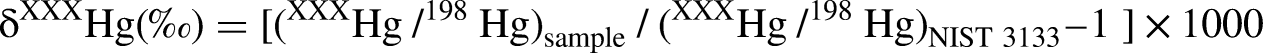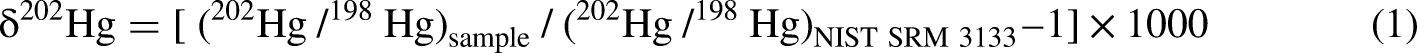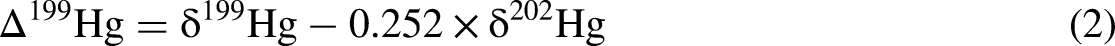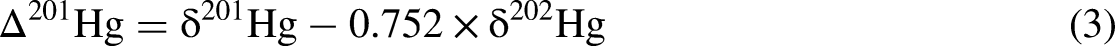Abstract
Mercury (Hg) is a critical pollutant in coal deposits, yet its sources and enrichment mechanisms remain debated. Mercury concentrations detected in ancient sedimentary deposits have frequently been employed as an indicator of volcanic activity, notably those associated with eruptions from large igneous provinces. Yet, this correlation requires careful evaluation, as multiple Hg sources may contribute to its enrichment in sedimentary environments. Potential Hg sources include volcanic emissions, hydrothermal fluids, anthropogenic activities, and other geogenic inputs. The examination of mass-dependent fractionation and mass-independent fractionation in laboratory experiments has increased the feasibility of employing mercury isotopic methodologies to deduce the origins of deleterious trace elements in environmental systems. The harmful trace elements Be, F, V, Cr, Cu, As, Se, Mo, Tl, Pb, U, and Hg show abnormal enrichment in the stratigraphic samples JY-3-1 and JY-3-9 from the Tengnan Coalfield. In the low-temperature hydrothermally altered coal seams of the Jinyuan Mine in southwestern Shandong Province, sulfide and clay minerals are likely the main carriers of mercury. This study aims to indirectly infer the sources of other harmful trace elements strongly correlated with mercury enrichment. A nuanced interpretation of Hg isotope data is necessary due to the complexity of multiple-source mixing, post-depositional modifications, and isotopic fractionation processes. Through the integration of mercury isotope analysis with a suite of analytical techniques, including geochemical analysis, mineralogical studies, and geological surveys, the results indicate that the sources of harmful trace elements in the No. 3 coal of the Jinyuan Mine are predominantly influenced by volcanic activity, atmospheric precipitation, and terrigenous sediment input, with a minor contribution from coal-forming plants. The application of Hg isotopes allows for more effective regulation of high-Hg coal usage, enabling policymakers to implement stricter emission controls, promote cleaner coal technologies, and establish Hg-specific air quality standards.
Introduction
Mercury has a high volatility and is the only metal element in nature that can exist in the form of gas phase (Hg0). After being discharged into the environment, mercury mainly exists in three forms: gaseous elemental (Hg0), inorganic (HgII), and methyl mercury (MeHg) (Gustin et al., 2020). Complex mutual transformation occurs between these different forms. Elemental gaseous mercury (Hg0) predominantly constitutes over 95% of the total atmospheric mercury pool. Its inherent chemical inertness grants Hg0 an atmospheric residence time spanning from 0.5 to 2 years. This extended atmospheric persistence facilitates its transboundary transport, achieving global or at least hemispheric dispersion before eventual deposition (Bagnato et al., 2007; Edwards et al., 2021; Mason, 2009; Pyle and Mather, 2003; Schroeder and Munthe, 1998; Selin, 2009). While positive mercury (Hg) anomalies are frequently attributable to volcanic inputs, the remobilization and recycling of Hg within the surface environment can obscure the identification of its primary source when relying solely on Hg concentration data in sediments. Given the relative abundances and mass-dependent differences of these isotopes, researchers are able to elucidate the sources and biogeochemical pathways of mercury by examining isotopic ratios. This approach allows for the precise tracking of mercury's environmental cycling and transformation (Blum and Johnson, 2017). Mass-dependent fractionation (MDF) follows the classic mass fractionation theory (Bigeleisen and Mayer, 1947; Criss, 1999; Urey, 1947). In contrast, odd-mass-independent fractionation (MIF) is mainly caused by photochemical processes (Blum et al., 2014) with little effects linked to other reactions. Thus, odd-MIF provides clear source constraints of sedimentary Hg (Blum et al., 2014; Jiskra et al., 2021; Kwon et al., 2020; Štrok et al., 2015). However, the photoreduction of mercury species during atmospheric transportation can alter the isotopic characteristics of volcanic mercury, leading to positive Δ199Hg remainings in the water system, while negative Δ199Hg may be released into the atmosphere in the form of Hg0 (Bergquist and Blum, 2007; Blum et al., 2014).
As shown in the BP Statistical Review of World Energy (Global, 2022), the grow in demand and consumption of coal has reached its highest level in recent years. China and India have dominated the market, while Europe and North America have shown an increase in coal consumption after nearly a decade of continuous decline. This growing reliance on coal has significant implications for atmospheric Hg emissions, as coal combustion remains the dominant anthropogenic source of Hg pollution worldwide. In addition, coal affected by magma or low-temperature hydrothermal solution often leads to abnormal enrichment of some trace elements, especially harmful trace elements such as F, Hg, Zn, Pb, Cu, Se, and As. In geologically active areas such as eastern Russia and southwestern China, mercury in hydrothermal fluids typically relies on coal matrix for storage, and local mercury concentrations can be enriched by several orders of magnitude (Dai et al., 2012b; Yudovich and Ketris, 2005; Zhuang et al., 2006). From this, starting from the evidence of mercury isotopes, we can trace the migration patterns of mercury in various geological evolution processes, and provide evidence for tracing and deducing geochemical processes of harmful elements in coal. Despite significant research on coal geochemistry, there is still a lack of studies focusing on the combined application of mercury isotopes and trace element analysis to comprehensively trace pollution sources. Early Permian coal is an important sedimentary mineral resource in North China. Many studies have been conducted on coal in the southwestern region of Shandong, mainly focusing on sedimentary environments, tectonic magmatic evolution (Wang, 1990), trace element geochemistry, and mineral composition and mineralogical anomalies (Guo et al., 2020; Liu et al., 2005, 2007; Wang et al., 2016; Zeng et al., 2000). However, detailed literature on the abnormal enrichment of harmful trace elements in coal seams and their geological controls remains sparse. This study aims to bridge this gap by integrating mercury isotope analysis with geochemical and mineralogical investigations to provide a more precise understanding of trace element sources and their depositional processes in coal seams.
In this investigation, we present detailed analyses of mercury (Hg) concentrations and isotopic compositions within No. 3 coal from Shandong Province, Eastern China, dated to the Early Permian period. This epoch is critically recognized for offering an unparalleled insight into the key evolutionary process of northern temperate ecosystems. Utilizing an extensive integrated geochemical dataset, our objective is to delineate the origins of detrimental trace elements via mercury isotopic signatures. By leveraging a combination of isotopic tracing and geochemical modeling, this study offers a novel perspective on the mechanisms governing mercury enrichment in coal. The sophisticated interpretation of mercury isotope data from this era elucidates the intricate geochemical dynamics and environmental stressors that have impacted ecosystem resilience and collapse, thereby highlighting the complexity inherent in these ancient ecological frameworks. By integrating mercury isotope analysis, this study provides insights that extend beyond the Tengnan Coalfield, offering implications for global mercury cycling, particularly in coal-dependent economies such as China, India, and the United States.
Geological setting
During the Early Paleozoic to Mesozoic, the North China Craton (NCC) has relatively complete geological records (Figure 1(b)) (Li et al., 2010; Zhao et al., 2012). It is the major tectonic unit of the Chinese Mainland (Figure 1(a)), has received extensive international attention, and is a natural laboratory for geological evolution research. The research area of this study, Tengxian Coalfield, is located in the eastern part of NCC, the southwest plain area of Shandong Province (Figure 1(c)), it contains the North China type Carboniferous-Permian coalfield and covers an area of approximately 1398 km2. The coalfield is a gradual descending alluvial plain from northeast to southwest and divided into Tengbei Coalfield and Tengnan Coalfield by the Tengxian anticline in northeast direction. Currently, there are 25 production mines in Tengnan Coalfield, and the boundary of the coalfield is generally dominated by faults. Outside the coalfields to the east and north of the coalfields are the low Ridge Mountains. The study sections examined are from Jinyuan Mine, located in the southeastern boundary of Tengnan Coalfield. The lower part of the Shanxi Formation was deposited in a transitional zone between marine and terrestrial environments, which later shifted to a continental sedimentary setting during the subsequent deposition.
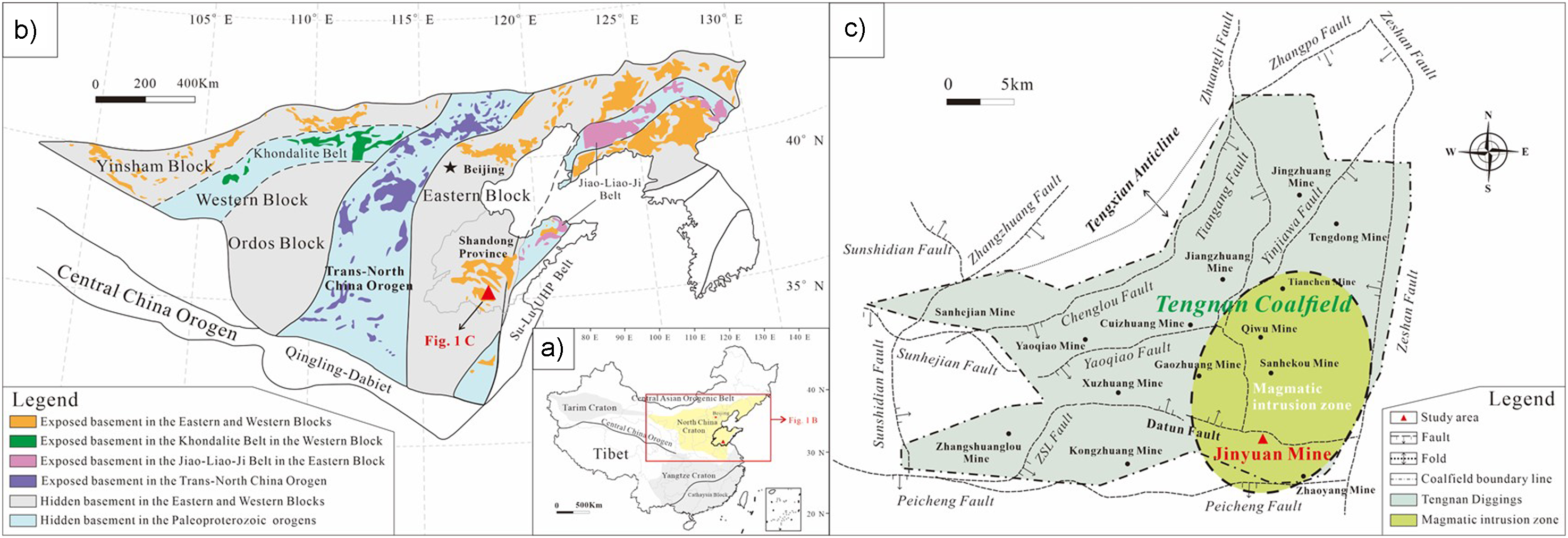
(a) Schematic tectonic map of China showing the major Precambrian blocks and intervening fold belts. (b) Tectonic subdivision of the North China Craton (after Zhao et al., 2012). (c) Geological map of Tengnan Coalfield in Shandong Province, North China.
Frequent and intense volcanic activity occurred in the southwest of Shandong Province during the later period of the Yanshan Movement. With a large scale and wide range of magmatic bodies, most coal seams in the area have been damaged by magmatic rocks to varying degrees. These volcanic activities likely played a significant role in the geochemical evolution of the Tengnan Coalfield, influencing the distribution of harmful trace elements. Volcanic ash deposition could have introduced a variety of trace elements, including Hg, As, Se, and Pb, into the coal-forming environment. Additionally, hydrothermal fluids associated with volcanic activity may have led to the remobilization and enrichment of these elements in coal seams. Tengnan Coalfield is also affected, especially in the southeast of the study area. From south to north, magmatic activity gradually weakened. Additionally, after entering the Jurassic, due to the addition of regional magma and the superposition of heat sources, the thermal metamorphism of magma in the study area was strengthened. The deepening of coal metamorphism is reflected in the change of vitrinite reflectance (Ro). The vitrinite reflectance (Ro) of the No. 3 coal in the Shanxi Formation of Tengbei Coalfield ranges from 0.66% to 0.69%. In this study, the measured Ro value of the No. 3 coal seam in Tengnan Coalfield is 0.757%, which is higher than that of Tengbei Coalfield, indicating that Tengnan Coalfield has undergone regional thermal metamorphism due to the influence of southern magmatic heat.
Being a complex organic rock, coal is influenced by various factors shaping its geochemical characteristics, including the type of original vegetation, climatic conditions, depositional environment, and subsequent geological processes. Notably, the elemental enrichment pattern in the No. 3 coal of the Tengnan Coalfield within the Shanxi Formation in North China is distinct compared to other regional coals. The coal seams of the Shanxi Formation, primarily composed of Paleozoic sediments such as mudstone and sandstone, have undergone specific genesis and evolutionary processes that critically determine the distribution and concentration of trace elements. It is important to recognize that the extent of trace element enrichment in coal is not solely a function of the inherent elemental content, but also heavily dependent on the mineralogical composition and structural characteristics of the coal. For example, the presence of sulfide minerals can enhance the accumulation of elements like mercury and zinc, whereas the type and maturity of the organic matter also play a pivotal role in elemental enrichment. Consequently, a comprehensive evaluation of trace element enrichment in coal must consider an array of factors, including the geological setting, genesis, and geochemical processes.
Geochemical evidence from Hg isotopes further supports the impact of volcanic activity on trace element distribution. The enrichment of mercury and other elements in certain coal layers, particularly JY-3-1 and JY-3-9, suggests a link to volcanic emissions. These emissions, followed by atmospheric deposition and hydrothermal interactions, likely contributed to the localized concentration of these elements in the coalfield. The isotopic signatures observed in this study align with volcanic-derived Hg, reinforcing the hypothesis that past volcanic activity played a crucial role in the geochemical anomalies within the Tengnan Coalfield.
Samples and methods
Sample collection
The Taiyuan and Shanxi Formations of Late Paleozoic are the major coal-bearing strata in the Tengnan Coalfield. This study systematically collected coal samples every 20 cm along the profile of the upper section of the No. 3 coal seam in the Jinyuan Mine, to analyze the geochemical characteristics at different depths. A total of 11 fresh samples from the Early Permian were collected, including 10 coal plies and one roof sample. The number of collected samples was limited by underground mining conditions. As the No. 3 thick coal seam was sampled sequentially from the roof downward, the vertical space available for sampling restricted the total number to 11. While additional samples would be ideal, the current dataset effectively captures the key geochemical trends within the seam. Furthermore, statistical consistency in elemental distribution across the sampled intervals supports the adequacy of this dataset for understanding coal geochemistry in the Tengnan Coalfield. The sample collection followed the Chinese Standard Method GB/T Standard 482-2008.
The recovered coal bench samples from the JY-3 area were immediately wrapped in moisture-resistant bags and sealed in plastic containers to prevent oxidation and contamination, ensuring the accuracy and reliability of their geochemical characteristics for subsequent analysis. The collected samples were sieved into two parts with different particle sizes, one part is 18-40-mesh particle sample, mixed with epoxy resin and curing agent (5:2) for preparation of coal petrographic polished sheet, and the other part was a 200-mesh powder sample for coal quality analysis and geochemical analysis.
Mercury concentrations and isotopes measurement
We measured the content of Hg element in the sample using the Milestone DMA-80. This method was chosen because it allows for direct mercury measurement in solid samples without requiring complex sample digestion, thereby minimizing contamination risks and maintaining sample integrity. The relative difference in mercury concentration between replicated samples and coal ash mixtures is within 10%, indicating that the instrument is stable and provided uniform results (Zheng et al., 2018). The coal samples were subjected to crushing and grinding until the particle size was reduced to less than 200 mesh, ensuring sample uniformity and providing a reliable foundation for subsequent geochemical analyses, then immediately sealed in plastic bags to prevent mercury volatilization. The crucible carrying the samples should be empty and burned at 850 °C for 15 min before use to eliminate any residual Hg. During testing, weigh 0.01 g of the sample and place it in a sample loading crucible. The crucible and sample should then be in the automatic sampler, which can directly analyze solid samples for fully automatic analysis.
Mass bias of the Multi-Collector Inductively Coupled Plasma Mass Spectrometry (MC-ICP-MS) was corrected using standard-sample with NIST SRM 997 Thallium as internal standard solution and NIST 3133 Hg as external standard solution. MC-ICP-MS was selected because of its high precision and ability to measure isotopic fractionation with minimal interference, making it the most suitable technique for tracing mercury sources in geochemical studies. During the mercury isotope measurement, two thallium isotopes were simultaneously measured. UMAlmaden and Fluka Hg were used as the standard materials for isotope analysis, and the laboratory mercury standard solution (Beijing National Standard Material Center, China) was used as the control standard for concentration measurement. The mercury isotope experiment was completed at the Key Laboratory of Institute of Geochemistry, Chinese Academy of Sciences.
MDF follows the general principle that lighter isotopes react slightly faster than heavier isotopes during physical and chemical processes, such as volatilization, adsorption, and redox reactions. This fractionation is quantified using the delta (δ, ‰) notation as defined in the following equation (Blum and Bergquist, 2007):
Major and trace element measurement
The high-temperature ash sample (815 °C) obtained from industrial analysis for measuring ash content is combined with boric acid and compacted in a mold to create the sample for testing. The major elements present as oxides in the sample are detected using X-ray Fluorescence (XRF). This technique was chosen for its ability to rapidly and nondestructively determine major-element compositions, making it particularly suitable for coal characterization. To determine the content of As and Se elements in coal, hydride generation-atomic fluorescence spectrometry was employed. The content of F is determined using high-temperature combustion hydrolysis–fluoride ion selective electrode method. The remaining 16 harmful trace elements were analyzed using a microwave digestion system coupled with ICP-MS, which was chosen for its high sensitivity and exceptionally low detection limits, ensuring precise quantification even at trace concentrations. Finally, the distribution of content distribution and enrichment characteristics of the measured elements are analyzed.
These analytical techniques were carefully selected to ensure that the results provide a comprehensive geochemical characterization of the coal samples, allowing for a better understanding of mercury and trace element behavior in sedimentary environments.
Results
Coal quality and chemistry
Previous studies have shown that the yield of dry-based ash in the samples collected from No. 3 coal in the Shanxi Formation of Tengnan Coalfield varies greatly, ranging from 3.24% to 29.08%, with an average value of 11.35%. Except for some coal seams located at the top and middle bottom of the coal seam, the ash yield of most coal seam samples is relatively low (<10%). The sulfur content of No. 3 coal in Tengnan Coalfield ranges from 0.02% to 1.35%, with an average content of 0.44%, which corresponded to low-sulfur coal based on the classification by Chou (2012). Except for the total sulfur content of samples JY-3-1 and JY-3-9, which are as high as 1.35% and 1.04%, the remaining sulfur content is relatively low (Table 1). The yield of volatile matter (dry ashless basis) ranges from 33.32% to 47.71%, with small fluctuations and an average value of 38.95% (Guo et al., 2020).
Concentrations of major-element oxides (wt%) in the coal bench samples and roof samples from the (No.3) in the Jinyuan Mine, Tengnan Coalfield, determined by XRF analysis.
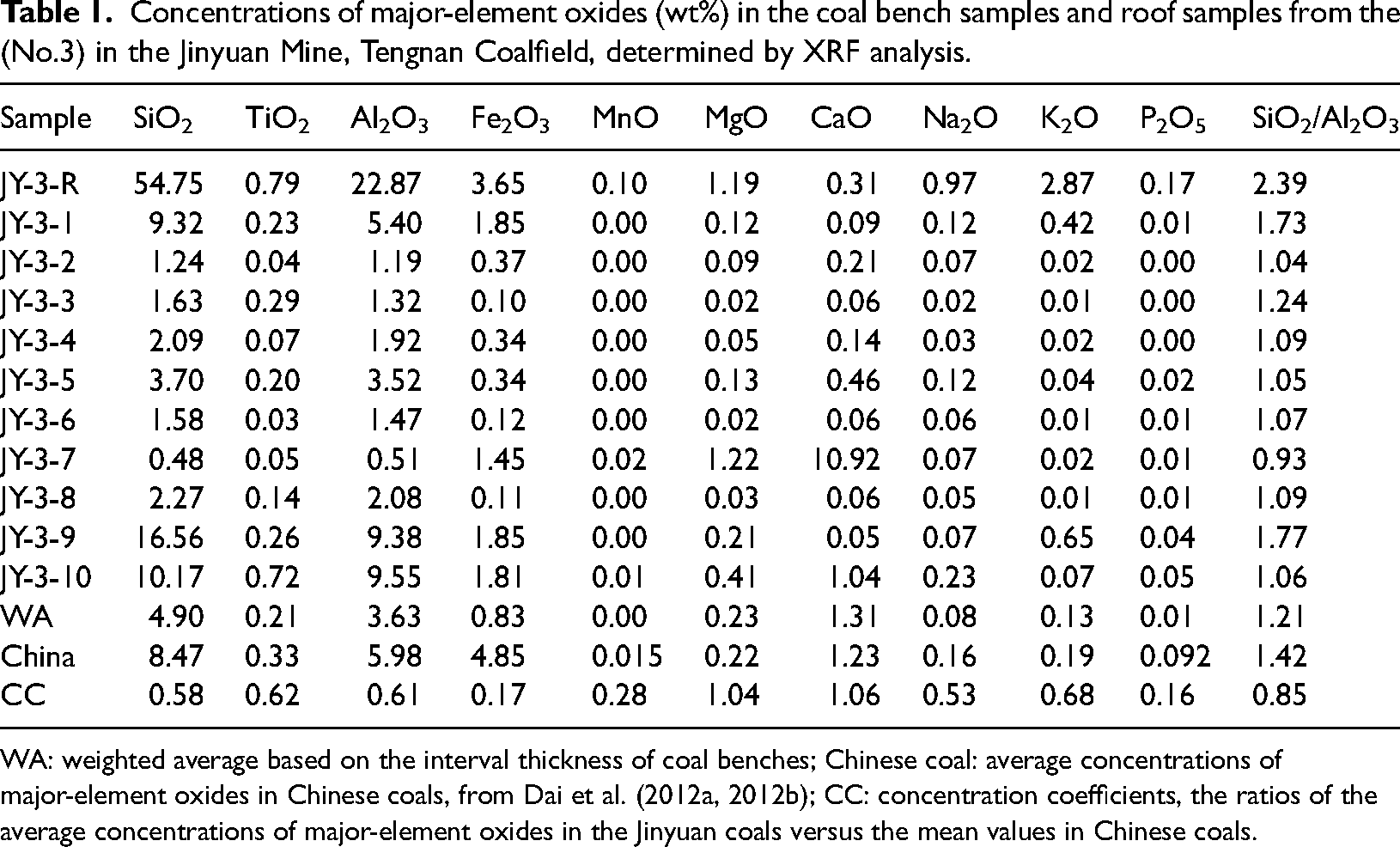
WA: weighted average based on the interval thickness of coal benches; Chinese coal: average concentrations of major-element oxides in Chinese coals, from Dai et al. (2012a, 2012b); CC: concentration coefficients, the ratios of the average concentrations of major-element oxides in the Jinyuan coals versus the mean values in Chinese coals.
Concentrations of major-element in the coals and non-coal samples
In the No. 3 coal of the Tengnan Coalfield, SiO2 and Al2O3 are the predominant constituents. While the average levels of MgO and CaO slightly exceed the background levels of constant elements in Chinese coal (hereinafter referred to as “Chinese coal”), the average concentrations of other major elements are comparatively lower. Some coal bench samples show an anomalous enrichment of constant elements. For instance, in coal samples JY-3-1, JY-3-9, and JY-3-10, the concentrations of SiO2 are recorded at 9.32%, 16.56%, and 10.17%, respectively, surpassing those found in Chinese coal. Similarly, in samples JY-3-9 and JY-3-10, the concentrations of Al2O3 are 9.38% and 9.55%, respectively, also exceeding those in Chinese coal. Additionally, in sample JY-3-7, the concentrations of MgO and CaO significantly exceed those observed in other coal strata and Chinese coal (Table 1).
Regarding SiO2 and Al2O3 content in the No.3 coal of the Tengnan Coalfield, the SiO2 content ranges from 0.48% to 16.56%, with an average of 4.90%, while the Al2O3 content ranges from 0.51% to 9.55%, averaging at 3.63%. Traditionally, quartz and clay minerals act as the primary carriers of Si and Al in coal. In this study, the SiO2/Al2O3 ratio in the No.3 coal of the Tengnan Coalfield varies from 0.93% to 1.76%, with an average of 1.20%. This value is notably lower than the average for Chinese coal (1.42) and is close to the theoretical value for kaolinite (1.18). Quantitative mineral data show that, except for sample JY-3-7, kaolinite predominates in all other samples, exceeding 40% on average. Elevated SiO2/Al2O3 values typically indicate the presence of non-clay silicate minerals (such as quartz) in coal. The higher SiO2/Al2O3 values noted in the No. 3 coal of the Tengnan Coalfield are particularly conspicuous in samples JY-3-1 and JY-3-9, where the proportions of quartz measure 21.5% and 19.8%, respectively, surpassing other minerals in these samples apart from kaolinite. Moreover, the quartz content in samples JY-3-1 and JY-3-9 significantly exceeds that in other samples. Conversely, sample JY-3-7 has the lowest quartz content, resulting in the lowest SiO2/Al2O3 ratio. Therefore, the SiO2/Al2O3 ratio indicates that kaolinite is the predominant silico-aluminate mineral in the coal of the Tengnan Coalfield (Table 1).
Concentrations and isotopic compositions of mercury
The concentrations of Hg in the No. 3 seam range from 14.27 ppb to 952.13 ppb with a mean of 177.87 ppb (Table 2). In comparison, the average mercury concentrations compiled for Chinese (Dai et al., 2012a) and world (Ketris and Yudovich, 2009) coal deposits are 163 ppb and 100 ppb, respectively. Most samples yield Hg concentrations lower than these baseline for evaluating Hg anomalies, except for two peaks at JY 3-1 (952.13 ppb) followed by JY 3-9 (553.36 ppb) (Figure 8). In this study, δ202Hg values from the No. 3 seam are primarily negative, ranging from −3.73‰ to −1.59‰ with an approximately 1.98‰ variation (Table 2). In contrast, the mean δ202Hg values in global coals from various geological periods display a comparatively constrained range (−1.35 ± 0.85‰ to −1.06 ± 0.70‰) without a discernible temporal trend (Sun et al., 2016). The broader δ202Hg variability observed in the No. 3 seam suggests more complex fractionation processes, potentially influenced by localized geochemical conditions, including depositional environment, post-depositional diagenesis, and hydrothermal or volcanic inputs. The Δ199Hg values range from −0.14 to 0.21‰ with an approximately 1.61‰ variation (Table 2). Notably, Δ199Hg values in Permian coals (−0.04 ± 0.16‰, n = 102) are generally higher than those from other geological periods (Sun et al., 2016). The Δ199Hg range observed in the No. 3 seam falls within this broader Permian trend, indicating that its isotopic composition may reflect characteristic depositional and geochemical conditions of the Permian. In contrast to MDF, the MIF effect of mercury stable isotopes provides crucial insights into mercury sources and transport pathways, due to the limited MIF pathways available (Blum et al., 2014; Yin et al., 2016) (Figure 2).
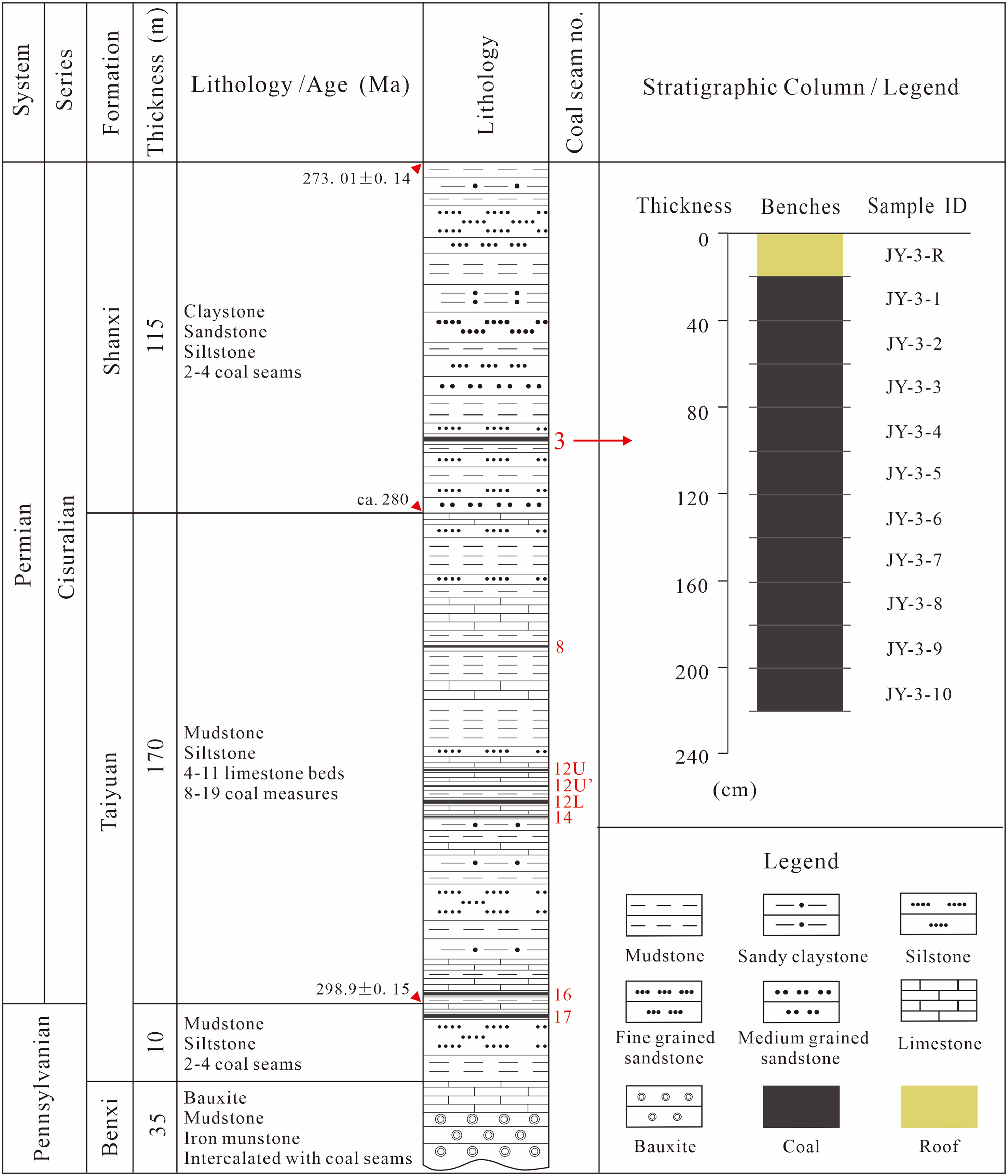
Stratigraphic section of the Tengxian Coalfield.
Hg concentrations, δ202Hg, δ201 Hg, δ200 Hg, δ199 Hg, Δ201Hg, Δ200Hg, and Δ199Hg values of coal-bearing section (No.3) in the Jinyuan Mine, Tengnan Coalfield.
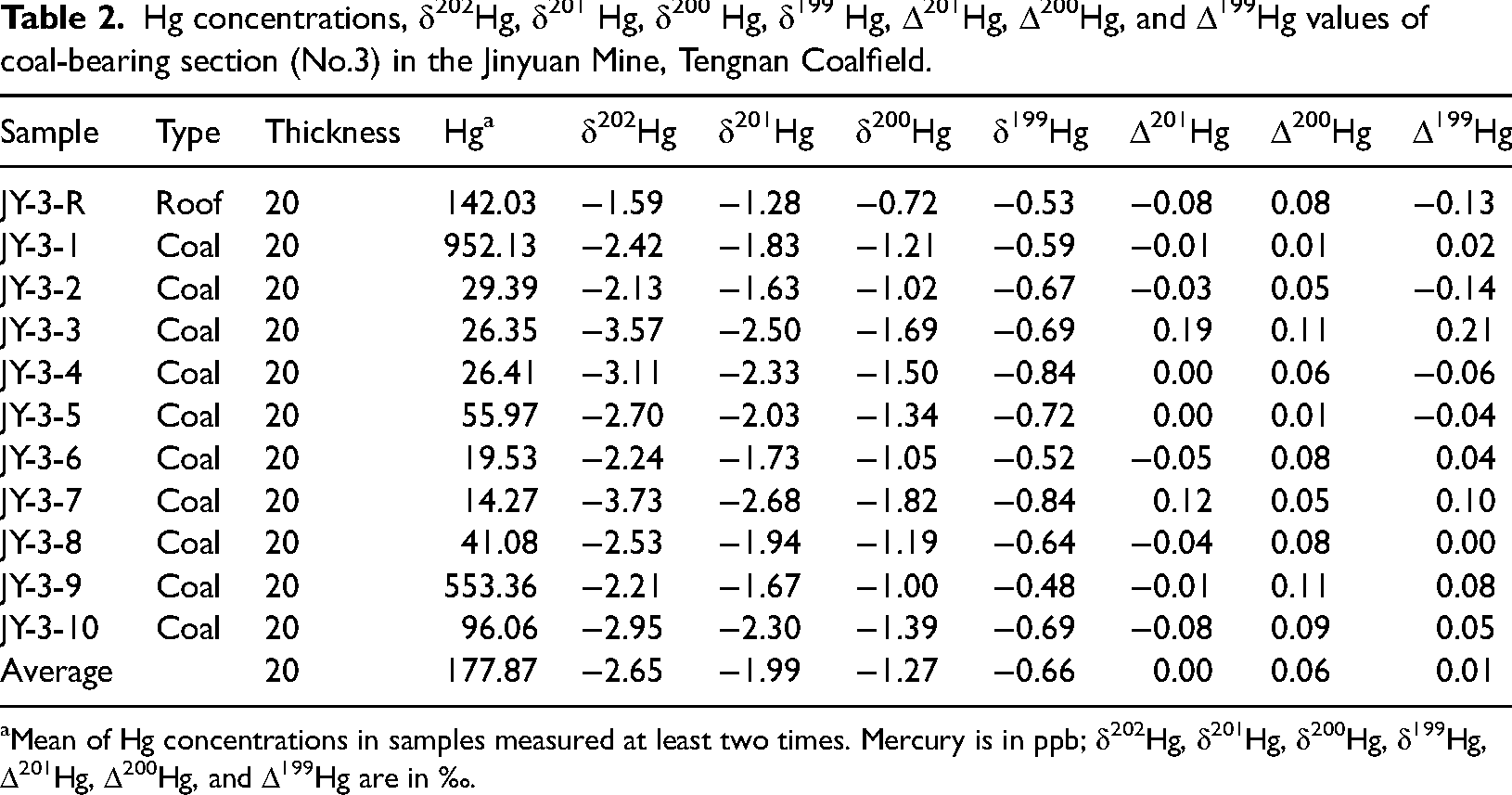
Mean of Hg concentrations in samples measured at least two times. Mercury is in ppb; δ202Hg, δ201Hg, δ200Hg, δ199Hg, Δ201Hg, Δ200Hg, and Δ199Hg are in ‰.
Enrichment characteristics of harmful trace elements
Enrichment and geochemical controls of trace elements in No. 3 coal seam
To gain a more comprehensive understanding of the overall characteristics of harmful trace element concentrations in the No. 3 seam of Tengnan Coalfield, the indicator (concentration coefficients (CC)) proposed by Dai et al. (2015) to evaluate the enrichment degree of trace element content in coal, which is the ratio of trace element concentration in coal to the average value of trace elements in world coal, is used to calculate the enrichment degree of harmful trace elements in the No. 3 seam.
Analysis of 12 harmful trace elements in No. 3 coal revealed that they are abnormally enriched in coal seams JY-3-1 and JY-3-9, with significantly higher content compared to world coal and Chinese coal (Table 3). When compared to the global coal average, the enrichment of elements in the No. 3 coal of the Tengnan Coalfield Shanxi Formation is relatively low. Among the 20 harmful trace elements studied, the enrichment coefficients of Be (0.43), Co (0.23), Ni (0.45), Zn (0.18), As (0.13), Cd (0.29), Sn (0.29), Sb (0.23), and Tl (0.30) are all less than 0.5, indicating a depletion in their enrichment. On the other hand, elements such as F (0.55), V (0.96), Cr (0.79), Cu (0.76), Mo (0.62), Ba (0.76), Hg (1.81), Pb (0.73), Th (0.62), and U (0.57) have enrichment coefficients ranging from 0.5 to 2, suggesting a normal enrichment level. Only the enrichment coefficient of Se reaches 2.33, classified as mild enrichment (2 < CC ≤ 5). Stratified coal samples JY-3-1 and JY-3-9 exhibit anomalous enrichment phenomena for these harmful trace elements mentioned above (Figure 3).
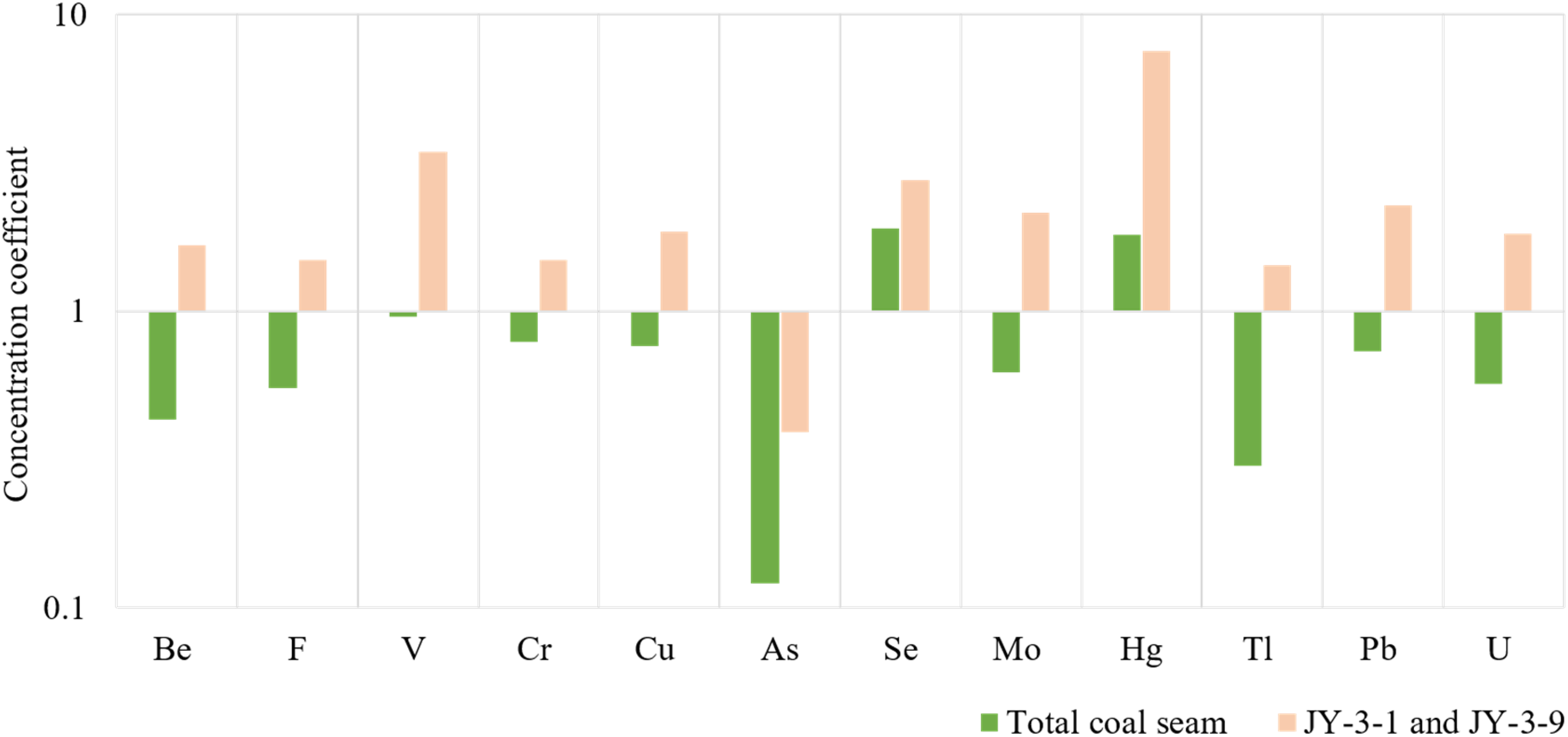
Concentration coefficients (CC) of total coal seam versus mean of JY-3-1 and JY-3-9 in the coal-bearing section (No. 3) from the Jinyuan Mine, Tengnan Coalfield. CC, the ratio of averages for harmful trace elements concentration in samples versus averages for world coals.
Concentrations and enrichment factor of selected harmful trace elements (μg/g) in the coal-bearing section (No. 3).
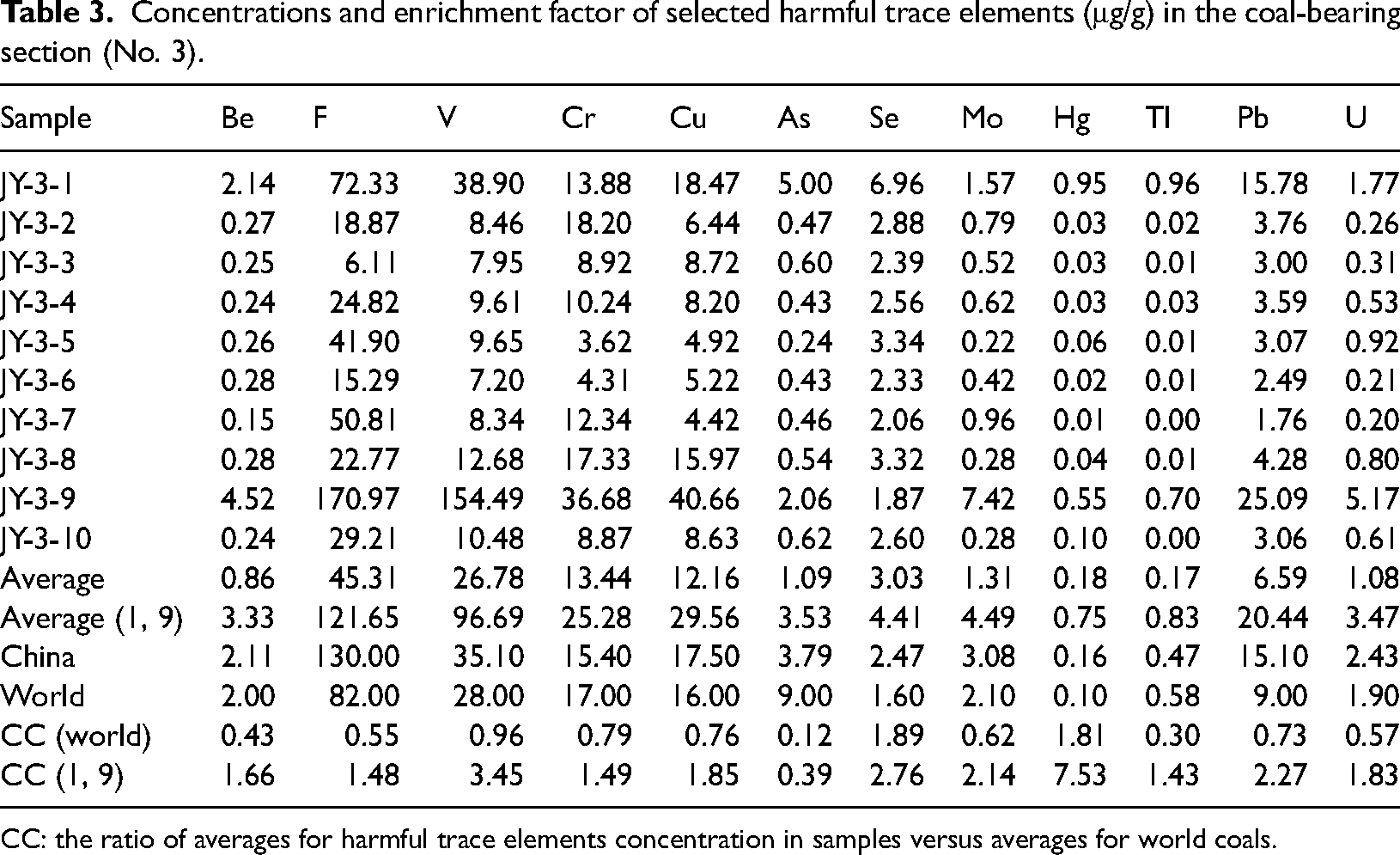
CC: the ratio of averages for harmful trace elements concentration in samples versus averages for world coals.
By comparing the CC values to both the world and Chinese coal baselines, we observe that certain elements, particularly Hg and Se, exhibit significantly higher enrichment levels in the Tengnan Coalfield relative to the global average, which may suggest localized geochemical controls, such as hydrothermal fluid activity or volcanic input. The higher Hg concentrations in JY-3-1 (9.52) and JY-3-9 (5.53) align with previous studies that indicate magmatic intrusions may introduce elevated Hg levels into coal seams (Ghosh et al., 2013). In addition, the enrichment of Ba, F, Cr, Zn, Mo, Pb, and U in JY-3-9 suggests that post-depositional geochemical processes, such as fluid migration and diagenetic mineralization, may have further contributed to trace element redistribution.
Geochemical correlations and the potential of Hg as a proxy for trace element origins
The harmful trace elements Be, F, V, Cr, Cu, As, Se, Mo, Tl, Pb, U, and Hg demonstrate pronounced enrichment in the stratigraphic samples JY-3-1 and JY-3-9 from the Tengnan Coalfield. There is a significant correlation among these elements, notably with mercury (Hg), suggesting intricate geochemical interactions and potential natural enrichment processes (Figure 4). Consequently, we consider the possibility of indirectly deducing the origins of other harmful trace elements that show strong correlations with Hg by studying the geochemical pathways of mercury. This investigative approach presupposes that these elements might originate from analogous geological processes or share similar geochemical environments, potentially involving common sources or geotransport mechanisms. It is imperative to acknowledge that although some elements may correlate with mercury enrichment, this does not necessarily mean their sources are identical or consistent. Various elements might display differences in geochemical behavior and sources, so using the origin of mercury as a proxy for the sources of other elements might be subject to certain limitations. It is important to recognize that while some elements exhibit strong correlations with mercury, their sources and geochemical behaviors may not be identical. Differences in solubility, affinity for specific mineral phases, and mobility under varying redox conditions could influence their ultimate distribution in coal seams. Therefore, using Hg as a proxy for other trace elements requires careful consideration of geochemical context and mineralogical constraints.
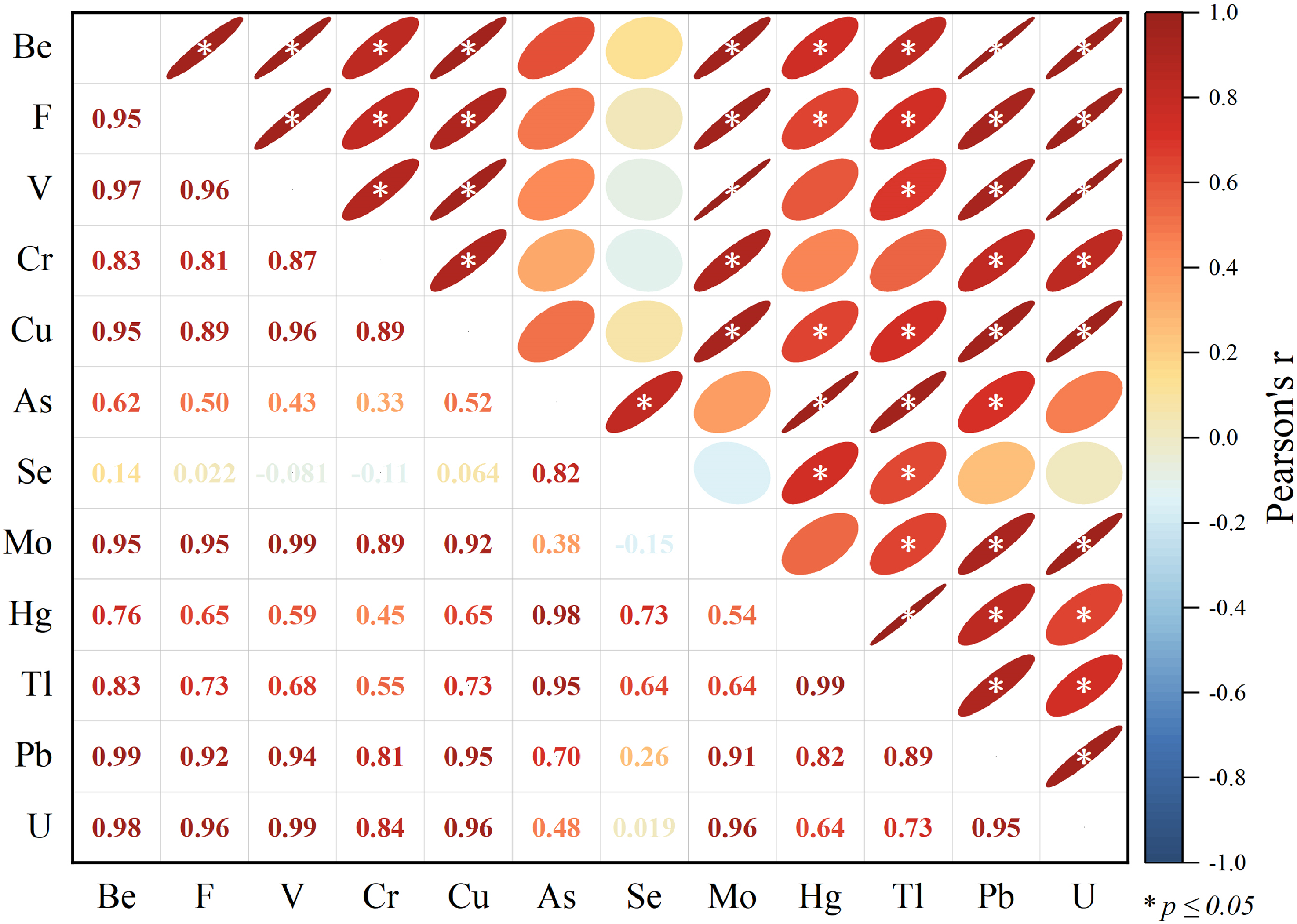
Pearson's r correlation coefficients for harmful trace elemental pairs at Jinyuan. Red and blue ellipses (upper right) and numbers (lower left) represent positive and negative correlations, respectively. Asterisks denote correlations significant at the 3σ level (p ≤ 0.05).
To develop a more comprehensive understanding of the sources of these harmful trace elements, it is essential to amalgamate data from diverse fields such as geology, geochemistry, mineralogy, and environmental science. We plan to utilize methods including geochemical analyses, isotope studies, mineralogical examinations, and geological surveys to elucidate the origins, migration pathways, and enrichment mechanisms of these elements within the Earth's internal cycle.
Discussion
During the Cisuralian epoch (298.9 ± 0.15–273.01 ± 0.14 Ma), the North China Block occupied northern tropical to subtropical paleolatitudes, strategically positioned along the periphery of the Paleo-Asian Ocean (PAO) (Figure 5(a)) (Boucot et al., 2013). This specific geographic configuration is integral for comprehending the significant climatic transformations that defined the shift from an icehouse to a greenhouse state. The vicinity to the PAO was likely a determinant factor in shaping regional climate patterns and engendering distinctive geochemical signatures within the sedimentary deposits. These prevailing conditions present a prime opportunity to examine the complex dynamics between marine and terrestrial forces during an era marked by extensive global climate evolution. Particularly, the application of mercury isotopic analysis offers profound insights into volcanic activity, terrestrial weathering, and oceanic exchanges, all of which are essential for elucidating the underlying mechanisms that facilitated the Earth's transition from a colder, glacially dominated environment to a warmer, more humid greenhouse regime. We utilize mercury isotope geochemical modeling and geological data to simulate the sources and migration pathways of harmful trace elements in the Jinyuan mining area during the Early Permian period (Figure 5(b)). This approach could advance our understanding of the geochemical behaviors of these elements and their consequent environmental impacts.
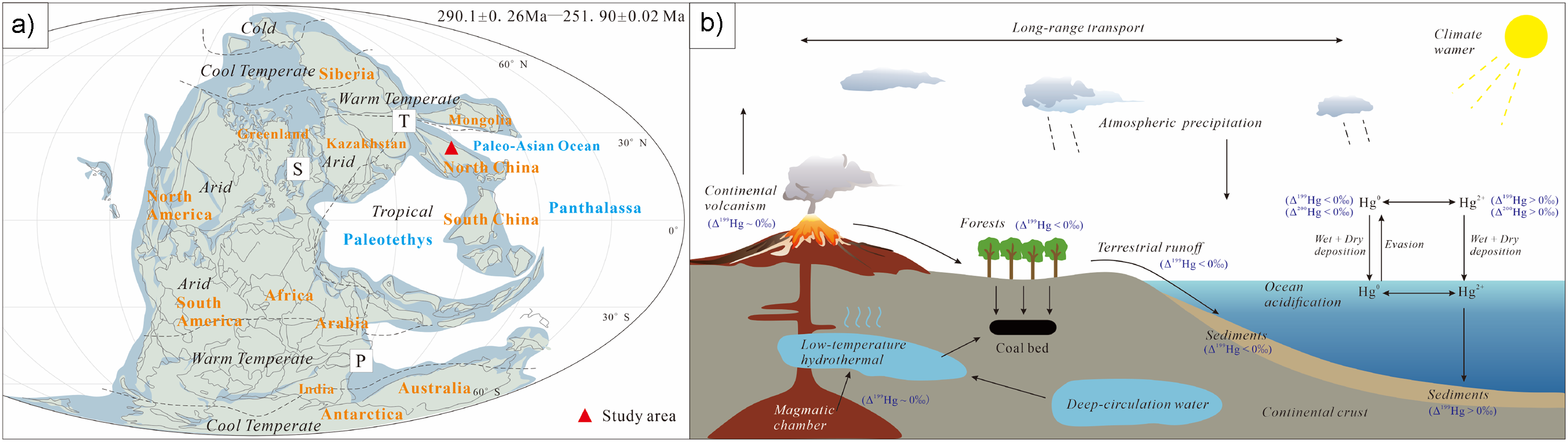
(a) Reconstruction of Artinskian–Lopingian paleogeography (after Boucot et al., 2013) showing the study area and major volcanic provinces. P: Panjal Traps; S: Skagerrak-centered large igneous province (LIP); T: Tarim LIP. (b) Schematic diagram illustrating harmful trace elements cycle under enhanced volcanic emissions in atmosphere, oceans, and land during the Early Permian.
The dominant host of mercury in Jinyuan Mine
Sulfide minerals
Mercury in coal is generally regarded as geochemically stable due to its strong binding with organic functional groups and its high affinity for sulfide minerals, which significantly limit its mobility within the coal matrix (Yudovich and Ketris, 2005). The pyrite content in the LTA of coal seam samples JY-3-1 and JY-3-9 is significantly higher compared to other coal samples, suggesting that these samples may have undergone specific mineral enrichment processes during deposition, which could be closely related to the depositional environment, temperature, and redox conditions of the coal seams. The correlation coefficient between mercury concentration and total sulfur (TS) is notably high (r = 0.93, p < 0.05) (Figure 6). Additionally, in coals from Indonesia, Romania, Mongolia, and parts of China, the correlation between total sulfur and mercury (R² = 0.31, p < 0.01, n = 95) (Sun et al., 2016; Tewalt et al., 2010; Yin et al., 2014) suggests that their accumulation and retention within the coal matrix may be governed by comparable geochemical processes. This highlights the significant role of the mercury-sulfur coexistence in geochemical cycling. The strong correlation between Hg and Fe (r = 0.67, p < 0.05) further supports the hypothesis that mercury is mainly associated with pyrite (FeS2), which serves as an effective host for Hg sequestration during diagenesis. The occurrence of vein-fill pyrite (Figure 9(a)) and chloritized kaolinite (Figure 9(b)) suggests potential remobilization of mercury-bearing sulfides due to hydrothermal fluid influence.
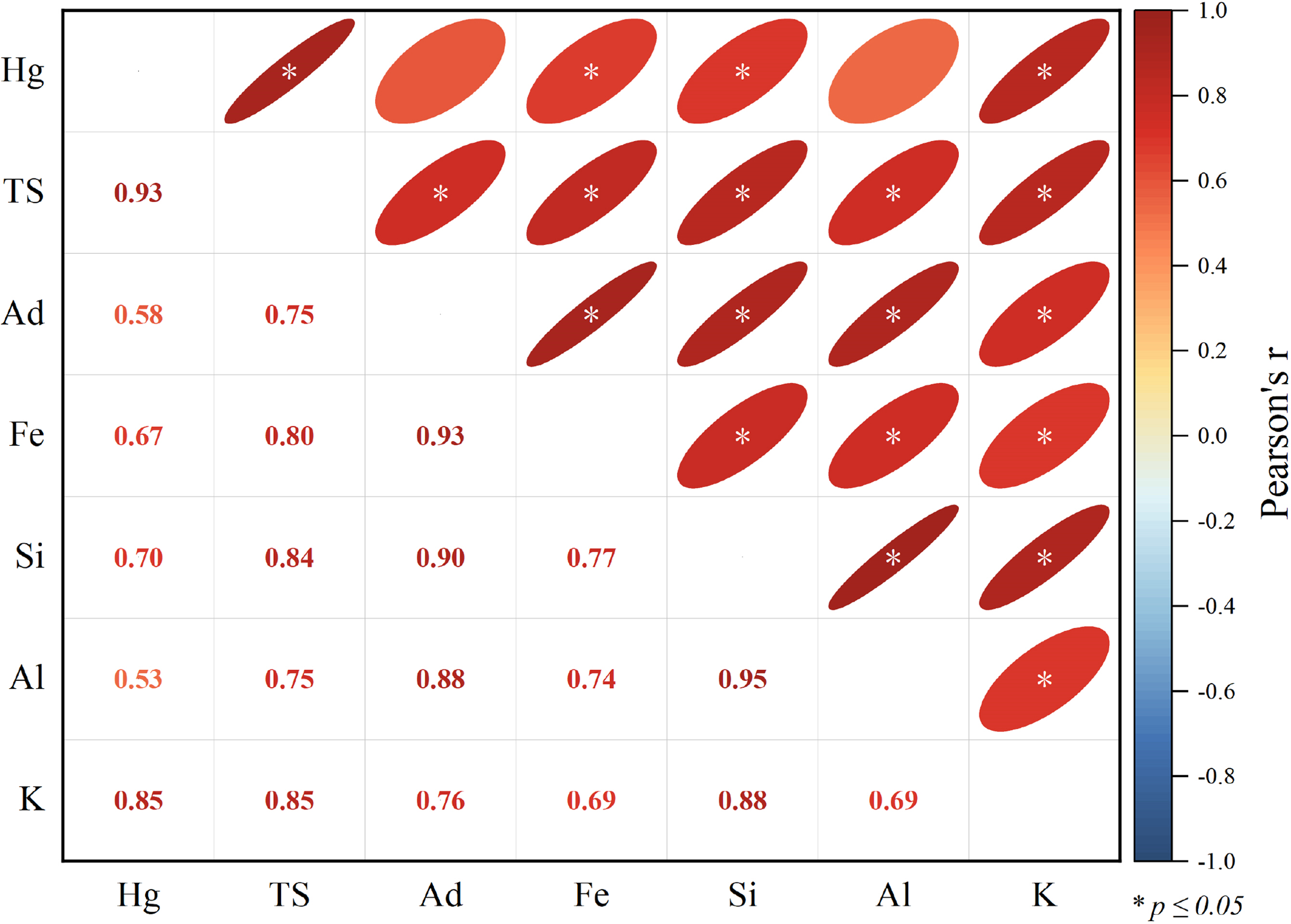
Pearson's r correlation coefficients for TS, Ad, SiO2, Al2O3, K2O, Fe2O3 (%), and Hg (ppb) pairs at Jinyuan. Red and blue ellipses (upper right) and numbers (lower left) represent positive and negative correlations, respectively. Asterisks denote correlations significant at the 3σ level (p ≤ 0.05).
Additionally, the correlation between Hg concentration and K (r = 0.85, p < 0.05) and Fe (r = 0.67, p < 0.05) concentrations is high in this coal seam (Figure 6). Meanwhile, TS is positively correlated with K (r = 0.85, p < 0.05) and Fe (r = 0.80, p < 0.05). Many pyrite and sulfate minerals (Figure 7(a) to (f)) can be observed under an electron probe micro-analyzer equipped (EPMA; EDS), indicating that pyrite is likely the main host of mercury in coal. Pyrite is predominantly observed in samples JY-3-1 and JY-3-9, exhibiting diverse morphologies (Figure 7(a) to (f)). Pyrite occurs as isolated or clustered nodules, euhedral to subhedral crystals, cavity fillings, and massive forms, indicating a syngenetic or early diagenetic origin. These results highlight that sulfide-hosted Hg is a dominant phase in the studied coal seams, but the contribution of clay minerals should not be overlooked.

EPMA back-scattered electron images for the distinct occurrences of pyrite (Py) from samples JY-3-1 and JY-3-9. Qz: quartz; Ill: illite; Kln: kaolinite.
Clay minerals
In the No. 3 seam, mercury concentrations exhibit a positive correlation with Ad (r = 0.58, p < 0.05), Si (r = 0.70, p < 0.05), K (r = 0.85, p < 0.05), and Al (r = 0.53, p < 0.05) concentrations (Figure 6). Notably, samples JY-3-1 and JY-3-9 exhibit elevated mercury levels alongside significant concentrations of illite and quartz (Guo et al., 2020). Due to their expansive surface area and numerous reactive sites, terrigenous sediments, particularly clay minerals and other alumina-rich components, possess a significant capacity for heavy metal adsorption (Altın et al., 1998; Uddin, 2017). The strong Hg–K and Hg–Al correlations suggest that clay minerals serve as important secondary reservoirs for mercury in the coal seam. The observed TS–Hg relationship, in combination with elevated Hg levels in JY-3-1 and JY-3-9, implies that mercury initially adsorbed onto clay minerals may have undergone post-depositional redistribution, leading to secondary fixation in sulfide minerals.
These characteristics suggest that clay minerals may serve as primary carriers for harmful trace elements within the samples. These correlations might not only signal mercury bound to clay but also indicate a post-depositional redistribution of terrigenous mercury to sulfides, as evidenced by even more robust total sulfur-mercury (TS–Hg) relationships (Figure 6). In marine environments, after sedimentation, mercury bound to particles can desorb from clay minerals during diagenesis and be absorbed by other sedimentary components such as organic matter and sulfides from pore water (Figure 5(b)) (Shen et al., 2020; Them et al., 2019).
Hg isotope evidence: Identify the source of harmful trace elements during the Early Permian
This article that uses δ202Hg, Δ199Hg, and Δ200Hg represents MDF, odd-MIF, and even-MIF for mercury isotopes. The interpretation of MDF data is often fraught with complexity and uncertainty, making source tracing particularly challenging (Bergquist and Blum, 2007; Blum et al., 2014). In this study, δ202Hg values are primarily negative, with a variation range of 1.6‰ (−3.73 ‰–−2.13 ‰ ± 0.04 ‰) (Table 2). This expands the negative characteristic range of δ202Hg (−2.64 ‰∼0.77 ‰) in Chinese coal (Sun et al., 2014) summarized by predecessors. The two mercury-enriched coal layers (JY-3-1, JY-3-9) have relatively high δ202Hg characteristics (−2.42 ‰, −2.21 ‰) compared to the middle coal layer (Table 2). However, these ranges are not the only diagnostic ranges for specific sources or processes. Consequently, we refrain from interpreting the MDF data from our study sections. Studies on the even-MIF of mercury isotopes have predominantly been reported in atmospheric precipitation. This suggests that the isotopic composition of Hg in the atmosphere may be influenced by a range of climatic and environmental factors, particularly during the precipitation process. Nonetheless, to fully understand the geochemical pathways influencing Hg isotope variations, we incorporate a discussion on photochemical, microbial, and geochemical redox processes that may have contributed to the observed isotope trends.
Volcanic activity
Mercury enrichment in coal deposits is primarily attributed to volcanic emissions, with subsequent modifications by hydrothermal activity. Within the corpus of mercury anomalies, discernible parallels and disparities emerge. Distinct large igneous province (LIP) occurrences or discrete phases within a singular LIP event may exhibit divergent profiles of mercury enrichment. Natural emissions of volcanic and/or hydrothermal Hg typically exhibit Δ199Hg values close to zero, whereas terrestrial reservoirs, such as soil and biomass, which accumulate gaseous elemental mercury (Hg0), tend to display slightly negative Δ199Hg values (Figure 5(b)) (Beckers and Rinklebe, 2017; Blum et al., 2014; Cooke et al., 2020; Jin et al., 2022). Conversely, the Δ199Hg values observed at JY-3-1 (0.02‰) and JY-3-9 (0.08‰) fall within the range typical of volcanogenic Hg (0.02 ± 0.06‰ (Yin et al., 2016)). Whether from terrestrial volcanoes or submarine hydrothermal systems, their initial Δ199Hg values are typically close to 0‰, which results in similar mercury isotopic signatures (Blum and Bergquist, 2007; Percival et al., 2015; Shen et al., 2019a; Sherman et al., 2009; Sial et al., 2016). As mentioned above, Δ199Hg in (No. 3) of the Jinyuan Mine sections either approach zero or exhibit slight positive or negative offsets (Figure 8), indicating that the primary source of mercury is volcanic emissions, which, upon undergoing photochemical reduction in the atmosphere, results in varying degrees of isotopic fractionation (Shen et al., 2019b).
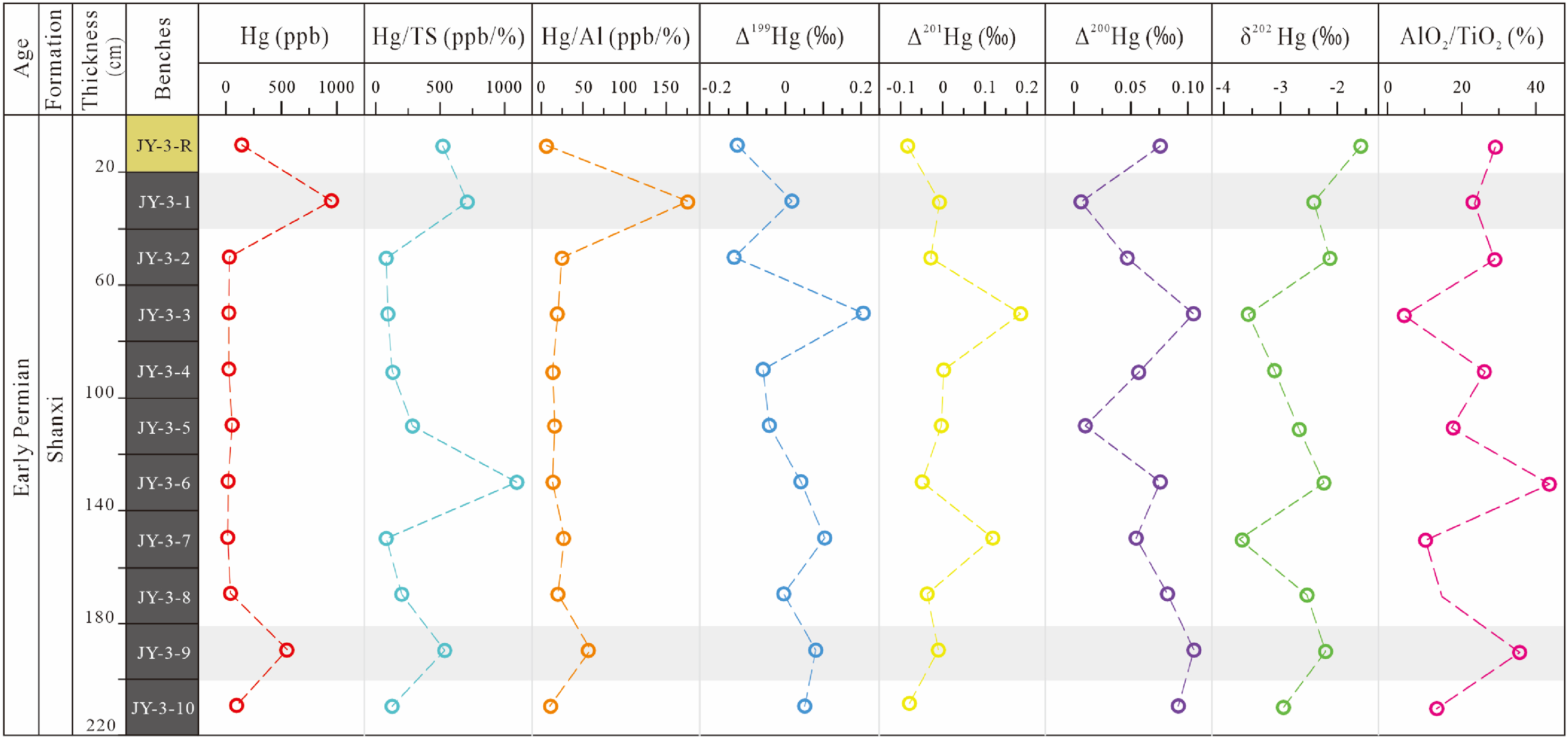
Vertical variations of Hg, Hg/TS, Hg/Αl, Δ199Hg, Δ201Hg, Δ200Hg, δ202Hg, ΑlO2/TiO2 in No. 3 coal of Jinyuan Mine.
During volcanic eruptions, volcanic debris and mineral particles can be transported to nearby areas by wind or water currents, including coal deposition environments. Such eruptions span tens to hundreds of thousands of years, facilitating the rapid release of mercury into the atmosphere, followed by its prompt deposition within strata over a concise temporal window (Figure 5(b)). In sample JY-3-9, the presence of detrital zircon (Figure 9(d)) typically indicates that these zircons, originating from volcanic ash, have undergone abrasion during transport and have been deposited as detrital grains within the sedimentary matrix. Additionally, Grasby et al. (2019) proposed that the enrichment of various metals is closely linked to volcanic eruptions, particularly elements such as arsenic, cadmium, cobalt, chromium, copper, manganese, lead, antimony, selenium, vanadium, and zinc. In this study, the CC for harmful trace elements of samples were calculated (Figure 3). According to the data, the coal strata JY-3-1 and JY-3-9 were enriched with most harmful trace elements compared with other samples.
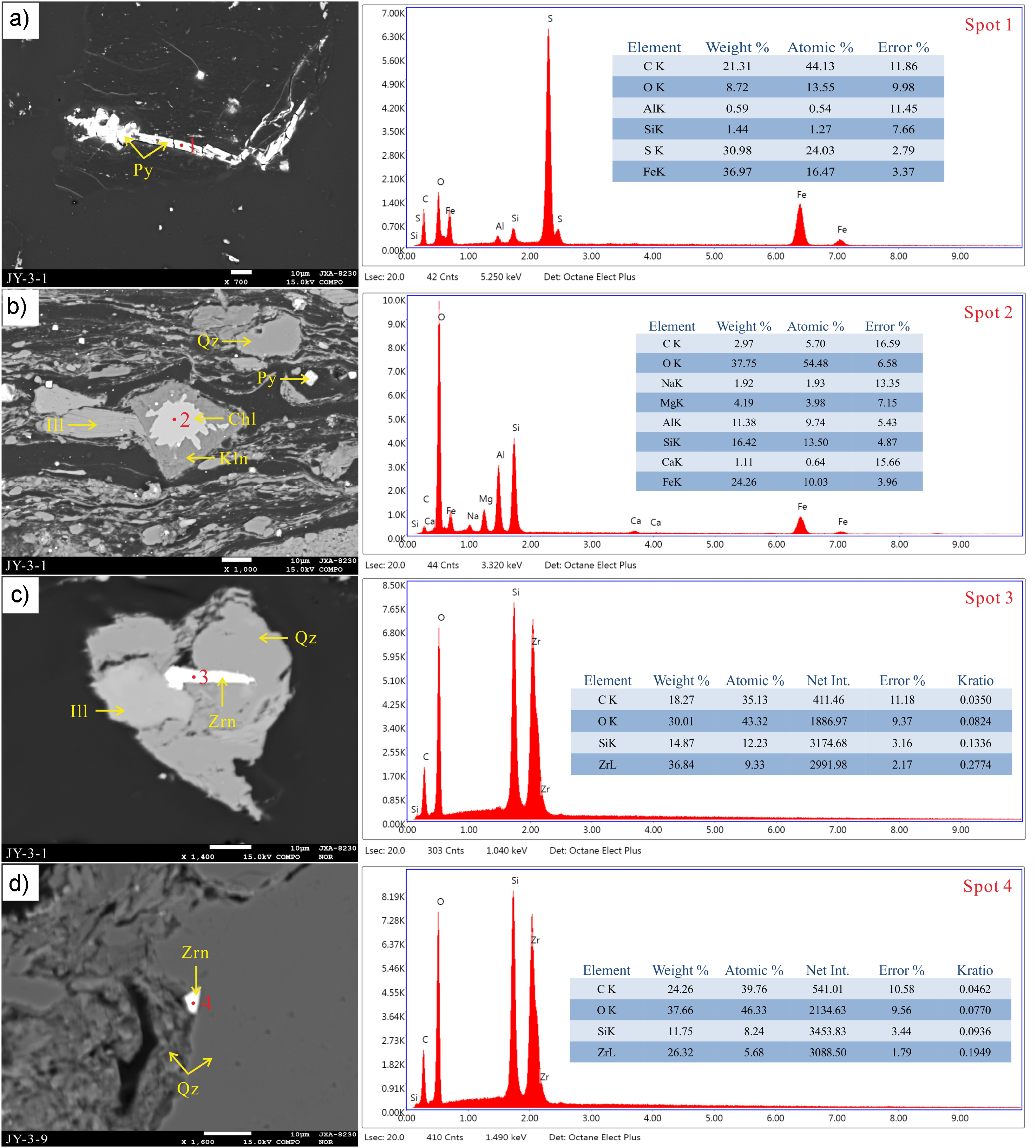
EPMA back-scattered electron images and selected EDS spectra of pyrite (Py), quartz (Qz), illite (Ill), kaolinite (Kln), chlorite (Chl), and zircon (Zrn) of samples JY-3-1 and JY-3-9.
Under electron probe microscopy, we observed a minor occurrence of chloritization in kaolinite in sample JY-3-1 (Figure 9(b)). Chloritization typically occurs during diagenesis and post-diagenesis, resulting from the interaction of iron-rich hydrothermal fluids with kaolinite or the deposition of these fluids. This transformation indicates that the sample has undergone significant burial and elevated thermal metamorphic conditions, suggesting a shift in the environmental conditions from acidic to neutral or alkaline (Dai et al., 2014; Dill, 2016). Concurrently, the presence of fracture-filling pyrite (Figure 9(a)) generally signifies hydrothermal activity. Such activity may be associated with regional volcanic events or other geological thermal events (Deditius et al., 2014; Kusebauch et al., 2018; Pokrovski et al., 2021; Reich et al., 2013; Schoonen and Barnes, 1991). Hydrothermal fluids migrate through fractures, depositing sulfide minerals such as pyrite at lower temperatures. This phenomenon indicates the existence of localized reducing conditions, implying that the sample may have experienced significant hydrothermal alteration. Additionally, we detected vein-like zircon (Figure 9(c)) and abundant secondary vein-like carbonate minerals, along with paragonite, in sample JY-3-9. The formation of these minerals is intricately linked to the migration of hydrothermal fluids carrying mineral substances through fractures and faults, subsequently depositing under relatively low temperature and pressure conditions.
However, the vitrinite reflectance of these two samples did not exhibit significant changes compared to other coal measures (Guo et al., 2020). This observation suggests that the multiple stages of hydrothermal fluid influencing the mineral anomalies in the Jinyuan Mine were not characterized by high temperatures. These hydrothermal fluids, known for their high reactivity, are capable of dissolving mercury and other metals through leaching from intrusive and surrounding rocks, providing a crucial pathway for mercury enrichment. Hydrothermal intrusion into coal beds occurs during the early stages of peat accumulation and diagenesis, as well as in the later stages along faults and fractures during post-depositional and metamorphic processes. This intrusion results in significant enrichment of mercury and other harmful elements, such as arsenic, selenium, antimony, and thallium, often by several orders of magnitude.
Atmospheric precipitation
The photoreduction process of aqueous Hg2+ in atmosphere or aquatic environments results in negative odd-MIF signals (Δ199Hg and Δ201Hg) in the elemental mercury (Hg0) produced, while positive odd-MIF is seen in the remaining Hg2+ species (Bergquist and Blum, 2007; Blum et al., 2014; Costa and Liss, 2000; Yin et al., 2014, 2015, 2016). In contrast, significant even-MIF signatures of mercury (Δ200Hg) have been purportedly exclusively engendered during the photooxidation of gaseous Hg0 within the tropopause (Chen et al., 2012). In this context, the oxidized forms of Hg2+ exhibit discernible positive Δ200Hg signals, while gaseous Hg0 manifests negative Δ200Hg values (Figure 5(b)) (Chen et al., 2012; Demers et al., 2013; Gratz et al., 2010; Rolison et al., 2013; Zerkle et al., 2020). The Δ199Hg/Δ201Hg ratio characteristics of odd isotopes have been widely used to identify and distinguish processes that cause non mass fractionation in nature. A plot of Δ199Hg versus Δ201Hg in all our samples shows a Δ199Hg/Δ201Hg regression slope of 0.87 (n = 11, r = 0.70) (Figure 10), which is consistent with the Δ199Hg/Δ201Hg values observed during the photoreduction of inorganic Hg2+ associated with dissolved organic matter in aquatic environments. This suggests that the photoreduction mechanism plays a significant role in the study area, and the mercury isotopic fractionation effect is likely consistent across different environmental conditions (Bergquist and Blum, 2007; Chandan et al., 2015; Estrade et al., 2009; Sun et al., 2016; Zheng et al., 2018, Zheng and Hintelmann, 2009). Furthermore, the high Hg/TS ratio in JY-3-1 and JY-3-9 often points to increased mercury fixation by sulfides (Figure 8). This also association is typical in environments where reducing conditions facilitate the formation of mercury sulfides (Bian et al., 2022, 2024; Kim et al., 2022).
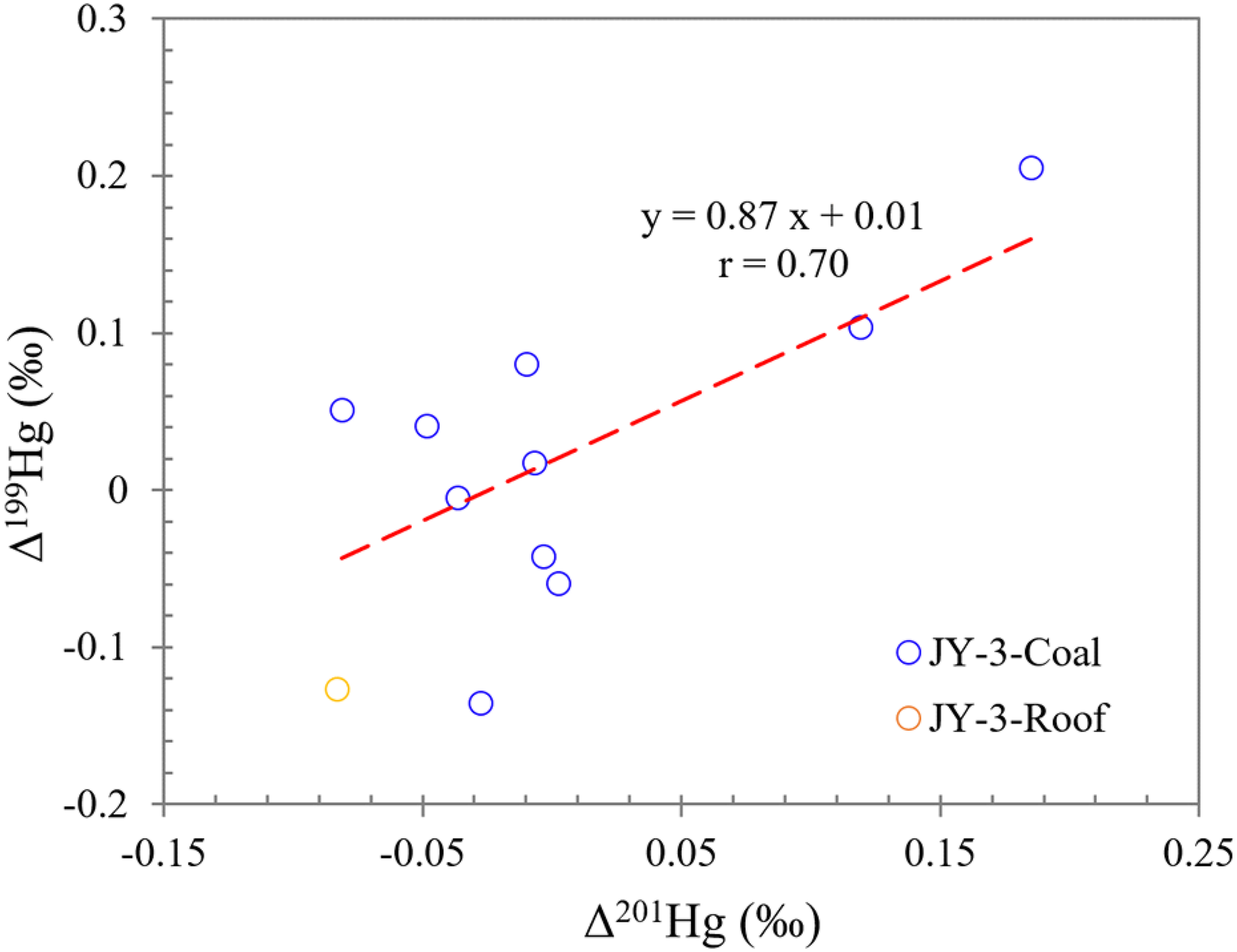
Crossplots of Δ199Hg versus Δ201Hg.
In all of our samples, the Δ199Hg and Δ200Hg values are positive. Specifically, in sample JY-3-1, Δ199Hg and Δ200Hg are recorded at 0.02‰ and 0.01‰, respectively, whereas in sample JY-3-9, these values are 0.08‰ and 0.11‰, respectively. The higher Δ199Hg and Δ200Hg values in sample JY-3-9 may indicate a more dynamic photochemical environment, potentially associated with higher rates of organic matter degradation or more intense solar irradiation at this location. Conversely, the lower values observed in sample JY-3-1 suggest a more constrained geological setting or limited photochemical activity. One hypothesized mechanism involves the photoreduction of Hg2+ within a sulfide-rich photic zone. In oxic surface seawater, the photoreduction of oxygen-bound Hg2+ tends to be the predominant process, resulting in positive MIF signals in the residual Hg2+. Conversely, in euxinic seawater where sulfur-bound Hg2+ prevails, photoreduction typically generates negative MIF signals. This disparity in photoreduction dynamics can contribute to the observed negative shift in MIF within euxinic sediments (Motta et al., 2020; Zheng and Hintelmann, 2010). All these signatures of MIF indicate that the mercury and associated harmful trace elements in our samples, regardless of their origins, underwent aqueous photoreduction processes prior to sedimentation.
Terrigenous input
In principle, atmospheric gaseous mercury (Hg0) can enter the terrestrial reservoir through two primary mechanisms. Initially, it undergoes a photoreaction, leading to its conversion into oxidized mercury, specifically Hg2+, subsequently deposited through either dry or wet deposition processes. Alternatively, mercury can directly be absorbed by plants or soil, where it undergoes oxidation, prior to its integration into the ocean via surface runoff (Thibodeau and Bergquist, 2017). This latter route constitutes the terrestrial influx of mercury, known as the terrestrial input source contributing to mercury levels (Holmes et al., 2010). The MIF caused by photochemical reduction yields a positive Δ199Hg signature within the remaining Hg2+ reservoir (Figure 8). This signature is then preserved in sedimentary environments through wet or dry deposition processes. As seen in the analysis of mercury-bearing carriers mentioned above, mercury is also present in clay minerals in addition to sulfide minerals. In sedimentary rocks, the Al2O3/TiO2 ratio serves not only as a provenance indicator but also as a reflection of the mineralogical maturity of the source rocks (Sugitani et al., 1996). Elevated ratios typically indicate origins from felsic rocks, which are rich in aluminum-bearing minerals such as clays and feldspars. The congruence of this ratio with the δ202Hg profile underscores the significant influence of terrigenous sediments on the concentration of harmful trace elements in coal (Figure 8). These results align with those observed through Electron Probe Microanalysis (EPMA) (Figures 7 and 9). It is important to note that the elevated Hg/Al ratios observed in the JY-3-1 and JY-3-9 samples may indicate an increase in terrigenous input, characterized by a relative depletion of alumina-rich minerals, such as clays. This suggests a preferential enrichment of mercury relative to more stable crustal elements like aluminum. Such a trend could reflect differential transport or depositional processes that selectively concentrate mercury, potentially implicating environmental influence that alter the natural geochemical baseline (Liu et al., 2023; Shen et al., 2020).
Additionally, vegetation predominantly sequesters mercury by absorbing Hg0 from the atmosphere. Previous research has indicated that terrestrial reservoirs accumulating Hg0 typically exhibit slightly negative Δ199Hg values. Recent experiments on forest ecosystems have indicated that leaf mercury bound with sulfur ligands produces negative Δ199Hg values (−0.4‰ to −0.2‰) (Demers et al., 2013; Sun et al., 2016; Yin et al., 2014) within plant foliage. It follows that the elevated mercury layers JY 3-1 (0.02 ‰) and JY 3-9 (0.08 ‰) in the study area (Table 2) remained unaffected by the presence of indicator plants.
Conclusions
The Jinyuan coal is characterized by low ash content and high volatility, making it a type of A-bituminous coal. The overall content of oxides of major elements in coal is relatively low, with SiO2 and Al2O3 being the main components. The average values of MgO and CaO are slightly higher than the Chinese coal average, while the average contents of other constant elements are lower than the Chinese coal average. The Al2O3/TiO2 ratio suggests that the sedimentary parent rock of the No. 3 coal in the Tengnan Coalfield is of intermediate acidity. The harmful trace elements Be, F, V, Cr, Cu, Se, Mo, Tl, Pb, U, and Hg show abnormal enrichment in the stratigraphic samples JY-3-1 and JY-3-9 from the Tengnan Coalfield.
In the low-temperature hydrothermally altered coal seams of the Jinyuan Mine in southwestern Shandong Province, sulfide minerals and clay minerals are likely the principal carriers of mercury. While the mercury bound to clay provides clear correlational signals, it is hypothesized that due to their more pronounced total sulfur to mercury (TS–Hg) relationships, these phases may reflect the redistribution of terrigenous mercury into sulfides post-deposition. Integrating mercury isotope analysis with a suite of analytical techniques, such as geochemical analysis, mineralogical studies, and geological surveys, has elucidated that the sources of harmful trace elements in the No. 3 coal of the Jinyuan Mine are primarily influenced by volcanic activity, atmospheric precipitation, and input of terrigenous sediments, with a minor contribution from coal-forming vegetation. This comprehensive approach enhances our understanding of the intricate interplay between geological processes and the distribution of trace elements in sedimentary environments. This comprehensive approach enhances our understanding of the intricate interplay between geological processes and the distribution of trace elements in sedimentary environments.
The isotopic approaches used in this study provide a framework for identifying mercury sources in other coalfields affected by volcanic and hydrothermal processes. Based on our Hg isotope data, the observed fractionation patterns indicate a strong influence of volcanic and hydrothermal sources, which may contribute to higher volatility and reactivity of Hg during combustion. The potential for Hg release from these enriched seams underscores the need for effective emission control technologies in coal-fired power plants, such as activated carbon injection (ACI) and flue gas desulfurization (FGD) systems, which have been proven effective in reducing Hg emissions (Streets et al., 2009; Zhang et al., 2016). Our study provides valuable geochemical insights that can inform clean coal utilization strategies. By identifying specific coal seams with elevated Hg concentrations and characterizing their isotopic signatures, this research contributes to targeted coal management practices, including selective mining, coal blending, and advanced mercury removal technologies. Additionally, the application of Hg isotopes in source attribution can aid policymakers in designing more effective environmental monitoring programs and regulatory measures to mitigate mercury pollution. Further research is needed to integrate Hg isotopic analysis with comprehensive atmospheric and environmental monitoring programs. Expanding the database of Hg isotopic compositions in coal from different geological settings will enhance our understanding of global mercury cycling and its environmental implications. Moreover, interdisciplinary approaches combining geochemistry, atmospheric science, and environmental policy are essential to developing holistic strategies for mercury pollution control.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Natural Science Foundation of Hebei Province (D2021402017), Science and Technology Project of Hebei Education Department (JZX2023010), and Central guide local fund for scientific and technological development of Hebei Province (246Z4103G).