the interface of the lowest portion of the tumor and stroma. Pattern of tumor infiltration at the invasive front has been evaluated in a few previous studies. 7,9 An invasive pattern of infiltration is associated with higher risk of LN metastasis and poor disease-free survival. 7 Another relatively recently described histological parameter is the presence of tumor budding. Tumor budding has been shown to be associated with high incidence of LN metastasis and relapse in colorectal, esophageal, endometrial, and lung cancers. 10 -13 A few previous studies have evaluated the role of tumor budding in oral cavity cancers and found its association with higher incidence of nodal metastasis, recurrence, and poor overall survival. 14 However, pattern of tumor invasion and budding are not well-established prognostic factors and are not mentioned as required criteria for reporting of oral cavity malignancies according to College of American Pathologist guideline. This is because of scarcity of literature available on invasive tumor front. Histological prognostic factors have been rarely evaluated in Indian patients. 8,15,16 This study was conducted to evaluate the role of histological parameters including pattern of invasion (POI) and tumor budding to determine the risk of LN metastasis in cases of OSCC and to determine the risk of recurrence and death in early-stage (T1/2, N0, M0) OSCC in north Indian patients.
Patients and Methods
This was a retrospective study. All the cases of buccal mucosa and oral tongue SCC who underwent resection with cervical LN dissection in our institute from July 2012 to December 2017 were included. Cases of carcinoma alveolus, lip, gingivobuccal sulcus, and floor of mouth were excluded from the analysis, as they are very infrequently encountered in our database. Cases where patient received preoperative radiotherapy or chemotherapy or where LN dissection was not performed or where slides were not available for review were excluded. The size of the tumor was noted from the surgical pathology report, and the pT stage was determined accordingly. 3 All the slides were retrieved from the archive and reviewed by 3 investigators (D.C., V.M., and V.B.) who were blinded to the clinical data. The following histological parameters were evaluated in each case degree of differentiation, depth of invasion, worst pattern of invasion (WPOI), tumor budding, PNI, LVE, HLR, and nature of tumor stroma.
The tumors were graded histologically as well, moderately and poorly differentiated according to their degree of differentiation. 17 Perineural invasion was determined as a nerve being surrounded or infiltrated by the tumor. Tumor invasion within arterial, venous, or lymphatic channels qualified for LVE. Host lymphocyte response was evaluated at the invasive tumor front. Host lymphocyte response was graded semi-quantitatively as none, mild, moderate, and heavy. A diffuse band-like lymphoid infiltrate with or without lymphoid aggregate qualified for heavy HLR. Only a few lymphocytes at the tumor interface was graded as mild HLR. Lymphoid infiltrate in between the 2 groups was graded as moderate HLR. The stromal response was graded as loose, desmoplastic, and hyalinized. The depth of invasion was measured as the distance between the lowest part of the adjacent normal mucosa and the lowest part of the tumor. The depth was measured in millimeter using slide caliper, and it was graded as D1 (≤5 mm), D2 (>5 mm, ≤10 mm), and D3 (>10 mm).
The invasive tumor front was evaluated for pattern of invasion (POI) and tumor budding. The POI was determined as described in the literature previously and classified as 5 patterns. 18,19 Pattern 1 was defined as broad, pushing margin of tumor with a smooth outline. Pattern 2 was defined as broad, pushing finger-like projection. Pattern 3 represents invasive tumor islands with >15 cells per island. Pattern 4 represents invasive tumor islands with less than 15 cells per island. Pattern 5 was defined by the presence of tumor island outside the main tumor at a distance of >1 mm (Figure 1). In a given case showing multiple patterns of invasion, the score was determined by the highest pattern present, even if present focally (WPOI). Among these 5 patterns, POI 4 and 5 were classified as invasive pattern, whereas POI 1 to 3 as cohesive pattern. 18,19 Tumor budding was defined as single cell or a cluster of <5 tumor cells present in the stroma at the invasive tumor front (Figure 2). 7 Number of tumor buds per high-power field (HPF, ×40) was counted at the area of maximum concentration.
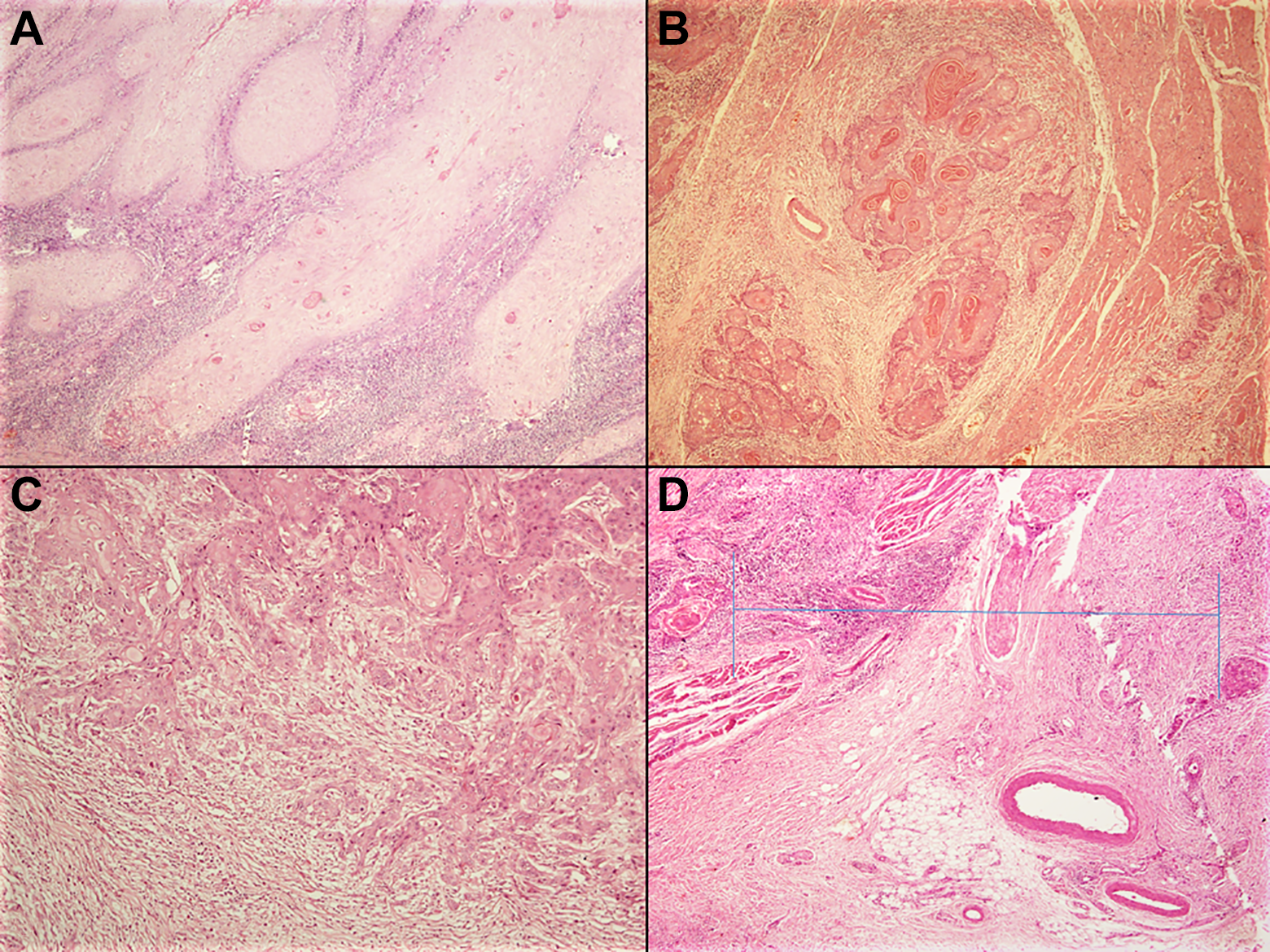
A, Pattern 2 invasion seen as broad, pushing finger-like projection (hematoxylin and eosin, ×40). B, Pattern 3 represents invasive tumor islands with >15 cells per island (hematoxylin and eosin, ×40). C, Pattern 4 represents invasive tumor islands with less than 15 cells per island (hematoxylin and eosin, ×40). D, Pattern 5 was defined by presence of tumor island outside the main tumor at a distance of >1 mm (hematoxylin and eosin, ×40).
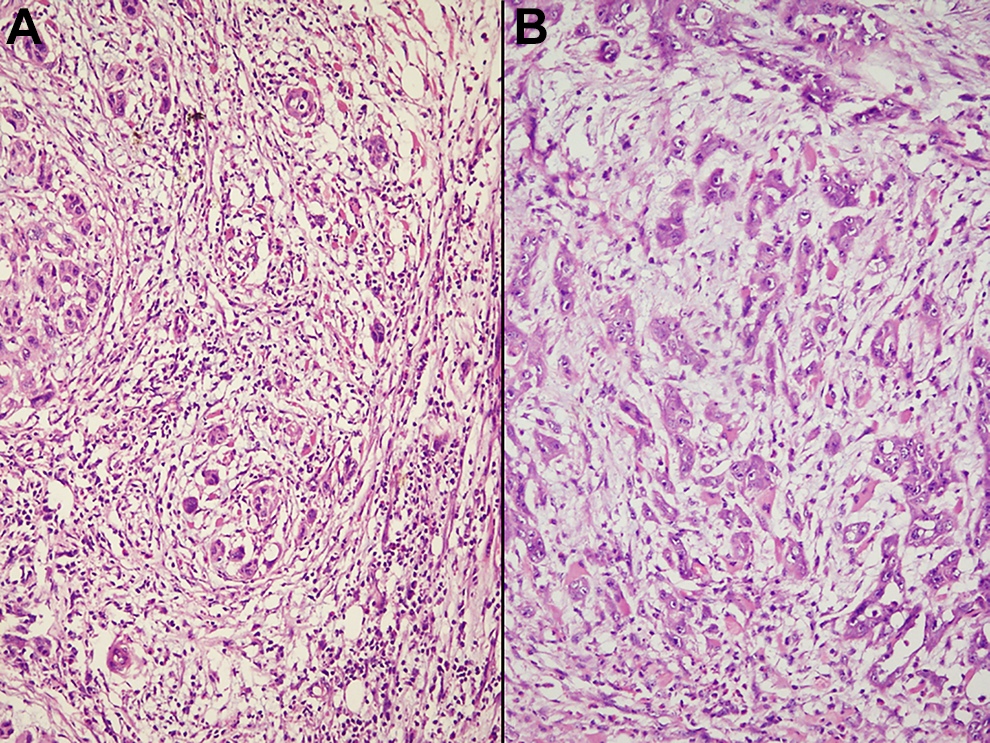
A, Invasive tumor front showing multiple tumor budding (cluster of less than 5 cells) with moderate lymphoid infiltrate (hematoxylin and eosin, ×200). B, Invasive tumor front showing a few tumor buds in a myxoid stroma with minimal lymphoid infiltrate (hematoxylin and eosin, ×200).
Survival Analysis
All cases of early cancer (T1/2, N0, M0) were further analyzed. Patients with available follow-up information were evaluated for any relationship between the histological parameters and overall survival. Follow-up till December 2018 was obtained (minimum 1-year follow-up).
Statistical Analysis
Univariate analysis with the Mann-Whitney and the Fisher exact tests (for categorical data) and unpaired t test (for continuous data) were used to assess the variables in predicting LN metastasis and overall survival (in early-stage cancers). The factors that were significant were then entered into multivariate analysis using a multiple logistic regression model to detect independent predictors. All tests were 2-sided with significance considered at P < .05. All statistical analyses were carried out using SPSS software, version 21 (SPSS Inc, Chicago, Illinois).
Results
During this period, a total of 147 cases of oral cavity cancers were operated in our institute. Out of these, neck dissection was not performed in 3 cases and slides and blocks were issued to the patients in 4 cases as they opted to continue treatment in some other institute. Among the different sites, anterior two-thirds of tongue (72 cases, 52.2%) and buccal mucosa (54 cases, 36.2%) were the 2 most common sites, while other sites were less commonly affected (lip—7 cases, retromolar trigone—4 cases, floor of mouth—1 cases, and gingivobuccal sulcus—2 cases). Only buccal mucosa and tongue SCC were included for analysis, as other sites were very rarely affected. Thus, a total 126 cases were included in the study for analysis. There were 104 male and 22 female patients (M–F ratio 4.7:1). The age of the patients ranged from 22 to 78 years (average: 47.2 years). There were 57 (45.2%) T1, 48 (38.1%) T2, 14 (11.1%) T3, and 7 (5.6%) T4a tumors. There were 14 (11.1%) well-differentiated, 109 (86.5%) moderately differentiated, and 3 (2.4%) poorly differentiated carcinomas. Average number of LNs sampled in each case was 12 (±3.6). Lymph node metastasis was identified in 48 (38.1%) cases, while it was free in 78 (61.9%) cases. The LN stage was as follows: 78 N0, 18 N1, 28 N2b, and 2 N3b. All cases showed a tumor-free margin.
The different histological parameters predicting the risk of LN metastasis have been shown in Table 1. By univariate analysis, the different histological parameters that showed significant correlation with risk of LN metastasis included histological grade (P = .0391), invasive pattern of tumor invasion (P < .001), presence of tumor budding (P < .001), presence of PNI (P < .001), and LVE (P < .001). Other clinical and histological parameters such as age, sex, maximum tumor dimension, T stage of tumor, depth of invasion, tumor stroma, and host lymphocytic response did not show any significant correlation with risk of LN metastasis (P > .05). On multivariate analysis, invasive pattern of tumor invasion and presence of tumor budding were the 2 most significant factors for predicting LN metastasis. Similar observations were also noted when buccal mucosa and tongue cancers were analyzed separately (Table 2). The WPOI remained the most important predictor of LN metastasis in both groups. The risk of LN metastasis was significantly higher in patients with ≥3 buds/HPF. The LN stage (N stage) also showed significant correlation with the average number of tumor buds per HPF (Table 3).
Comparison of Different Parameters Between Lymph Node Positive and Negative Cases.
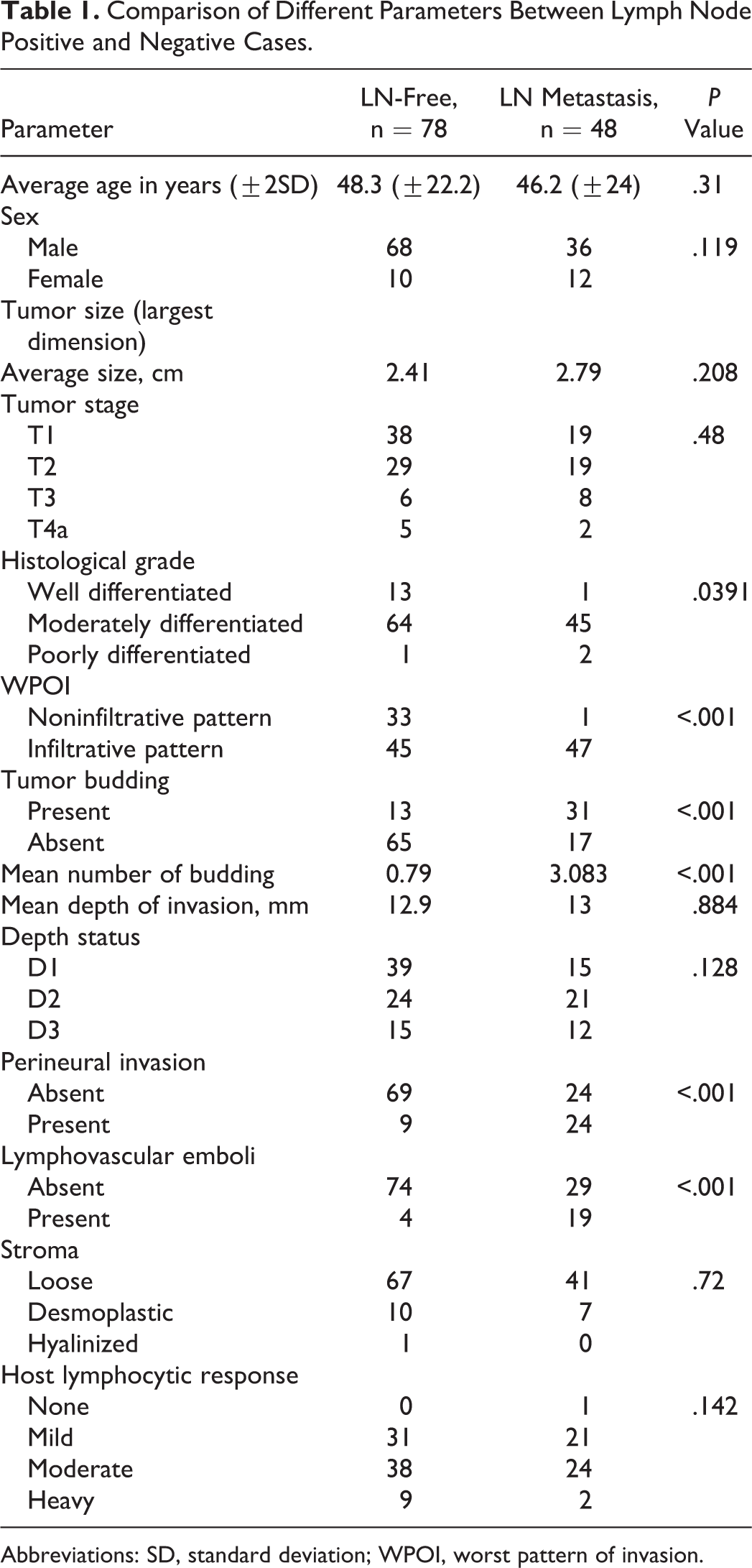
Abbreviations: SD, standard deviation; WPOI, worst pattern of invasion.
Histological Prognostic Factors in Predicting Nodal Metastasis in Buccal Mucosa and Tongue Cancers.
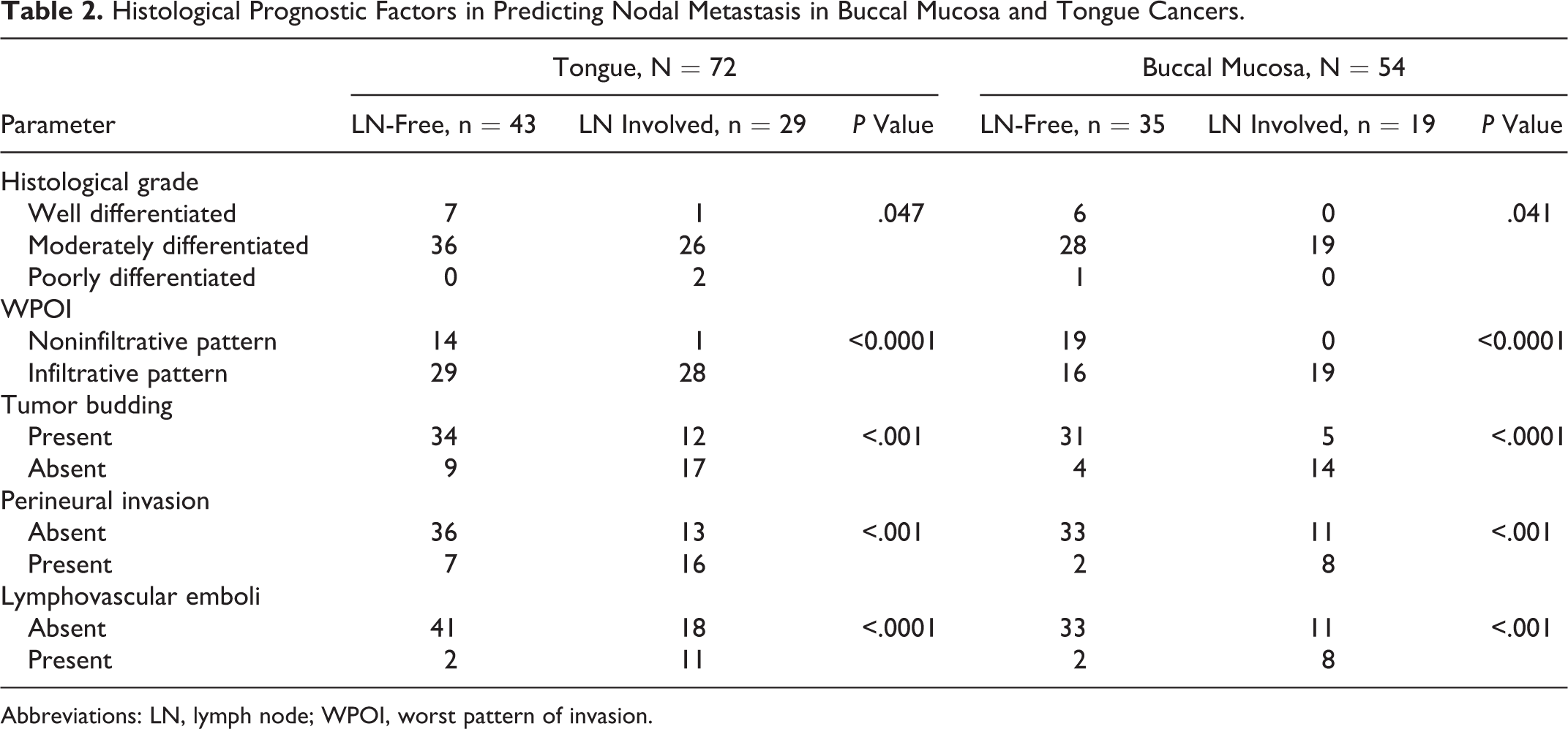
Abbreviations: LN, lymph node; WPOI, worst pattern of invasion.
Relationship of Number of Tumor Budding and Lymph Node Involvement Status.
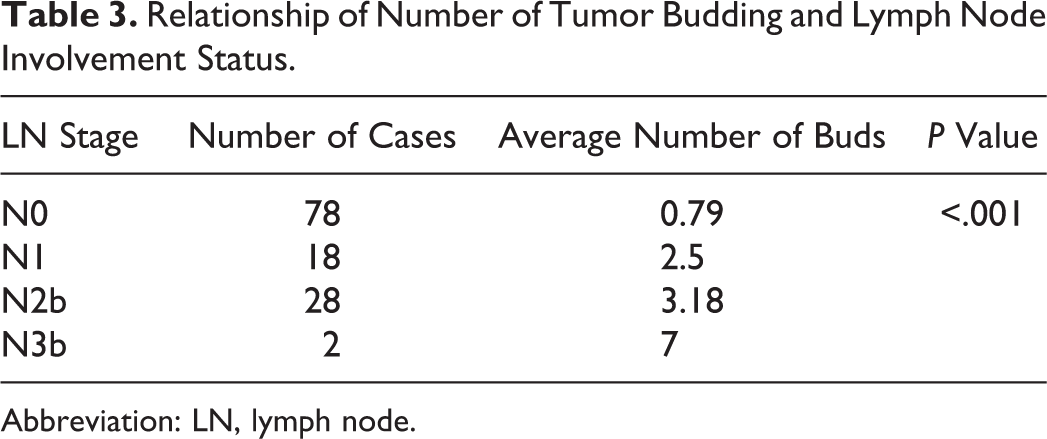
Abbreviation: LN, lymph node.
There were 67 cases that qualified for early-stage tumor (T1/T2, N0, M0). Among these, follow-up information was available for 48 cases, while the rest of the patients were lost to follow-up. The average duration of follow-up was 28.7 months (ranging from 12 to 78 months). Eight patients died within this period due to disease, while 2 of them had recurrence. No clinical or histological parameter showed any statistically significant correlation with patient outcome (Table 4). The mean overall survival of patients with invasive pattern of infiltration was shorter than patients with cohesive pattern of infiltration (37.7 vs 44.4 months), but this difference was not statistically significant (P = .436). Patients with presence of tumor budding had shorter overall survival (28.5 months) compared to patients without tumor budding (44.1 months), but this difference was not statistically significant (P = .153). Similarly, other histological parameters such as depth of invasion and LVE also didn’t show any significant influence on patient survival (Figure 3).
Comparison of Different Clinical and Pathological Factors Determining Outcome in Early Oral Cavity Cancers (T1/2, N0, M0).
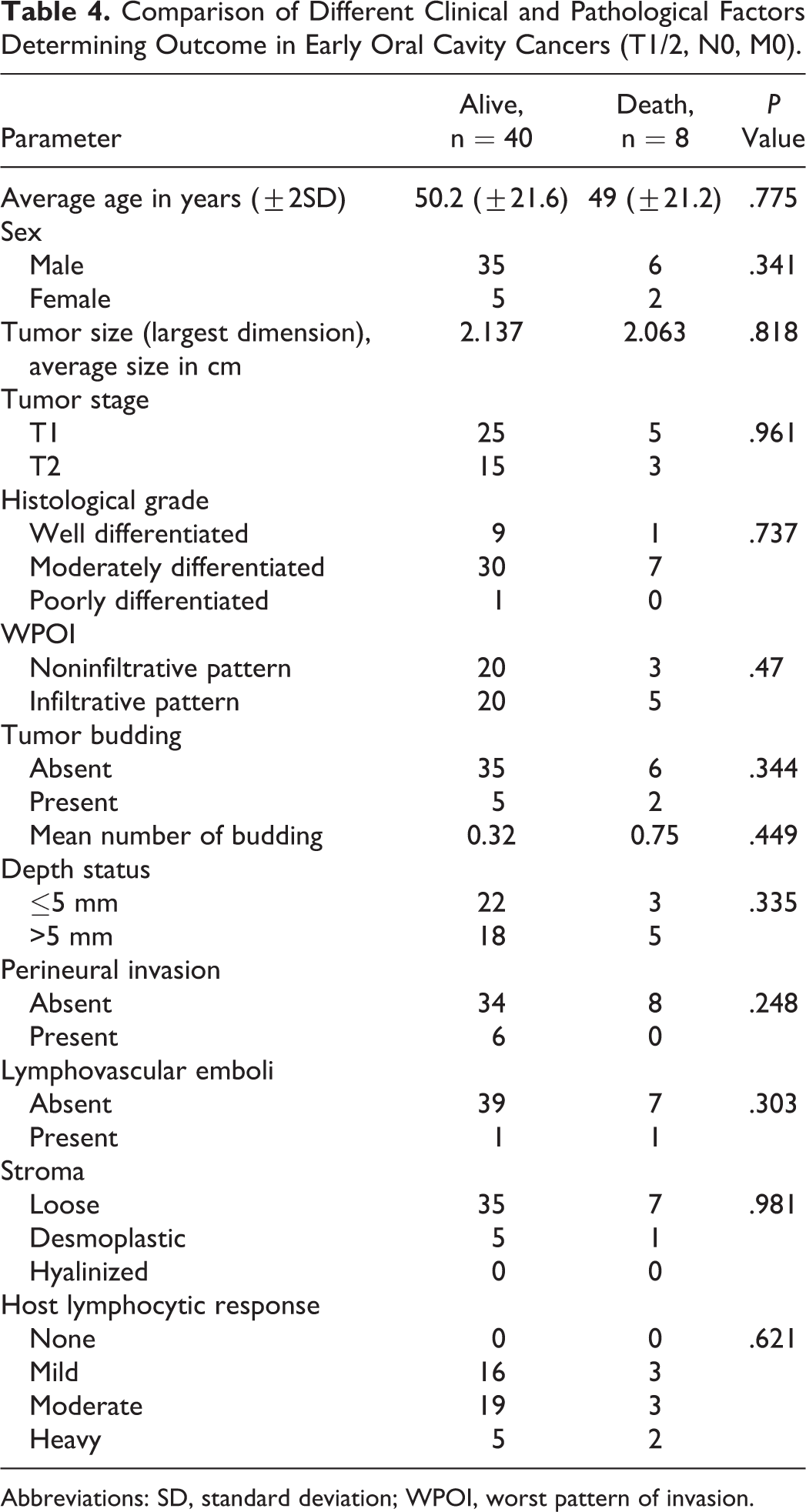
Abbreviations: SD, standard deviation; WPOI, worst pattern of invasion.
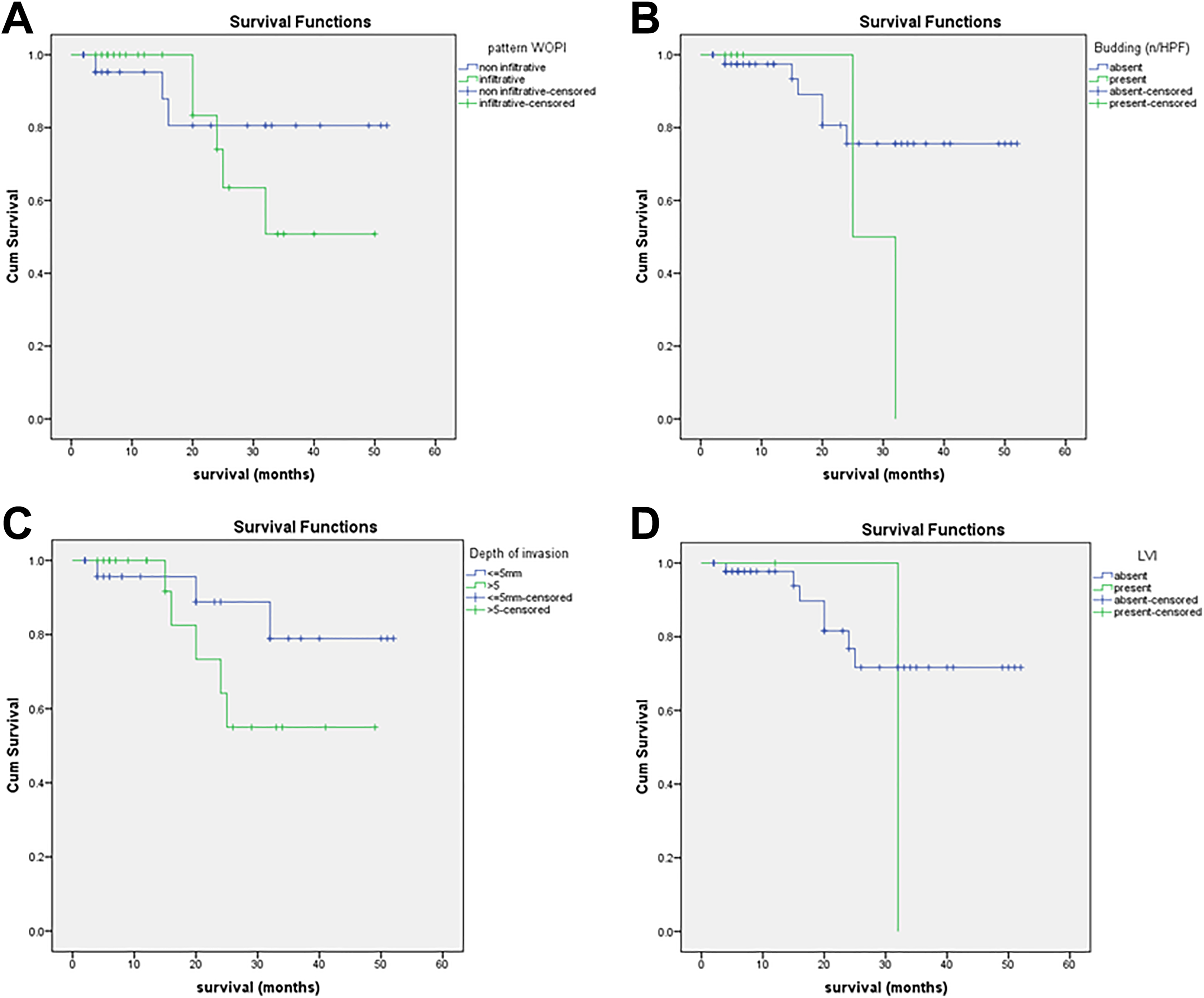
Kaplan-Meier survival analysis curve showing effect of pattern of invasion (A), tumor budding (B), depth of invasion (C), and lymphovascular invasion (D) on survival.
Discussion
The incidence of oral cavity carcinoma is steadily increasing worldwide due to tobacco consumption and has become a public health problem in developing countries such as India. In general, the prognosis of these patients remains poor. The prognosis of oral cavity cancers depends on multiple clinical and pathological parameters.
The management of OSCC is mainly guided by its clinical stage (Tumor node metastasis [TNM]). Lymph node metastasis is considered as an independent poor prognostic factor for OSCC. Histological examination of the excised cervical LNs is the gold standard to detect the presence of LN metastasis. However, clinical examination and radiological investigations such as computed tomography scan is routinely used to determine LN metastasis, with variable sensitivity. Previous studies have evaluated various histological parameters that can predict cervical LN metastasis in OSCC. Invasive tumor front is an area of recent research interest in OSCC. 20 Pattern of invasion and tumor buds are 2 important parameters determining the risk of LN metastasis. Various previous studies have described that the presence of tumor budding is an independent risk factor for LN metastasis in oral cavity SCC. 14 Tumor budding is an expression of 2 properties of malignancy, which include loss of cellular cohesion and active invasive movement. 21 Tumor budding is an inexpensive and easy to evaluate histological parameter that shows good interobserver agreement. 14 A strong correlation between the number of tumor buds and LN metastasis has been observed in OSCC. 8,22 Different studies have evaluated the cutoff value of tumor buds for predicting LN metastasis. Most of the studies have evaluated tumor buds using ×20 magnification and found 5 buds or more are significantly associated with higher chance of metastasis. 7,21 However, ×20 magnification may not be available in all microscope produced by different manufactures, whereas ×40 magnification is universally present. Thus, we evaluated the number of tumor buds per ×40 field and found ≥3 buds are significantly associated with risk of LN metastasis in OSCC irrespective of tumor stage and site. We also found the number of tumor buds was significantly associated with N stage. Although some authors recommend to use different tumor budding criteria for different subsites within oral cavity, we evaluated them together because according to the current cancer staging system of World Health Organization and American Joint Committee on Cancer, carcinoma of these sites are staged using same criteria and shows similar biological behavior. 3,17 Using different cutoff for budding for different sites will complicate the reporting format, and it will be difficult to maintain uniformity. Even when evaluated separately, ≥3 buds/×40 field was associated with higher risk of LN metastasis in carcinoma of both buccal mucosa and oral tongue. Tumor budding can be evaluated in preoperative biopsies, which can predict the risk of LN metastasis and occult metastasis with high sensitivity. 22,23
Worst pattern of tumor invasion (pattern 4 and 5) are associated with higher risk of LN metastasis. Pattern of invasion is strongly associated with risk of LN and distant metastasis. Invasive POI is able to predict the risk of occult LN metastasis efficiently. 24 In a large cohort of cT1-2 N0M0 patients, who had no LNs metastasis based on clinical and imaging findings, presence of invasive POI was significantly associated with cervical LN metastasis at resection. 8 We also found a significant correlation between invasive POI and LN metastasis over all clinical stages both on univariate and multivariate analyses. However, some studies have reported contradictory findings. Lundqvist et al and Kane et al didn’t find any adverse effect of WPOI on risk of LN metastasis. 25,26 Our study, contrary to previous few studies, found no association between depth of invasion and risk of LN metastasis in OSCC across all stages. This could be due to relatively smaller sample size in this study.
Early-stage OSCC (T1-2, N0, M0) comprises a heterogeneous group. Despite improvement in treatment, the outcome remains unsatisfactory in a subset of patients with early-stage OSCC who might need more aggressive treatment. The ability to stratify early-stage tumors into high- and low-risk categories would represent a major advancement in their management. There is search for histological parameters that can predict an unfavorable outcome in these patients. Unfavorable POI, tumor budding, and depth of invasion have been documented as poor prognostic factors in OSCC in some previous studies. 27,28 In a multicentric study involving a large patient cohort, Almangush et al found high-risk tumor budding (≥5/×20 field), depth of invasion ≥4 mm, and high-risk POI are associated with poor prognosis in early-stage oral tongue SCC. 7 These results were reflected in other studies as well. 9,19 In this study, we found in patients with early-stage OSCC, depth of tumor >5 mm, presence of tumor budding, and invasive POI were associated with a shorter overall predicted survival, although these parameters didn’t reach a statistical significance. This may be due to relatively small number of cases for which follow-up information was available and a relatively limited follow-up period. The previous studies have used variable cutoff for depth of invasion. While most of the previous studies used 4 mm or more depth of invasion as predictor of poor prognosis, most of the recent studies found 5 mm as the most useful cutoff for early-stage OSCC. 29 -31 We also found >5 mm depth of invasion was associated with a shorter survival in early-stage OSCC, while there was no significant difference of survival with ≥4 mm invasion. Although majority of the previous studies have described invasive POI and tumor budding as independent poor prognostic factors for OSCC, some studies failed to show any association. 25 This may be due to different inclusion criteria and shorter period of follow-up.
Although all cases in this study had a tumor-free surgical margin, tumor margin doesn’t seem to bear a strong prognostic significance in T1 or T2 OSCC. 32,33 Multiparametric histological risk assessment score (HRS) have been evaluated for predicting the survival of T1-T4 OSCC. Although some studies have found HRS useful, other studies have found it very cumbersome or not of much prognostic significance. 34,35 Thus, it is not widely followed any more. Other histological parameters such as LVE and PNI are well-known risk factors for OSCC in all stages and are mandatory part of reporting format. 8 We also found significant association of LVE and PNI with the risk of LN metastasis, although tumor budding and invasive POI were the 2 most important risk factors on multivariate analysis.
To conclude, the prognosis of OSCC is predicted by various histological parameters that are easy to assess on routine hematoxylin and eosin–stained sections. These parameters such as tumor budding, POI, depth of invasion, LVE, and PNI can provide extremely valuable prognostic information. Tumor budding and POI are easy and reliable predictive factors in early-stage OSCC and are associated with higher risk of cervical LN metastasis in all stages, associated with a poorer outcome. Thus, based on the findings of this study and the previous studies, we recommend that these parameters should be routinely evaluated both in resection and in preoperative biopsy specimens and should be a part of standard reporting format for OSCC, which may help in individualization of treatment in these patients.
Limitations
The study is limited by small sample size, relatively short follow-up period, and small number of patients with available follow-up information.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
