Abstract
Microorganisms are one of the main driving forces of the cycle of carbon and other life elements in the underground environment. The natural environment is the comprehensive result of these microorganisms. In contrast, the study of coal reservoir microorganisms is mostly under laboratory conditions, which limits people's understanding of the symbiotic relationship between microorganisms, the interaction between microorganisms and the environment, and the distribution differences of microbial communities in the region. Similarly, the carbon cycle of the underground environment driven by essential microorganisms in coal reservoirs cannot be further studied. The geochemical process of underground methane generation and oxidation is critical in discussing the production and consumption of biomethane in the underground environment and the metabolic behavior of microorganisms. For this reason, we conducted biogeochemical tests and microbial sequencing on the water produced by coalbed methane wells in the south of the Qinshui Basin to analyze and improve the understanding of the distribution difference and metabolic behavior of microbial communities in coal reservoirs. The concentration of Cl− and HCO3− in the detention environment in the study area increases, while the concentration of SO42−, NO3−, NO2−, and Fe3+ decreases with the increase of coal seam depth, reflecting the distribution difference of hydrochemical environment and redox conditions of the underground reservoir in the study area. The results of microbial sequencing showed microbial methanogenesis in the study area, but it could also be consumed by microbial oxidation simultaneously. The microbial communities related to methane production and consumption had diversity distributions similar to geochemical parameters and geographical patterns. Methanogens and dissolved inorganic carbon isotopes confirmed the potential of in situ methane generation. Still, biomethane's enrichment and accumulation conditions and the impact of aerobic/anaerobic oxidation of methane need further study.
Introduction
Microorganisms are one of the most active and powerful geological agents in lithospheric materials’ weathering, transportation, and deposition. Microbial action has also affected the environmental change and material cycle of the earth's supergene system, profoundly changing the evolution process, so it plays a vital role in the global life element cycle (Iverach et al., 2017; Singh et al., 2012; Schweitzer et al., 2019; Su et al., 2022). Methane is the single-carbon compound with the highest reduction degree, and its role in the global carbon cycle and greenhouse effect cannot be underestimated (Li et al., 2019; Li et al., 2020a; Li et al., 2023). Methanogens are strict anaerobic archaea located at the end of the anaerobic digestion food chain. Methanogens use these single-carbon or double-carbon products to produce methane through multiple metabolic pathways (Beckmann et al., 2019; Fu et al., 2022; Zhou et al., 2022). Methane-oxidizing bacteria (MOB) widely distributed in different environments can oxidize organic matter or methane through electron acceptors such as oxygen, sulfate, nitrate, and metal elements. These processes are integral to the global carbon cycle and control the global carbon balance (Fu et al., 2021; Li et al., 2022a).
Microorganisms are the main factor driving the underground biogeochemical cycle. The commercial development of unconventional natural gas and the clean utilization of coal resources have promoted people's research on biomethane production and consumption process in coal reservoirs (Bao et al., 2019; Zhao et al., 2022). The accumulation of methane depends on the methanogenesis of methanogens and methane oxidation by other heterotrophic bacteria. Generally, the accumulation of methane is the comprehensive result of the metabolism of these types of microorganisms (Zhao et al., 2022). Therefore, methanogens, sulfate-reducing bacteria, nitrifying bacteria, and other microorganisms control the carbon cycle and the production and consumption of biomethane. Still, the research on microbial communities related to the carbon cycle in coal reservoirs is insufficient. Although methane isotope composition can characterize the source of methane, microbial oxidation also affects the isotope data. Therefore, it is necessary to identify the distribution and behavior of microbial communities related to the carbon cycle in combination with microbial data. This study combined microbial sequencing with geochemical indicators such as sulfate and nitrate and dissolved inorganic carbon isotope to speculate the metabolic activities of microorganisms related to the carbon cycle in carbon reservoirs.
Underground environmental conditions significantly affect the composition and distribution of carbon cycle-related microbial communities. The effect of oxygen on microorganisms depends on the tolerance of enzymes in microbial cells to oxygen. For example, several coenzymes in methanogens cells are extremely sensitive to oxygen, and oxygen-enriched conditions make their enzyme activity rapidly decline or even inactivate (Guo et al., 2019; Li et al., 2020b; Li et al., 2022b). Therefore, the methanogenic process in an anaerobic environment is the primary source of biomethane. In addition, the discovered anaerobic oxidation mainly includes sulfate-reduced methane anaerobic oxidation, nitrate or nitrite-reduced methane anaerobic oxidation, and Fe3+ and Mn4+ reduced methane anaerobic oxidation (Beckmann et al., 2019). Sulfate-reducing bacteria and acetoclastic methanogens can use acetic acid as an electron donor under anaerobic conditions. Acetoclastic methanogens are extremely sensitive to sulfate concentration because acetic acid is more used by sulfate-reducing bacteria to oxidize to carbon dioxide than to produce methane in the presence of large amounts of sulfate. At the same time, sulfate-reducing bacteria will also use hydrogen, resulting in hydrogen partial pressure lower than the lowest concentration available for carbon dioxide reduction methanogens (Li et al., 2019; Schweitzer et al., 2019). Therefore, the influence of the underground environment on microbial metabolic function is significant.
The generation and oxidation of methane have high spatial heterogeneity, manifested by the differential distribution of carbon cycle-related microorganisms. At present, most of the research focuses on describing the response of microorganisms to a single environmental factor under laboratory conditions (such as SO42−, NO3−, NO2−, Fe3+, and Mn4+) or the abundance and structure of microbial communities at a certain point (Su et al., 2018; Vinson et al., 2019; Yang et al., 2018; Zandt et al., 2018). It is urgent to strengthen in-depth research on the geographical distribution, functional characteristics, and response characteristics to multiple environmental factors of microorganisms related to the carbon cycle at the regional scale. In addition, because most microorganisms in the natural environment cannot be cultivated, the traditional methods that rely on culture cannot fully describe the abundance and diversity characteristics of microbial communities in situ. It is also difficult to explore the impact of environmental factors on carbon cycle-related microbial communities and methane production potential in space (Li et al., 2022). With the development and application of metagenome and macrotranscriptome technologies, these culture-independent microbial ecology research methods can reveal the abundance, diversity, and metabolic function of microorganisms related to the carbon cycle in the underground environment more objectively at the molecular level.
The southern part of the Qinshui Basin is one of the areas where CBM resources have been successfully commercialized in China, with more than 1000 CBM surface wells (Li et al., 2020a; Shi et al., 2021). In this study, the Shizhuangnan Block is taken as the research area, and the No. 3 coal seam, currently the primary gas producer, is taken as the target reservoir. The Shizhuangnan Block is a monoclinic structure from southeast to northwest, and the hydrodynamic strength gradually weakens from southeast to northwest. High-yield wells are mainly distributed in the west of the study area. The reservoir environment with regular distribution of redox environment is an ideal area for studying microbial communities’ geographical distribution and metabolic function. Through geochemical testing and microbial sequencing of water samples from coalbed methane wells in the Shizhuangnan Block, this study uses geochemical indicators and microbial data to reflect the geographical distribution and metabolic function of microorganisms related to the carbon cycle in the in situ environment of coal reservoirs, thus clarifying the production potential of biomethane in the underground environment, and providing a scientific basis for the utilization and transformation of carbon in the underground space.
Study area
The Qinshui Basin is China's earliest and most successful area for commercializing coalbed methane (Fu et al., 2019; Li et al., 2019; Li et al., 2020b; Li et al., 2022c). As Carboniferous-Permian Basin, the Qinshui Basin has the coal-bearing strata, including the Taiyuan and Shanxi Formations in the Upper Pennsylvanian and Lower Permian. Refer to relevant documents for specific stratigraphic conditions (Li et al., 2020, 2022). The Shizhuangnan Block in the southern Qinshui Basin strikes NNE and dips to the west. Jinhuo fault is located southeast of the study area at a high altitude. Sitou fault and other normal faults developed in the northwest have a sealing effect on the reservoir. The No. 3 coal seam gradually decreases from east to west. The surface water or atmospheric precipitation supply can be accepted in Jinhuo fault, and infiltration and runoff occur from southeast to northwest (Shi et al., 2021). Due to the sealing effect of Sitou fault in the northwest of the Shizhuangnan Block, the hydrodynamic force is gradually weakened from southeast to northwest, thus forming the hydrogeological characteristics with clear runoff conditions from southeast to northwest in the study area. The western part of the study area has a high degree of mineralization in the reservoir, which is also the main distribution area of high-yield wells. The redox environment of the reservoir in the study area is ideal for studying the geographical distribution of microbial communities and their environmental response (Li et al., 2020; Shi et al., 2021). This study collected water samples from coalbed methane wells in the Shizhuangnan Block of Qinshui for geochemical testing and microbial sequencing, focused on the spatial distribution and biogeochemical characteristics of microbial communities related to the carbon cycle in the underground in situ environment in the Shizhuangnan Block, analyzed the response of the microbial abundance and distribution to the electronic receptors, and further clarified the production potential of biomethane in the underground in situ environment.
Geochemical analyses
Eight water samples were collected from different coalbed methane wells in the Shizhuangnan Block (Figure 1). Rinse the sampling bottle with the collected water sample three times before sampling. The sampling bottle is filled with these water samples and sealed to ensure the air in the bottle is discharged. The water samples for geochemical testing and biological sequencing are packed in 2L sampling bottles and 50 mL centrifuge tubes. The centrifuge tube is stored in a 4°C incubator for refrigeration. Conductivity (EC), oxidation-reduction potential, dissolved oxygen, temperature (T), and pH are measured on-site to ensure the accuracy of data. Microbial sequencing and geochemical testing should be completed within 1 week. The ion test is based on the coal industry standard “Water Quality Analysis of Coal Mine Water” (MT /T894-2000). Stable isotope gas mass spectrometer is used for the inorganic carbon isotope test (model: MAT253). Inductively coupled plasma atomic emission spectroscopy is used for the cation test, and ion chromatography is used for the anion test. Total dissolved solids (TDS) are the sum of major anions and cations.
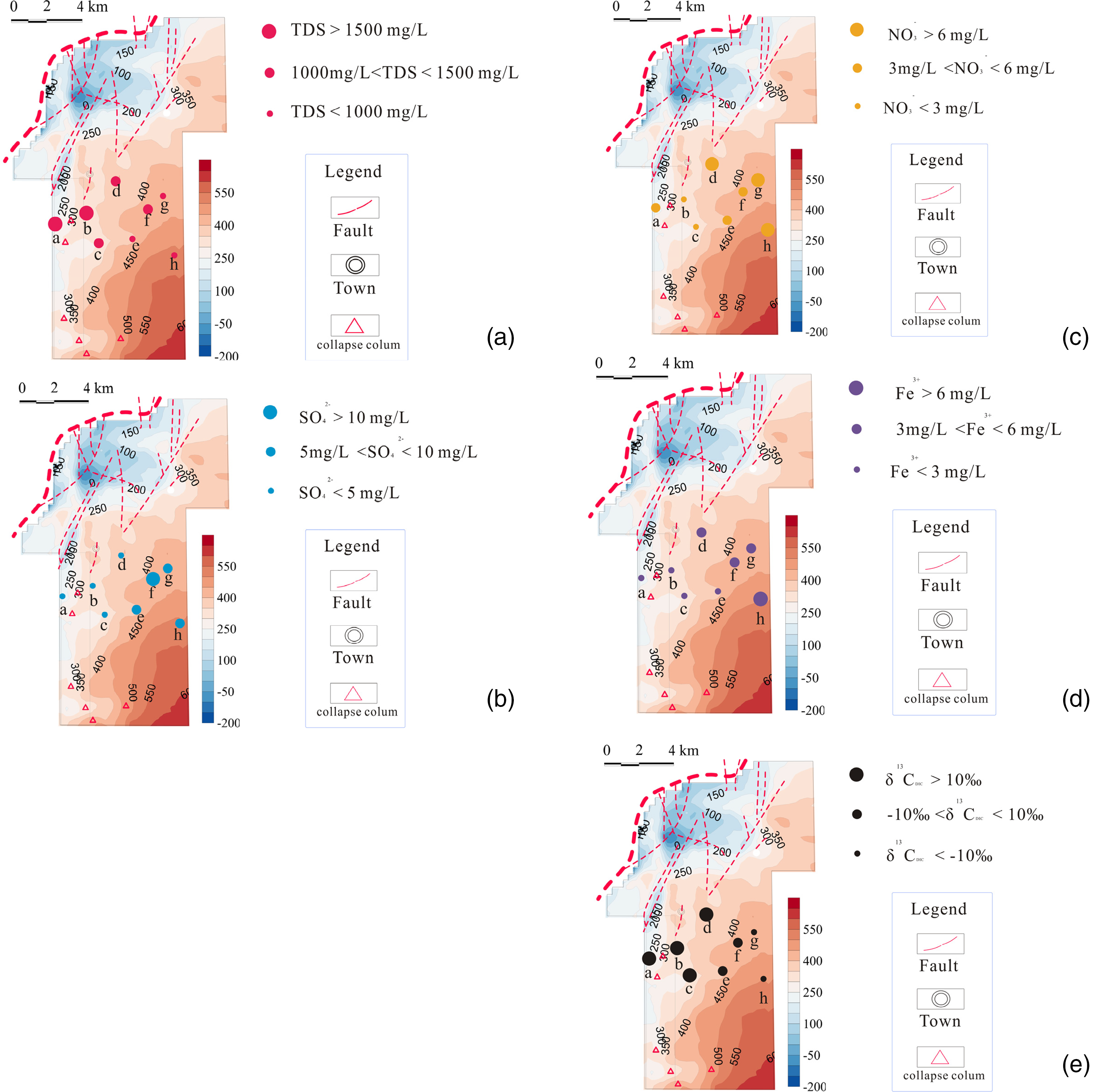
The distributions of significant ion concentration, total dissolved solids, and dissolved carbon isotope in the Shizhuangnan block: (a) total dissolved solid (mg/L); (b) SO42− (mg/L); (c) NO3− (mg/L); (d) Fe3+ (mg/L); and (e) δ13CDIC (‰).
Gene extraction and sequencing analysis
To ensure the integrity of the microbial community in situ, the water samples collected from the surface coalbed methane wells in the study area with a 50 mL centrifuge tube were stored in a 4°C incubator with dry ice until the gene extraction was completed. Biomass is extracted from filtered water samples for DNA extraction in the laboratory. Before extraction, DNA was first precipitated with polyethylene glycol, then washed with 70% ethanol and resuspended. According to the manufacturer's specifications, the DNA for each sample was extracted with a Qubit Assay Kit, which was used as a target for Illumina sequencing. The sublibrary was amplified according to the 16S Metagenomic Sequencing Library Preparation Protocol. To obtain sufficient biological information and avoid errors caused by over-amplification, the initial PCR cycle number was adjusted to 20 times. The PCR products were purified by magnetic bead capture kits and were quantified by fluorometric kits. A MiSeq platform is used to perform Index PCR on the amplicons, following the manufacturer's instructions. Illumina sequences were checked for quality and bioinformatics analysis.
Operational Taxonomic Unit (OTU) to classify the sequence according to the needs of phylogeny or population genetics to analyze a taxonomic unit to understand the number of strains and genera in the sequencing results. Sequences with a higher than 97% similarity are identified as a specific OTU. To obtain the species classification information of each specific OTU, it is necessary to classify and analyze the OTU representative sequence and count the community composition of the sample at different levels. To ensure the uniformity and depth of subsequent analysis, the OTU data of each sample is maintained at a low sequence number.
The 16S rRNA and functional gene (mcrA, mmoX, pmoA, and dsrA) were measured by qPCR (quantitative real-time PCR). The 16S rRNA of bacteria and archaea were amplified by the primer pairs 519F/907R, and sequence fragments dsrA and mcrA were amplified by the primer pairs ME1F/ME3R and 1F/500R. The qPCR of pmoA was measured by the pmoA primer pair A189F and mb661R. The qPCR amplification was measured under the following reaction cycle: 95°C for 3 min, 30 cycles of 95°C for 30 s, 65°C for 45 s, and 65°C for 45 s. The fragments of mmoX were quantified by the primer pairs mmoXB-1401b and mmoX-ms-945f. The qPCR amplification of mmoX was measured under the following reaction cycle: 95°C for 3 min, 45 cycles of 95°C for 1 min, 50°C for 1 min, and 70°C for 1 min. Different dilutions of DNA extracts have performed the gene of bacteria and archaea (1: 1000, 1: 100, and 1: 10) in triplicate. The PCR products were examined by gel electrophoresis (2% agarose). The specificity of the reaction was confirmed by PCR products, which be identified by agarose gel electrophoresis and melting curve analysis. The r2 value of the standard curve is > 0.99. All PCR products were extracted and eluted into 20 µL DNA elution buffer, and DNA solutions were quantified with 2 µL DNA.
Results and discussion
Regional distribution of geochemical indicators in the southern Qinshui Basin
Geochemical results indicators of the coal reservoir
Because groundwater flows through the coal reservoir and undergoes water–rock interaction, the geochemical characteristics of the produced water from coalbed methane wells can be used to effectively identify the environmental characteristics of the reservoir (Fu et al., 2019, 2022). Common ions include Fe3+, Ca2+, Mg2+, Na+, K+, Cl−, SO42−, CO32−, HCO3−, and NO3−. Among them, Fe3+, SO42−, and NO3− are closely related to microbial action, and they are anaerobic electron receptors of typical organic matter and methane (Iverach et al., 2017; Shi et al., 2021). As shown in Table 1, the concentration range of Fe3+ is 0.59∼6.93.57 mg/L, the concentration range of SO42− is 0.68∼12.05 mg/L, and the concentration range of NO3− is 1.67∼8.72 mg/L.
Ion concentration (mg/L), total dissolved solids (mg/L), and dissolved carbon isotope (‰) in the samples from the Shizhuangnan Block.
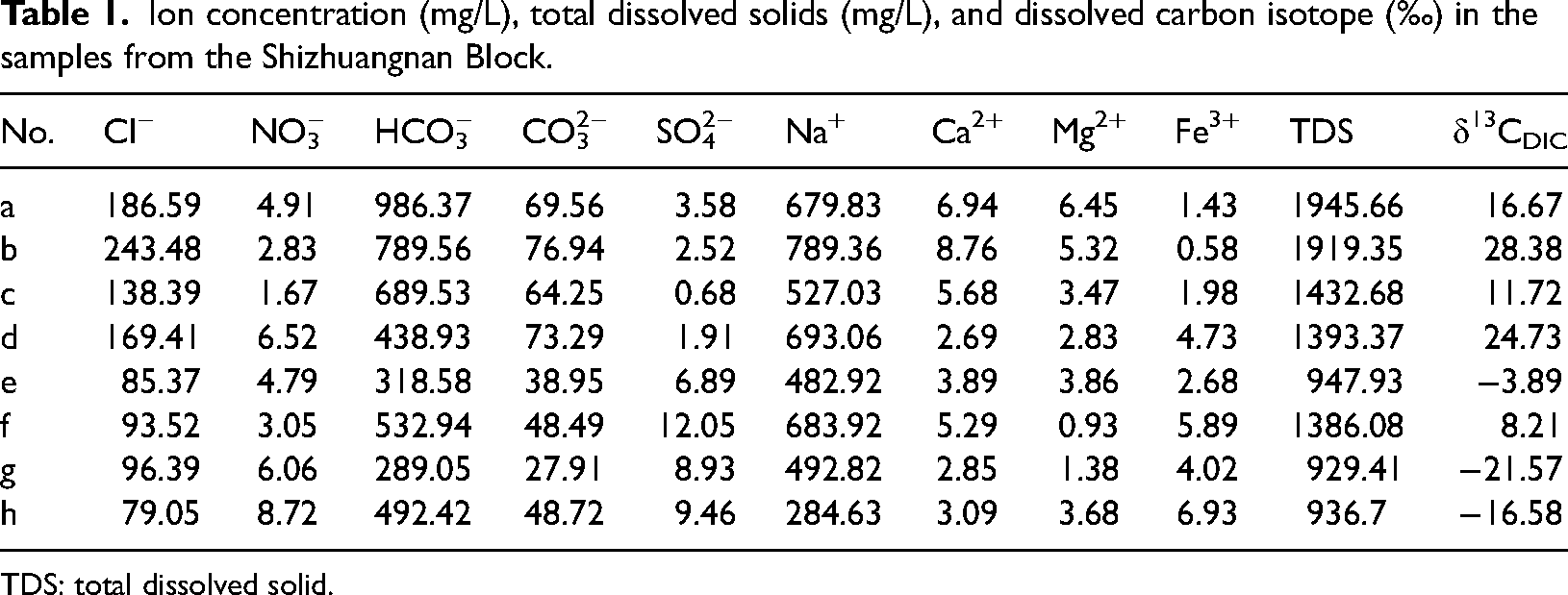
TDS: total dissolved solid.
As shown in Figure 1(a), the size of the circle represents the relative concentration of ions. In the study area, atmospheric precipitation or surface water flows from southeast to northwest through the east Sitou fault. The detention environment in the northwest area strengthens the water–rock effect, making the TDS of reservoir water in this area higher. It can be seen that along the runoff direction from southeast to northwest, the mineralization degree in the southeast region is significantly lower than that in the northwest detention area. On the contrary, as shown in Figure 2(b), (c), and (d), the Fe3+, SO42−, and NO3− content in the southeast runoff area is higher than in the northwest detention area. In groundwater runoff, the dissolved oxygen in the water body gradually decreases, and the aerobic environment transits to the anaerobic environment. The concentration of oxidants Fe3+, SO42−, and NO3− decreases from southeast to northwest, indicating that with the underground runoff of coal reservoir water, Fe3+, SO42−, NO3−, etc., are used as electron receptors for anaerobic oxidation of microorganisms for iron reduction, sulfate reduction, and denitrification, resulting in the consumption of these anaerobic electron receptors.

Bacteria and archaea profiles and relative abundances measured by Illumina sequencing.
The regional distribution of δ13CDIC values
As shown in Figure 1(e), the distribution range of δ13CDIC value in the produced water from coalbed methane wells in the Shizhuangnan Block is −21.57‰ to 28.38‰. Dissolved inorganic carbon (DIC) in groundwater generally comes from dissolving carbonate minerals and carbon dioxide. However, the DIC value of coal seam water may be affected by microbial methanogenesis (Iverach et al., 2017; Li et al., 2019). The value of δ13CDIC is positive and abnormal (δ13CDIC > 10‰) indicates that methanogens preferentially use 12C to produce methane; when the value of δ13CDIC is positive (0‰>δ13CDIC < 10‰), The δ13CDIC value is mainly related to the dissolution of carbonate minerals and carbon dioxide and begins to be affected by methanogenesis; when the δ13CDIC value is negative (−10‰>δ13CDIC < 0‰), the coalbed seam water is wholly derived from the dissolution of carbonate minerals and carbon dioxide. When δ13CDIC value negative abnormality(δ13CDIC < −10‰), it means δ13CDIC value is related to microbial oxidation (Shi et al., 2021). The sampling time avoids the rainy season, so in the shallow reservoir of the southeast study area, the influence of atmospheric precipitation or surface water runoff on the δ13CDIC value can be ignored.
The structure of the study area determines the hydrodynamic conditions and then affects the distribution of the hydrochemical field and the regional distribution of microorganisms. Previous research results show that the value of δ13CDIC in the rainy season is generally negative, and within the study area, there is no significant difference in the distribution of δ13CDIC values (Li et al., 2020). The sampling time should avoid the impact of the rainy season, and the selected wells can ignore the effects of hydraulic fracturing and other engineering factors. The values of δ13CDIC of well a, b, c, and d in the northwest of the study area exceed 10 ‰, indicating strong methanogenic metabolism. The value of δ13CDIC of well f in the northwest region is between 0‰ and 10 ‰, meaning slight microbial methanogenesis. The value of δ13CDIC of well e in the central and southeast areas is between −10 ‰ and 0 ‰, indicating that the reservoir water is mainly from the dissolution of carbonate minerals and carbon dioxide and is not affected by microorganisms. The value of δ13CDIC of g, h wells in the southeast region is lower than—10 ‰, which can be inferred to be related to microbial oxidation.
The regional difference distribution of δ13CDIC in the study area is related to microbial methanogenesis and methane oxidation. In the past, the gas and water samples from the Shizhuangnan Block were analyzed for gaseous and dissolved methane isotopes, respectively. The results show that the gas from coalbed methane wells in the study area is mainly thermogenic. However, dissolved methane isotopes indicate biomethane in the study area, but microbial methanogenesis is not enough to form gas reservoirs (Li et al., 2019, 2020, 2022). To verify the regional distribution characteristics of biogeochemistry in the above study area and identify the distribution differences of microbial communities on the regional scale. The results and discussion are as follows.
Microbiological response investigation for the coal reservoir environments
Carbon-related microbial community and functional genes in the Shizhuangnan Block
The biomass in the produced water of coalbed methane wells collected in the study area was sequenced after PCR amplification. As shown in Figure 2, microbial communities of the samples were composed of bacteria (78–91%) and archaea (9–22%). The bacterial and archaeal composition did not vary significantly among these samples. If the microbe's relative abundance was <1%, it was related to other bacteria or archaea. In all samples, Proteobacteria (Gammaproteobacteria, Betaproteobacteria, Deltaproteobacteria, and Alphaproteobacteria) were the most abundant bacteria, accounting for more than 70% of all microorganisms. Firmicutes, Bacteroidetes, Actinobacteria, Spirochaetes, Chloroflexi, and Euryarchaeota were also present at higher relative abundances. The several most highly abundant microorganisms related to methanogenesis, denitrification, and sulfate reduction were selected (Li et al., 2023; Shi et al., 2021). Three orders of methanogens were in the study area, including Methanobacteriales, Methanomicrobiales, and Methanosarcinales. High δ13CDIC in the west of the study area is related to the metabolism of methanogens. Although methanogens are also distributed in the eastern part of the study area, a low δ13CDIC value indicates that the activity of methanogens is inhibited, which may also be caused by aerobic/anaerobic oxidation of microorganisms (Iverachetal., 2017). The samples a, b, and d had relatively high Euryarchaeota (Methanobacteriales, Methanomicrobiales, and Methanosarcinales), while the remaining samples may only comprise certain methanogens. Denitrifying bacteria included Burkholderiales and Rhodospirillales. Sulfate-reducing bacteria had Desulfovibrionales and Desulfuromonadales.
Microbial methanogenic potential and limited SRB and methanotrophs for methanogenesis in the Shizhuangnan Block
To further verify the biogeochemical characteristics of coal reservoir water in the above study area, the 16S rRNA genes of bacteria and archaea were sequenced, and specific functional genes (mcrA, pmoA, mmoX, and dsrA) were located. As shown in Figure 3, microbial abundances were estimated between 103 and 105 mL−1 throughout all groundwater samples.
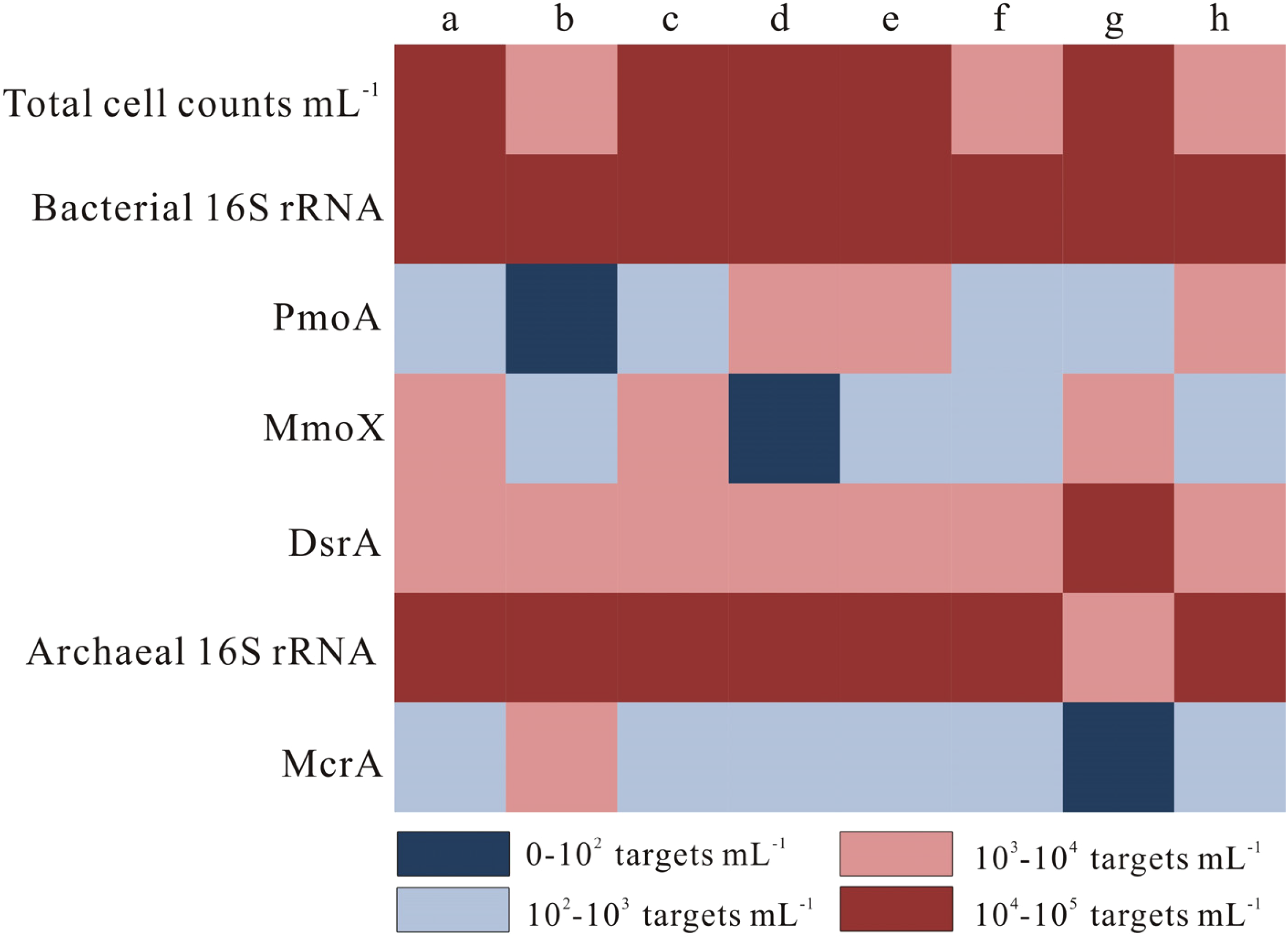
Total cell concentration and 16S rRNA genes of bacteria and archaea and functional genes for CH4 production (mcrA), sulfate reduction (dsrA), and aerobic CH4 oxidation (pmoA and mmoX) measured by PCR.
The mcrA gene about archaea was measured in most of the samples, which encodes the methyl coenzyme M reductase. And the mcrA gene not only means methanogens but also states the existence of anaerobic methanotrophs. The sulfate content of reservoir water in the Shizhuangnan Block is 0.68∼12.05 mg /L. Microbial community sequence and gene analysis show that SRB is ubiquitous in all samples, and its relative abundance accounts for 3% to 9% of all microorganisms (Figures 2 and 3). Some studies also show that sulfate-reducing bacteria can oxidize methane with other electronic receptors (Gomes et al., 2017; Treude et al., 2014). Sulfate-reducing bacteria have an advantage in competing with methanogens for substrates (such as acetate and H2), keeping the substrate at a low content range and inhibiting the growth of methanogens (Beckmann et al., 2019). As shown in Figure 3, Deltaproteobacteria has a high abundance of reservoir environment in the study area. Desulfovibrionales, Desulfulomonadales, and Syntrophobacterales are closely related to acetate oxidation and sulfonate-reducing bacteria. The increase of sulfate and other reducing agents and the presence of sulfate-reducing bacteria confirmed that the activity of methanogens was inhibited, and biomethane might not be effectively accumulated.
As shown in Figure 3, relative to bacterial 16S rRNA abundance (3.6 × 104–6.2 × 104 targets mL−1), the functional gene abundance (6.5 × 102–8.1 × 103 targets mL−1) of aerobic MOB (pmoA) is high. As shown in Figure 3, the proportion of microbial community of MOB is not <7%. At the order level, Rhzobiales may be Alphaproteobacteria's main aerobic CH4 oxidizers, including Methylocella palustris and Methylosinus acidophilus belonging to the type-II methanotrophs. It should be noted that there may also be aerobic MOB in microaerophilic and anaerobic environments (Iverach et al., 2017). The mmoX gene targeted with relatively high abundance usually means a soluble CH4 monooxygenase. And the pmoA gene is a biomarker for MOB, which confirmed the possibility of aerobic oxidation in the eastern part of the study area.
According to previous studies, the dissolved oxygen in the water produced by coalbed methane wells in the Shizhuangnan Block varies widely (Li et al., 2019, 2020; Shi et al., 2021). Atmospheric precipitation or surface water supply, drilling, and fracturing of coalbed methane wells have increased the oxygen content of the originally sealed reservoir, resulting in a certain abundance of aerobic MOB, which may consume dissolved methane in the sample water in the underground environment, wellbore or drainage outlet. The increased dissolved oxygen leads to the massive reproduction of aerobic bacteria and inhibits the formation of methanogenic archaea. At the same time, the widespread presence of sulfate-reducing bacteria and other anaerobic methanogens also leads to the consumption of biomethane, that is, biomethane in the study area may be consumed by aerobic and anaerobic microorganisms at the same time so that the biomethane produced by in situ methanogens is not effectively preserved. According to the δ13CDIC data, the Shizhuangnan Block exists anomaly positive δ13CDIC (samples a, b, c, and d) and anomaly negative δ13CDIC (samples g and h). The difference between them is more than 20, indicating that there are simultaneous microbial methanogenesis and anaerobic oxidation of methane.
Conclusions
The symbiotic relationship between microbial communities under natural conditions determines the material cycle of the regional environment. The cooperation or competition relationship between microorganisms in the underground environment cannot be fully considered under laboratory conditions. The monoclinic structure of the Shizhuangnan Block in the south of Qinshui Basin and the gradual reduction of coal seam elevation from the southeast to the northwest provides an ideal place for studying biogeochemistry and element cycling of coal reservoirs in the study area. This study used geochemical data and microbial sequencing to evaluate microbial communities’ diversity and regional distribution related to carbon cycling in coal reservoirs in the southern Qinshui Basin.
The microbial community structure and metabolic function differ in the study area, especially the geographical distribution of microbial processes such as organic matter decomposition, methane generation, and sulfate reduction, which participate in the carbon cycle. The sealing effect of the Sitou fault makes the TDS in the northwest of the study area generally increase compared with the southeast. At the same time, Fe3+, SO42−, NO3−, and other oxides are gradually consumed, and the change of δ13CDIC confirmed the methanogenesis of microorganisms in the underground environment. There are methanogens in the study area, but the abundance and species of methanogens vary with the reservoir environment. The methanogenic species in the northwest under reduction conditions are significantly more than those in the southeast of the study area. The gene sequencing results showed that aerobic oxidation of methane and anaerobic sulfate reduction were the possible causes of methane consumption in coal reservoirs in the study area.
The mixing of atmospheric precipitation or surface water, as well as the environmental differences between the runoff area and the detention area, may be the reasons for the differences in the microbial distribution in the study area, providing evidence for the study of biogeochemical effects at the regional scale, and providing a basis for promoting the production of coalbed methane by in situ environmental microorganisms. More importantly, in most cases, the rate of the complex polyphase macromolecular structure of coal metabolized by microorganisms in a situ environment is slow. The content of simple organic matter available to methanogens is low. That is, the hydrolysis and fermentation process of coal restricts the enrichment and accumulation of biomethane. In addition, heterotrophic microorganisms in some environments compete with carbon dioxide-reductive methanogens and acetic acid-decomposing methanogens for available substrates to further reduce the metabolic intensity of methanogens. The slow production efficiency of biomethane also makes it conditional on being fully oxidized, eventually leading to the existence of methanogens and their metabolic evidence in coal reservoirs and the inability to enrich biogenic gas. In the future, it is necessary to comprehensively evaluate the potential and conditions of microbial methanogenesis and reservoir formation, deeply study the ideal in situ environment suitable for microbial methanogenesis, and find feasible methods to transform the underground environment significantly improve the degree and efficiency of microbial methanogenesis.
Footnotes
Acknowledgements
We would like to thank the China United Coalbed Methane Corporation for providing the production well date.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Foundation for High-level Talents of Anhui University of Science and Technology, the NSFC-Shanxi Coal-based Low Carbon Joint Fund of China, Open Foundation of the Key Laboratory of Universities in Anhui Province for Prevention of Mine Geological Disasters, National Natural Science Foundation of China, State Key Laboratory of Petroleum Resources and Prospecting, China University of Petroleumum, Scientific Research Foundation for Young Teachers of Anhui University of Science and Technology, University-level key projects of Anhui University of Science and Technology, the National Natural Science Foundations of China (grant number U1910205, 2022-MGDP-07, 42102216, XCZX2021-04, xjzd2020-05, 41972173).
