Abstract
Age-related macular degeneration (AMD) is a chronic degenerative retinal disorder that leads to gradual central vision loss. Approximately 0.37% of older adults in the global population experience permanent visual impairment, including blindness, due to late-stage AMD (Jong, 2006; Wong et al., 2014). Concerningly, the prevalence of late-stage AMD is predicted to surge by 86% between 2014 and 2040, reaching a staggering 9.92 million affected individuals worldwide, compared to 4.59 million in 2014 (Wong et al., 2014).
AMD is linked to sleep disruptions, depression, anxiety, and worse quality of life, all indicating significant psychosocial impairment (Cimarolli et al., 2015; Hassell et al., 2006; Khurana et al., 2016; Maynard et al., 2017; Mitchell & Bradley, 2006). Sleep disturbances may manifest as trouble falling asleep or staying asleep and the experience of daytime drowsiness (Roth & Roehrs, 2003). Sleep disturbances are common, with estimated rates ranging from 8% to 22% (Cappuccio et al., 2010; Ohayon, 2002). Roughly 50% of adults over 65 years of age experience insomnia symptoms, reflecting the increased sleep difficulties that are common with advanced age (Cappuccio et al., 2010). Sleep disturbances can affect both health and overall quality of life, resulting in diminished physical well-being and cognitive abilities (Chattu et al., 2018; Lee et al., 2009; Worley, 2018). Both excess and insufficient sleep are linked to adverse health outcomes such as hypertension and cardiovascular disease, which are also associated with increased AMD risk (Cappuccio et al., 2011; Chakravarthy et al., 2010; Hyman et al., 2000). Evidentially, sleep disturbances impose
by answering questions on this article. For more information, visit:☑ Earn CEs Online
Several mechanisms might explain the relationship between AMD and sleep dysfunction. Visual impairment from AMD can disrupt the sleep cycle (Adeoti & Akang, 2010). The retina is instrumental in sleep regulation via photoentrainment. Intrinsically photosensitive retinal ganglion cells (ipRGCs) depend on light perception to regulate the suprachiasmatic nucleus, our body's internal clock, which is responsible for governing the sleep–wake cycle (Maynard et al., 2017). Sleep disturbances in individuals who have AMD could also stem from disrupted melatonin production. Melatonin is mainly produced by the pineal gland and is key to the regulation of the sleep–wake cycle (Hardeland et al., 2006). Nocturnal serum melatonin levels decrease with advancing age and are associated with age-related diseases like AMD (Bubenik & Konturek, 2011; Chattoraj et al., 2009; Iguchi et al., 1982). Reduction in melatonin levels in AMD patients is concerning, since melatonin protects against oxidative stress, a significant risk factor implicated in the development and progression of AMD (AREDS, 2000; Chakravarthy et al., 2010). The link between AMD and sleep dysfunction involves a complex and bidirectional interplay, where each condition can potentially influence and exacerbate the other. The relationship between AMD and sleep is not fully understood, since existing research suggests poor sleep quality is linked to AMD, while other studies indicate no significant difference in sleep quality between patients with and without AMD. Despite the significance of this relationship, no previous meta-analysis has investigated the effects of AMD on sleep quality. Therefore, we aim to explore the relationship between AMD and sleep quality to enhance the management of AMD and improve patients’ quality of life.
Methods
Search Strategy
This systematic review and meta-analysis adhered to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines (see Appendix A). We conducted comprehensive searches of the CINAHL, EMBASE, and MEDLINE databases up to August 1, 2023, as outlined in Appendix B. In addition, we manually searched for relevant poster presentations or abstracts from conferences organized by the American Academy of Ophthalmology, Canadian Ophthalmological Society, and the Association for Research in Vision and Ophthalmology. Grey literature was also reviewed, including sources from ProQuest Dissertations and Theses Global and ClinicalTrials.gov. Detailed information on the gray literature searches can be found in Appendix B.
Inclusion Criteria
Studies were included if they investigated the effects of AMD in sleep of adults 18 years and older. Although AMD is primarily a disease that is associated with aging, it may occur in individuals as young as 30 years of age. An inclusion criterion of patients above 18 years of age ensures all studies documenting these cases are included in our analysis. Despite the poorer prognosis associated with wet AMD compared to dry AMD, no distinction was made due to the limited studies available. The effects of AMD on sleep were assessed by looking at measures including, but not limited to, sleep time, sleep quality, insomnia, and the Pittsburgh Sleep Quality Index (PSQI) scores. Insomnia was diagnosed either by clinical diagnosis by a physician, self-reported measures, or standardized questionnaires. Included in the review were case series, clinical trials, cohort studies, comparative studies, economic studies, observational studies, and randomized controlled trials.
Exclusion Criteria
Case reports, case series, letters to editors, commentaries, review articles, systematic reviews, and meta-analyses were excluded. If sufficient study details and data were provided, then conference abstracts were included. Studies that were not published in English were omitted to prevent potential inconsistencies that could arise during translation. This study did not impose any restrictions on geographical location or publication year.
Study Selection
We uploaded the collected literature into Covidence, a systematic review tool, where an automated duplicate check was conducted, followed by a manual verification by one of the reviewers. The screening process was conducted in three stages. Initially, title and abstract screening (levels 1 and 2) included all studies addressing the effects of AMD on sleep. The full-text screening (level 3) focused on studies that examined at least 20 adult eyes and reported outcomes related to sleep duration, quality, insomnia, and PSQI scores. Two reviewers independently assessed the studies at each stage, resolving any discrepancies through discussion. If an agreement could not be reached, a third reviewer was consulted. The Cohen's kappa (κ) coefficient was calculated for each screening stage to ensure reliability. Figure 1, presented as the PRISMA flowchart, depicts the number of studies at each screening stage.
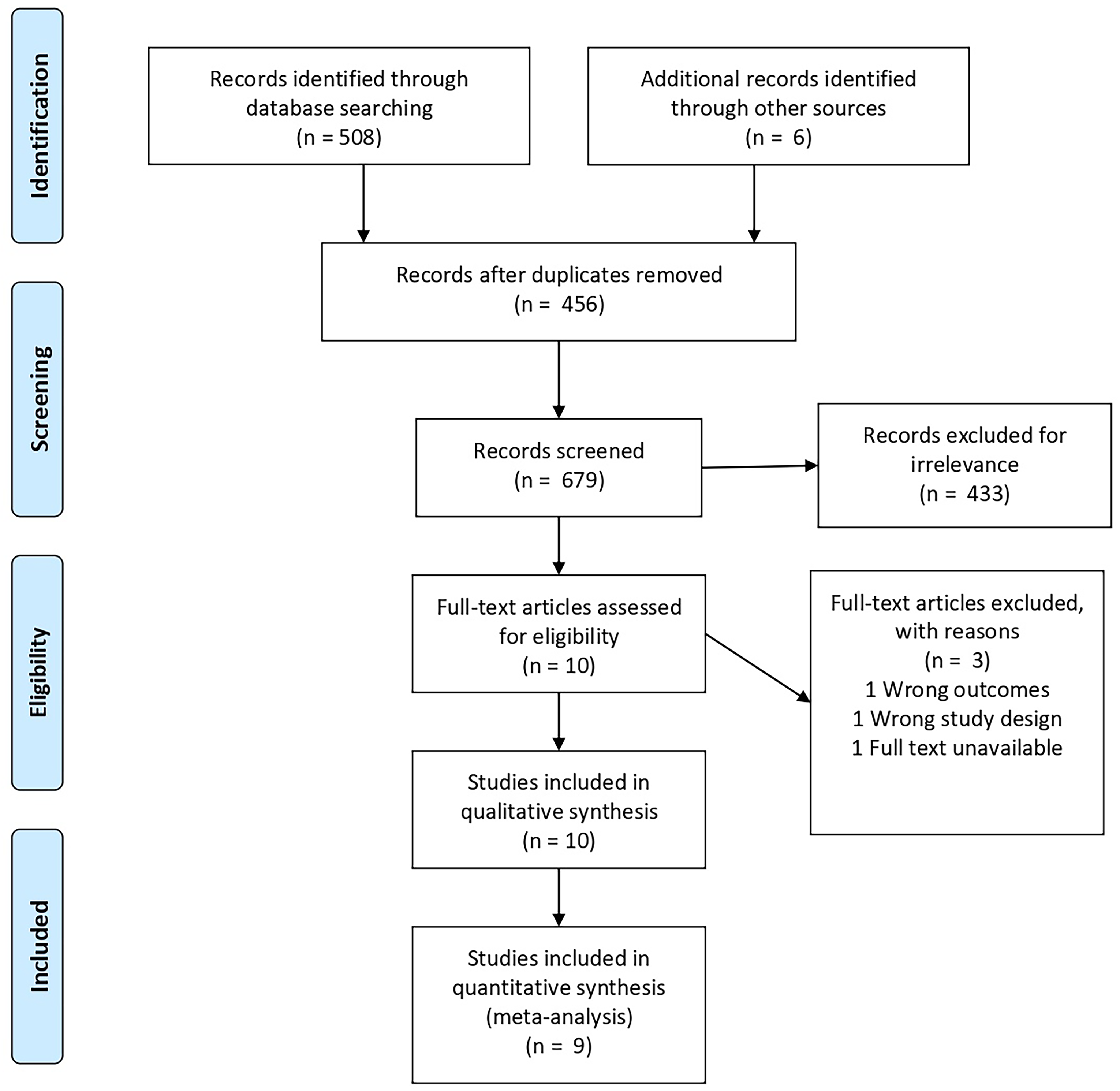
Prisma Flowchart. Note: The flowchart is for the systematic review outlining the number of database searches, the number of abstracts screened, and full-text articles reviewed for study eligibility, and reasons for exclusion. From: Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G., & the PRISMA Group. (2009). Preferred reporting items for systematic reviews and meta analyses: the PRISMA statement. PLoS Med, 6(6), Article e1000097. https://doi.org/10.1371/journal.pmed.1000097.
Risk of Bias Assessment
A modified Downs and Black Checklist (Downs & Black, 1998) was utilized for assessing the risk of bias. To ensure methodological integrity, each article included in this systematic review and meta-analysis underwent a rigorous quality evaluation.
Data Collection Process
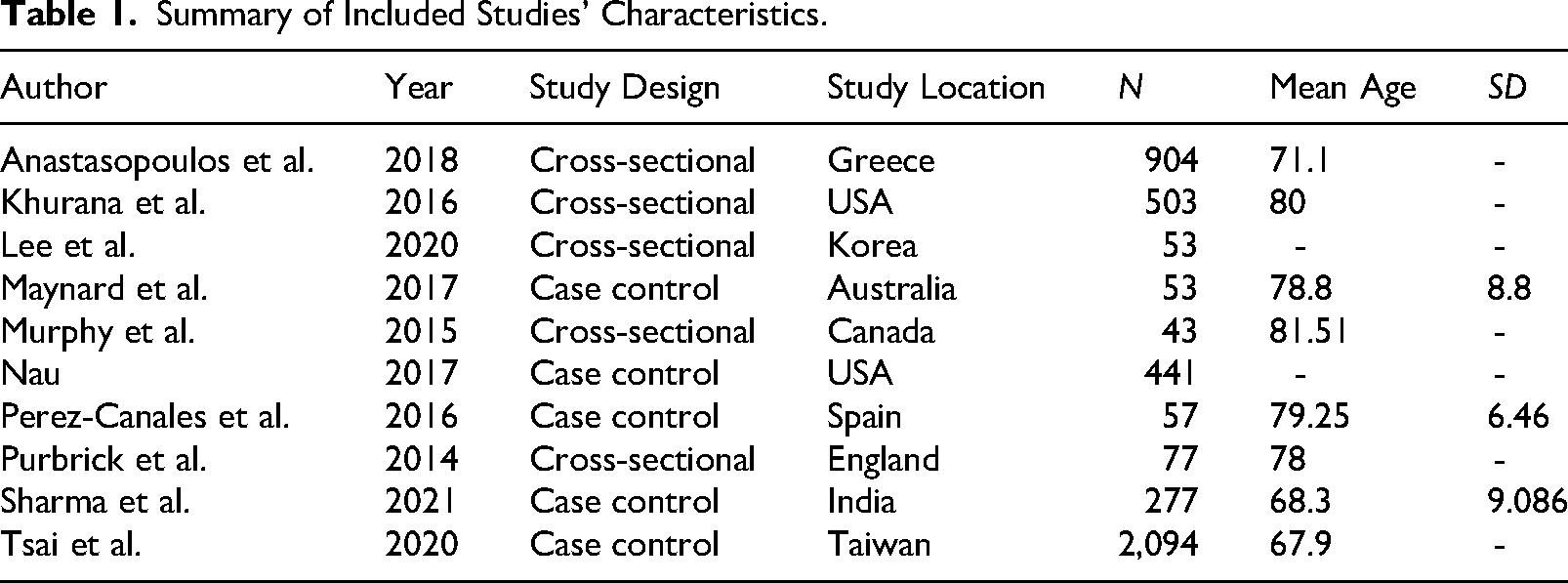
Table 1 displays the study characteristics of the 10 studies extracted. This included author, study design, year of publication, study location, sample size, sleep duration, prevalence of insomnia, and PSQI scores. Studies were categorized based on their risk of bias assessment scores: those scoring 15 or below were classified as low quality, scores from 16 to 19 were considered medium quality, and scores over 20 were deemed high quality. Given the limited evidence available, no low-quality studies were excluded from the analysis. For a detailed explanation of the risk of bias assessment, please see Appendix C.
Summary of Included Studies’ Characteristics.
Statistical Analysis
We used STATA 15.0 software (STATA Corporation, College Station, TX) to perform the meta-analysis. The proportion metric was employed to evaluate the effect size or treatment outcome. To assess the degree of heterogeneity among studies, we calculated the I2 value, which signifies the variance attributable to heterogeneity rather than random chance. The Chi-squared test was used to determine whether the observed variances among studies were likely random. Significant heterogeneity was indicated by a low p-value in conjunction with a high Chi-squared statistic relative to its degrees of freedom. Depending on the level of heterogeneity, we chose either fixed-effect or random-effect models. Forest plots and funnel plots were utilized to visually display the results and to identify any potential publication bias.
Results
Search Results
We identified 626 articles from online databases and grey literature. Among these, 170 duplicates were removed. Following a review of titles and abstracts, we excluded 434 papers due to irrelevance. This left 22 articles for an in-depth full-text review, of which 10 were chosen for data analysis. One study was not incorporated into the meta-analysis due to noncomparable data. Therefore, the meta-analysis included nine studies with a total of 4,501 subjects. Articles were excluded if they were deemed irrelevant or did not pass the screening criteria. The Cohen's kappa (κ) values were 0.79 for title and abstract screening and 0.38 for full-text review.
Study Characteristics
Table 1 provides a breakdown of the demographic characteristics of the 10 studies that qualified for our review, including the one study that was disqualified from the meta-analysis. Five of these studies were case-control studies, while the remaining five were cross-sectional studies. The research was conducted in various global locations, such as Australia, Canada, India, Korea, Spain, and the United States. Study populations varied in size, from 43 to 2,094. Although not every study disclosed a mean age for participants, the majority focused on older populations, with average ages spanning from 67.9 to 81.51 years.
Impact of AMD on Sleep Quality Using PSQI Scores
PSQI serves as a self-assessment tool for evaluating sleep quality and disruptions (Buysse et al., 1989). Comprising 19 questions, this instrument examines seven sleep facets: perceived sleep quality, time taken to fall asleep, total sleep time, regular sleep efficiency, sleep interruptions, reliance on sleep medications, and daytime challenges. These individual component scores are aggregated to produce a global score, which can vary between 0 and 21. A higher score signifies poorer sleep quality. A PSQI score greater than 5 is generally considered to indicate poor sleep quality and suggests that the individual has significant sleep disturbances or difficulty sleeping.
Figure 2 demonstrates heterogeneity in the studies analyzing the effect of AMD on PSQI scores: >5 (I2 = 69.8%, p = .445). Studies reported a significantly higher prevalence of PSQI scores among patients with AMD: >5 (ES 0.52, 95%; CI: [0.38, 0.65]).
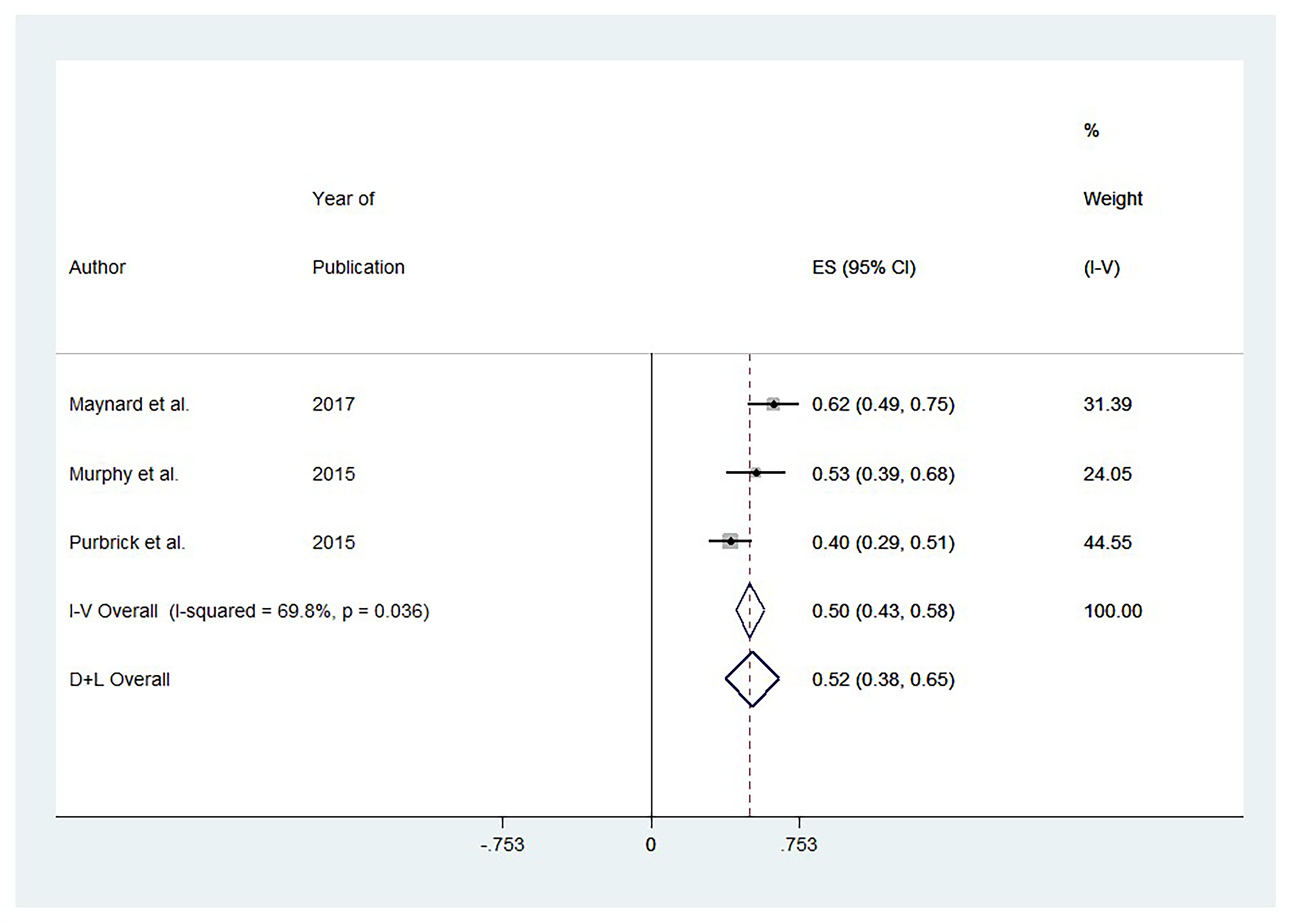
Forest Plot of Pooled Effect Size (ES) for Pittsburgh Sleep Quality Index (PSQI). Note: Score comparing age-related macular degeneration (AMD) patients versus controls. Note: Positive ES indicates a higher (worse) PSQI score in AMD.
Impact of AMD on Insomnia
Figure 3 reveals heterogeneity among the studies evaluating the impact of AMD on rates of insomnia (I2 = 79.7%, p = .026). Studies reported a significant impact on insomnia among patients with AMD: ES 0.02, 95%; CI: (0.01, 0.03).
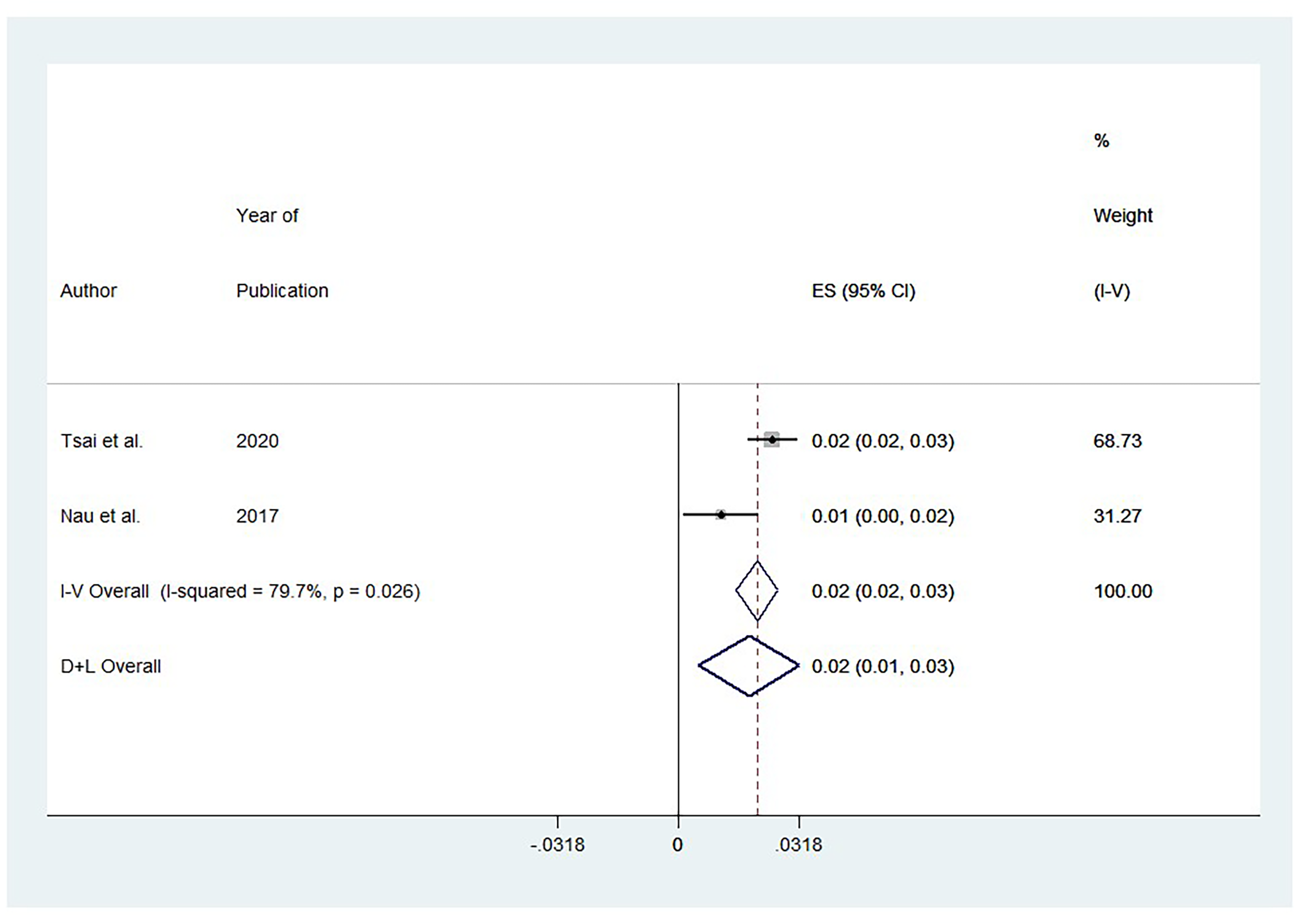
Forest Plot of Pooled Effect Size (ES) for Insomnia Comparing AMD Patients Versus Controls. Note: Positive ES indicates greater insomnia burden in AMD.
Impact of AMD on Sleep Time
Figure 4 shows heterogeneity among studies investigating the effects of AMD on sleep time (I2 = 92.0%, p = .000). Studies reported a nonsignificant impact on sleep time among patients with AMD: ES 0.04, 95%; CI: −0.17, 0.25).

Forest Plot of Pooled Effect Size (ES) for Sleep Duration Comparing AMD Patients Versus Controls. Note: Positive ES indicates longer sleep duration in AMD.
Impact of AMD on Odds Ratio for Sleep Time Over 8 Hours
Figure 5 indicates there was no significant heterogeneity between studies examining the impact on odds ratio for sleep time over 8 hr (I2 = 0.0%, p = .445). Studies reported a nonsignificant odds ratio for sleep time over 8 hr among patients with AMD: ES 0.99, 95%; CI: (0.64, 1.52).
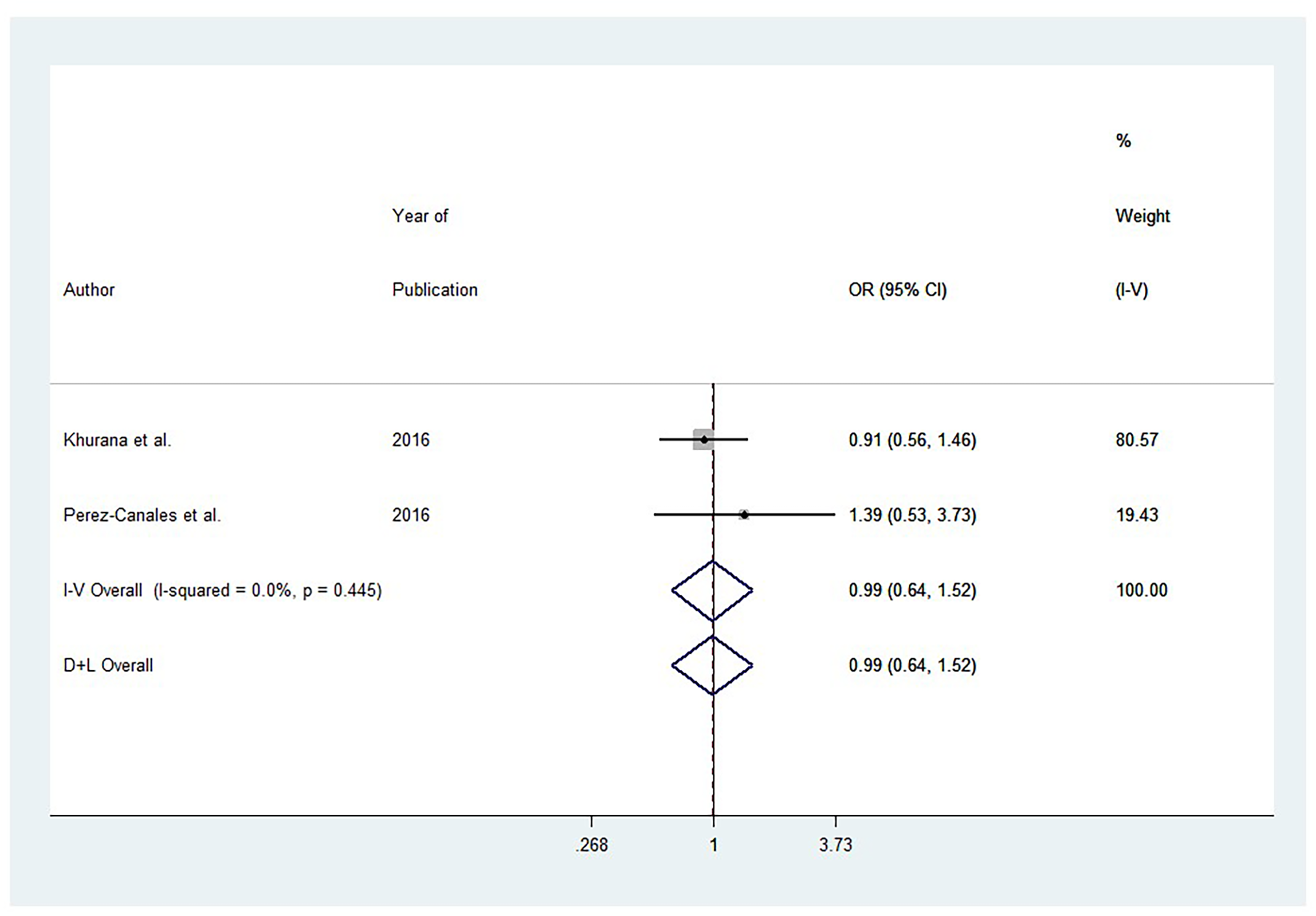
Forest Plot of Pooled Odds Ratio (OR) for Long Sleep Duration (>8 hr) Comparing AMD Patients Versus Controls. Note: OR > 1 indicates higher odds of sleeping >8 hr in AMD.
Assessment of Study Quality and Publication Bias
The majority of the studies evaluated using the Modified Downs and Black Checklist (Downs & Black, 1998) achieved scores of 15 or higher, indicating they were “good” quality (n = 9). Only one study scored between 11 and 14 (n = 1), categorizing it as of poor quality. Articles of lower quality were not excluded from our analysis due to limited evidence. Detailed information on the risk of bias assessment is provided in Appendix C.
Publication Bias
Figures 6 through 9 illustrate funnel plots for PSQI scores above 5 (Figure 6), insomnia prevalence among AMD patients (Figure 7), the average sleep duration (Figure 8), and the odds ratio for sleep exceeding 8 hr (Figure 9). The plots for these figures show studies spread from the top left to the bottom right. A visual inspection of the funnel plots did not reveal any noticeable asymmetry. The few studies at the bottom suggest that smaller studies with nonsignificant findings might be unpublished.
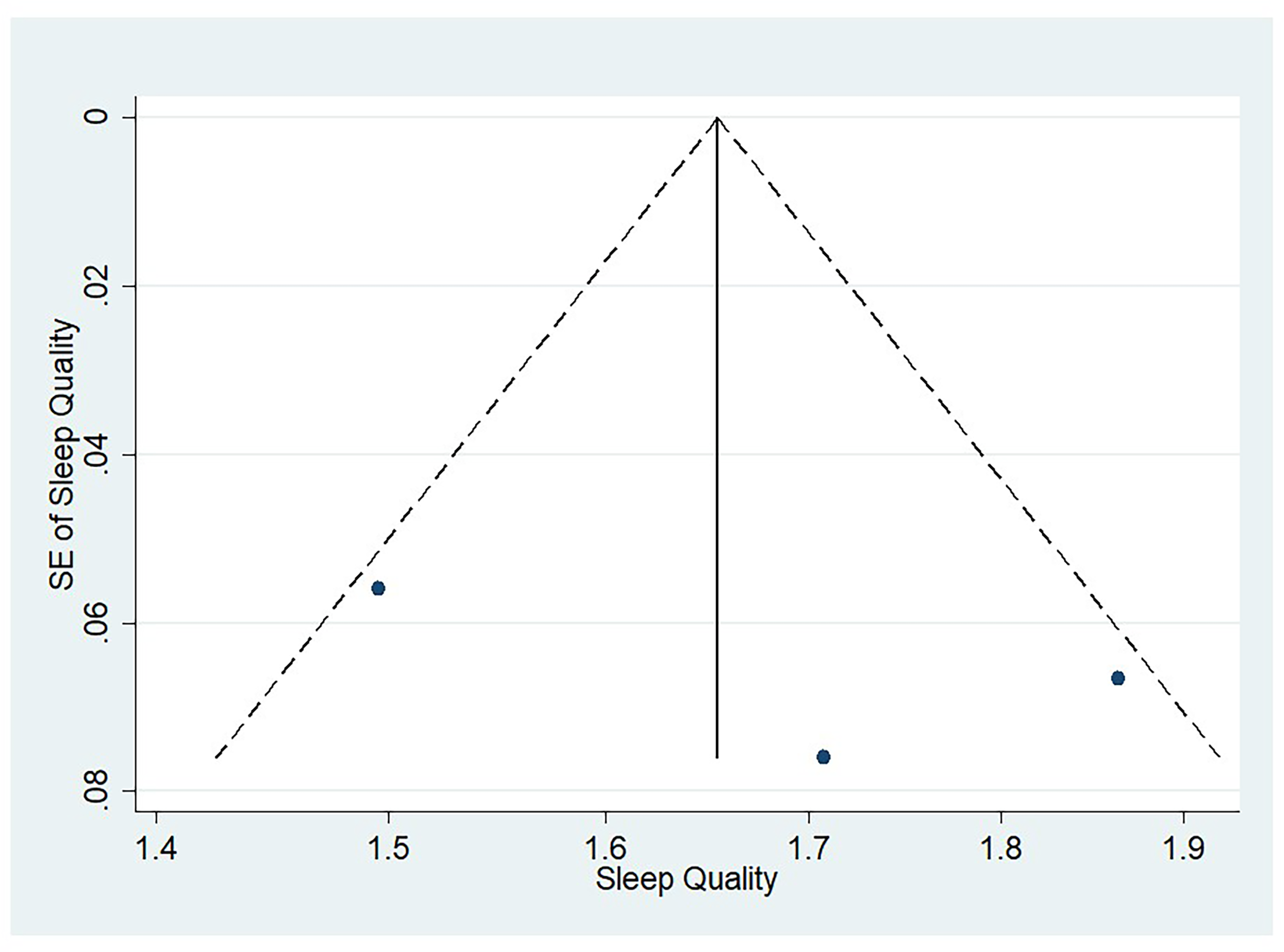
Funnel Plot for Studies Evaluating Pittsburgh Sleep Quality Index (PSQI) Score in AMD.
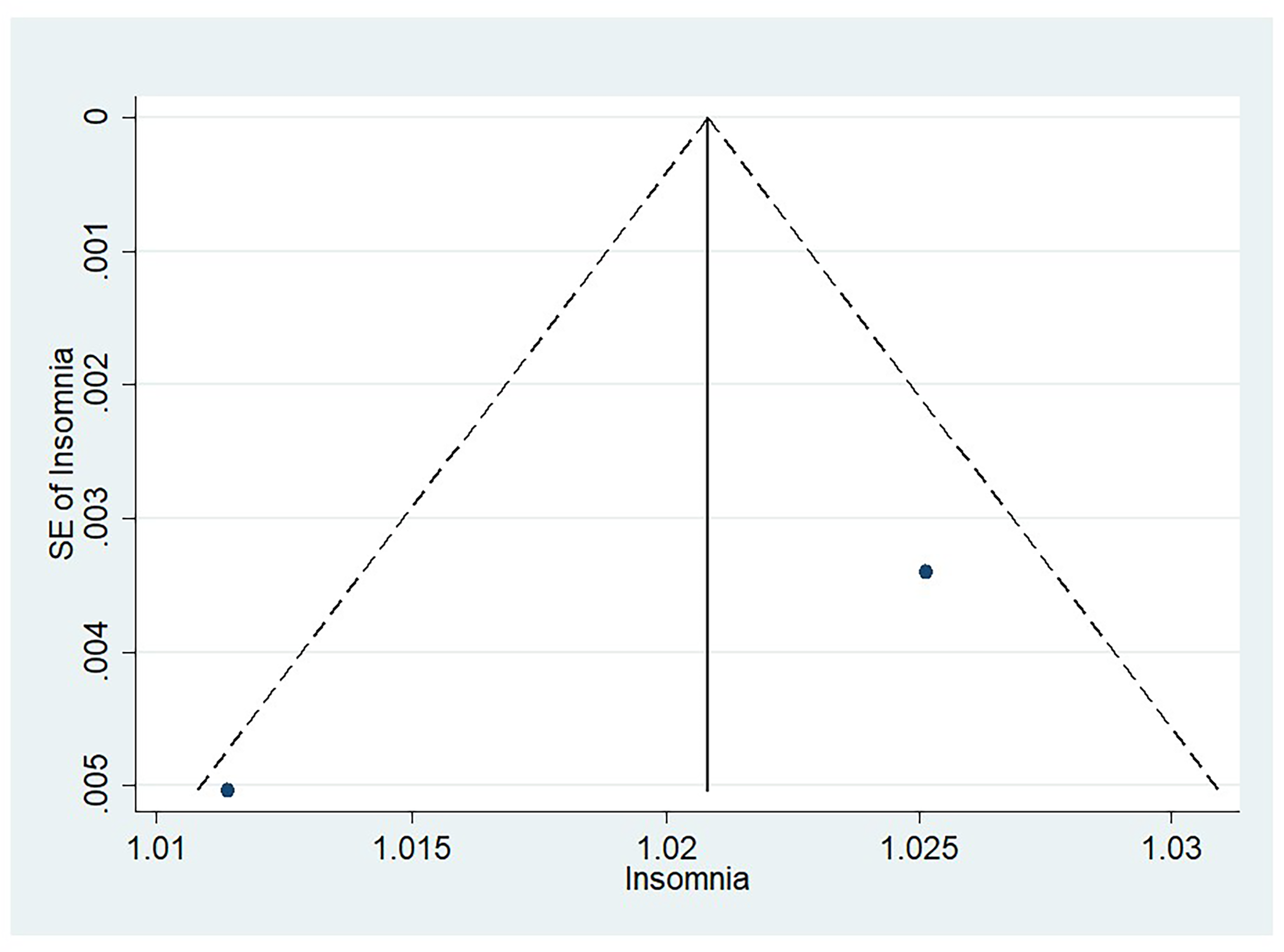
Funnel Plot for Studies Evaluating Insomnia in AMD.
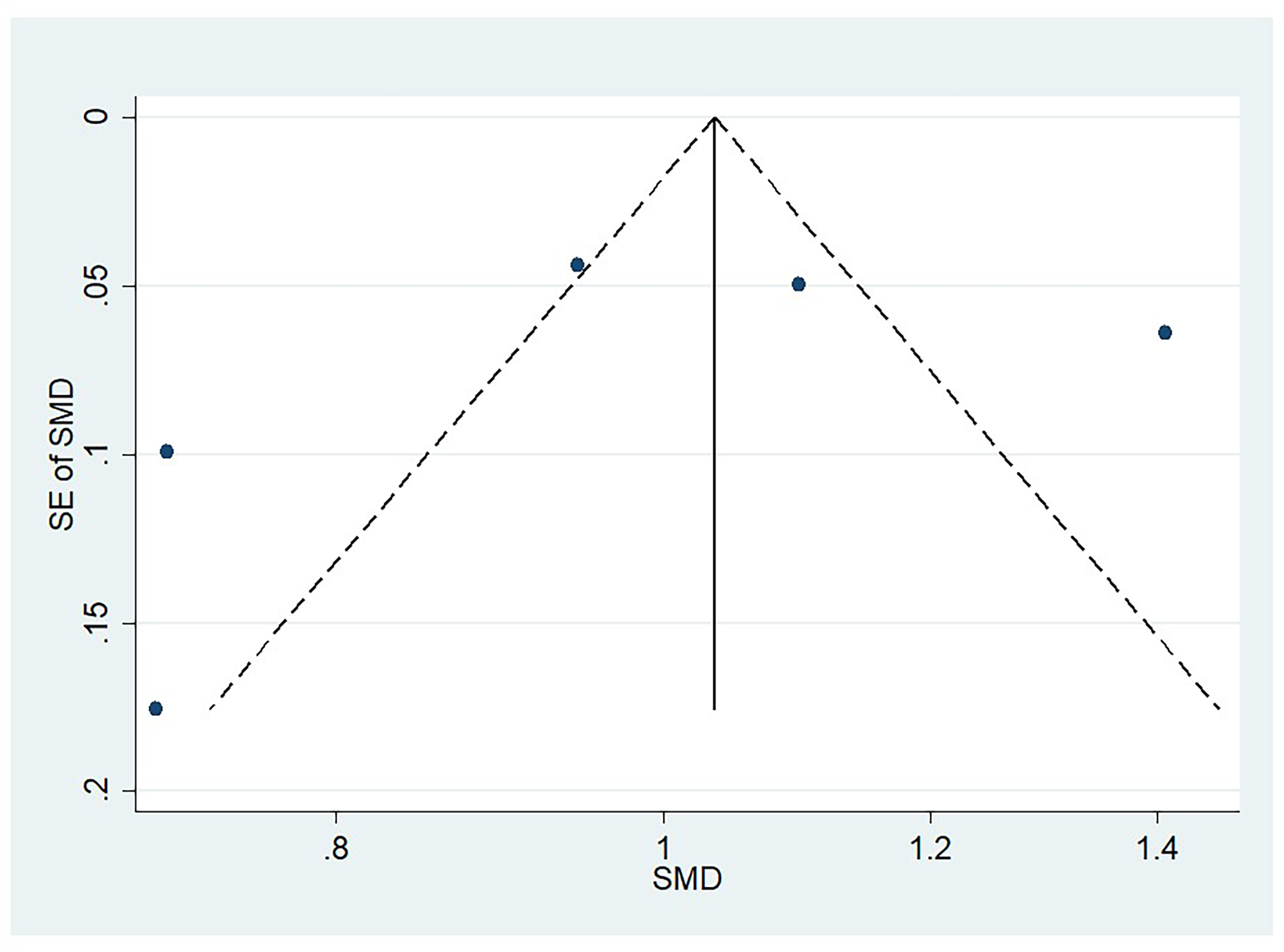
Funnel Plot for Studies Evaluating Sleep Duration in AMD.
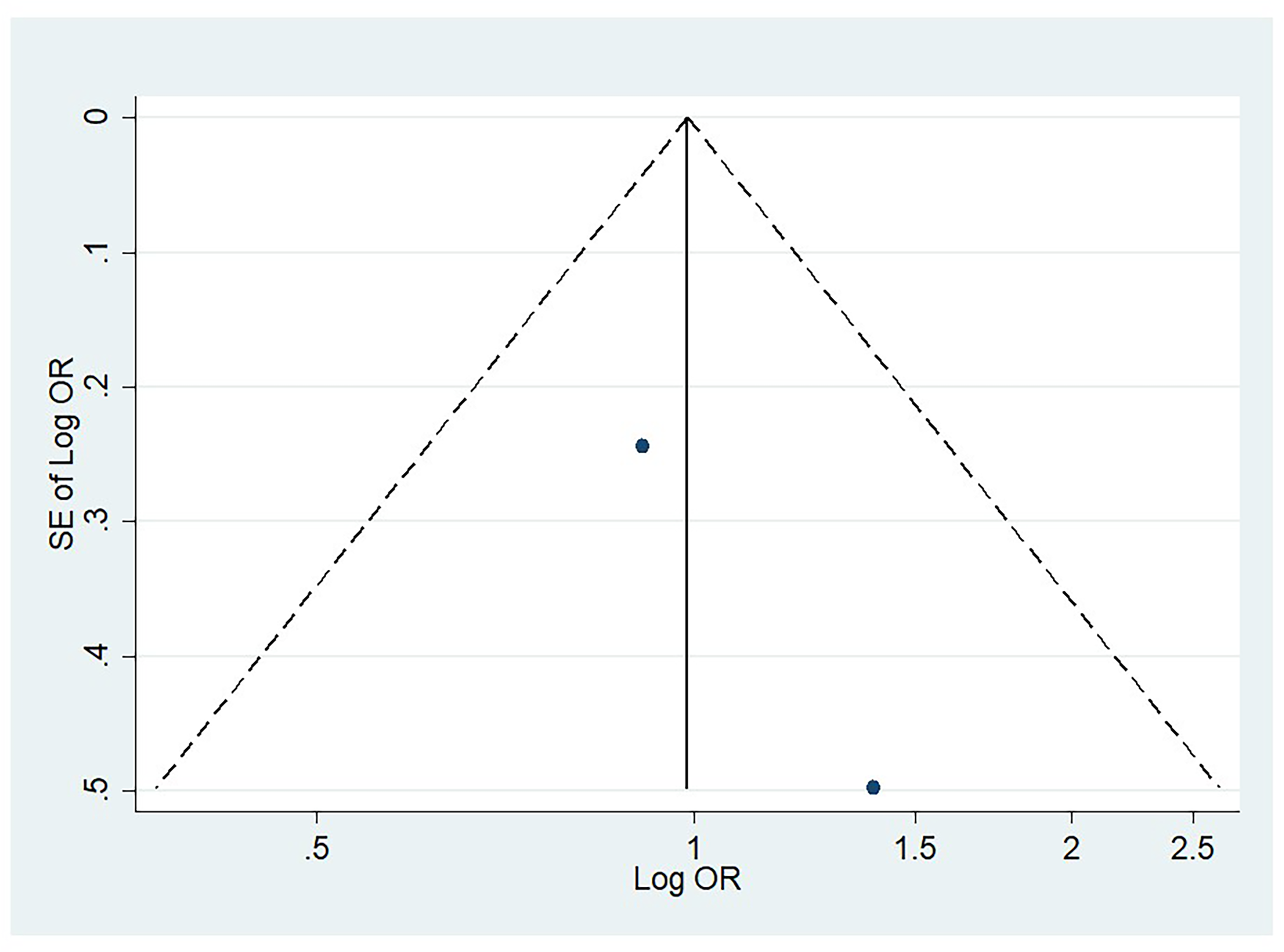
Funnel Plot for Studies Evaluating Odds of Long Sleep Duration (>8 hr) in AMD.
Discussion
Background
Our study entailed a meta-analysis exploring the relationship between AMD and sleep quality. Our meta-analysis revealed a significant increase in the prevalence of poor sleep quality among AMD patients (Maynard et al., 2017; Murphy et al., 2015; Purbrick et al., 2014). Similar observations of poor sleep quality have been observed in visually impaired patients with eye conditions like diabetic retinopathy and glaucoma (Chew et al., 2020; Chin et al., 2020). The impact of AMD on mental health, including depression, anxiety, stress, and reduced quality of life, could further deteriorate sleep quality (Cimarolli et al., 2015; Hassell et al., 2006; Mitchell & Bradley, 2006). Chronic illness diagnosis has been found to heighten patient stress and arousal, leading to less restorative sleep (Parish, 2009).
Analysis
Our meta-analysis revealed an increased prevalence of insomnia among AMD patients (Nau, 2017; Tsai et al., 2020). It is important to note that the definition of insomnia does not rely on a specific number of hours of sleep, since each person has unique sleep requirements. Instead, insomnia is characterized by various sleep changes, such as a prolonged amount of time needed to fall asleep, frequent nighttime awakenings, or extended periods of wakefulness during sleeping periods (Roth, 2007). There was no significant correlation found between AMD and sleep duration, including sleep time exceeding 8 hr (Khurana et al., 2016; Maynard et al., 2017; Murphy et al., 2015; Purbrick et al., 2014; Tsai et al., 2020). This finding is consistent with a previous study that also did not establish any significant link between sleep duration and AMD (Hanscom & Kreiger, 1980). Additionally, a different study also reported no correlation between sleep duration and AMD, but, interestingly, it observed longer sleep times (greater than 8 hr) among individuals with geographic atrophy or significant visual impairments (Khurana et al., 2016).
Impact of AMD on Sleep Quality
Age-related macular degeneration (AMD) can significantly affect a person's mental health, leading to poor sleep quality. Depression and anxiety, often associated with AMD, are well-known contributors to sleep disturbances (Senra et al., 2019). Additionally, psychosocial stressors such as difficulty performing daily activities, loss of autonomy, and impaired social interactions further degrade sleep quality in AMD patients (Lane et al., 2018; Taylor et al., 2016). The persistent fear of worsening vision, potential blindness (Enoch et al., 2024), and anxiety about treatments like anti–vascular endothelial growth factor (VEGF) injections exacerbate these stressors (Senra et al., 2022), resulting in disrupted sleep. Furthermore, visual hallucinations, which can be distressing, may also contribute to poor sleep quality (Carpenter et al., 2019; Pang, 2015). Addressing the mental health challenges faced by AMD patients is crucial in managing their sleep quality and overall well-being.
Physiology of Sleep in AMD
The increased prevalence of poor sleep quality and insomnia in AMD patients provides strong evidence of underlying sleep dysfunction in this population. This finding has also been observed in several studies involving visually impaired individuals (Lockley et al., 1997; Lockley et al., 2007; Mistlberger & Skene, 2005; Revell & Skene, 2010; Sack et al., 1992). One potential cause of poor sleep quality and insomnia in AMD patients is the disruption of the circadian rhythm. In humans, the circadian rhythm governs various physiological processes in a near 24-hr cycle and is regulated by the suprachiasmatic nucleus (SCN) (Saper et al., 2005). This central clock in the brain synchronizes with the solar day through light information received from intrinsically photosensitive retinal ganglion cells (ipRGCs), which, in turn, influences the secretion of melatonin, a hormone critical for sleep and circadian rhythm regulation (Berson et al., 2002; Czeisler, 1995; Hillman et al., 2006; Martínez-Águila et al., 2021). Degeneration of the macula may disrupt the photic signals transmitted to the SCN, leading to irregular melatonin synthesis and subsequent sleep disturbances. Consequently, this disruption in the circadian rhythm could be a significant contributing factor to the observed poor sleep quality and insomnia seen in AMD patients.
Melatonin secretion is disrupted in individuals with AMD. Research has demonstrated that nocturnal melatonin levels in the serum decrease with age (Bubenik & Konturek, 2011; Iguchi et al., 1982; Jung et al., 2010). This decline has been linked with age-related diseases, such as AMD (Bubenik & Konturek, 2011; Jung et al., 2010). Arylalkylamine N-acetyltransferase (AANAT), a crucial enzyme in melatonin synthesis and the regulation of circadian rhythm, is found to be decreased with AMD (Julio & Clifford, 1986; Rosen et al., 2009). Moreover, a study has reported approximately 40% lower levels of urinary 6-sulfatoxymelatonin, a reliable biomarker of melatonin synthesis, in AMD patients (Rosen et al., 2009). Lower levels of melatonin could contribute to an increased prevalence of insomnia and poor sleep quality in AMD patients.
It is important to consider that sleep disturbances can affect the development and progression of AMD. Poor sleep quality can cause oxidative stress, a key contributor to AMD, which can lead to retinal damage and chronic inflammation (AREDS, 2000; Chakravarthy et al., 2010; Everson et al., 2005). Melatonin, known for its antioxidant properties, plays a protective role by mitigating oxidative stress in the retina (Morvaridzadeh et al., 2020). Furthermore, fragmented sleep has been linked to increased atherosclerotic risk, potentially compromising blood flow and oxygenation to the eye and exacerbating AMD progression through inflammatory-mediated pathways (Vallat et al., 2020). Hence, addressing sleep disturbances and promoting healthy sleep patterns may hold potential benefits in managing AMD and its associated complications. This bidirectional relationship between sleep disturbances and AMD highlights the complex interplay and mutual influence of these factors on pathophysiology. It may also be the reason that sleep quality appears to be affected in patients with AMD, but not the sleep quantity. Reason may suggest that only patients with reduced sleep quality, despite adequate duration, experience the negative consequences of oxidative stress, leading to the development of AMD. This observation may give further insight into the directionality of the relationship between AMD and sleep. However, more research is needed to elucidate the specific reason that there appears to be a detriment of sleep quality but not duration in patients with AMD.
Limitations
The limitations of this study include a lack of adjustments for common risk factors for AMD, such as age, smoking status, and genetic polymorphisms. Additionally, there were no adjustments for common risk factors associated with insomnia or poor sleep, including age, gender, occupation, and general health status. These unaccounted factors may influence the associations found between AMD and sleep quality, potentially limiting the reliability and generalizability of the results. Future research should consider these variables to provide a more comprehensive understanding of the relationship between AMD and sleep disturbances.
Another limitation is that we were unable to differentiate between mild and severe AMD (e.g., geographic atrophy). This lack of differentiation restricted our ability to assess potential differences in sleep duration, as seen in another study showing longer sleep duration among those with severe visual impairment and geographic atrophy in AMD (Pérez-Canales et al., 2016). As previously stated, we were also unable to selectively identify the relationship between wet and dry AMD, with regard to their impact on sleep duration, quality, or both. Our inability to distinguish the findings between the disease variants serves as a potential limitation, given that there may be differences between how strongly either form is correlated with an impact on sleep (Hackett et al., 2018).
Another limitation is that the relationship between sleep and AMD is intricate and bidirectional, making it challenging to determine causal factors definitively. Psychosocial factors further complicate the understanding of this relationship, making it difficult to discern whether AMD affects sleep or vice versa. The relationship between sleep and AMD is complex, and AMD can cause sleep disturbances. It is crucial to be vigilant about sleep issues caused by AMD and seek medical attention to address and manage them effectively to preserve vision and prevent worsening outcomes.
Conclusion
Although AMD is primarily a degenerative retinal disorder, its implications extend beyond the eye. The condition can profoundly affect mental health and quality of life, contributing to depression, anxiety, and social isolation. Exploration of cognitive behavioral therapy and melatonin supplementation to improve sleep quality in this population may be warranted. Understanding AMD's broader effects emphasizes the need for comprehensive patient care that includes psychological support and interventions to improve overall quality of life, not just ocular health.
Supplemental Material
sj-docx-1-jvb-10.1177_0145482X261425228 - Supplemental material for The Effects of Age-Related Macular Degeneration on Sleep Quality and Sleep Duration: A Meta-Analysis
Supplemental material, sj-docx-1-jvb-10.1177_0145482X261425228 for The Effects of Age-Related Macular Degeneration on Sleep Quality and Sleep Duration: A Meta-Analysis by Edward Tran, Vivian Phu, Mohamed Aly, Nirmit Shah, Cindy Hutnik and Monali S. Malvankar-Mehta in Journal of Visual Impairment & Blindness
Supplemental Material
sj-docx-2-jvb-10.1177_0145482X261425228 - Supplemental material for The Effects of Age-Related Macular Degeneration on Sleep Quality and Sleep Duration: A Meta-Analysis
Supplemental material, sj-docx-2-jvb-10.1177_0145482X261425228 for The Effects of Age-Related Macular Degeneration on Sleep Quality and Sleep Duration: A Meta-Analysis by Edward Tran, Vivian Phu, Mohamed Aly, Nirmit Shah, Cindy Hutnik and Monali S. Malvankar-Mehta in Journal of Visual Impairment & Blindness
Supplemental Material
sj-docx-3-jvb-10.1177_0145482X261425228 - Supplemental material for The Effects of Age-Related Macular Degeneration on Sleep Quality and Sleep Duration: A Meta-Analysis
Supplemental material, sj-docx-3-jvb-10.1177_0145482X261425228 for The Effects of Age-Related Macular Degeneration on Sleep Quality and Sleep Duration: A Meta-Analysis by Edward Tran, Vivian Phu, Mohamed Aly, Nirmit Shah, Cindy Hutnik and Monali S. Malvankar-Mehta in Journal of Visual Impairment & Blindness
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Glaucoma Research Society of Canada
Supplemental Materials
Supplemental materials for this article are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
