Abstract
The migration and diffusion of coalbed methane (CBM) are impeded by adsorption in pore channels, resulting in a significant portion of CBM being trapped in the reservoir and thereby reducing the efficiency of CBM extraction. Investigating the adsorption and diffusion behavior of CBM in reservoirs is crucial for optimizing CBM extraction. In this study, we developed a theory based on the basic geological characteristics of coal beds and the adsorption state of CBM, focusing on the transition state of adsorption gas diffusion of surface diffusion gas molecules. We explored the impact of various factors on the diffusion coefficients and adsorption capacity of CBM, elucidating the underlying reasons for variations in CBM adsorption and diffusion through molecular dynamics simulations. Our findings reveal that CBM undergoes simultaneous adsorption and desorption on the surface of coal beds, with adsorption conforming to the Langmuir monomolecular layer adsorption. Increasing reservoir temperature leads to a reduction in methane adsorption and a gradual increase in diffusion coefficient. For instance, at 80 °C, methane adsorption decreases to 0.5 mg/m3, while the diffusion coefficient increases to 4 × 10−9 m2/s. Moreover, the presence of water in coal beds significantly weakens CBM adsorption capacity, with a water content of 0.5% resulting in a minimal adsorption capacity of 0.5 mg/m3. Additionally, increasing coal bed pressure enhances CBM adsorption capacity but decreases the diffusion coefficient. For example, a reservoir pressure of 30 MPa increases CBM adsorption capacity to 0.65 mg/m3, while reducing the diffusion coefficient to 2.5 × 10−9 m2/s. Overall, our study demonstrates that geological conditions, such as temperature, water content, and pressure, play crucial roles in shaping CBM adsorption and diffusion behavior. By understanding these environmental factors, we can effectively manipulate CBM diffusion and adsorption capacity, thereby providing essential insights and foundational data for CBM exploitation.
Keywords
Introduction
Coalbed methane (CBM), a distinct gas adsorbed in coal, is typically extracted from reservoir depths deeper than those of conventional natural gas wells (Dong et al., 2020). At equivalent reservoir depths and pressures, coal beds exhibit two to three times greater methane sequestration capacity compared to conventional gas reservoirs (Mohamed and Mehana, 2020). As conventional oilfield energy sources such as oil and natural gas gradually deplete, the development, production, and processing of CBM have emerged as a new industry and research focus in the oil and gas sector (Wang et al., 2017). However, the behavior of CBM differs from that of natural gas in conventional oilfield reservoirs, with CBM diffusion in coal bed micropores and adsorption on solid surfaces being key factors influencing CBM production (Cai et al., 2014).
Diffusion, the migration of methane molecules under gas concentration gradients, is closely related to natural gas desorption on coal bed surfaces (Zhang et al., 2016). Based on the molecular mean free range and porous medium pore size, diffusion models include Fick diffusion, Knudsen diffusion, and transition diffusion, with current research mainly focusing on the Knudsen diffusion model in coal methane diffusion studies (Meng and Li, 2016).
Numerous scholars have conducted extensive investigations into CBM adsorption and diffusion in coal seam fractures or on coal seam surfaces (Dong et al., 2017). Li et al. (2022) experimentally examined the effects of pressure and temperature on gas diffusion, finding that diffusion capacity increased with temperature under constant pressure. Zhang et al. determined methane gas diffusion coefficients in coal micropores and observed that coal reservoir pressure, methane concentration, and water content affected these coefficients (Pillalamarry et al., 2011). Pillalamarry et al. studied and evaluated methane adsorption and diffusion characteristics in Illinois basin coals, observing negative correlations between diffusion coefficients and pressure in low-pressure ranges and positive correlations with surface coverage and pressure (Zhang et al., 2017).
Although these studies contributed valuable insights into methane adsorption and diffusion in CBM, each lacked comprehensive detail for specific CBM mining operations (Zhang et al., 2017). Nevertheless, their findings provide a foundation for practical gas diffusion applications in coal. Additionally, Liu et al. explored how coal seam conditions affect CBM extraction and diffusion, finding that increased coal seam temperature significantly reduced CBM diffusion coefficients, resulting in a 25% decrease in extraction efficiency. Substantial CBM entrapment in reservoir fractures due to strong pore adsorption presents significant challenges for efficient CBM extraction (Wang et al., 2024).
This study addresses gaps in existing CBM research by establishing a theory of adsorbed gas diffusion transition state for surface-diffusing gas molecules. We explore diffusion characteristics and the effects of various factors such as reservoir pressure, temperature, water content, and fracture width on methane gas diffusion. Furthermore, we propose correlations between diffusion coefficients and these factors, shedding light on differences in CBM adsorption and diffusion through molecular dynamics simulations. Our findings provide practical data support for effectively guiding CBM exploitation.
Materials and methods
Test samples
Coal briquettes for the experiments were sourced from two coal seams in the Ordos region of the Inner Mongolia Autonomous Region. The lumps were subsequently pulverized and sieved to obtain fines ranging from 50 to 70 mesh, with an average particle size of ∼1.32 × 10−4 m. The pulverized coal was exposed to reservoir temperature (65 °C) and humid conditions in an environmental chamber for 70 h to reach moisture equilibrium.
Test apparatus
The experimental setup for studying the diffusion and adsorption of CBM in reservoir rock is illustrated in Figure 1. Initially, both ends of the core holder are saturated with CBM, with no pressure differential across the coal sample. Additionally, the core holder is equipped with temperature and pressure sensors to regulate and monitor the temperature and pressure conditions. Throughout the experiment, it is crucial to maintain a constant pressure differential across the core and peripheral pressure of the core holder while investigating the gas diffusion coefficient and adsorption capacity of the natural gas equilibrated within the coal seam fractures (Busch et al., 2004).
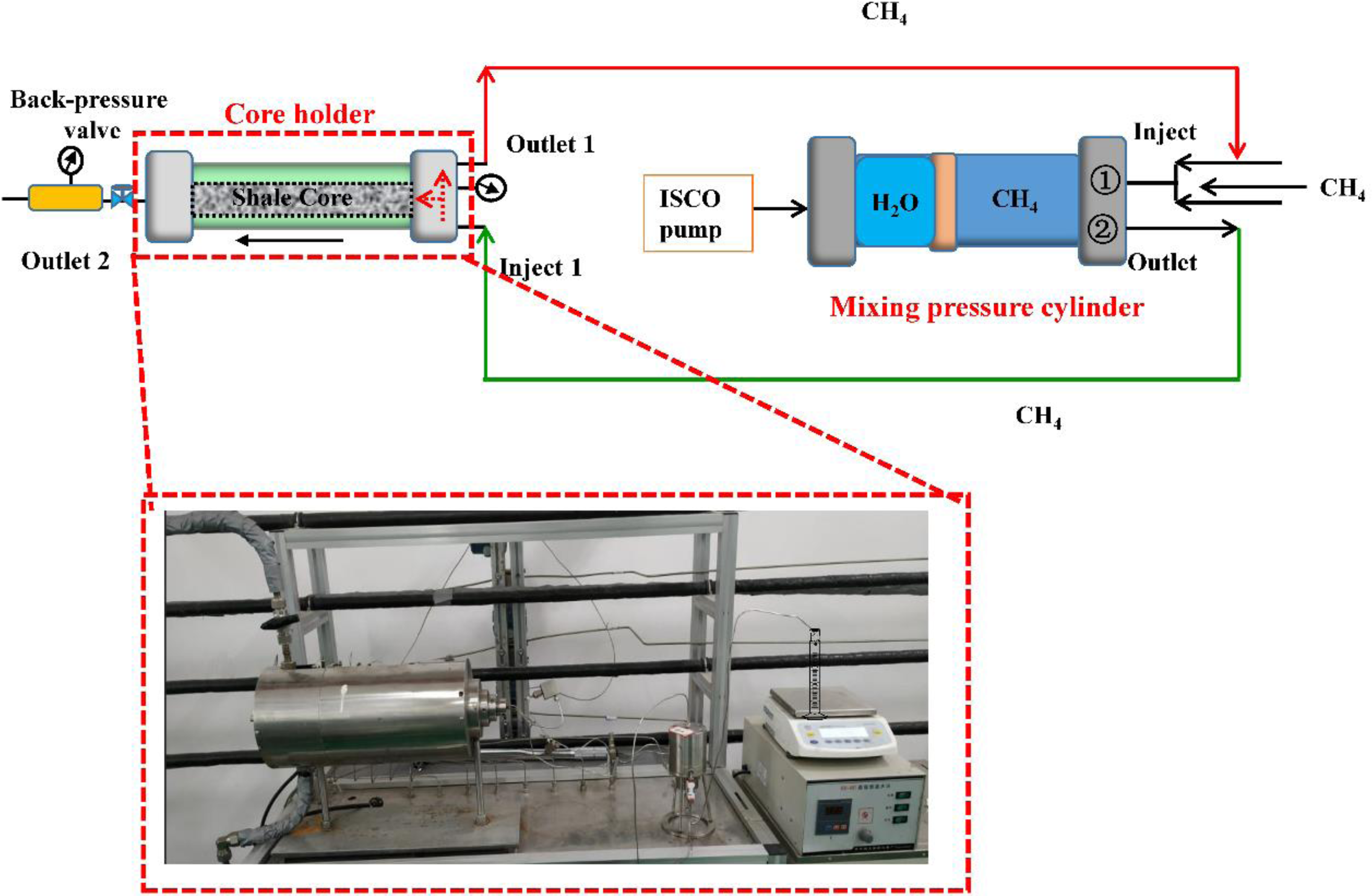
Equipment used for expansion and adsorption of coalbed methane.
The entire experimental setup mainly consists of a compression pump, a pressure-resistant steel cylinder, and a core holder. Firstly, the pressurized methane gas enters the pressure-resistant steel cylinder through the inlet on the right (①), which is mainly used to ensure the appropriate pressure of methane-simulating reservoirs. The pressurized methane, upon completion of pressurization, is injected into the core holder through the outlet of the pressure-resistant steel cylinder (②) under the action of the ISCO pump. These pressurized methane gases continuously enter the core under the thrust force to realize the adsorption and diffusion experiments of CBM. The experimental temperature is controlled by the heating wire around the core holder to achieve a constant temperature environment and temperature regulation, while the water content needs to be pre-saturated in a mist environment (Wang and Liu, 2016).
Adsorption isotherms
The isotherm theory is commonly used to explain the adsorption or diffusion properties. The Langmuir, Freundlich, and Brunauer–Emmett–Teller models are common types of models for adsorption of substances, with Langmuir models being widely accepted in the petroleum industry and CBM studies (Zhu et al., 2020).
The Langmuir adsorption isotherm assessment equation used in this study is shown in equation (1) (Xiangchun et al., 2012):
According to equation (1), this study can quantify the adsorption behavior and adsorption capacity of CBM on the coal bed surface.
Measurement of diffusion coefficients
The adsorption and diffusion coefficients of CBM on the surface of a coal seam can be evaluated and measured by using equation (2) (Yang et al., 2019):
Results and analysis
Influence of water content of coal briquettes on diffusion and adsorption of CBM
The CBM diffusion experiments were conducted on samples with an in situ temperature of 80 °C and a pressure of up to 30 MPa. The influence curves of different coal block water contents on the diffusion and adsorption performance of CBM in the cracks of coal blocks are illustrated in Figure 2, and it is evident that the pore diffusion of CBM is directly proportional to the water content of coal blocks. The data in Figure 2 indicate that increasing the water content of coal briquettes directly enhances the diffusion performance and extraction of CBM in the cracks of coal briquettes, while significantly reducing the adsorption capacity of CBM. Specifically, at 0% water content of coal briquette, the adsorption amount of CBM reached 0.9 mg/m3, with a diffusion coefficient of only 1.5 × 10−9 m2/s. As the water content gradually increased to 0.5%, the changes in the adsorption amount and diffusion coefficient of CBM were minimal, with the diffusion coefficient only elevated to 1.9 × 10−9 m2/s. However, at 1% water content of coal briquettes, there was a rapid desorption and a significant increase in the diffusion ability of CBM, with the adsorption amount reduced to 0.75 mg/m3, and the diffusion coefficient increased to 2.4 × 10−9 m2/s. The diffusion ability of CBM due to the 1% water content of coal briquettes was 2.1 times higher than that of 0.5% water content of coal briquettes, and the reduced adsorption of CBM predicted a notably increased extraction rate of CBM. Overall, the data in Figure 2 demonstrate that the CBM adsorption capacity is significantly reduced after gradually increasing the water content of coal briquettes to 1%, while the diffusion coefficient is increased, leading to a marked improvement in the extraction rate of CBM.
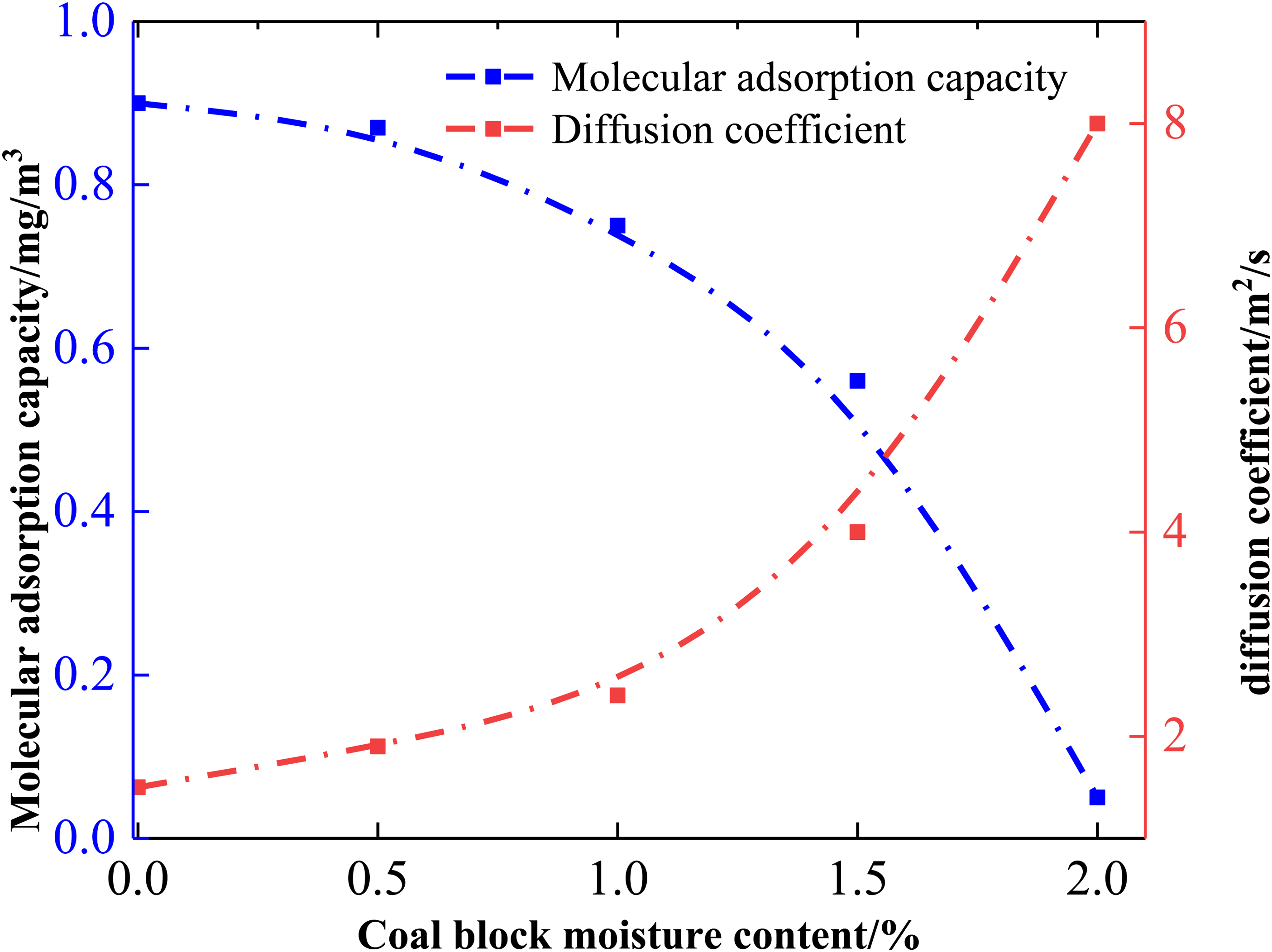
Curve of the influence of different coal briquette water content on the adsorption amount and diffusion coefficient of coalbed methane (CBM).
The reason for the decrease in adsorption due to the water content of coal briquettes may be related to the repulsive force between the water molecules in the coal briquettes and the natural gas molecules. As shown in Figure 3, when the water content of the coal block is 0%, both the natural gas molecules and the coal block molecules contain a large amount of carbon C element. Coal briquettes with the same polarity as natural gas can have a strong attraction (principle of similar dissolution), which means that natural gas molecules can have a strong adsorption capacity on the surface of coal briquettes (Wang et al., 2021). Molecular simulation data show that 0% water content of coal briquettes will form very short intermolecular interaction bonds (13 nm) with natural gas molecules and very large bond energies (35 × 10−5 kJ/mol). However, after the presence of water molecules (0.5%) in the coal block, there is a great intermolecular repulsion between water molecules and natural gas because of the difference in polarity between water molecules (element H) and natural gas molecules (element C). The chemical bond (16 nm) between the natural gas and the lump of coal gradually becomes longer, while the bond energy (30 × 10−5 kJ/mol) also appears to decrease significantly. Part of the CBM molecules adsorbed on the briquettes will gradually detach from the briquettes due to the presence of water molecules, and the diffusion coefficient increases as a result. The increase of water content in the coal briquette directly accelerates the degree of detachment and diffusion of natural gas molecules from the surface of the coal briquette, which is macroscopically manifested in the increase of the CBM extraction rate.
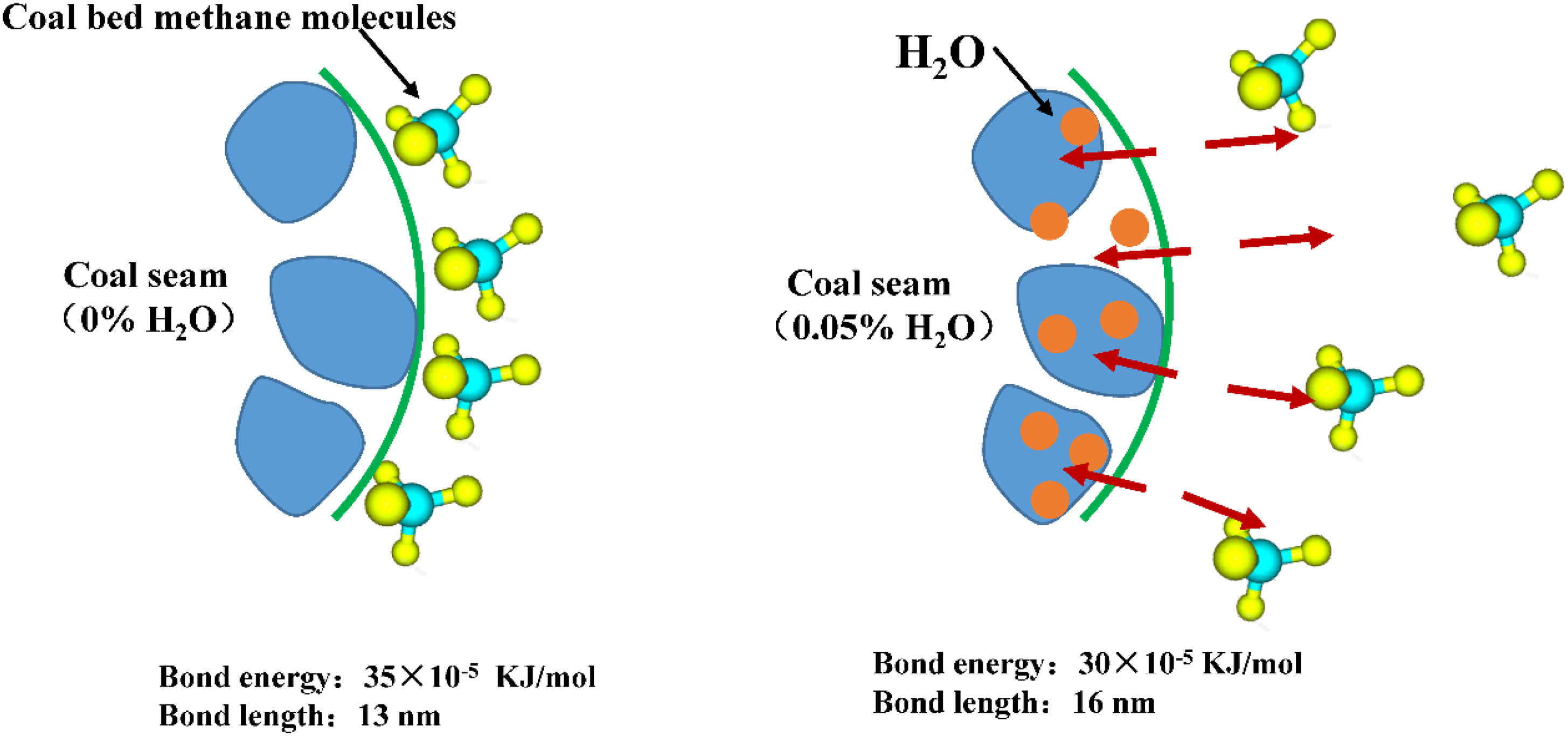
Schematic diagram of the force between coalbed methane (CBM) molecules and water molecules under different water contents of coal briquettes.
Influence of seam pressure on CBM diffusion and adsorption
Coal bed pressure, as a typical geological condition, is an important factor in analyzing the adsorption of CBM in the diffusion coefficient, and Figure 4 analyzes the changes of different coal bed pressures on the diffusion performance of CBM. The data in Figure 4 show that with the gradual increase of reservoir pressure, the diffusion coefficient decreases to different degrees, while the adsorption amount of CBM rises due to the increase of reservoir pressure (Yu et al., 2021).
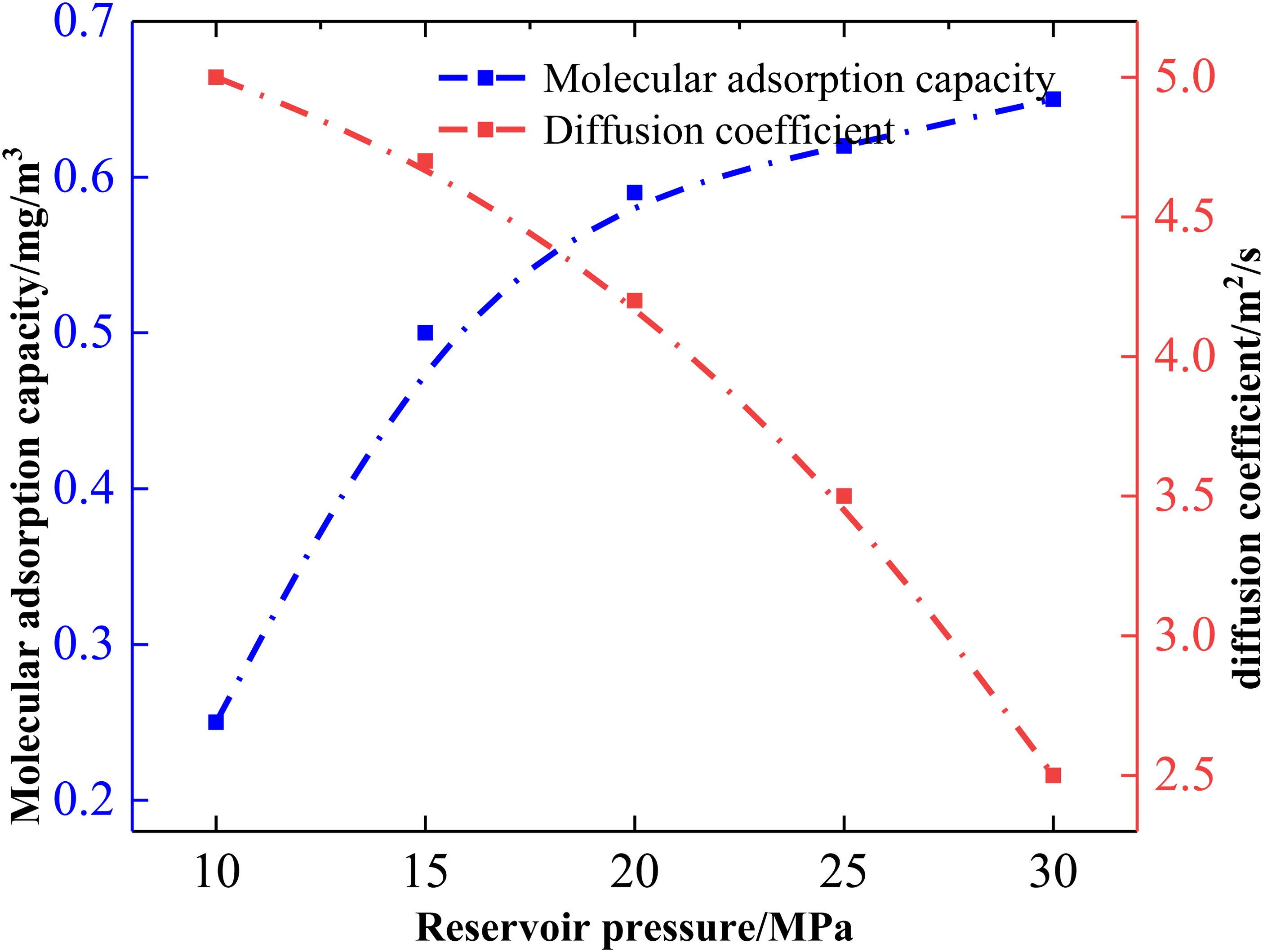
Effect of different reservoir pressures on adsorption and diffusion coefficients of coalbed methane.
However, the influence curve of reservoir pressure on the diffusion and adsorption of CBM shows different stage changes, with reservoir pressures below 15 MPa having a significant impact on the diffusion coefficient and adsorption amount of CBM. At 10 MPa, the coal seam pressure can cause the adsorption amount of CBM to reach 0.25 mg/m3, with a natural gas diffusion coefficient of only 5 × 10−9 m2/s. Lower reservoir pressures exhibit a larger adsorption amount of natural gas molecules in coal seam pores and a smaller gas diffusion coefficient. However, the adsorption amount and diffusion coefficient of CBM will change significantly after the reservoir pressure reaches 15 MPa, with the above two values changing by +0.25 mg/m3 and −1 × 10−9 m2/s, respectively. Meanwhile, as the reservoir pressure increases from 15 to 30 MPa, the adsorption amount of CBM gradually increases with the increasing reservoir pressure, while the diffusion coefficient decreases with the increasing reservoir pressure. A reservoir pressure of 30 MPa will increase the adsorption amount of CBM to 0.65 mg/m3, while the diffusion coefficient decreases to 2.5 × 10−9 m2/s. A reservoir pressure of 35 MPa will further increase the adsorption amount of CBM from 0.65 mg/m3 at 30 MPa to 0.70 mg/m3, and the diffusion coefficient will decrease from 2.5 × 10−9 m2/s at 30 MPa to 1.5 × 10−9 m2/s at 35 MPa. Higher reservoir pressure enhances the adsorption capacity of CBM molecules on the coal surface, making it difficult for CBM to desorb from the coal surface. Therefore, higher reservoir pressure is not conducive to CBM extraction, and economic efficiency will be greatly affected (Long et al., 2021).
The bi-dispersion model is considered to analyze the mechanism of the effect of different reservoir pressures on CBM adsorption and diffusion, with both coal bed pore and macroporous diffusion decreasing with increasing adsorption. The decrease in diffusion coefficient with increasing reservoir pressure and CBM adsorption may be attributed to two reasons: (a) Reservoir pressure increases the volume and rate of coal matrix swelling, which has an effect on decreasing the pore size inside the coal block because adsorptive swelling may narrow the entrance of some micropores and increase the resistance to diffusion of gas molecules (Xiang et al., 2023). (2) Strong repulsion between adsorbed molecules and increased surface coverage. Because the natural gas molecules in the pore roar channel of the coal block are squeezed by the pressure of the reservoir, the gas between the coal bed and the surface of the coal block will also be squeezed by the outside world and gradually approach (Hao et al., 2022). The constantly approaching CBM molecules are difficult to detach from the surface of the briquette, and the CBM molecules close to the surface of the briquette also reinforce the force of adsorption on the surface of the briquette under the repulsion of other molecules. The double force increases the adsorption capacity of CBM on the surface of the briquette, and the diffusion capacity of CBM molecules decreases (Figure 5).
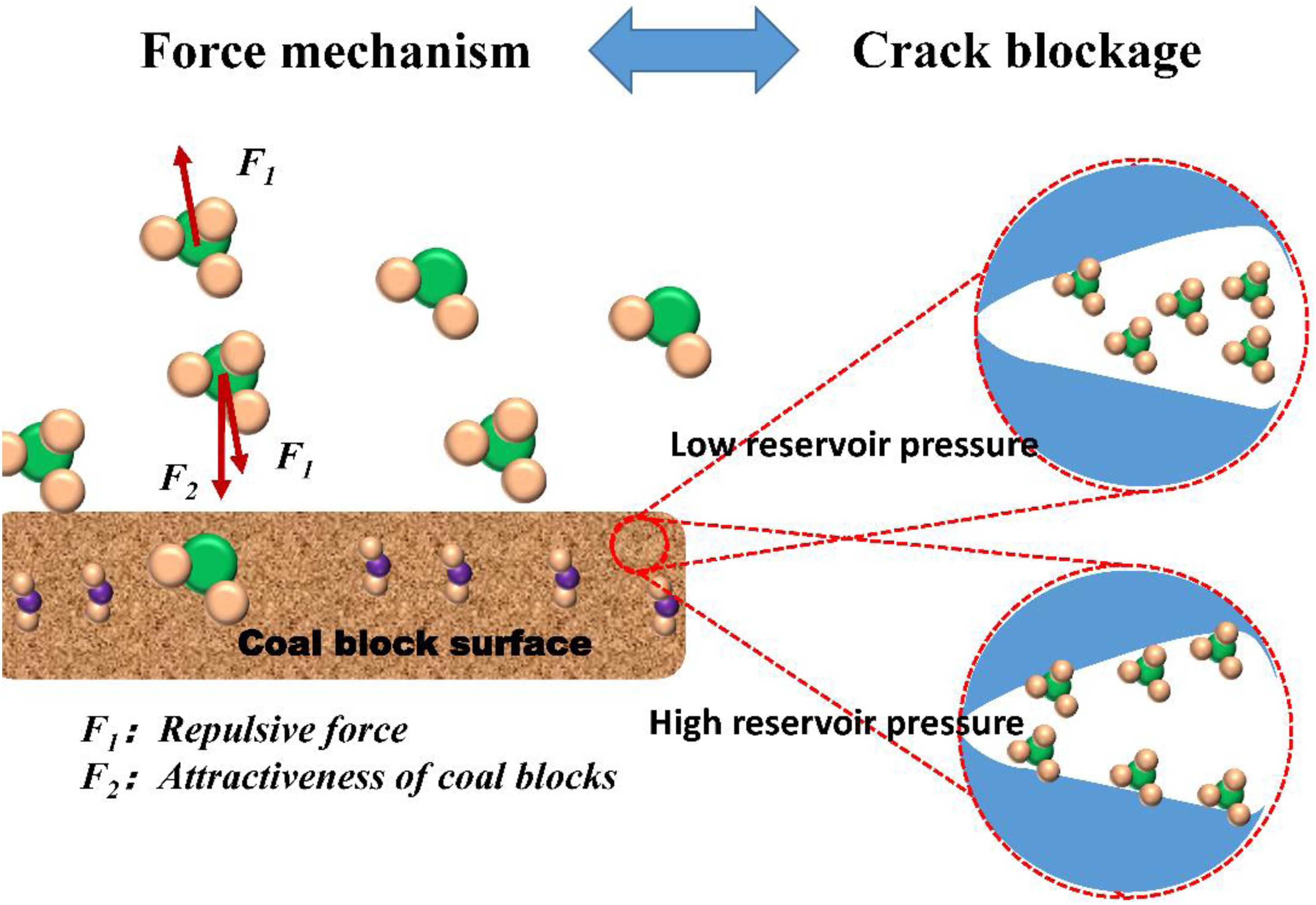
Simulation of adsorption of coalbed methane (CBM) molecules in the pores of coal block under different reservoir pressures.
Influence of reservoir temperature on adsorption and diffusion of CBM
The above studies have shown that the reservoir pressure becomes an important factor affecting the adsorption and diffusion of CBM in the pore channel, and the reservoir temperature also affects the flow and diffusion of CBM in the pore channel under certain conditions, and the related data are shown in Figure 6. In Figure 6, with the increase of reservoir temperature, the adsorption capacity of CBM in the pore channel and the diffusion coefficient increased obviously. However, when the reservoir temperature is below 60 °C, the adsorption of CBM in the pores decreases relatively strongly. The adsorption amount decreases from 1.05 mg/m3 at 40 °C to 0.72 mg/m3 at 60 °C, with a decrease of only 0.33 mg/m3. The diffusion coefficient decreases from 1.7 × 10−9 m2/s at 40 °C to 1.9 × 10−9 m2/s at 60 °C. However, reservoir temperatures higher than 60 °C can slightly reduce the adsorption amount of CBM in the pores, with the adsorption amount rapidly decreasing from 0.72 mg/m3 at 60 °C to 0.65 mg/m3 at 80 °C, a decrease of only 0.07 mg/m3. Meanwhile, the diffusion coefficient increases to 2.5 × 10−9 m2/s at 80 °C. The increase in reservoir temperature benefits the desorption and diffusion of CBM on the coal surface or in the pores. Moderately increasing the reservoir temperature provides the necessary geological conditions for the desorption and rapid diffusion of CBM (Fang et al., 2023).
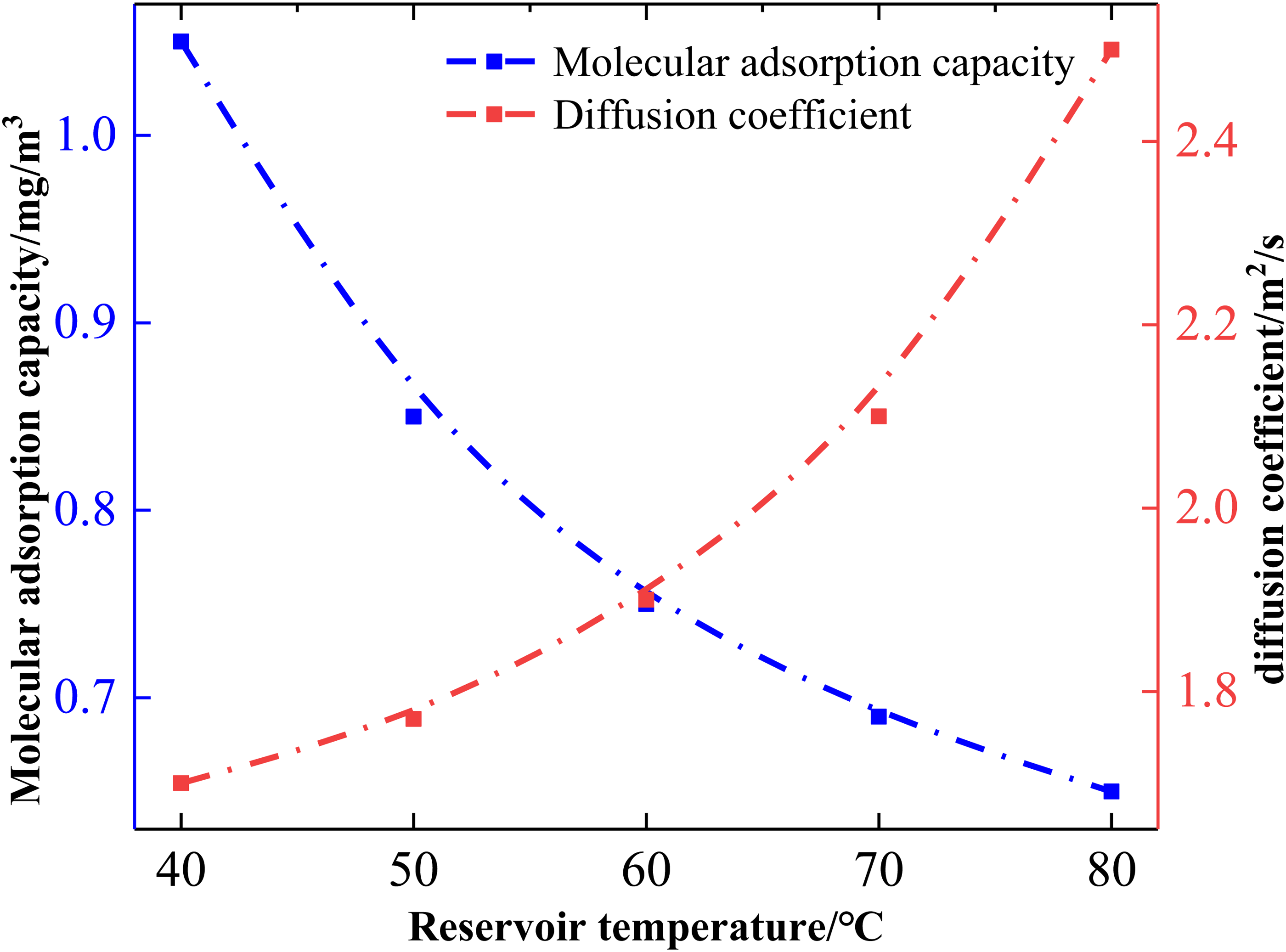
Effect of adsorption and diffusion coefficients of coalbed methane on pore surface at different reservoir temperatures.
The effect of reservoir temperature on the adsorption and diffusion of CBM in the pore channels can be explained by the Arrhenius equation (Liu et al., 2019):
When the temperature of the reservoir increases, the energy and vigor of each molecule in the pore channel of the coal block increase significantly. The molecules with increased energy will move more vigorously and appear similar to the Brownian irregular collision phenomenon (Yu et al., 2018). Due to the increased vigor of the molecules, the CBM molecules that were close to each other kept colliding and moving away from each other due to the effect of vigor, which led to an increase in the energy of desorption of CBM molecules from the surface of the briquette. In addition, the CBM molecules adsorbed on the surface of the briquette at low temperatures have increased vigor and repulsive force due to the increase in temperature, and these forces will also prompt the CBM molecules to accelerate the detachment from the surface of the briquette and realize the obvious increase of the diffusion coefficient. In summary, the increase in molecular vitality caused by larger reservoir temperature is the main microcosmic reason for the increase in the molecular diffusion coefficient (Figure 7).
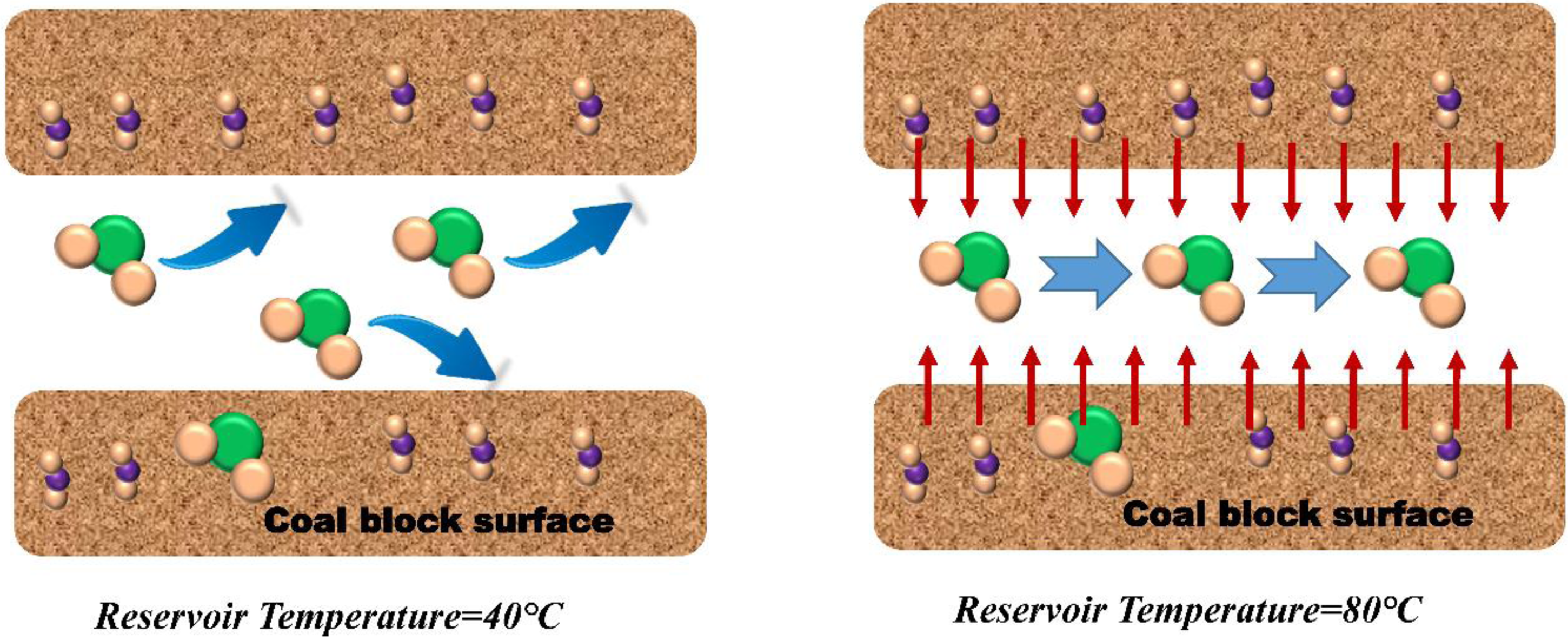
Simulation of adsorption and diffusion of coalbed methane on pore surface at different reservoir temperatures.
Influence of coal block pore width on CBM adsorption and diffusion
Oilfield reservoir fractures can affect the oil and gas recovery rate and fracturing efficiency, and the pore width of coal briquettes has also become an important influence on the adsorption diffusion and recovery rate of CBM; as shown in Figure 8, as the pore width of coal briquettes continues to increase, the adsorption amount of CBM on the surface of the briquettes is also gradually reduced. When the width of the briquette pore channel is 0 mm, seamless briquettes significantly reduce the adsorption amount of CBM in the pore channel (0 mg/m3), and at the same time, when the width of the briquette pore channel is increased from 0 to 2 mm, the adsorption amount of CBM in the pore channel rises from 0 to 0.5 mg/m3. The effect on the adsorption amount of CBM is obviously smaller when the width of the briquette pore channel is <2 mm. However, the adsorption amount of CBM in the pore channel increased from 0.5 to 2.0 mg/m3 as the width of the coal block pore channel increased from 2 to 4 mm. In addition, the difference of diffusion coefficients of different coal block pore channel widths on the adsorption amount of CBM in the pore channel can be obtained in Figure 8, and the diffusion coefficients in the pore channel of the coal block pore channel were weaker when the width of the pore channel of the coal block pore channel was lower than 2 mm. This also means that the mobility of CBM is poor, and the recovery rate of CBM is reduced accordingly. The diffusion coefficient of CBM in the borehole shows a significant increase when the width of the coal block borehole is larger than 2 mm. The diffusion coefficient increases from 1.5 × 10−9 to 2.2 × 10−9 m2/s when the width of the briquette aperture is increased from 2 to 4 mm. The data in Figure 4 also show that the increase in the width of the briquette aperture not only promotes the adsorption of the CBM on the surface, but also promotes the diffusion of CBM in the aperture. The pore width of coal briquettes shows a strong similarity to the adsorption and expansion of CBM. At the same time, the influence of the pore width of coal briquettes on the adsorption and diffusion of CBM showed a trend of change opposite to that of other factors.
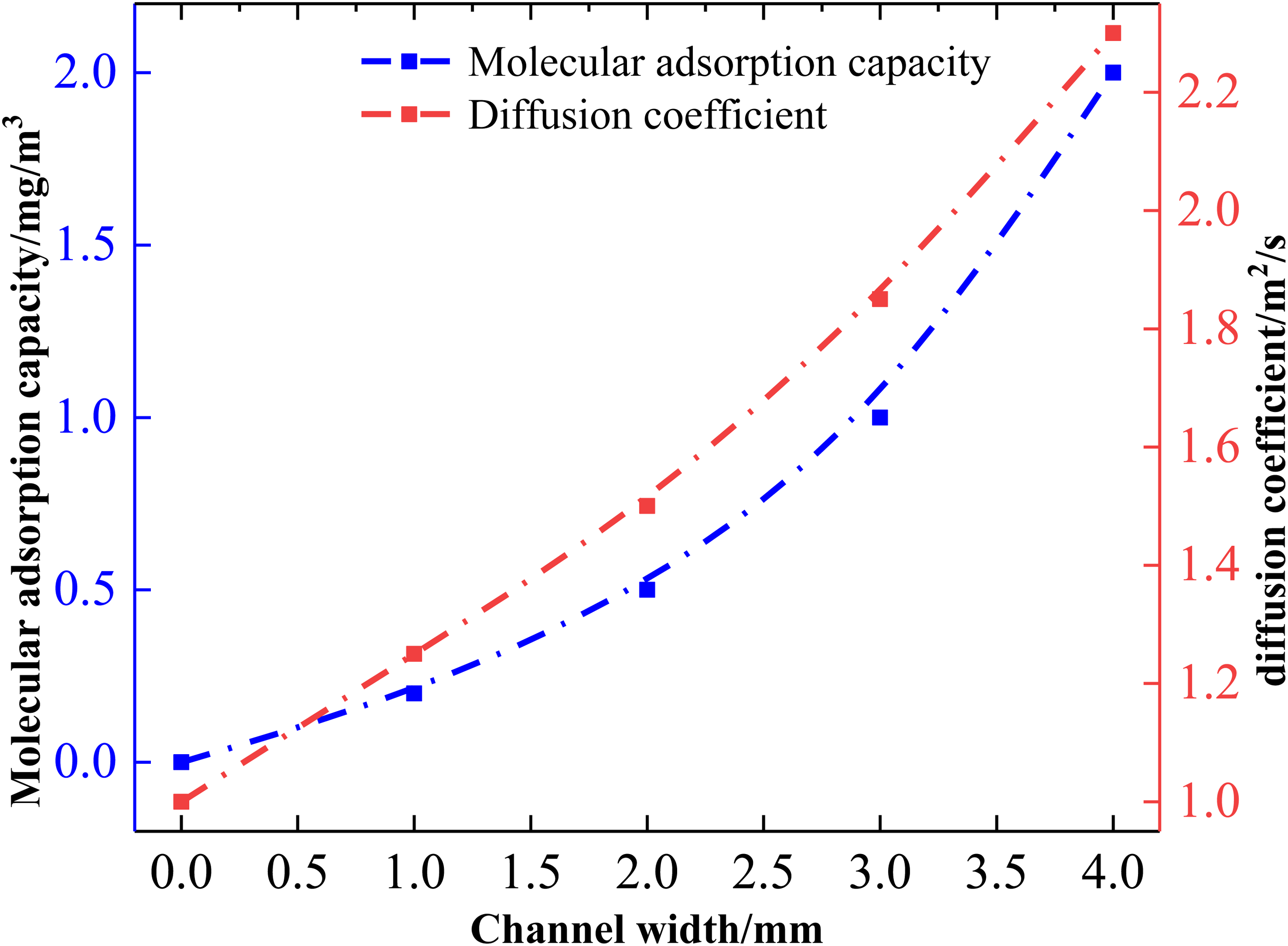
Effect of adsorption and diffusion coefficients of coalbed methane on the pore surface with different pore widths in different coal seams.
When the coal block is solid (no cracks or pores), CBM molecules cannot enter the cracks by themselves to realize diffusion and leakage; however, with the increase of the pores in the coal block, a large number of CBM molecules can enter the coal block through the pores. In addition, as the width of the pore channel increases, the flow rate of a large number of CBM molecules through the pore channel increases, and the diffusion ability is significantly enhanced. At the same time, as the width of the pore channel of the coal block increases, the surface area of the pore channel gradually increases, and more CBM molecules are adsorbed on the pore channel due to the increase of the surface area of the pore channel. The increase of the pore surface area directly leads to the increase of CBM adsorption capacity. Therefore, although moderately increasing the width of the pore channel of a coal briquette increases the molecular adsorption capacity of CBM, the gas diffusion capacity, and the CBM recovery rate are also greatly improved.
Discussion
As a reliable energy source in the future, CBM, which occurs during the mining process of coal seams, can provide a certain energy supplement for human production and life. Therefore, exploring the adsorption and diffusion law of CBM on the surface of coal briquettes can help to improve the recovery rate of CBM. However, some specific factors can enhance the adsorption of CBM, which is not conducive to the diffusion of CBM in the cracks of coal seams. Finding the most suitable mining conditions and geological factors becomes an important method to study the desorption of CBM at present.
CBM with lower adsorption and larger diffusion coefficient can only have a very good CBM recovery rate, which is also the ideal coal bed screening target block as a collection of CBM. The coal bed blocks explored in this study have shown that coal beds with smaller water content reservoir pressure are more favorable for CBM extraction and pore expansion, and moderately increasing the reservoir temperature can also reduce the transportation and desorption of CBM. Meanwhile, coal seams with larger porosity are more suitable for CBM extraction.
Conclusion
In this study, based on the above-mentioned defects in CBM research, we established the theory of adsorbed gas diffusion transition state of surface-diffusing gas molecules and explored the diffusion characteristics and effects of different reservoir pressures, reservoir temperatures, water contents of coal beds, and fracture widths on methane gas diffusion. Meanwhile, this study also proposed the correlation between the diffusion coefficient and each of the above factors and revealed the reasons for the differences in the adsorption over diffusion of CBM gas by molecular dynamics simulation methods. The results of experimental and molecular dynamics simulation studies show that when the water content is lower than 0.1%, the reservoir temperature is 80 °C, and the reservoir pressure is 30 MPa, it is possible to have a very small CBM adsorption amount (0.65 mg/m3) and a large diffusion coefficient (2.5×10−9 m2/s). The excess CBM in the process of coal seam mining is a wasteful energy source, moderate improvement of mining conditions can enhance the diffusion capacity and recovery rate of CBM, which will help to improve the energy structure and energy security of China.
Footnotes
Acknowledgments
The authors would like to show sincere thanks to those techniques who have contributed to this research.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.



