Abstract
This was a randomized controlled trial of low thermal damage device versus traditional electrosurgery in children 3 to 17 years old with a clinical diagnosis of sleep disordered breathing, obstructive sleep apnea with adenotonsillar hypertrophy, or recurrent adenotonsillitis. Pain score (Wong-Baker FACES pain scale) was recorded each morning before eating, drinking, or administering pain medication for 14 days postoperatively. Seventy-five children were enrolled. There was no difference in the rate of decrease in pain scores. A significant interaction between rate of pain decrease and number of pain medication doses was present (P < .0001). Median number of pain medication doses was greater with electrosurgery (36, range: 7-49) versus low thermal device (21, range: 2-124; P = .001). Pain scores reached 0 after a median of 7 days (95% confidence interval [CI], 5.2-8.6) for low thermal device and 9 days (95% CI, 8.0-10.0) for electrosurgery (P = .67). One child randomized to electrosurgery was withdrawn due to hospitalization for postoperative bleed. In children, low thermal device results in significantly less pain medication used during the postoperative period than electrosurgery.
Introduction
Tonsillectomy is the second most common surgical procedure in children younger than 15 years. 1 It is estimated that 583 000 outpatient tonsillectomy procedures were performed in children in the United States in 2006, accounting for 16% of all ambulatory surgeries. 1,2 In children 0 to 6 years old, the diagnosis of airway obstruction or sleep disturbance accounts for the majority of surgical indications, with adenotonsillectomy being the dominant (∼90%) procedure performed (vs 10% for tonsillectomy alone). 2
Surgical techniques for tonsillectomy include traditional electrosurgery (ES), microdebrider, laser dissection, harmonic scalpel, PlasmaBlade, and coblation. 3 We compared traditional ES (Valleylab; Medtronic, plc, Minneapolis, Minnesota) with low-temperature pulsed radiofrequency energy dissection “low thermal device (LTD)” (pulsed-electron avalanche knife [PEAK] PlasmaBlade TnA tonsil and adenoid tissue dissection device; Medtronic, plc) in children 3 to 17 years old undergoing adenotonsillectomy. Use of low-temperature pulsed radiofrequency energy has been developed for efficient tissue dissection with minimal thermal damage. 4 -9 The goal was to compare pain scores during the 14-day postoperative period. To our knowledge, there are no known published prospective clinical trials of the PEAK PlasmaBlade on pain outcomes in pediatric patients undergoing adenotonsillectomy. 9,10
Patients and Methods
Trial Design
This was a prospective, randomized, single-blinded, pilot study in pediatric patients undergoing adenotonsillectomy. The Baptist Medical Center institutional review board for Wolfson Children’s Hospital in Jacksonville, Florida, approved the study. Parents provided written permission, and children aged 7 years and older provided written assent. The trial is listed at www.clinicaltrials.gov (NCT02533999). Surgeries were performed at Baptist Health Wolfson Children’s Hospital or Wolfson Surgery Center.
Patients
Patients aged 3 to 17 years scheduled for adenotonsillectomy with a clinical diagnosis of sleep disordered breathing, obstructive sleep apnea 11 with adenotonsillar hypertrophy, or recurrent adenotonsillitis were enrolled between October 2013 and June 2015. Children were excluded who had craniofacial malformation, previous adenotonsillectomy, suspected lymphoma, developmental delay, diabetes or any other disease that slows wound healing, peritonsillar abscess, expressive language disorder, hematologic wound healing disorder or necrotizing dermatosis, implanted electric device, mucopolysaccharidosis, use of nonsteroidal anti-inflammatory drugs in the 2 weeks prior to the surgery, allergy to medication(s) used in this protocol, other planned surgical procedure during the study period, chronic pain disorder, or planned hospital admission. In addition, children with a diagnosis of bleeding disorders or a family history of bleeding disorders were excluded.
Study Procedures
Participants were randomized to LTD or ES using a randomized block design (4 participants per block established by the study biostatistician). Randomization was stratified by surgeon, age (3-10 years and 11-17 years), and surgery site (Baptist Health Wolfson Children’s Hospital or Wolfson Surgery Center). Parents, their child, and the research study team were blinded to surgical method until all data were collected from the study (6 months following surgery). The surgeon, anesthesiologist, and operating room staff were blinded to the procedure until immediately prior to surgery. The operating and recovery room personnel were trained on study procedures and the importance of not revealing the surgery method to parents or the research team. Only anesthesiologists who agreed to a standardized anesthesia and pain protocol were assigned to the study participant. All surgeries were performed by 3 board-certified, fellowship-trained pediatric otolaryngologists who agreed to use standardized instrument settings.
Prespecified stopping criteria for the study were established. If the rate of postoperative hemorrhage, defined as level II or level III according to Gallagher et al, 12 exceeded 2 participants in the same surgical group during the first 5 days postoperatively after the first 30 patients were enrolled, the study would be stopped and unblinded. 1,3,13
Surgical Methods
Settings and methods for ES and the LTD were standardized. For the PEAK PlasmaBlade, the setting was set to 2 for coagulation and 1 for cutting, and for adenoids, to 7 for coagulation and 7 for cutting. For ES, the setting was standardized to 12 W for tonsils and 30 W for adenoids, both in coagulation mode.
Children randomized to one surgical method who required rescue by the other method were excluded from further participation in the study and replaced; these data are reported in the study “Results” section. Children requiring hospital admission were removed from the study and replaced.
Postoperative Study Procedures
Prior to surgery, the parent(s)/caregiver and patient were instructed on completing the 14-day diary that included subjective assessment of pain, pain medication use, fluid intake, diet, and activity level to be completed on the day of surgery through 14 postoperative days. Patients received a prescription for 0.1 mg/kg hydrocodone with acetaminophen to be used every 6 hours alternating with ibuprofen at 3-hour intervals for the first 24 to 48 hours for moderate to severe pain as needed. Then parents were instructed to use hydrocodone with acetaminophen, or acetaminophen alone alternating with ibuprofen every 6 to 8 hours, as needed, as pain lessened. A $40 gift card was mailed to the family upon receipt of the diary card after 14 days. The study team called the parent on days 1, 3, 5, 7, and 14 postoperatively to assess the status of the participant. After 30 (4) days following surgery, the study coordinator contacted the parent by telephone to obtain information on incidence of late complications and to assess the parent’s perception of the 30-day postoperative period. At 6 months postprocedure, parents were mailed the Glasgow Children’s Benefit Inventory (GCBI) 14 and were compensated with a $10 gift card for returning the completed GCBI questionnaire.
Outcome Measures
Pain
The child was asked by the parent to grade severity of pain daily using the Wong-Baker FACES pain rating scale 15 each morning before eating, drinking, or taking analgesics. The parent recorded each instance of pain medication use, including medication name, dose, and time administered.
Bleeding
Total blood loss during surgery was determined by the method described by Walner et al 16 in which the operating nurse recorded the blood loss based on the amount in the suction canister or suction tubing at the conclusion of the procedure.
Diet and activity
Diet and activity level were scored on the diary card each morning for the prior 24-hour period. Normal diet was defined as consumption of the types and amount of food such that another family member would not be able to recognize that the patient had undergone throat surgery. Dietary progression from liquid to soft and solid food was documented. Diet was scored as 1 (liquids and soft diet only), 2 (some solids), 3 (mostly solids), and 4 (normal diet). 13 Normal activity was defined as carrying out the same types and amounts of daily activity as before surgery, even if still associated with fatigue. Activity was scored as 1 (none), 2 (very little), 3 (mostly normal), and 4 (normal). 13
Statistical Analysis
Sample size justification
This was an exploratory study to compare mean pain scores between LTD and ES as no previous prospective clinical trials had compared the effect of LTD and ES on pain outcomes. A sample size of 70 participants was planned for this study in order to have 60 participants (30 in each group) complete all data collection (15% anticipated attrition rate). We considered this number sufficient based upon sample sizes included in previously published studies that compared surgical techniques on pain outcome with the Wong-Baker FACES in pediatric patients undergoing tonsillectomy. 17 -20
Data analyses
Demographic and baseline characteristics were summarized by surgical group. Categorical variables were summarized using frequencies and percentages, and quantitative variables were described by presenting mean and standard error of mean (SE) or median (interquartile range), where appropriate. A χ2 or Fisher exact test, whichever appropriate, was used to compare the distribution of categorical variables between surgical groups. A 2-sample t test or Mann-Whitney U test was used to compare the mean or median between 2 surgical groups. A nonparametric local smoother local regression (LOESS) was used to discern the pain reduction in 2 surgical groups over time. A mixed-effects model was used to compare the trend of pain reduction between 2 surgical groups. Pain scores from postoperative day 1 to day 14 were used as the response variable. To account for the individual-level heterogeneity in the baseline (day 1) pain scores as well as in the reduction of pain over days, intercept and the time (that represents days 1-13) were used as random variables. An unstructured correlation of the 2 random coefficients was used. In the variable surgical groups, time and interaction of these 2 variables were used as fixed effects in the basic model. Later, the variable daily doses were used in the model for comparing the pain reduction over time between 2 surgical groups. In addition, the Mann-Whitney test was used to compare median days to no pain and median number of postsurgery pain medication doses between the 2 surgical groups. To evaluate the long-term benefit, a 2-sample t test/Mann-Whitney U test were used to compare mean/median scores of the 24-item GCBI questionnaire between the 2 groups. The GCBI score was calculated according to the method by Kubba et al. 14 Model assumptions were checked before statistical analyses. All tests were performed against a 2-sided alternative at α = .05 significance level. Data were analyzed using statistical software SAS version 9.3 (SAS Institute, Cary, North Carolina).
Results
Participant demographics are shown in Table 1. Of 75 children randomized, 38 were allocated to LTD and 37 to ES. In the LTD group, 32 children were included in the analysis (3 lost to follow-up, 3 discontinued by investigator); in the ES group, 30 children were included in the analysis (5 lost to follow-up, 1 did not receive allocated intervention, and 1 discontinued due to hospitalization secondary to bleeding). Due to a large number of children not meeting all inclusion and exclusion criteria, 2 amendments were made within the first month of enrollment: increasing the originally planned age range from 3-6 years to 3-17 years and removing recurrent tonsillitis as exclusion criteria. The principle reason provided for declining participation was that the parent wanted the surgeon to select the method that was best for their child rather than by randomization.
Demographic and Baseline Characteristics by Surgical Groups.
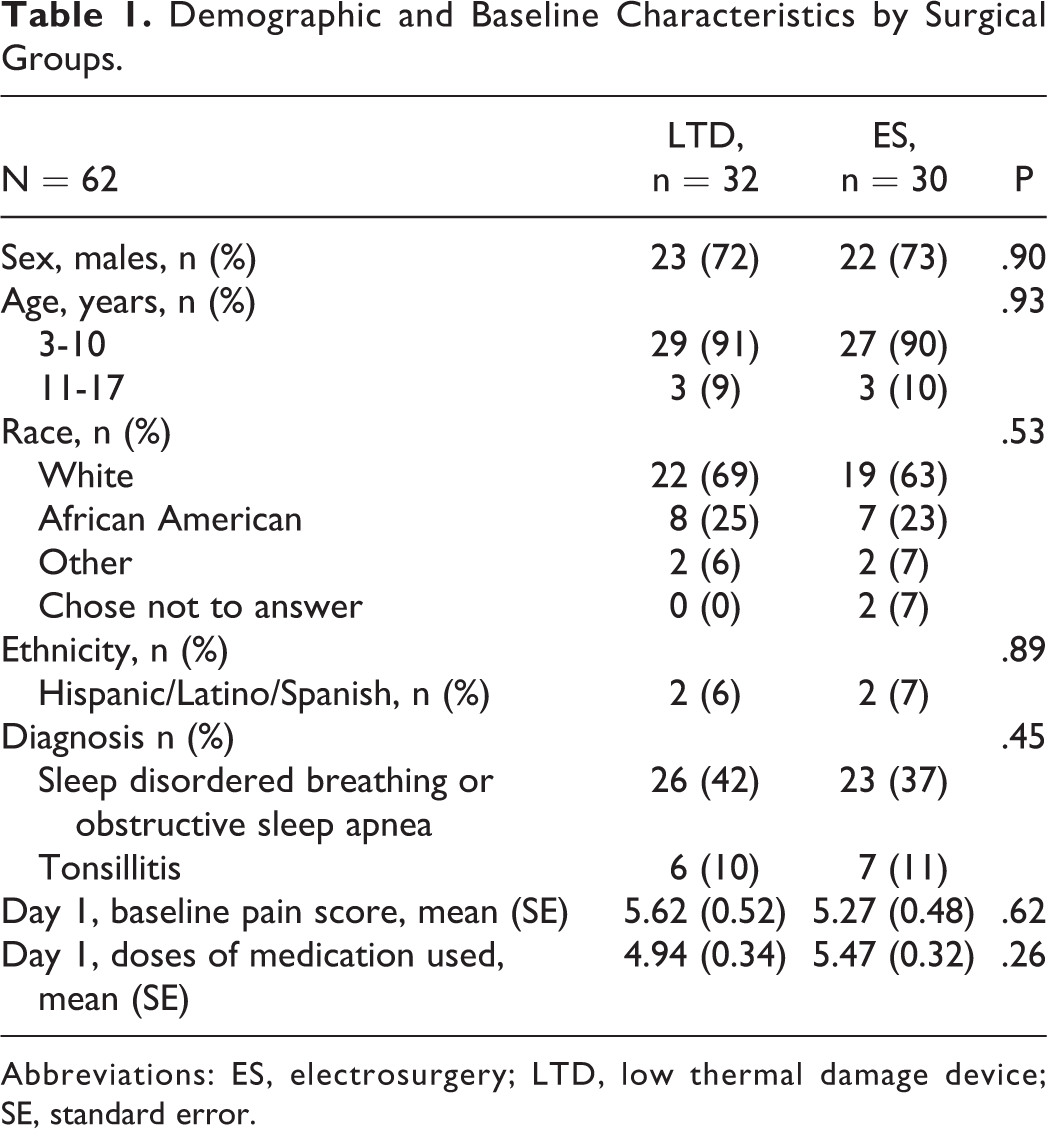
Abbreviations: ES, electrosurgery; LTD, low thermal damage device; SE, standard error.
Pain
The mean daily pain score over days was reduced by 0.52 (SE, 0.07), but there was no difference in the overall mean reduction between the 2 surgical methods (P = .48) even after adjustment for the daily uses of pain medication (P = .96). The median number of days (95% confidence interval [CI]) to having no pain was 7 days for the LTD group (95% CI, 5.2-8.6) and 9 days for the ES group (95% CI, 8.0-10.0; P = .67; Figure 1). There was a significant difference in the number of doses of pain medication used between the 2 surgical methods. The median number of pain medication doses in the LTD and ES groups was 21 (range: 2-124) and 36 (range: 7-49), respectively (P = .001; Figure 2). The mean number of doses (standard deviation) in the LTD and ES groups was 24.8 (22.0) and 33.3 (11.3), respectively (P = .001). One outlier (included in the analysis) in the LTD group used 124 doses of pain medication and was substantially greater than the maximum number (48 doses) reported for all other participants. There were significantly fewer doses of non-narcotic analgesics used on day 1 in both groups; on day 2, there were significantly fewer doses of non-narcotic use in the LTD group than narcotic use by either method, but on days 4, 6, 7, and 8, non-narcotic use was significantly greater in the ES group than narcotic use in ES or any drug use in the LTD group, and on days 9 and 10, non-narcotic use in the ES group was significantly greater than narcotic use in either group (Figure 3).
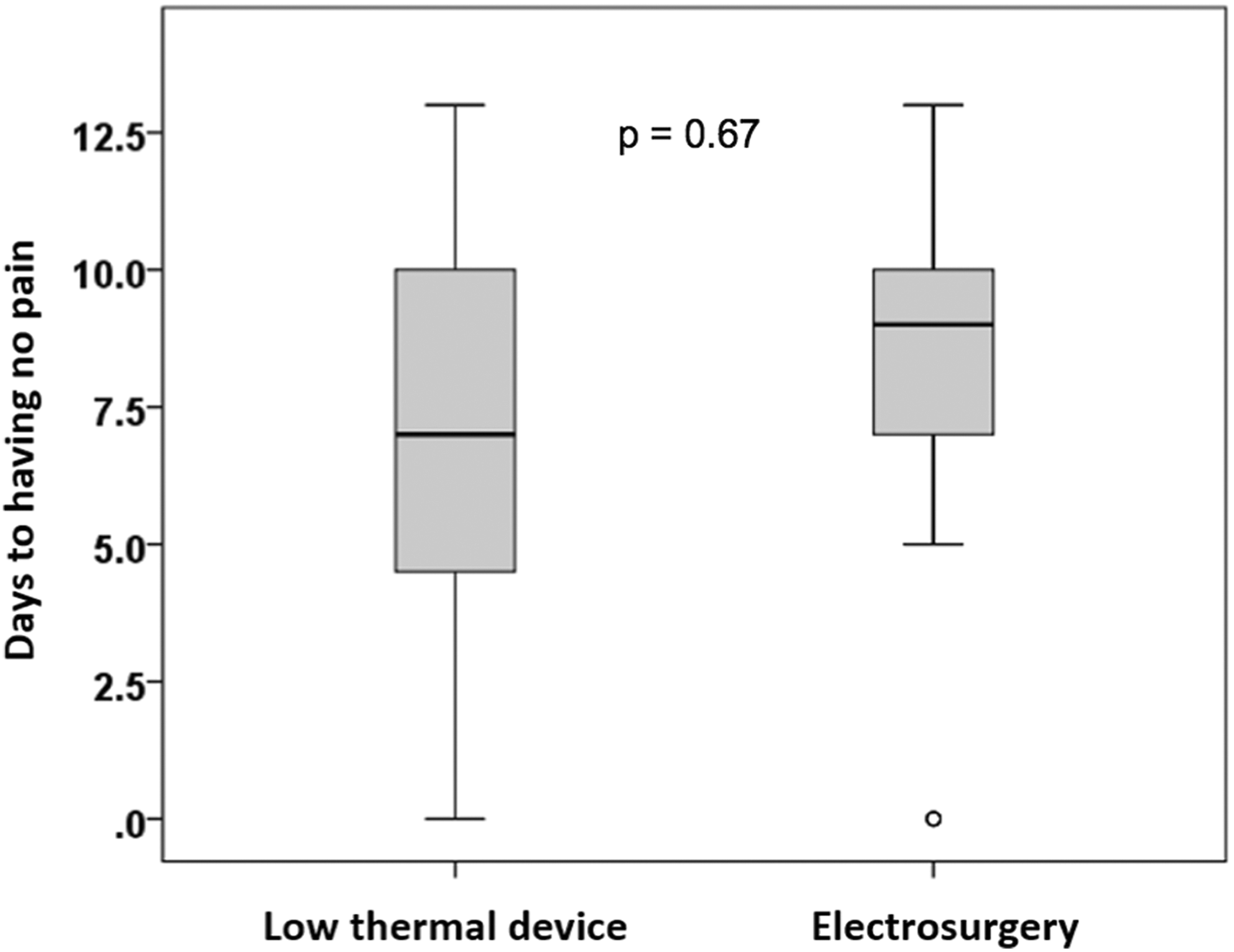
There was no difference in the median number of days to no pain during the 14-day postoperative period with the low thermal damage device versus traditional electrosurgery (P = .67).
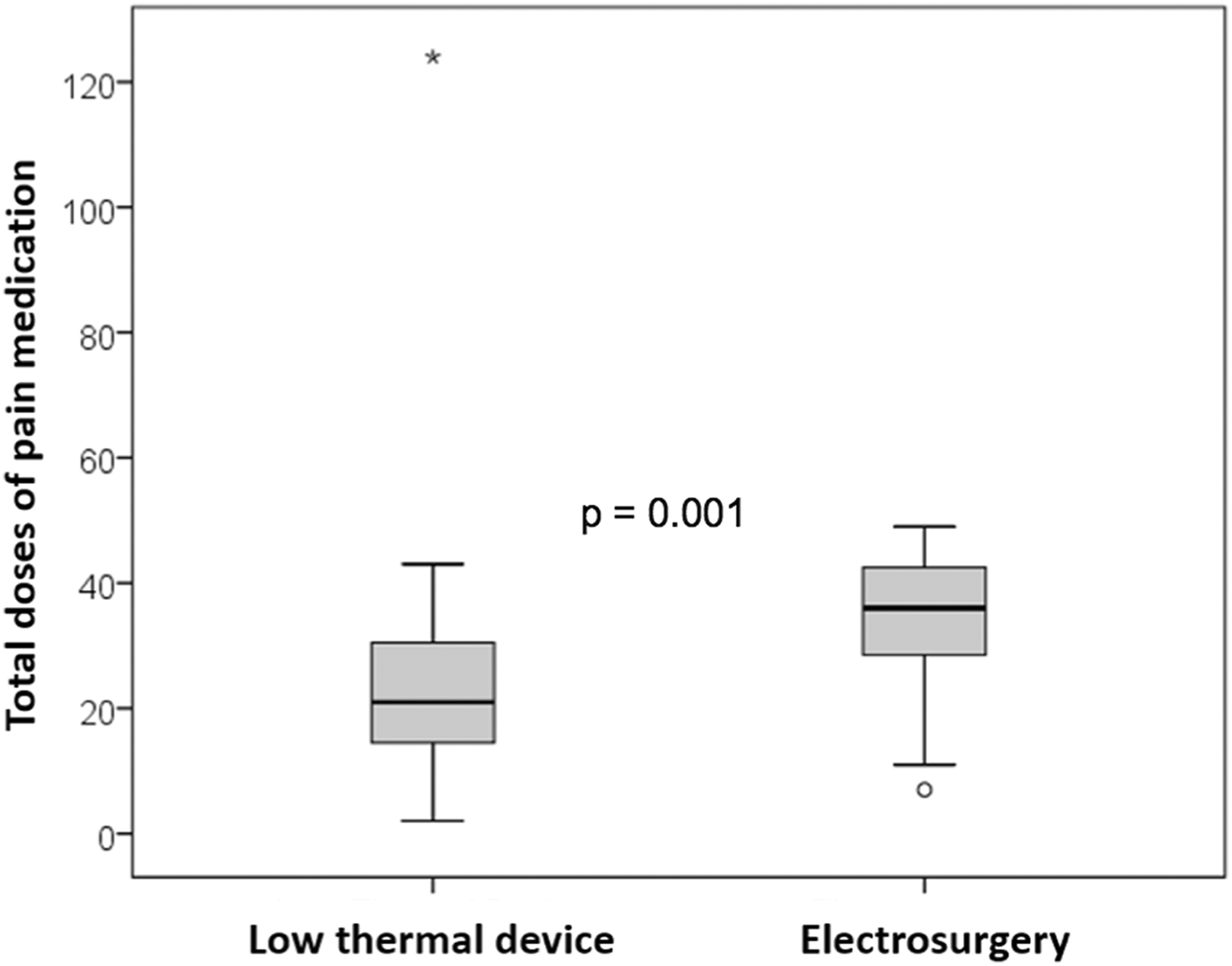
The median number of doses of analgesics used during the 14-day postoperative period with the low thermal damage device was significantly less than with traditional electrosurgery (P = .001). There was one outlier in the low thermal damage device group that used 124 doses of pain medication.
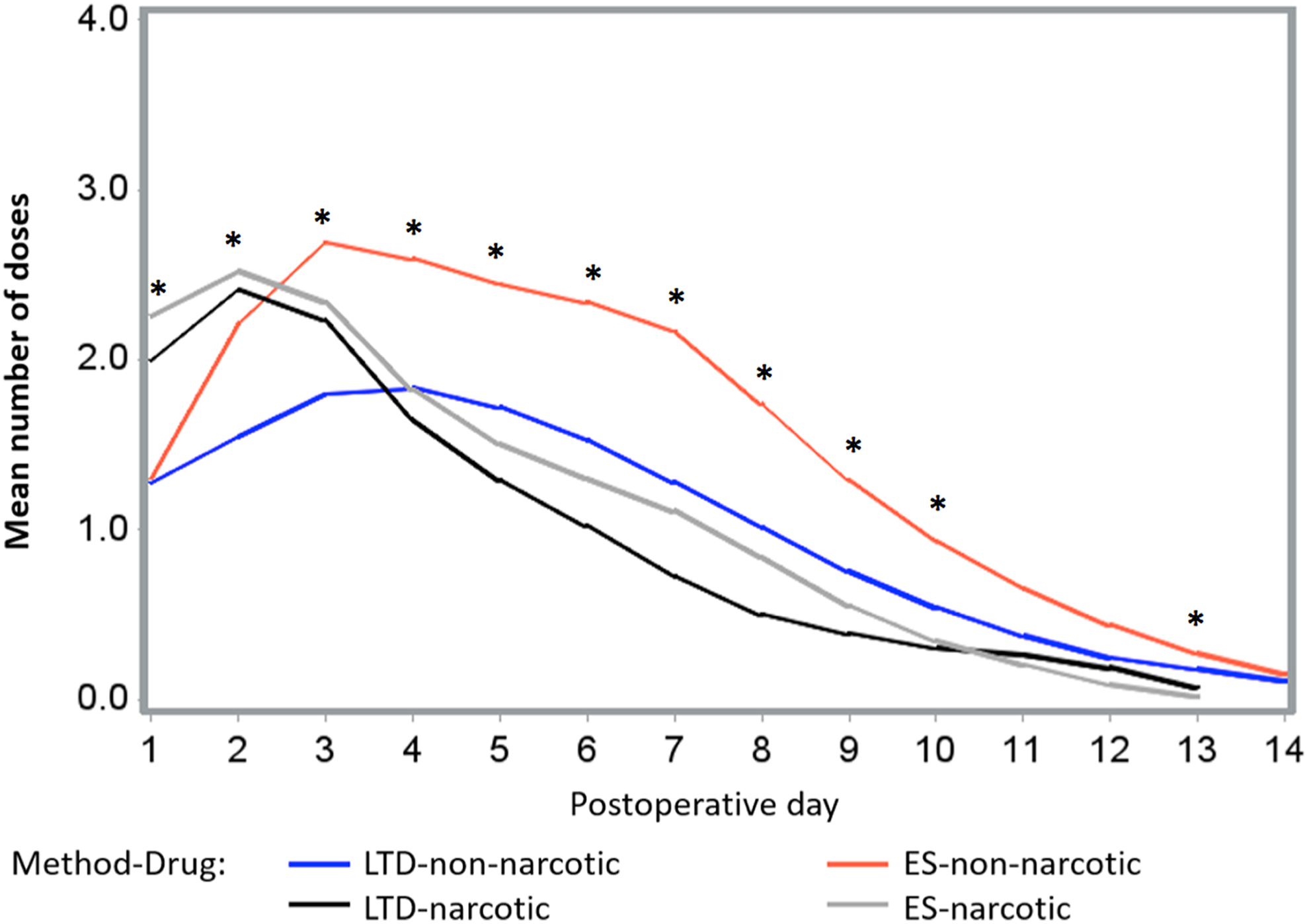
The mean number of doses of narcotic and non-narcotic analgesics used during the 14-day postoperative period with the low thermal damage device (LTD) and traditional electrosurgery (ES). On day 1, narcotic analgesic use in both groups was greater than non-narcotic use (P < .05). On day 2, narcotic use in both groups was greater than non-narcotic use in the LTD group (P < .05). On day 3, non-narcotic use in the ES group was greater compared to non-narcotic use in the LTD group (P < .05). On day 5, non-narcotic use in the ES group was greater than narcotic use in either group (P < .05). On days 4, 6, 7, and 8, there was greater non-narcotic use in the ES group compared with narcotic use and compared to any analgesic use in the LTD group (P < .05). On days 9 and 10, non-narcotic use was greater in the ES group compared to narcotic use in either group (P < .05). On day 13, non-narcotic use in the ES group was greater than narcotic use in the ES group (P < .05).
Bleeding
There was no difference in bleeding rates between devices. One child (10-year-old female) randomized to ES who had been eating well with minimal pain returned to the emergency department 5 days postoperatively because the mother noted a “large brown spot” to the left tonsil. There was no bleeding noted in the emergency department and the child was discharged. A short time later after eating dinner, oozing blood was noted and she returned to the emergency department and was hospitalized. No surgical intervention was necessary. No further bleeding occurred and the child was discharged the next day.
Adverse Events
Two other children (both randomized to the LTD) returned to the emergency department (5 and 2 days following surgery) due to fever and low fluid intake. No treatment was given in the emergency department and families were reminded to increase fluid intake.
Other Outcomes
There were no differences between groups for return to normal diet and activity, intraoperative blood loss, or the GCBI (data not shown).
Discussion
Tonsillectomy and adenoidectomy are associated with severe pain in children for up to a week and with moderate pain requiring analgesics for up to 14 days following surgery. 1,17 Thus, the 500 000 tonsillectomies performed per year in the United States equates to over 7 million days per year in which children experience postoperative pain following this procedure. 1,2 Several comparative studies of surgical instruments and variations in instrument settings have been studied to identify procedures that will reduce pain in children. 9,10,13,17 -21
The PEAK PlasmaBlade TnA tonsil and adenoid tissue dissection device was developed to reduce tissue damage by minimizing exposed electrode surface area, pulsing radiofrequency energy, and using plasma-mediated electroconduction combining cutting and hemostatic activity. 9 High-frequency pulses of radiofrequency energy (approximately 40 microseconds) with a burst rate of less than 1 kHz generates the formation of electrical plasma along the edge of a thin (nominally 12.5 μm), 99.5% insulated electrode 7 ; these features enable an operating temperature of 40°C to 100°C for the PlasmaBlade. 7 These lower temperatures reduce thermal collateral tissue damage and are expected to reduce pain experienced following surgery such as with tonsillectomies.
Traditional monopolar ES is the most common technology used for tonsillectomy. It emerged as a preference to “cold techniques” due to reported decreased intraoperative hemorrhage rates and ease of use. 9 Through application of radiofrequency energy, the natural resistance of tissues to the flow of current through the circuit increases the electrode temperature (250°C-350°C), generates heat, and creates a tissue cauterization effect. This effect may be modulated by changing power setting, electrode size and position (using the point, tip or flat of a blade), and time of tissue contact. 22 Unfortunately, this increased energy delivery has been correlated with increased pain and odynophagia. 23
As traditional ES is commonly used for adenotonsillectomy in our institution, we compared this technique to the LTD in children 3 to 17 years old who were undergoing adenotonsillectomy without any other concurrent surgical procedures including myringotomy.
Our primary outcome was difference in pain scores using a validated pain scoring instrument, the Wong-Baker FACES scale. 15 Parents were instructed to ask their child “to choose the face that best describes how they are feeling” for scoring the level of pain intensity each morning prior to any eating, drinking, or analgesic intake. We asked parents to keep detailed diary records of type, dose, and time of each analgesic administered. We found that the rate of pain decrease was not different between LTD and ES, with children taking a median of 7 to 9 days to experiencing no further pain. However, we found a significant interaction between rate of pain decrease and the number of analgesic doses. There were significantly more doses of pain medication used with ES (median 36 vs 21 with LTD, P = .001 [median difference of 15 fewer doses with LTD]), with non-narcotic use significantly greater with ES compared to LTD and narcotic use in both groups for several days postoperatively. This is a clinically important difference to parents and physicians as it could represent less risk of adverse effects and lower out-of-pocket expense for non-narcotic medication use with LTD.
Further, we found no difference in time to resuming normal diet or activity levels between participants randomized to LTD versus ES. Families were reminded of the target fluid intake based on their child’s weight during the follow-up phone calls. There were no differences in the 6-month follow-up between groups as assessed by the GCBI.
The primary limitation of this study is the small sample size, which may preclude generalizability. A larger prospective study is warranted to further evaluate diet and activity outcomes. Even so, this sample size has been used in several studies of similar design and outcomes. 13,17 -19 Another limitation was that we had to adjust the original inclusion criteria (children aged 3-6 years with an obstructive diagnosis) early on to include 7- to 17-year-old children and those with a diagnosis of recurrent tonsillitis. Older children and those with recurrent tonsillitis may experience greater pain than young children with obstruction. 24 -28 In this study, 22 children were older than 6 years (14 and 8 in the LTD and ES groups, respectively) and 13 children had recurrent tonsillitis (6 and 7 in the LTD and ES groups, respectively). Thus, it is unlikely that older age or diagnosis explains the greater pain medication use in the ES group. The one child with excessive pain medication use was a 15-year-old with a diagnosis of obstruction in the LTD group (Figure 2). The strengths are that the study was blinded to the parents and the study team, and the surgeons were committed to using the procedure and specified instrument settings to which the patient was randomized. The study coordinators were extensively trained in research procedures to ensure careful data collection. The frequent phone contact by the study team during the first week postoperatively aided in retention of participants.
Conclusion
Postoperative pain after adenotonsillectomy can lead to poor oral intake, dehydration, and readmission to the hospital, and analgesic use can expose children to potential adverse effects. Our study found that using the LTD for adenotonsillectomy in children aged 3 to 17 years results in significantly fewer doses of pain medication used compared to ES. Less analgesic usage is suggestive of decreased postoperative pain, provides cost savings, and offers assurance to physicians and families for limiting potential analgesic-related adverse effects.
Footnotes
Authors’ Note
Trial Registration: www.clinicaltrials.gov (NCT02533999). https://clinicaltrials.gov/ct2/show/NCT02533999?term=NCT02533999&rank=1. Kathryn Blake contributed to concept and design, interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content, obtaining funding, administrative, technical, and material support, supervision, and gave final approval of the version to be published. Jobayer Hossain contributed to concept and design, statistical analysis and interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content, and gave final approval of the version to be published. Bret Chafin contributed to concept and design, acquisition of data, critical revision of the manuscript for important intellectual content, technical support, and gave final approval of the version to be published. Angela Black contributed to acquisition of data, critical revision of the manuscript for important intellectual content, technical support, and gave final approval of the version to be published. Stefanie Schrum contributed to concept and design, acquisition of data, critical revision of the manuscript for important intellectual content, technical support, supervision, and gave final approval of the version to be published. Gary Josephson contributed to concept and design, acquisition of data, interpretation of data, critical revision of the manuscript for important intellectual content, obtaining funding, technical support, supervision, and final approval of the version to be published. All authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Dr Kathryn V. Blake and Dr Jobayer Hossain had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Dr Hossain (Nemours/Alfred I. duPont Hospital for Children) conducted and was responsible for the data analysis. Data from this study were presented as an oral presentation by Dr Josephson at the American Society of Pediatric Otolaryngology Spring Meeting, May 20-22, 2016, in Chicago, Illinois.
Acknowledgments
The authors gratefully acknowledge the contribution of the children and parents who volunteered for this research. In addition, the efforts of Deanna Seymour, RN, BSN, Jenny Batalla, BS, CCRP, Brooke Riber, RN, BSN, Stacey Gray, CCRP, Linda Wheeler, BA at Nemours Children’s Specialty Care, and the operating staff of Baptist Health Wolfson Children’s Hospital or the Wolfson Surgery Center in the conduct and data acquisition for this study are acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by an unrestricted research grant from Medtronic, plc. Medtronic supplied the funding for the study and the PEAK PlasmaBlade TnA tonsil and adenoid tissue dissection devices. Medtronic did not have input into the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. The authors were required to submit the manuscript to Medtronic, plc. for “review and approval [prior to submission to a journal] to protect its rights in patentable or copyrightable material and to check for technical correctness”.
