Abstract
We conducted a retrospective study of the long-term functional results of surgery for head and neck paragangliomas. Our study population was made up of 9 patients—4 men and 5 women, aged 22 to 59 years (mean: 46.6; median: 51)—who had undergone surgical excision of a head and neck paraganglioma from January 2002 through December 2006 in the ENT Department at Pugliese-Ciaccio Hospital in Catanzaro, Italy. Of the 9 paragangliomas, 4 were carotid body tumors, 2 were glomus tympanicum tumors, and 3 were glomus vagale tumors. None of the cases was bilateral or hereditary. Complete tumor resection was achieved in 8 patients; in the remaining patient, a small amount of intradural residual vagus nerve paraganglioma had to be left in situ. The internal carotid artery was preserved in all 4 resections of carotid body tumors. There was only 1 case of postoperative lower cranial nerve deficits, which occurred in a patient with a carotid body tumor. Follow-up ranged from 12 to 53 months (mean: 37.2; median: 36), and no recurrences were documented. Our small sample showed that surgical treatment of head and neck paragangliomas provided excellent tumor control with low postoperative morbidity, even in patients with large tumors. A wait-and-scan policy may be more appropriate for patients at an advanced age or who are otherwise at high surgical risk, as well as for those whose tumors have recurred following radiotherapy.
Introduction
Paragangliomas are uncommon hypervascular neuroendocrine tumors that develop in the extra-adrenal para-ganglion tissue; they represent 0.03% of all neoplasms. 1 Paragangliomas account for only 0.6% of all tumors of the head and neck region. 2 When they do occur in the head and neck, the most common locations are the carotid body at the bifurcation of the common carotid artery (carotid body tumor), the jugular bulb (glomus jugulare), the tympanic plexus (glomus tympanicum), and the vagus nerve ganglia (glomus vagale). Approximately 80% of these tumors are either carotid body tumors or glomus jugulare tumors. 3 Glomus vagale tumors are much less common (2.5 to 4.5%).4,5 Paragangliomas are the second most common neoplasm of the temporal bone, second only to acoustic schwannomas. 6 Glomus tympanicum tumors are the most common neoplasms of the middle ear. 7
Paragangliomas are classified on the basis of their location, innervation, and microscopic appearance. In the head and neck, these well-circumscribed masses usually exhibit innocuous clinical manifestations as nontender, slowly growing, soft-tissue lesions of the neck and skull base. Cranial nerve paralysis is a common symptom, particularly with lesions arising near the skull base. On magnetic resonance imaging (MRI), these tumors typically have a salt-and-pepper appearance with prominent flow voids. Intense enhancement is almost always noted following the administration of contrast material. For larger paragangliomas, preoperative angiography is required for surgical planning and often for preoperative embolization.
Most paragangliomas are benign. The prognosis is directly related to the location of the tumor. Patients with a carotid body paraganglioma have the best outcome, whereas those with skull base tumors have a less favorable prognosis because of the increased difficulty in achieving total resection. Although paragangliomas are usually benign, some cases of malignancy have been described. Rates of malignancy have been reported as 2 to 4% for jugulotympanic tumors, 6% for carotid body tumors, and 16 to 19% for vagal tumors. 8
Metastatic disease from cervical paragangliomas is usually confined to the neck, and it is said to be more common in cases of glomus vagale tumors than in cases of carotid body tumors.8–10 Most paragangliomas are solitary, but some patients develop multiple tumors. Multiple tumors are most often associated with carotid body tumors and least often with glomus tympanicum tumors. 11
It is now recognized that several types of these tumors are inherited through mutations of the genes that encode for the succinyl dehydrogenase enzymes SDHD and SDHB. Both genes are inherited in an autosomal dominant fashion, but the SDHD gene is subject to maternal imprinting while the SDHB gene has incomplete penetrance. Patients with the inherited gene almost invariably have multiple tumors. 12
The peak incidence of vagus nerve ganglia, jugular bulb, and tympanic plexus paragangliomas occurs in the fifth and sixth decades of life. Female-to-male ratios are 2.7:1 for glomus vagale tumors and 4:1 to 6:1 for glomus jugulare and glomus tympanicum tumors.13,14 Carotid body paragangliomas are also more common in women. 15
Three management options are recognized: surgical resection, radiation therapy, and a wait-and-scan policy. Of these, surgery is the only curative treatment. However, because surgery may be complicated by significant morbidity, especially in patients with larger tumors, it is considered by some to be controversial. Imaging—either with computed tomography (CT) alone, MRI alone, or the two in combination—is essential for determining the optimal surgical plan for jugular bulb and vagus nerve paragangliomas.
A preauricular infratemporal approach is recommended for lesions confined to the jugular fossa alone; those masses that also extend into the posterior fossa require a combined infratemporal and posterior fossa approach. 16 Preoperative embolization has been acclaimed by many investigators as a useful adjunct in the surgical management of paragangliomas.17–19
Table. Selected patient characteristics
Moret and colleagues proposed that the vascularization of paragangliomas be classified into two categories: multicompartmental and monocompartmental (or unicompartmental).20,21 In a multicompartmental tumor, each compartment is hemodynamically independent—that is, individual feeding vessels opacify only the compartments that are supplied by them. In contrast, one or more feeding vessels may supply a monocompartmental paraganglioma, and each artery will supply the entire mass. Most paragangliomas (83%) have a multicompartmental pattern of vascularity. 18
In this article, we describe our study of the long-term outcomes of surgery in patients with a head and neck paraganglioma. We have placed special emphasis on tumor control and deficits of the lower cranial nerves. We also review the literature.
Patients and methods
For this retrospective analysis, we reviewed the records of 9 adults who had been treated surgically for a head and neck paraganglioma in the ENT Department of Pugliese-Ciaccio Hospital in Catanzaro, Italy, from January 2002 through December 2006. Our patient population was made up of 4 men and 5 women, aged 22 to 59 years (mean: 46.6; median: 51). In addition to demographic data, we complied information on each patient's clinical presentation, diagnostic workup, surgical treatment, and outcome.
Four of these patients had a carotid body tumor, 2 had a glomus tympanicum tumor, and 3 had a glomus vagale tumor (table). All of these tumors were solitary. No patient had a positive family history of paraganglioma. All patients had undergone CT, MRI, and bilateral carotid angiography before surgery.
All tumors were subjected to histopathologic examination to confirm the diagnosis, and intraoperative frozen-section analysis was undertaken to look for negative margins.
Postoperatively, all patients were followed as outpatients for 12 to 53 months (mean: 37.2; median: 36). Follow-up included clinical examinations as well as serial MRIs and, in 1 case, angiography.
Results
Carotid body tumors
Three of the 4 patients presented with a large, nontender neck mass that was located just anterior to the sternocleidomastoid muscle. The other patient presented with dysphagia secondary to hypoglossal nerve compression. None of the patients had a cranial nerve deficit before surgery, and none had received preoperative radiotherapy.
The four carotid body tumors were categorized according to the Shamblin classification system. 22 There was 1 type I tumor, 2 type II tumors (figures 1 and 2), and 1 type III tumor. Preoperative embolization was undertaken in all 4 patients, and all the tumors were removed completely via a cervical approach. Only 1 patient required repair of the internal carotid artery (by end-to-end anastomosis). Ligation was not necessary in any patient. No patient was treated with postoperative radiotherapy. The histologic examination found no signs of malignancy in any of the tumors.
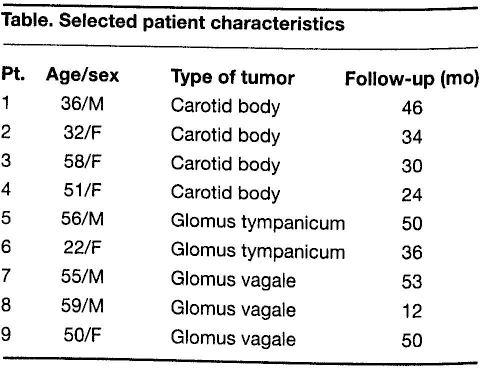
Patient 1: Images represent the preoperative angiogram (A) and CT (B) and the postoperative CT (C) of the 36-year-old man with a carotid body tumor.
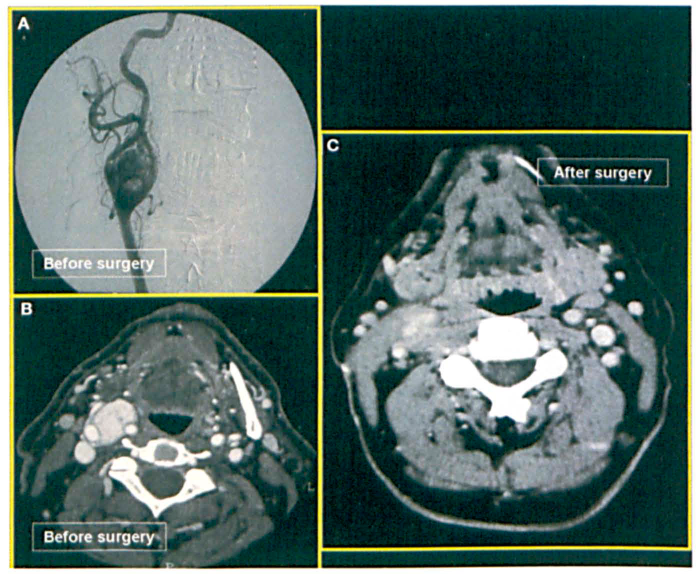
Patient 3: Images show the preoperative CT (A) and angiogram (B), the intraoperative view of the tumor (C), the excised tumor (D), the postoperative CT (E), and the surgical field following excision (F) in the 58-year-old woman with a carotid body tumor.
Postoperatively, 1 patient developed deficits of the glossopharyngeal, vagus, and hypoglossal nerves as a result of surgery. These deficits did not lead to aspiration. No recurrence was found over a follow-up of 24 to 46 months (mean: 33.5).
Glomus tympanicum tumors
Both patients with a glomus tympanicum tumor—a 56-year-old man (patient 5) and a 22-year-old woman (patient 6) presented with tinnitus and a reddish tumor in the area of the tympanic nexus; also, patient 6 complained of hypoacusis. The man had a class A1 tumor (figure 3), and the woman had a class C1 tumor (figure 4) according to the expanded Fisch classification system. 23
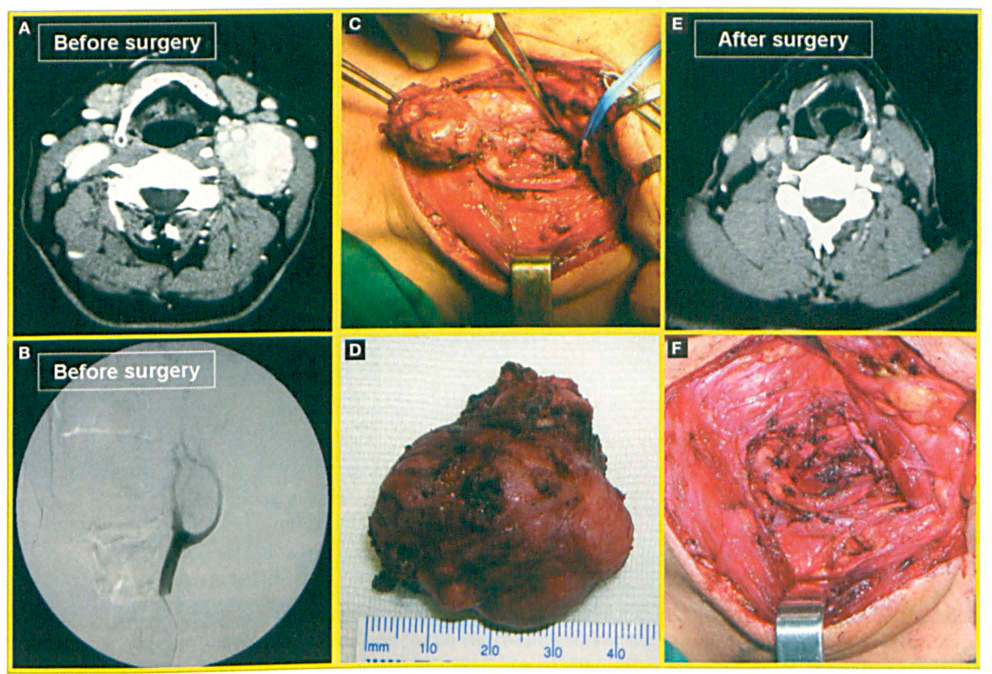
Patient 5: Images demonstrate the preoperative MRI (A) and CT (B), the surgical field prior to excision via a Fisch type A infratemporal approach (C), the preoperative angiogram (D), and the postoperative MRI (E) and CT (F) in the 56-year-old man with a glomus tympanicum paraganglioma.
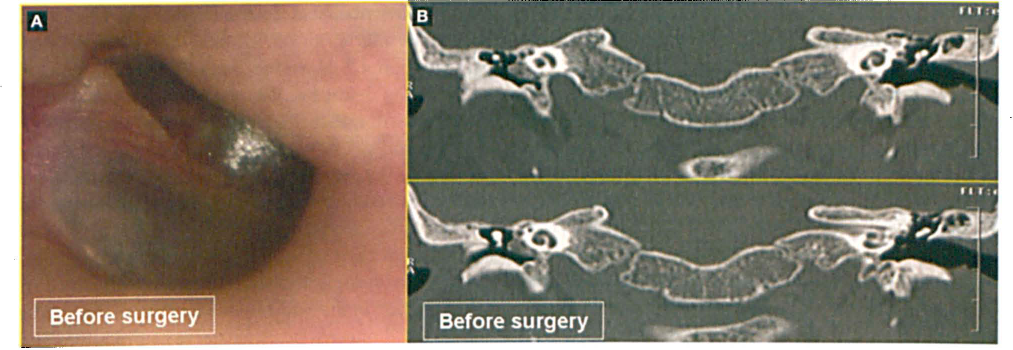
Patient 6: Images show the preoperative micro-otoscopic view (A) and CTs (B) in the 22-year-old woman with a glomus tympanicum tumor prior to enlarged antromastoidectomy.
Preoperative embolization was performed on both patients. The man underwent excision via a Fisch type A infratemporal approach without facial nerve transposition, and the woman was treated with an enlarged antromastoidectomy. According to the literature, an infratemporal approach without facial nerve transposition is an accepted method that yields excellent results, including long-term House-Brackmann 24 grade I or II facial nerve function in 72% of patients.25–27
Both tumors were removed completely. The histologic examination found no signs of malignancy in either. Patient 5 underwent a balloon occlusion test preoperatively and carotid angiography postoperatively. No postsurgical cranial nerve deficits or recurrences were seen over a follow-up of 50 and 36 months, respectively, for the man and woman.
Glomus vagale tumors
All 3 patients presented with a cervical tumor. Preoperatively, no patient exhibited any lower cranial nerve deficits. The volume of the 3 tumors ranged from 9.3 to 50 cm3 (mean: 21.2) (figure 5). The volume was calculated on the basis of MRI findings, as there is no generally accepted staging system for these tumors. Preoperative embolization was undertaken in all cases.
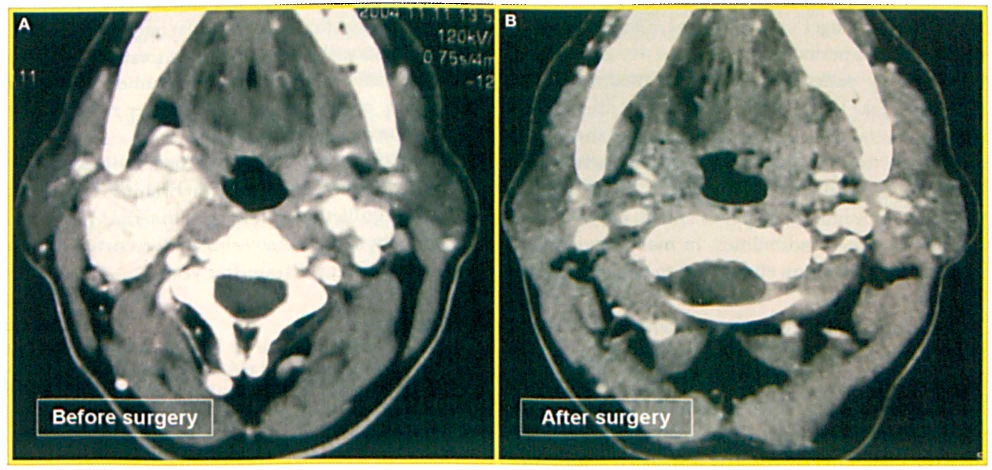
Patient 7: Images represent the pre- (A) and postoperative (B) CTs of the 55-year-old man with a glomus vagale tumor.
All 3 patients underwent excision via a transcervical approach. Complete removal was achieved in 2 patients, while a small amount of intradural residual vagus nerve paraganglioma had to be left in situ in the other. Histopathology was negative for malignancy in all cases. One patient (patient 7) complained of postoperative vertigo, and another (patient 9) experienced postoperative hemorrhage, which required a return to the surgical theater. No patient experienced vagus nerve palsy postoperatively. During follow-up periods of 12, 50, and 53 months (mean: 38.3), none of the 3 patients developed a recurrence.
Discussion
In our small series, the incidence of carotid body tumors was 44.4%, the incidence of glomus vagale tumors was 33.3%, and the incidence of glomus tympanicum tumors was 22.2%. Thus, glomus vagale tumors were over-represented in this small sample, and carotid body tumors were under-represented. Moreover, familial cases and cases of bilateral tumors and multiple tumors were not represented at all.
Differential diagnosis
Masses other than paragangliomas can arise in the carotid space of the suprahyoid and infrahyoid neck. The most common are nerve sheath tumors, nodal metastases, abscesses, and venous thrombi. Much rarer possibilities include lipomas, liposarcomas, and hibernomas. The most common possibilities in the differential diagnosis of a jugular foramen mass are nonneoplastic entities such as an asymmetrically enlarged jugular foramen, a high-riding jugular bulb, a dehiscent jugular bulb, and a jugular vein thrombus.
In addition to nerve sheath tumors and metastases, other neoplastic possibilities include meningiomas and miscellaneous primary bone lesions, such as multiple myeloma, lymphoma, and a mass representing Langerhans cell histiocytosis. The differential diagnostic considerations for a middle ear mass include both benign neoplasms (e.g., adenoma, endolymphatic sac tumor, choristoma, cholesteatoma, cholesterol granuloma) and malignant neoplasms (e.g., squamous cell carcinoma, adenocarcinoma, and sarcoma).
Carotid body paragangliomas often present as a slowly growing, nontender neck mass located just anterior to the sternocleidomastoid muscle at the level of the hyoid bone. Occasionally, the tumor mass may transmit the carotid pulse or demonstrate a bruit or thrill. Because of its location close to the carotid vessels and cranial nerves X through XII, tumor enlargement can cause progressive symptoms such as dysphagia, odynophagia, hoarseness, or cranial nerve deficits. Patients may relate a history of symptoms associated with catecholamine production, such as fluctuating hypertension, blushing, obstructive sleep apnea, and/or palpitations.28,29
Shamblin classification system
In 1971, Shamblin et al 22 introduced a classification system for carotid body paragangliomas based on tumor size:
Type I tumors are small and can be easily dissected away from the carotid vessels. One of our patients had a type I tumor.
Type II tumors are of medium size, and they adhere to or partially surround the vessels. In our 2 patients with a type II tumor, the mass had compressed the carotid vessels. These tumors were removed by careful subadventitial dissection.
Type III tumors are large, and they typically encase the carotid arteries. They require a partial or complete vessel resection and replacement. One of our patients had a type III tumor.
Fisch classification system
In 1982, Fisch 23 expanded his system for defining glomus tumors:
Class A tumors are characterized by limited localization within the tympanic cavity. Subclass A1 tumors are located around the promontory, and A2 lesions around the ossicular chain.
Class B tumors erode the wall of the hypotympanum but leave the cortical boundary of the jugular bulb intact.
Class C tumors are temporal glomus tumors in the strict sense because they are confined to the petrous temporal bone. Subclasses C1 through C4 designate the degree of extension to the carotid canal. C1 tumors are associated with a slight erosion of the posterior wall of the vertical canal segment; C2 tumors have completely eroded the vertical canal segment; C3 tumors also involve the horizontal segment; and C4 tumors extend to the cavernous sinus with involvement of the foramen lacerum.
Class D tumors are characterized by intracranial extension; the suffixes e and i are appended to denote tumors that are still purely extradural (De) and that already show intradural extension (Di).
Histologic characteristics
Histologically, the architecture of carotid body paragangliomas resembles the normal architecture of the carotid body. Pathologic examination reveals a characteristic biphenotypic cell line composed of chief cells and sustentacular cells with a peripheral fibrovascular stromal layer that are organized into a whorled (Zellballen) pattern. 30
Imaging studies
The cornerstone of paraganglioma assessment is fine-slice (1 mm) CT with a bone algorithm. MRI provides details about how soft tissue is affected by the tumor, and it may detect carotid bulb and jugular vein occlusion. Magnetic resonance angiography allows one to perform an anatomic study of paraganglioma irrigation. The role of Doppler ultrasonography is limited; it is useful for assessment and follow-up of carotid body tumors only.
Standard angiography can identify a tumor's vascular pedicle for embolization, which can reduce intraoperative bleeding. Embolization is usually performed 1 or 2 days before surgery. If a patient has to wait too long for surgery, inflammatory phenomena or even tumor revascularization can occur.
All imaging studies should be obtained bilaterally to rule out the existence of multicentric tumors.
Surgery
Surgical treatment has been the standard approach to cervical paragangliomas for many years. Resection of a carotid body paraganglioma carries the inherent risks of injury to the cranial nerves and carotid arteries, as well excessive blood loss. Cure rates of 89 to 100% have been reported.1,31
Advances in preoperative and operative techniques together with better postoperative care have reduced complication rates. Preoperative cranial nerve deficits have been reported in as many as 30% of patients with a glomus vagale tumor and 20% of those with a carotid body tumor.1,32
In our series, 1 of the 4 patients with a carotid body tumor experienced postsurgical lower cranial nerve deficits. The reported incidence of cranial nerve palsy as a complication of carotid body tumor surgery ranges from 10 to 40%.33–35 In one of these series, postoperative cranial nerve palsies developed in 11.1 % of patients with a solitary carotid body tumor and in 100% of patients with multiple tumors. 33 The latter group included patients with glomus vagale and jugulotympanic tumors, the resection of which is known to be associated with higher morbidity. 35 There was no correlation between tumor size and the incidence of cranial nerve palsies.
As would be expected, the rate of neural complications is higher in patients with Shamblin type II and III tumors. None of the 3 patients in our study with a glomus vagale tumor experienced postoperative vagus nerve palsy.
The rate of major vascular complications has dropped from 30% in the 1960s to less than 1%, according to a 2004 report by Köhler et al. 35 No major vascular complications were observed in any of our patients, except for 1 of the 3 patients with a glomus vagale tumor (patient 9), who experienced a hemorrhage that required a return to the surgical theater.
The risks of infratemporal and lateral skull base surgical approaches are significant. Among them are morbidity associated with angiography and embolization, the possibility of opening the jugular bulb, the possible need for tracheotomy, an increase in blood loss, the risk of cranial neuropathies, longer operative time, and a prolonged hospitalization period. 36
For elderly patients in particular, treatment decisions must take into account concomitant diseases, the many potential morbidities of surgery, and the impact of these factors on the patient's postoperative quality of life, especially for those patients with a glomus tympanicum tumor. This applies to both operative and disease-associated morbidities. In many reported series, patients older than 60 years often did not undergo surgical resection.
Our local control rate after surgery was 100%, as there were no recurrences over a mean follow-up of 37.2 months.
Radiotherapy
The main arguments in favor of radiotherapy as a first-line treatment are that (1) it is less invasive than surgery, (2) it is associated with fewer complications, and (3) it yields high rates of local control.37–39 Hinerman et al reported tumor control rates of 96 to 100% after 45 Gy of radiation therapy for cervical paraganglioma. 38 However, radiotherapeutic control is difficult to define because it is based on cessation of tumor growth rather than tumor disappearance.
Histologic examination has shown that chief cells are minimally affected by irradiation, although the distinctive vascular structure of the tumor is replaced by fibrous connective tissue. 14 This would seem to be a concern in patients with a potentially malignant tumor. 40 Other complications of radiation therapy include inflammation of the external auditory canal and middle ear, osteoradionecrosis, cranial nerve neuropathies, and direct injury to brain tissue. Furthermore, it must be remembered that if radiation therapy fails, surgery will be more difficult.33–35
In conclusion, the management of head and neck paragangliomas remains a challenge. Treatment must be individualized, with clinicians taking into account the patients age, tumor site and size, multicentricity, and preexisting cranial nerve deficits. The likelihood of tumor control is high regardless of whether treatment is by surgery or radiotherapy. However, there is no consensus regarding the choice between the two.
In general, patients with a solitary lesion that is easily removable with acceptable morbidity are better treated surgically. It may be reasonable in selected cases to choose a conservative approach and to postpone surgery until cranial nerve impairment becomes evident or other vital structures are threatened, because cranial nerve function can be preserved until late and because surgery of paragangliomas often leads to similar nerve impairment.
For patients with multiple tumors, treatment options are influenced by the location of the tumors, the age of the patient, comorbidities, and whether radiotherapy has been previously administered.
A policy of watchful waiting with interval imaging may be prudent in some cases. Most important, every patient must undergo lifetime follow-up with serial MRIs—and angiography, if necessary—because recurrences can develop after many disease-free years.
