Abstract
The aim of this study is to investigate the effect of gastric Helicobacter pylori colonization on nasal functions. The study enrolled patients (n = 100) who underwent endoscopy for gastroesophageal reflux disease. Patients with laryngopharyngeal reflux (LPR) were identified by Reflux Symptom Index (RSI) and Reflux Finding Score (RFS). Patients were divided into 2 groups: LPR (+) (n = 64) H pylori (+), RSI > 13, RFS > 7; LPR (−) (n = 36) H pylori (+), RSI < 13, RFS < 7. Visual analog scale (VAS), sinonasal outcome test-22 (SNOT-22), peak nasal inspiratory flowmeter (PNIF), mucociliary clearance (MCC), and olfactory tests were used to evaluate the nasal functions. The average VAS for nasal obstruction, PNIF, and MCC did not differ significantly between the LPR (+) and LPR (−) groups (P > .05). However, the average olfactory test scores were lower in the LPR (+) patients than the LPR (−) patients (P < .05). Also, the SNOT-22 scores were significantly higher in LPR (+) patients than in LPR (−) (P < .01). Nasal functions and symptom scores were also evaluated according to the H pylori grading. The PNIF, MCC, SNOT-22, and olfactory test results deteriorated as the gastric mucosal H pylori colonization increased (P < .05). In conclusion, nasal functions differed between LPR disease and GERD only, while the density of H pylori colonization in the gastric mucosa had an effect on nasal function.
Introduction
Laryngopharyngeal reflux (LPR) is an extra-esophageal manifestation of gastroesophageal reflux and causes distressing symptoms when the gastric contents stimulate and injure the laryngeal mucosa. Laryngopharyngeal reflux differs from classic gastroesophageal reflux disease (GERD), and the laryngeal complaints are more prominent. 1 Throat clearing, persistent cough, heartburn/dyspepsia, globus sensation (lump in the throat), and voice quality change are the main otolaryngologic manifestations of acid reflux. Laryngopharyngeal reflux constitutes 10% of patients presenting to an otolaryngologist and should not be underestimated.
The laryngopharyngeal epithelium is sensitive to damage caused by gastric acid. The acidic content reaching the upper airways or the esophageal–nasal reflex stimulated by vagal nerve response may result in the congestion of nasal mucosa and a pathologic increase in nasal mucus secretion. 2 While the larynx can produce some carbonic anhydrase isoenzymes, the pharyngeal mucosa cannot. 3 When the protective effect of the mucosa is eliminated, mucociliary dysfunction results in mucous stasis. Studies carried out recently have demonstrated the reflux as a potential exacerbating factor in upper airway diseases. 4 –6
Helicobacter pylori is a microaerophilic, Gram-negative spiral organism that has been shown to be the causative factor for stomach ulcers and gastritis. 4 Helicobacter pylori infection is widespread throughout the world. Helicobacter pylori not only colonizes the gastric mucosa but also the oral cavity, adenoids, tonsils, and pharyngeal mucosa. 4 –6 When H pylori infection occurs in the gastric antrum, it causes an increase in the acidity of gastric secretions. 5 The acidic content reaching the upper airways may deteriorate the nasal functions.
The current LPR treatment is based on the treatment of GERD, with acid suppression through H2 antagonists, proton pump inhibitors, lifestyle modifications, prokinetics, alginate, Reza band, and in selected cases surgery. However, H pylori eradication is associated with improved laryngeal reflux symptoms in refractory cases. 7 There have been significantly fewer studies investigating the relationship between H pylori and LPR. For this reason, we hypothesize that the colonization of H pylori in the gastric mucosa may affect the nasal functions in patients with LPR disease.
Patients and Methods
This study was carried out at a tertiary clinic with a diagnosis of GERD disease. The study enrolled patients who underwent endoscopy for GERD. The data retrieved from the medical records included patient age, sex, and presenting complaints. All study protocols and informed consent forms were collected and approved by the institutional review board.
Exclusion criteria include nasal septal deviation, previous nasal surgery, nasal polyposis, preexisting sinus disease or nasal allergies of any sort, preexisting subjective olfactory disturbance, smokers, intranasal drug abusers, and systemic disease such as diabetes mellitus and rheumatologic disorders. The current use of intranasal corticosteroid, antihistamine, decongestant and/or systemic corticosteroid, proton pump inhibitor, or histamine H2 receptor antagonists, alginate, and prokinetics as well as those who underwent fundoplication operation were also excluded from the study.
Nasal function tests were performed in all patients, together with a complete ear, nose, and throat examination. All patients underwent rigid and flexible endoscopic examination by the same otolaryngology specialist who was blinded to the symptoms of the patients. The patients had no allergic symptoms, and prick tests were negative.
Patients with LPR were identified by Reflux Symptom Index (RSI) and Reflux Finding Score (RFS) before upper gastrointestinal endoscopy. Both RSI > 13 and RFS > 7 were accepted as LPR as recommended by Belafsky et al. 8,9 The RSI was validated by Belafsky et al 8 and consists of 9 questions that are scored between 0 (no problem) and 5 (severe problem). This tool aims to assess symptoms caused by LPR. The RFS was also validated by Belafsky et al 9 and is used to evaluate endoscopic findings of the larynx. This tool is composed of 8 parameters, each of which has a special scoring method. An overall RFS equal to 7 or above shows a high correlation with reflux that has been documented by pH monitoring and could be accepted as pathologic.
Patients with GERD were prospectively included in the study. Patients were divided into 2 groups: LPR (+) (n = 64), RSI > 13, RF > 7; LPR (−) (n = 36), RSI < 13, RFS < 7. In the pre-endoscopy evaluation, 36 patients who had typical complaints regarding GERD but did not prioritize laryngeal complaints with RSI < 13 and RFS < 7 were included in the LPR (−) group, while 64 patients with a preliminary diagnosis of GERD (heartburn and/or acid regurgitation once a week or common) with possible LPR with RSI > 13 and RFS > 7 whose major complaints were laryngeal (such as voice related problems, cough, hoarseness, throat globe and cleaning, and difficulty in swallowing) were included in the LPR (+) group.
Gastric antral biopsy with histology is the most commonly employed method of testing and is considered by some to be the gold standard of H pylori diagnosis. 10 A biopsy specimen from the antrum for histopathologic examinations was fixed in neutral formalin solution and was embedded in paraffin for each patients. Two sections were stained with hematoxylin–eosin and 1 section was stained with Giemsa. The slides were microscopically examined for the bacterial density. The colonization of H pylori on the gastric epithelium was graded on a 4-point scale of none (grade 0), mild (grade 1), moderate (grade 2), or severe (grade 3), according to the guidelines of the Sydney system. 11 Histological grading was made independently by 2 pathologists who were blinded to the results of other tests. Patients with negative H pylori biopsies were excluded from the study. Patients underwent the following tests for nasal functions.
Obstruction and Olfactory Evaluation Tests
Visual analog scale (VAS) was used for all patients to show obstructive symptoms subjectively. There are numbers on the scale from 0 to 10, where 0 reflects total obstruction, while 10 represent a fully opened passage. The value which best reflects the nasal obstruction indicated the patient’s VAS result.
Sinonasal outcome test-22 (SNOT 22), which was validated by Hopkins et al, 12 was used to evaluate sinonasal symptoms. Data were collected by means of a self-administered questionnaire. The SNOT 22 is composed of 22 questions that are scored from 0 to 5. A lower score implies a better result.
Peak nasal inspiratory flowmeter (PNIF) measurement was performed using a nasal inspiratory flowmeter (Clement Clark International, Harlow, Essex, United Kingdom). The value during forced inspiration was expressed as liters per minute. The patients were asked to expire forcefully while sitting and inspire forcefully through the nose with an anesthesia mask placed over the mouth. Of the 3 consecutive measurements with a maximum difference of 10%, the highest measurement was recorded as the final value.
The Connecticut Chemosensory Clinical Research Center test was conducted in an odorless room under standard conditions using a commercially available smell test kit for the patients. This test is composed of n-butanol odor threshold test and odor identification test. 13 For both parts of the test, each nostril is tested separately by having the patient occlude the opposite nostril.
Detection threshold is measured using 9 serial dilutions of butanol. The strongest butanol concentration (bottle 0) was 4% butanol in deionized water. Each subsequent dilution (bottles 1-9) was 1:3 dilutions with deionized water. Each concentration is presented along with a water control in a double-blind, forced-choice paradigm. Threshold is defined as the dilution at which the butanol bottle is correctly identified in 4 consecutive trials. There are 7 olfactory stimuli (baby powder, chocolate, cinnamon, coffee, mothballs, peanut butter, and soap) and 3 stimuli (ammonia, Vicks, and wintergreen) used to test trigeminal nerve sensory function. Olfactory tests were conducted individually and were scored out of 7 (0: worst, 7: best olfaction). 13
Nasal mucociliary clearance (MCC) was assessed for all individuals by blinded researcher. Saccharin transit time test was used to measure the nasal MCC, as previously described. 14 Patients were seated in upright position. Granulated sodium saccharin (250 mg) was deposited under visual control. A saccharin granule was placed 2 cm inside the right nostril lateral to inferior turbinate by the tester. They were instructed to swallow every 30 seconds per minute with a chronometer. The time when the patients first percepted the sweet taste of the saccharin was recorded in minutes. Individuals were instructed not to breathe deeply, talk, cough, sneeze, or sniff during the test. Test results were compared within and between the groups at the end of the study.
Statistical Analysis
Statistical analyses of the data were conducted using IBM SPSS Statistics 22 (IBM SPSS, Istanbul, Turkey). Data were analyzed using descriptive statistical methods (mean and standard deviation). Shapiro-Wilk test was used to assess the normal distribution of the parameters. Parametric independent sample t test was used for the comparison of independent groups and Mann-Whitney U test was used for nonparametric data. Kruskal-Wallis test was used for the nonparametric comparison of more than 2 groups and Mann-Whitney U test was used for intergroup comparison. Results were evaluated using the 95% confidence intervals (CIs), and the level of significance was set at P < .05.
Results
The study group comprised 100 consecutive patients. Forty-four (44.0%) of them were male and 56 (56.0%) were female. The mean age was 40.84 ± 11.81 years (range: 19-67 years). Sixty-four (64%) of patients were in the LPR (+) group, while 36 of them were in the LPR (−) group. No significant difference was observed between the 2 groups with respect to age (P = .341) and gender (P = .946; Table 1). The colonization of H pylori on the gastric epithelium was mild in 46, moderate in 42, and severe in 12 cases (Table 2).
Evaluation of the Groups According to Age and Sex.a
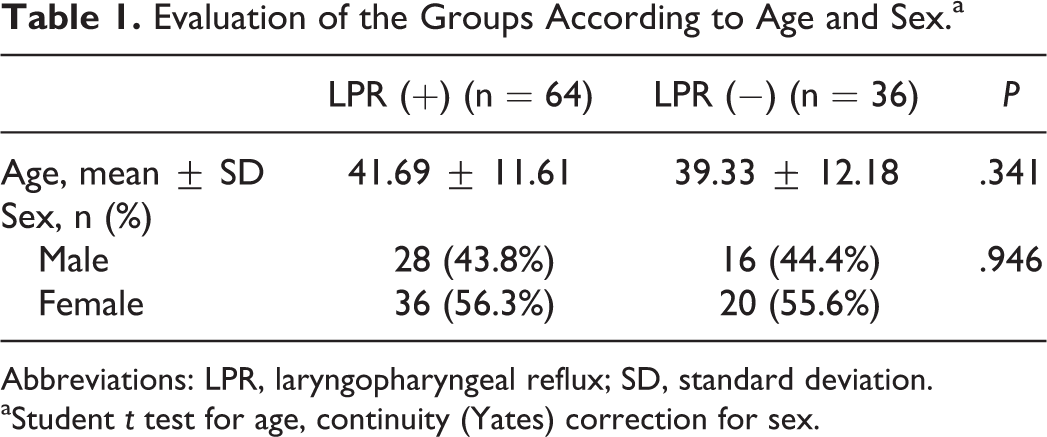
Abbreviations: LPR, laryngopharyngeal reflux; SD, standard deviation.
aStudent t test for age, continuity (Yates) correction for sex.
The Colonization of Helicobacter pylori on the Gastric Epithelium.

The average VAS for nasal obstruction, PNIF, and MCC measurements did not differ significantly between the groups (P > .05). However, the average olfactory test scores were lower in the LPR (+) patients than the LPR (−) patients (P = .035; P < .05). Also, the SNOT-22 scores were significantly higher in LPR (+) patients than in LPR (−) (P = .001; P < .01; Table 3).
Comparison of the Nasal Functions According to the LPR.
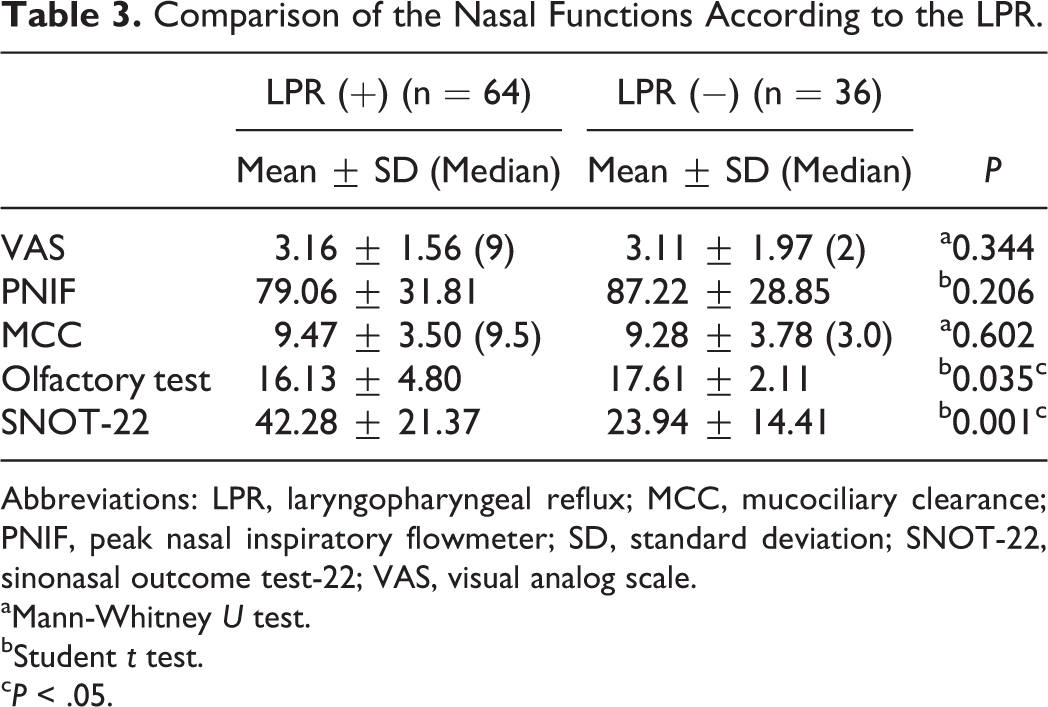
Abbreviations: LPR, laryngopharyngeal reflux; MCC, mucociliary clearance; PNIF, peak nasal inspiratory flowmeter; SD, standard deviation; SNOT-22, sinonasal outcome test-22; VAS, visual analog scale.
aMann-Whitney U test.
bStudent t test.
c P < .05.
Nasal functions were evaluated in the LPR (−) group according to the gastric H pylori colonization. The average VAS for nasal obstruction, PNIF, and MCC measurements did not differ significantly between the groups (P > .05). However, there was a significant difference in olfactory test results (P = .034; P < .05). “Moderate” colonization showed a significant higher results than mild group (P = .014; P < .05). There were no differences between the other groups (P > .05).
There was a significant difference in the SNOT-22 scores between the groups (P = .002; P < .01). The “moderate” group showed significantly low scores than both “mild” and “severe” groups (P < .05; P < .01). “Severe” group had the highest scores (P > .05; Table 4).
Evaluation of the Nasal Functions in LPR (−) Patients According to the Helicobacter pylori Colonization.
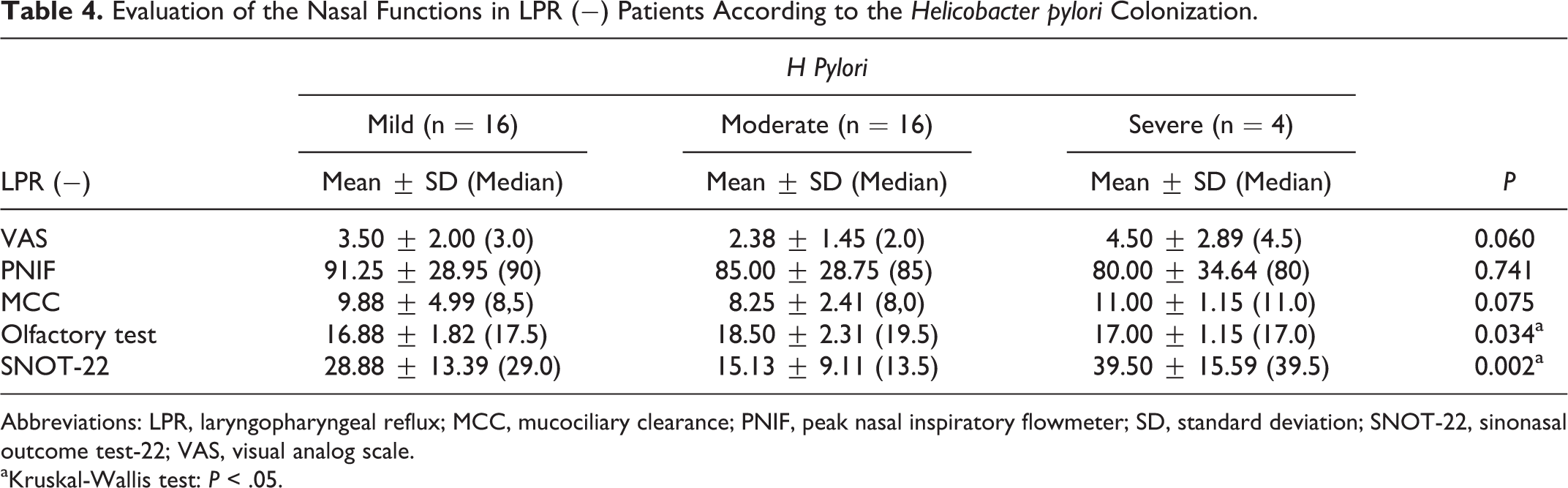
Abbreviations: LPR, laryngopharyngeal reflux; MCC, mucociliary clearance; PNIF, peak nasal inspiratory flowmeter; SD, standard deviation; SNOT-22, sinonasal outcome test-22; VAS, visual analog scale.
aKruskal-Wallis test: P < .05.
The average VAS for nasal obstruction did not differ significantly in LPR (+) patients between the groups according to the gastric H pylori colonization (P > .05). There was a significant difference in the PNIF measurements between the groups (P = .019; P < .05). “Mild” group had significantly higher measurements compared with both “moderate” and “severe” groups (P = .011; P = 0.049; P < .05).
There was a significant difference in the MCC time between the groups (P = .038; P < .05). “Severe” group had significantly higher measurements compared with both “mild” and “moderate” groups (P = .036; P = .008; P < .05).
There was a significant difference in the olfactory test scores between the groups (P = .046; P < .05). “Mild” group had significantly higher measurements compared with “severe” group (P = .012; P < .05; P < .05). There was no such difference in the results between “mild” and “moderate” groups for the control group (P > .05).
The SNOT-22 scores differed significantly between the groups (P = .006; P < .01). “Severe” group had significantly higher measurements compared with both “mild” and “moderate” groups (P = .012; P = .006; P < .01; Table 5).
Evaluation of the Nasal Functions in LPR (+) Patients According to the Helicobacter Pylori Colonization.
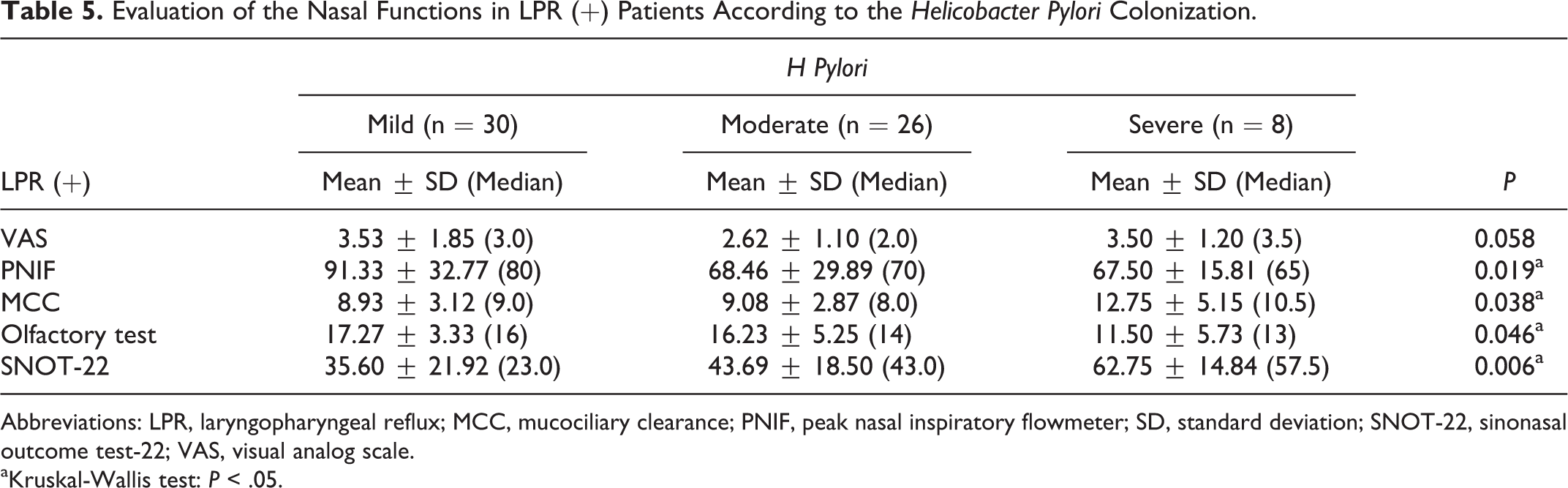
Abbreviations: LPR, laryngopharyngeal reflux; MCC, mucociliary clearance; PNIF, peak nasal inspiratory flowmeter; SD, standard deviation; SNOT-22, sinonasal outcome test-22; VAS, visual analog scale.
aKruskal-Wallis test: P < .05.
Discussion
This study examined whether H pylori colonization in the gastric mucosa has any effects on the nasal functions in patients with LPR disease. Laryngopharyngeal reflux is being diagnosed with increasing frequency and is regarded as a distinct disease. Helicobacter Pylori is frequently reported in patients with GERD undergoing endoscopic biopsies. However, the nasal function of these patients has not been studied comprehensively.
Studies carried out recently have demonstrated the reflux as a potential exacerbating factor in upper airway diseases. Dagli et al 15 revealed LPR had a negative effect on nasal resistance and nasal congestion. Bozec et al 16 also showed higher nasal resistance and nasal obstruction score in patients with LPR before treatment. Furthermore, Wong et al 2 revealed nasal functions decreased in GERD.
In our study, the average VAS for nasal obstruction, PNIF, and MCC measurements did not differ significantly between the groups (P > .05; Table 3). These findings did not supported previous studied. 15,17 Delehaye et al 17 found a statistically significant prolongation of the mucociliary transport time in 74% of patients with erosive esophagitis. Our study revealed LPR hadn’t any negative effect on nasal resistance, nasal congestion, and nasal clearance. However, the average olfactory test scores were lower in the LPR (+) patients than the LPR (−) patients (P < .05). Also, the SNOT-22 scores were significantly higher in LPR (+) patients than in LPR (−) (P < .05; Table 3). Katle et al 18 also reported increased SNOT-20 score in GERD. Nevertheless, nasal obstruction and MCC were not different between the groups in our study and this suggested there might be another factor for high symptom scores in the LPR (+) group.
When H pylori infection occurs in the gastric antrum, it causes an increase in the acidity of gastric secretions and eradication of H pylori in these patients is associated with improved reflux symptoms. For this reason, we investigate the effect of gastric H pylori colonization on nasal functions. Nasal functions and symptom scores were evaluated according to the H pylori colonization grading. It was found out that PNIF, MCC, SNOT-22, and olfactory test results deteriorated in LPR (+) patients as the gastric mucosal H pylori colonization increased (P < .05). The average VAS for nasal obstruction did not differ significantly in LPR (+) patients (P > .05). But this was a subjective finding and patients did not complain about the nasal obstruction as the study was conducted on patients without nasal disease.
“Mild” group had significantly higher PNIF measurements compared with both “moderate” and “severe” groups (P < .05). Also, “severe” group had significantly higher MCC measurements compared with both “mild” and “moderate” groups (P < .05). These results supported previous studies. 15,17 For this reason, we concluded that H pylori was a possible factor for this effect.
In addition, acid reflux may lead to high SNOT-22 scores, which indicated worsening of the nasal symptoms in the LPR group. “Severe” group had significantly higher SNOT-22 scores compared with both “mild” and “moderate” groups (P < .01; Table 5).
Ercan et al 19 previously reported that there was no relationship between gastric H pylori infection and LPR. However, it has recently been shown that eradication of H pylori may prolong disease-free intervals and, in any case, does not exacerbate reflux symptoms in patients with GERD. 20,21 This suggests that gastric colonization of H pylori may be a causative factor for LPR. However, it is still unclear if H pylori produces a direct damage to the nasal mucosa or produces damage as a result of acid reflux. These findings revealed that there was a positive association between rate of concomitant GERD with LPR and H pylori positivity.
In conclusion, nasal functions differed between LPR disease and GERD only, while the density of H pylori colonization in the gastric mucosa had an effect on nasal function.
Footnotes
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
