Abstract
The occurrence states of hydrogen sulfide in coal seams are crucial in preventing and controlling hydrogen sulfide emission in coal mines and the safe development of coal bed methane. In this study, the research status of the occurrence states of free-state, adsorbed-state, and water-soluble hydrogen sulfide in coal seams was systematically analyzed. H2S anomaly areas in China's coal seams are mainly located in the Carboniferous-Permian and Jurassic series of northern, eastern, central, and northwest regions of China. Bacterial sulfate reduction accounts for most of the hydrogen sulfide anomalies of low-rank coal, while thermochemical decomposition thermal desorption spectroscopy and thermochemical sulfate reduction may also result in hydrogen sulfide anomaly in medium- and high-rank coal. In contrast, magmatism-induced hydrogen sulfide anomalies are rarely found. Absorbed-state hydrogen sulfide anomalies are prevailing, while water-soluble and free-state hydrogen sulfide anomalies are relatively scarce. Coal seam's porosity mainly controls the hydrogen sulfide adsorption, pressure, coalification degree, pore volume, and specific area, while water-soluble hydrogen sulfide is influenced by pressure, sulfate-reducing bacteria, burn, porosity, fractures, water temperature, and hydrodynamic conditions. The fractures in coal seams, their burial depth, coal quality, coal rank, roof, and floor lithology are the main factors controlling the free-state hydrogen sulfide preservation. The absorbed-state hydrogen sulfide in coal seams is mainly mitigated by varying the ventilation mode, increasing the ventilation capacity, spraying alkali fog into the air, and injecting alkali liquid into coal seams for governance.
Introduction
Hydrogen sulfide (H2S) is one of the most harmful components of coal mine gas. With increased coal mining development scale and depth in recent years, the H2S emission in coal seams has become a serious threat to coal production safety (Lin et al., 2012). On the other hand, mining of high-sulfur coal at increasing burial depths aggravates hazards related to H2S emission (Chou, 2012; Fu et al., 2011; Liu et al., 2012, 2019). In particular, of China, with its prevailing coal-fired power generation, the occurrence state of H2S in coal seams has become very topical.
There exist a variety of occurrence states of H2S in coal seams. Free-state H2S generally exists in coal seams with well-developed fractures, weak hydrodynamic forces, and compact surrounding rocks. The adsorption-state H2S is adsorbed on the inner surface of coal seams’ pores and fractures. Water-soluble H2S is found in coal seam water. In case of water inrush accidents, high-concentration H2S, due to its high solubility, can get into the tunnel with the mine water, triggering secondary disasters and jeopardizing miners’ safety. Tan et al. (2020) conducted an in-depth survey on the causes, distribution, prevention, and control measures of H2S in China's coal seams, including the H2S harmful impact on the environment and health. Deng et al. (2019a) introduced the prevention and control technology of H2S in coal-bearing strata, airflow in tunnels, and underground water bodies, and analyzed the efficiency of different ventilation systems in the prevention and control of H2S, and systematized the main treatment methods, commonly used alkaline reagents and additives, and the major bottlenecks in this problem's solution. This survey presents the development history and research results on free-state, adsorbed-state, and water-soluble H2S, which are systematized in terms of distribution characteristics, formation genesis, occurrence state, and development trend of H2S occurrence in China's coal seams. The present study aims to provide insightful guidance for the prediction and treatment of H2S in coal mines.
Geneses of H2S anomaly in coal seams
Overall, anomalous enrichment of H2S in coal seams can be classified into three states: free-state, adsorbed-state, and water-soluble ones. The formation mechanisms mainly include biogenic formation, thermochemical formation, and magmatism (Berner, 1984; Chambers and Trudinger, 1979; Dai et al., 2002; Zhang et al., 2005). To be specific, the biogenic formation refers to bacterial sulfate reduction (BSR), while the thermochemical formation mainly refers to thermochemical decomposition (thermal desorption spectroscopy (TDS)) and thermochemical sulfate reduction (TSR) (Zhang et al., 2007, 2008).
BSR can be regarded as the main biogenic origin of H2S in coal seams. The occurrence of BSR activity should satisfy the following three basic conditions: availability of organic matter, sulfate, and sulfate-reducing bacteria (SRB) (Huang et al., 2016; Simonton and King, 2013). The anaerobic environment for the occurrence of the reduction is favorable for the storage and aggregation of H2S. Accordingly, BSR or BSR-including mixed causes can mainly account for H2S anomalies in China's coal seams. The abundance of BSR-induced H2S is generally <3%. Additionally, the formation medium condition should be suitable for the growth and reproduction of SRB (Amrani et al., 2008; Asaoka et al., 2018; Machel, 2001; You et al., 2009). Given this, BSR usually occurs in shallow coal seams.
Thermochemical formation mainly includes TSR and TDS. The former one is the main factor controlling the formation of anomalous H2S enrichment. High temperatures (exceeding 150 °C), sufficient organic matter, and sulfate are three basic conditions required for TSR. Spontaneous combustion of coal rock can directly affect the production of H2S in the TSR process. After being heated or baked to high temperatures, sulfur in the coal rock is partly oxidized to SO2 and partly dissolved in water to form sulfate, providing conditions for TDS and TSR to form H2S. The concentration of TDS-generated H2S is generally <2% (Sośnicka and Lüders, 2020; Vengosh et al., 2014; Zhang, 2007; Zhang et al., 2008).
Magmatic activities melt the rocks in the deep crust, and the generated volatile components, including H2S, enter into the coal seams after degassing separation. Therefore, the content of H2S under magmatism mainly depends on the magma constitutes and gas migration conditions, being very unstable. Furthermore, the formation's H2S can be preserved only under certain reservoir conditions (Wu et al., 2013).
The factors in coal seams that can affect the concentration of H2S include the total sulfur content in the coal, SRB, reservoir pressure, coalification degree, hydrodynamic condition, and the spontaneous combustion of coal rocks. Further identification of genetic types of H2S in coal seams should consider various factors, including coal-forming environment, thermal evolution history of coal rocks, the constituting characteristics of C and S isotopes, and gas components.
According to the National Coal Board coal classification standard (Spears et al., 1999), coal with a total sulfur content exceeding 2.5% is considered a high-sulfur coal. Besides, coals with H2S concentrations exceeding 1000 ppm referred to as high-concentration H2S anomalies. While most coal seams in East China fall into high-sulfur coal classification, high-sulfur Fenghuangshan and Tiexin Coal Mines in North China contain mostly medium- and low-concentration H2S anomalies. In contrast, among the Longtan Coal Mine, Binlang Coal Mine, and Guang’an Coal Mine in Southwest China, the first two mines have high-sulfur coals and high-concentration H2S anomalies, while the latter has medium-concentration one. In general, the H2S concentration in high-sulfur coals significantly exceeds those in medium- and low-sulfur coals, reaching as high as 57.14%.
Noteworthy is that pyrite is one of the common metal minerals in coal seams, and H2S is the basic condition for generating coal seam pyrite. Due to the aggressive chemical properties of H2S, iron ions are susceptible to its action, forming a relatively stable sulfide–pyrite, consuming a large amount of H2S in the gas reservoir. Because the valence of sulfur in pyrite is higher than that of H2S, it shows the sulfur isotope of pyrite. The composition of the sulfur isotope is higher than that of H2S (Zhao et al., 2021a).
Deng (2015) described the two main forms of pyrite produced by consuming H2S in coal as grain-shaped and raspberry-shaped ones:
A grain-shaped crystal pyrite is formed by the direct precipitation of H2S into raw pyrite. This occurs in a reducing environment with pH < 6.5, when the coal seam water contains sulfate-reduced saturated water-soluble H2S and Fe2+ ions, which concentrations are less than that of FeS. A raspberry-shaped pyrite is formed by evolution of pyrite of complex origin. In the coal seam water environment with pH > 6.5, when the dissolved S2− in the coal-forming environment is relatively abundant, the organic matter can react with SO42− to form H2S, and Fe3+ is reduced to Fe2+. At this time, Fe2+ reacts with H2S. The reaction produces FeS, which may continue to undergo several sulfide stages and eventually form a raspberry-shaped pyrite.
Deng (2015) studied the Zhunnan Coalfield and found that the measured sulfur isotope values in the study area were quite low, ranging from −14.5‰ to 11.6‰. Among them, the δ34S value range of pyrite in coal was 8.7‰ to 11.6‰, with an average of 10.2‰. The δ34S values of H2S gas in coal seams were negative and ranged from −14.5‰ to −9.4‰, with an average of −12.3‰. The δ34S value in the underground water body of the coal mine was −0.6‰, while the δ34S value measured in the crude oil of the regional boundary of Houxia was 14.17‰. Using the above-mentioned average values of 10.2‰ and −12.3‰, the total δ34S value could be assessed as δ34SPyrite–δ34SH2S = 10.2 – (−12.3‰) = 22.5‰ > 22‰. Thus, the regional H2S gas generally exhibited the characteristics of BSR genesis.
Wen (2018) conducted an in-depth study of the Huayingshan mining area and reported that most coal fields had H2S content above 2%, being high H2S reservoirs. The sulfur isotope value distribution range was mostly between +10‰ and +15‰. According to the relationship between the content of H2S of different genetic types and the δ34S value, H2S in this region has the characteristics of TSR origin.
Distribution characteristics of H2S anomaly areas in the coal seams
From China's coal mines with proven H2S geneses, a comprehensive analysis of the formation of H2S anomaly coal mines revealed that BSR was the main factor controlling the anomalous enrichment of H2S, among H2S abnormal coal mines, the share of pure BSR-induced ones was 42.31%, while shares of BSR/TSR and BSR/TDS/TSR ones in mixed BSR-related cases were 19.23% and 7.69%, respectively, as shown in Figure 1. It can be observed that the anomalies of free-state H2S and adsorbed-state H2S have coincident formation geneses, namely, BSR, magmatism, TSR, and the mixed BSR/ TSR and BSR/ TDS/ TSR causes. Except for magmatism, the anomaly of water-soluble H2S shows almost the same formation geneses with free-state and adsorbed-state H2S anomalies.
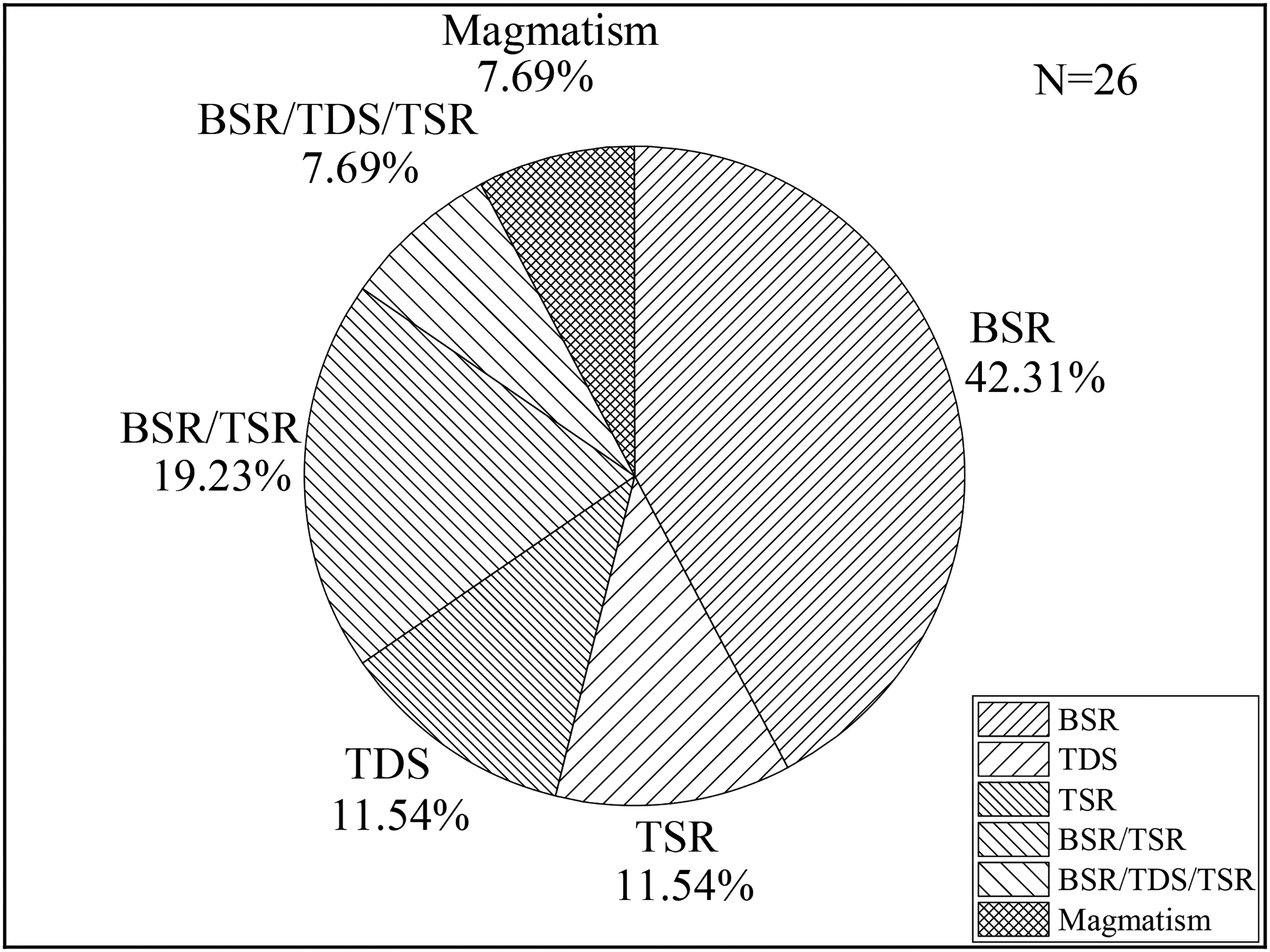
Geneses of hydrogen sulfide (H2S) anomalous enrichment in China's coal seams.
As shown in Table 1 and Figure 2, H2S anomaly areas in China's coal seams are mainly distributed in Inner Mongolia, Hebei, and Shanxi in North China, Shandong in East China, Sichuan and Chongqing in Southwest China, Xinjiang, Shaanxi, and Ningxia in Northwest China, and Henan and Hunan in Central China. H2S anomaly areas in coal seams are mainly found in Carboniferous-Permian and Jurassic coal series. In terms of anomaly genesis, BSR can primarily account for H2S anomaly in China, followed by the mixed genesis, while magmatism is rarely found (see Figure 1). As listed in Table 1, BSR genesis is mainly responsible for H2S anomalies in North and Northwest China. In contrast, H2S anomalies in East and Southwest China were formed mainly under magmatism and TDS (see Table 1).
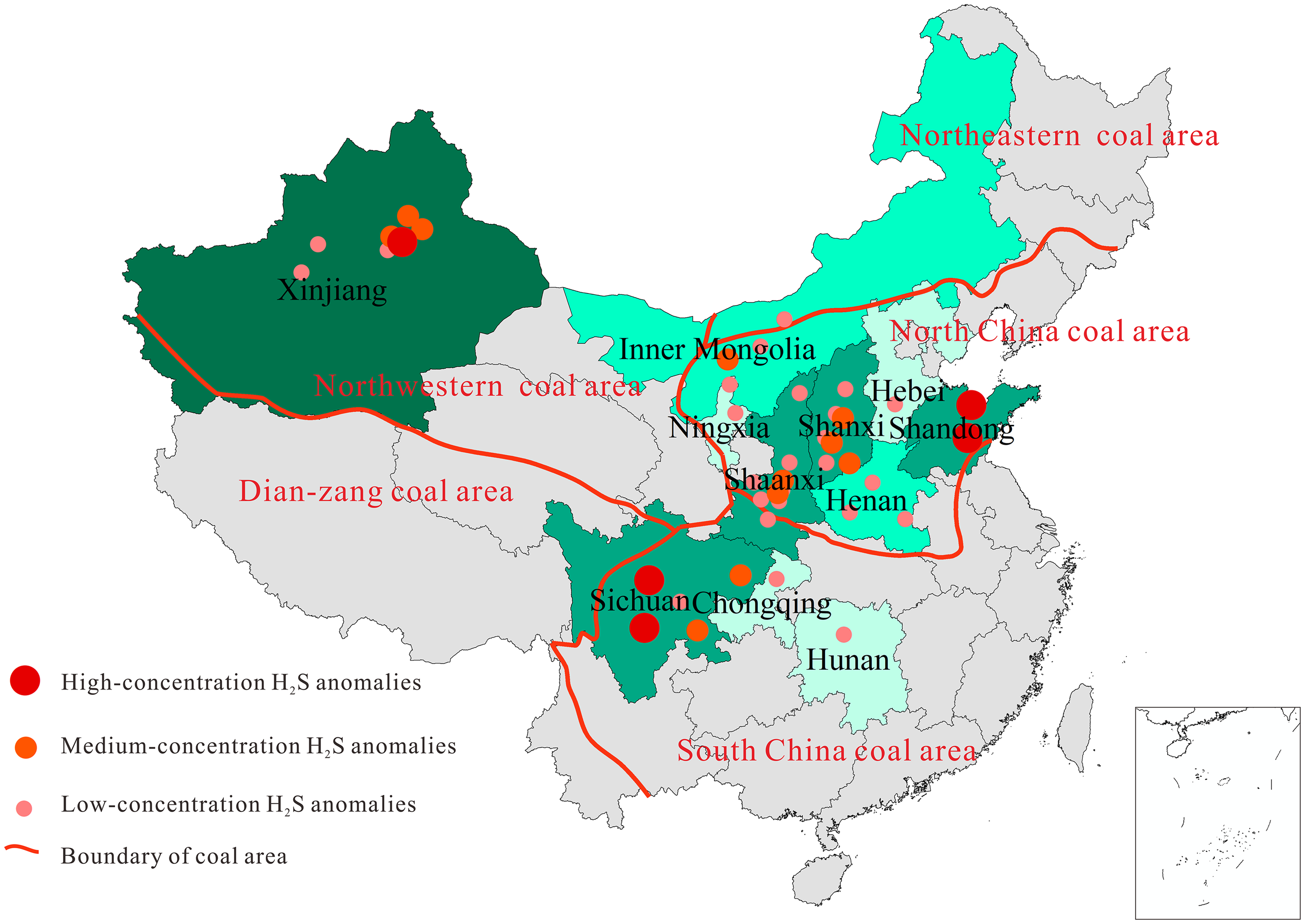
Distribution of hydrogen sulfide (H2S) anomaly areas in China's coal seams, with account of findings of Tang et al., 2015 and Liu et al., 2019.
Distribution characteristics of H2S anomalous areas in China's coal mines.
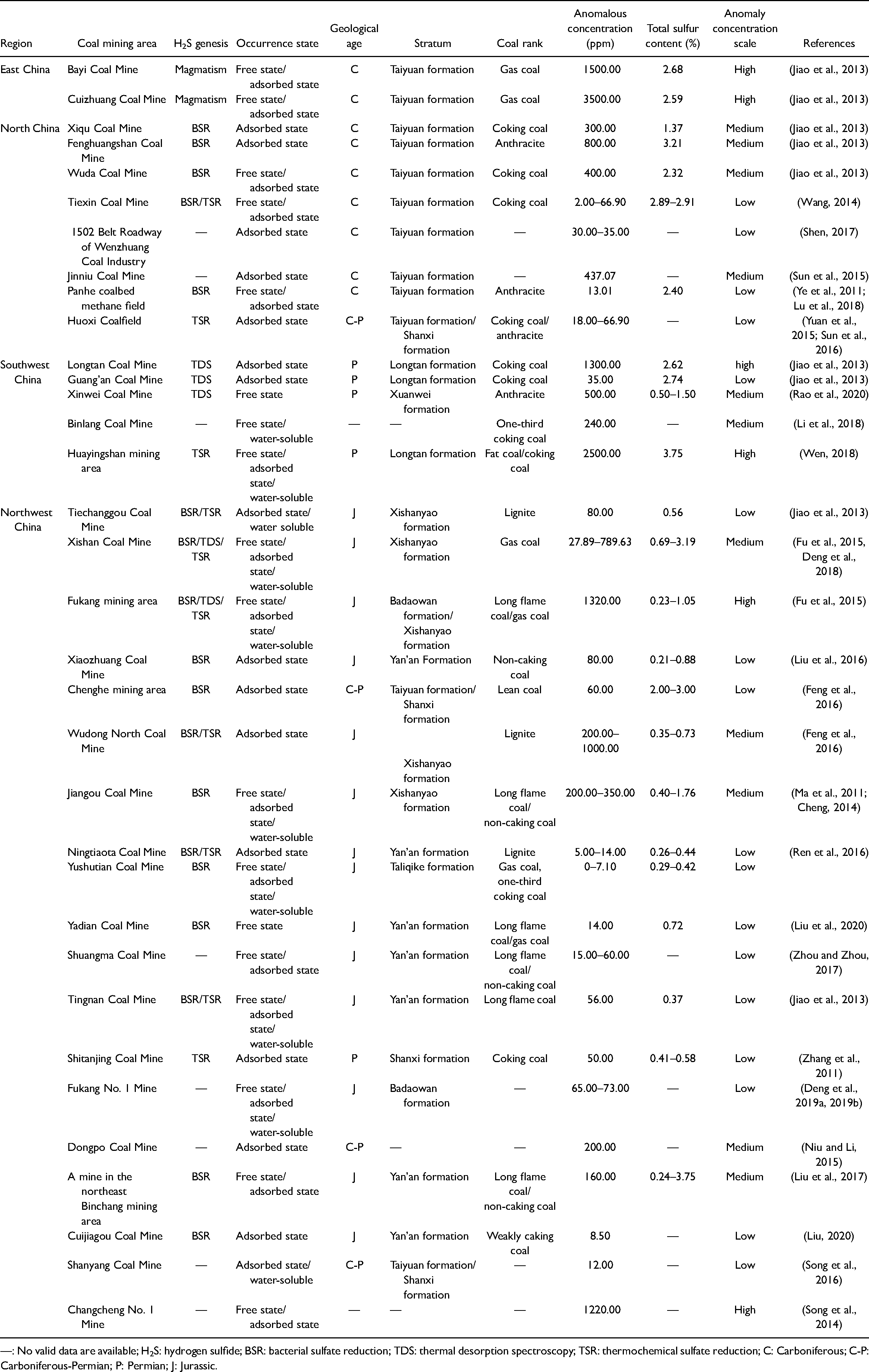
—: No valid data are available; H2S: hydrogen sulfide; BSR: bacterial sulfate reduction; TDS: thermal desorption spectroscopy; TSR: thermochemical sulfate reduction; C: Carboniferous; C-P: Carboniferous-Permian; P: Permian; J: Jurassic.
In terms of occurrence state, as shown in Table 1, H2S in China's anomalous coal seams exists in the adsorbed state; free-state H2S is relatively common in East and North China, while water-soluble H2S is scarce. H2S in free and water-soluble states is rarely found in Southwest China. In contrast, in Northwest China, H2S anomaly coal seams contain H2S in the adsorbed, water-soluble, and free states.
H2S anomalies in China are present in low-, medium-, and high-rank coal seams. However, these coal seams differ in coal type among different regions. Specifically, H2S anomaly coal seams in East China are mainly composed of gas coal, as shown in Table 1. H2S anomaly coal seams in Southwest China are mainly composed of fat coal, coking coal, and anthracite, while those in Northwest China are mainly composed of lignite, long flame coal, non-caking coal, gas coal, and gas-fat coal.
Based on the concentration of H2S in coal gas, H2S anomalies can be subdivided into high-concentration (>1000 ppm), medium-concentration (100–1000 ppm), and low-concentration (6.6–100 ppm) ones. Most of China's H2S abnormal coal seams are medium- and low-concentration ones, while high-concentration H2S anomalies are scarce. The H2S anomaly concentration scale is related to H2S genesis. As shown in Table 1, the H2S anomaly concentration scale in coal seams nationwide is dominated by medium- and low-concentration anomalies, while high-concentration ones are relatively rare. The abnormal concentration scale is related to the origin of H2S. The total sulfur content and abnormal concentration scale of H2S related to BSR and TSR are lower, while those related to magmatism are generally higher. In general, the total sulfur content and abnormal concentration scales in high-rank coals are generally higher than those in middle- and low-rank ones, and the total sulfur content and H2S concentration exhibit a pronounced positive correlation (Figure 3). For example, in North China, anthracite coals generally have higher total sulfur content and abnormal concentration scale coking coals, with several exceptions. The first one is the Fenghuangshan Coal Mine: although its total sulfur content is as high as 3.21%, its abnormal concentration scale is only moderately abnormal, not reaching a high-concentration anomaly level. The second one is the Xinwei Coal Mine, although its coal rank is anthracite, its total sulfur content is not high. The medium-rank coal (including fat coal and coking coal), with high total sulfur content and anomaly concentration, prevails in Southwest China (Zhao et al., 2021b). Coal seams in Northwest China mainly contain low- and medium-rank coals, including lignite, long flame coal, non-caking coal, gas coal, and gas-fat coal, with low and medium total sulfur content and anomaly concentration.
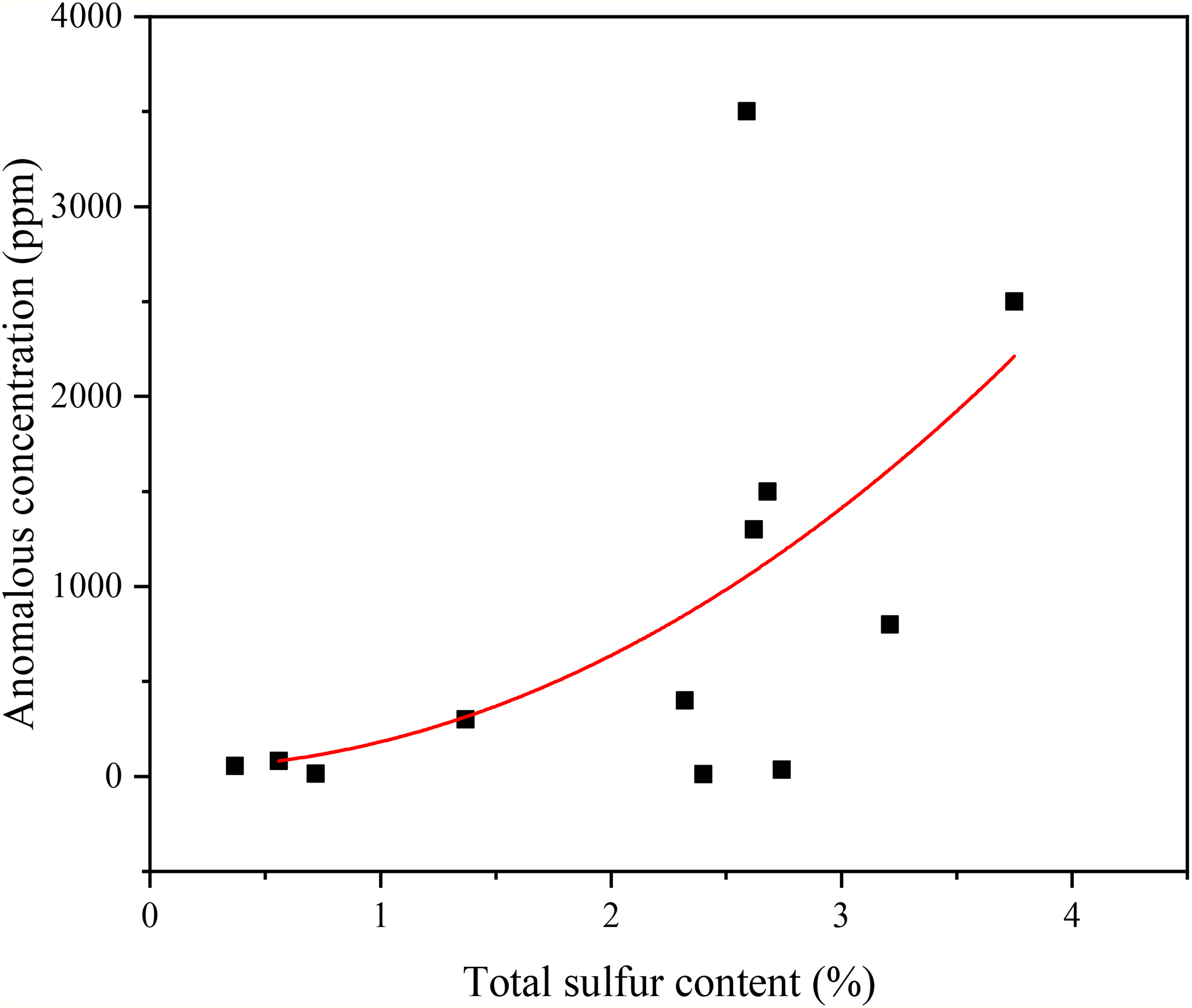
The relationship between total sulfur content in coal and hydrogen sulfide (H2S) concentration.
Free-state H2S occurrence
Free-state H2S is generally found in coal seams with well-developed fractures and weak hydrodynamic conditions. Besides, coal seam roofs and floors, as the overlying rocks, are compact in lithology. Numerous researchers have revealed dependencies between tectonic activities, burial depth, coal quality, and free-state H2S via field measurements and numerical simulations. Gas-collecting bags are commonly used to collect coal bed methane (CBM) samples with free-state H2S. After being separated by the chromatographic column, H2S is combusted in the reaction kettle. The combustion products react with ozone, and the amplification reaction produces chemoluminescence, which is detected by a photomultiplier. Accordingly, the content of H2S in a CBM sample can be analyzed in the data analysis module. This method is quite expensive and involves potentially harmful oxidizing agents, such as ozone (Cheng et al., 2013).
By performing in-situ measurements and numerical simulations on the content of H2S in coal seams, Fu et al. (2015) revealed the effects of burial depth and coal quality on H2S in coal seams of the Xishan Coal Mine, Xinjiang, China. They reported that the H2S content was in negative correlation with the contents of CBM, CH4, CO2, and N2. Besides, the content of H2S was also in negative correlation with moisture (Figure 4(a)) and ash yield (Figure 4(b)), and in positive correlation with the content of volatile yield (Figure 4(c)) and total sulfur (Figure 4(d)). However, no obvious correlation between the H2S content and the coal seam burial depth was detected (Figure 4(e)). Besides, they analyzed the genesis of the H2S anomaly and reported that the coal seam partly absorbed the generated H2S under magmatism of diabase at the late Yanshan Orogeny in the No. 3 Coal Seam of the Bayi Coal Mine in Zaozhuang, China. The remaining free-state H2S was distributed in pores and fractures of coal seams. Thus, anomaly enrichment areas of free-state H2S were formed on the west side of the dry rock wall with no water or faults (Song et al., 2016).
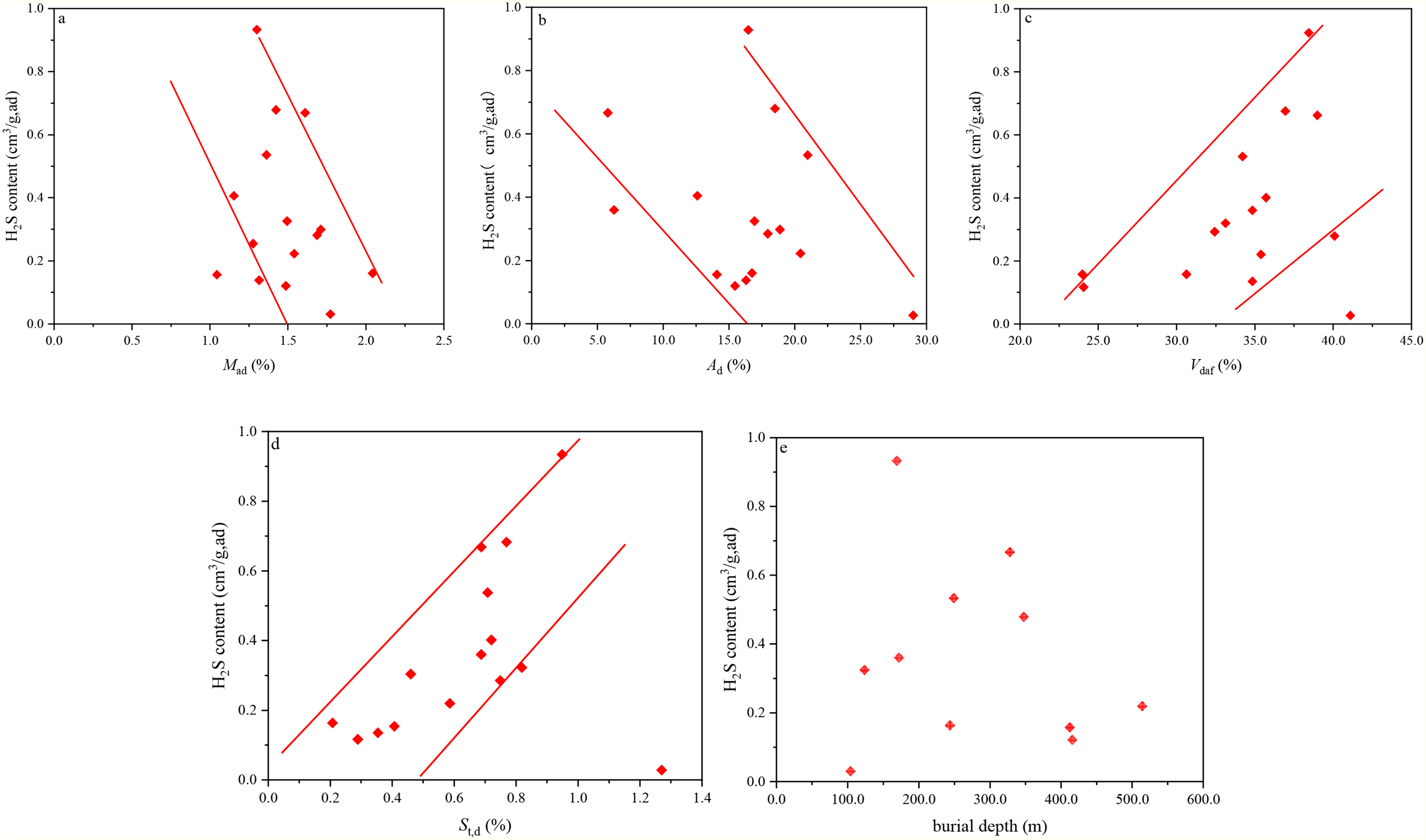
Measured H2S content versus water content (a), ash content (b), volatile matter yield (c), total sulfur content (d), and burial depth (e) in the Xinjiang Xishan Coal Mine (Liu, 2014).
Free-state H2S diffusion to the coal mining tunnels is quite high; therefore, minimizing the concentration of free-state H2S in tunnels is vital to ensure miners’ safety. Safe and convenient passive protective ways of reducing H2S hazards in mining tunnels imply more effective ventilation methods, increased exhaust air rate, spraying alkaline fog into the air for neutralization, and wearing anti-H2S masks. For example, in the +469 m B3+6 fully mechanized caving face on the east wing in the Wudong Coal Mine's western region (Gao, 2020), the mined-out area and the coal seam were above and below the working face, respectively. The measured concentration of H2S in the advanced detection hole reached 14,300 ppm. H2S in a free state would inevitably enter the tunnel's return flow during the working face's recovery process. The concentration of H2S was abnormally high (∼60 ppm) at the back of the working face. H2S in the adsorbed state could be further desorbed into a free-state one during the coal drawing process, increasing the latter's concentration and jeopardizing underground workers’ safety. The tunnel was ventilated with a 754.8 m3/min ventilation capacity to mitigate this problem, which dropped the concentration of H2S in the working face below 28 ppm. Besides, workers were obliged to wear anti-H2S masks for more effective personal protection. Another example is the Baozigou Mine in Gansu Jingchuan County, which is a low-concentration H2S anomaly coal mine (Jia et al., 2018). Before the treatment, the concentration of H2S in the working face was 90 ppm, which far exceeded the safety limit of 6.6 ppm. Some protective measures, such as spraying the alkaline liquid and improving the ventilation condition, were recommended to reduce the concentration of H2S to below 6.6 ppm. On the one hand, to avoid the appearance of large-vortex core regions in the working face, the air cylinder could be shifted by 3 m from the working face to ensure the migration of H2S toward the sidewall of the tunnel with the airflow rather than being taken to the vortex region. On the other hand, three high-pressure nozzles that were originally installed for absorbing free-state H2S and dust gushed from the drum during the coal cutting process could be removed, and six single high-pressure alkaline-liquid-sprayers should be reinstalled around the heading machine. As an optimal solution, two nozzles were set on the bottom of the cutting drum of the heading machine 5° toward the inside of the connecting rod, while two more nozzles were set on both sides of the front part of the heading machine at a spraying angle of 30° and 45°, respectively. The front nozzles absorbed the free-state H2S desorbed from the crushed coal near the absorbing air cylinder and H2S and dust in the convolutional airflow on the air return side, while the rear nozzles further purified the escaped H2S and dust. After adopting the above measures, the concentration of free-state H2S at a distance of 5 m from the heading machine's driving on the air intake side was reduced below 6.6 ppm. In particular, the concentration of free-state H2S on the air return side was reduced below 6.1 ppm, which satisfied the above safety requirements.
H2S in the adsorbed state
The adsorbed-state H2S is the main occurrence form of H2S in coal seams. At present, the H2S accumulation process is extensively explored via isothermal adsorption parallel tests, contrastive analysis, generalized gray-scale correlation analysis, and quantum chemical analysis.
Fu et al. (2011, 2015) conducted the H2S isothermal adsorption parallel tests under equilibrium water conditions, which revealed that the Langmuir curve could describe the H2S adsorption pattern by coal rock, i.e., the isothermal curve consisted of (i) rapid adsorption, (ii) relatively slow adsorption, and (iii) equilibrium adsorption stages, as shown in Figure 5. Moreover, the distribution of coal mines with abnormally high H2S concentration was controlled by tectonic structures (Meng and Li, 2018; Shen et al., 2018; Wang et al., 2018). For instance, the Fukang Coal Mine, located in the Mesozoic folded belt of the Bogeda Mountain between the Fukang and Yaomoshan Fractures, had H2S anomaly coal seams, mainly distributed in the protruded cambered part of the thrust nappe and well-developed tectonic coal regions. Similarly, the Choumeigou Mine, the Xinlong Mine, the Jinlong Mine, and the Kanglong Mine with H2S anomaly were located in the protruded cambered part in the middle thrust nappe, which were closed inverted anticline structures. The coal seams with high-concentration H2S anomaly were crushed, and gas escaping during the coal seam lifting process provided space for later adsorption of H2S. The coal seams were confined by the Nanchi steel reservoir fault, the Choumeigou reverse fault, the Ganhezi reverse fault, the Wugonggou fault, and the Xiaolongkou reverse fault. Accordingly, the late-adsorbed H2S was confined, which led to anomalous enrichment of absorbed-state H2S in the coal seams.
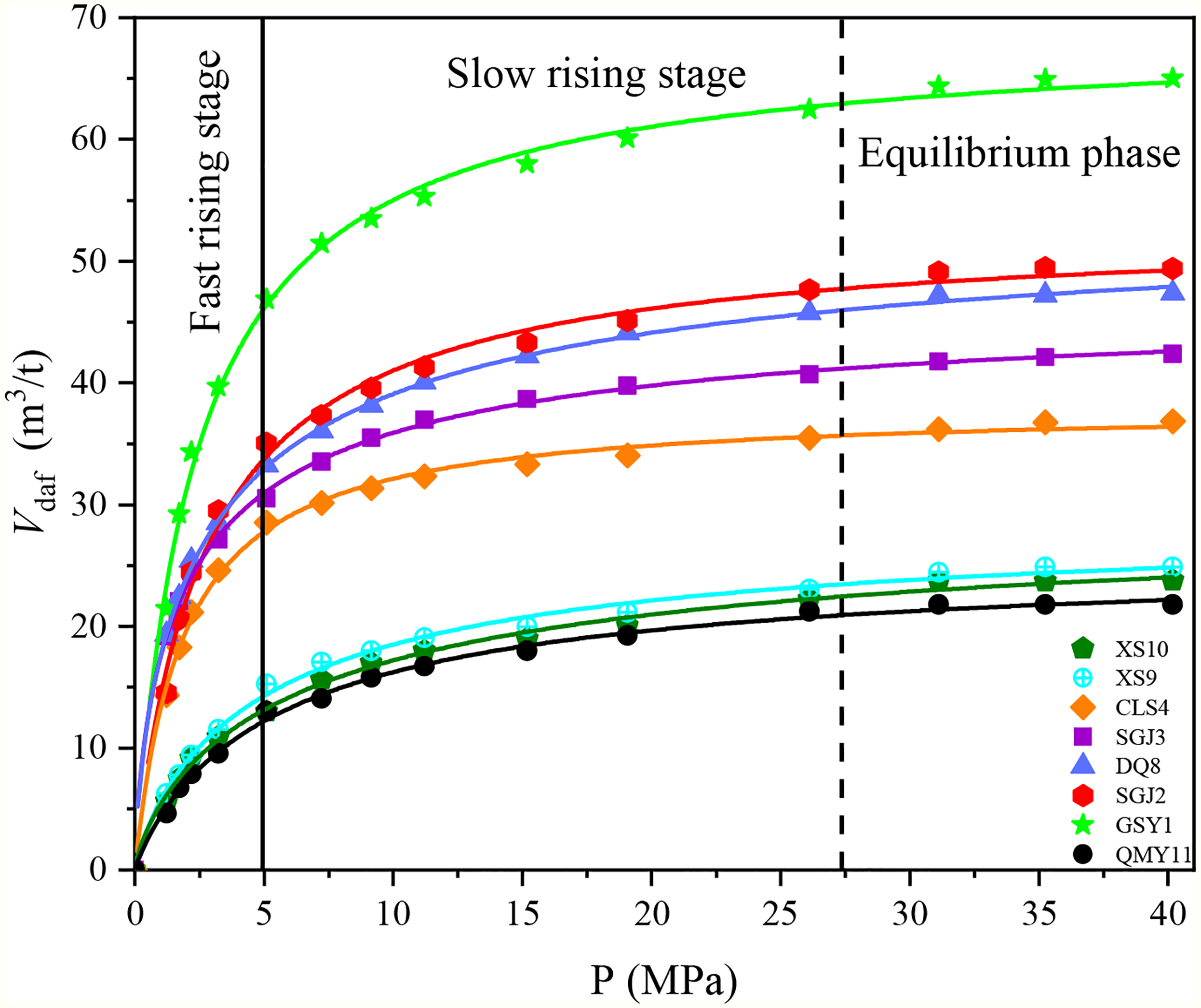
Isothermal adsorption curve of hydrogen sulfide (H2S) by coal at 30 °C: rapid adsorption, slow adsorption, and equilibrium stages (He et al., 2015).
Some scholars also carried out mercury injection tests and isothermal adsorption tests under equilibrium water conditions and analyzed the adsorption rules of H2S by coal at different ranks (He et al., 2015; Xue et al., 2016, 2017). It was found that pressure and coal rank were the main factors influencing the H2S-absorbing capacity of coal. As shown in Figure 6, coal's H2S-adsorbing capacity increased with pressure and the degree of coal metamorphism. Besides, H2S adsorption by coal also depended on the pore distribution characteristics of the coal sample. A larger number of micro- and transition pores contributed to the adsorption of H2S by coal, while medium and large pores were unfavorable for the adsorption (Cheng et al., 2017; Guo et al., 2007; Luo et al., 2014).
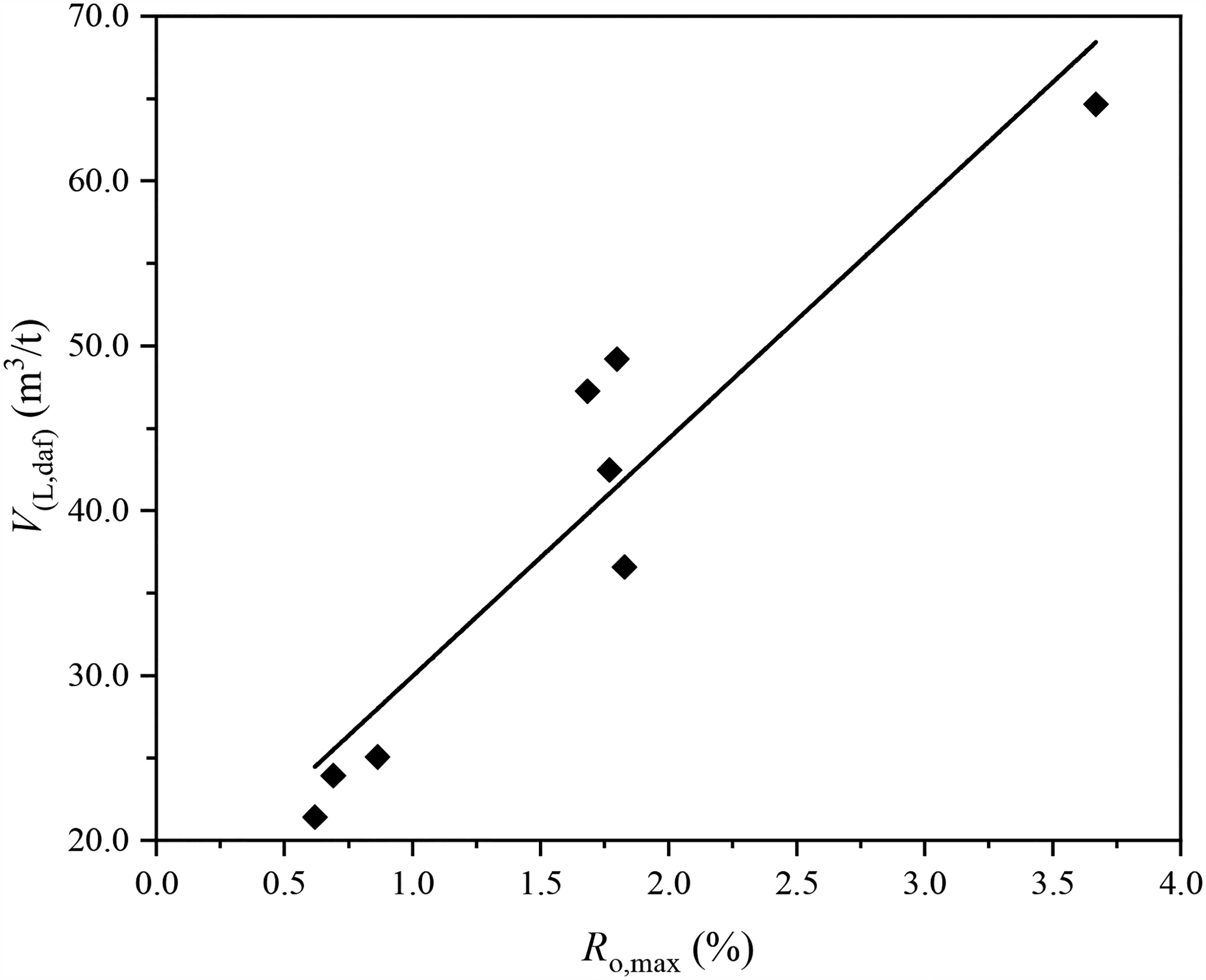
Maximum reflectivity of the vitrinite versus hydrogen sulfide (H2S) adsorption by coal (He et al., 2015).
Lin et al. (2017) and Zhang (2018) analyzed the effects of coal seam's thermal evolution temperature, adsorption characteristics, pore characteristics, total sulfur content, and the reducibility index on anomalous H2S enrichment. They elaborated a method of quantitative determination of the generalized relation degree of various factors. The effect of the studied factors on anomalous H2S enrichment was ranked in the decreasing order as follows: the reducibility index, the content of total sulfur, the adsorption constant, evolutional thermal temperature, the Brunauer–Emmett–Teller (BET) specific surface area, and the burial depth. Further analysis revealed that large fractional dimensions, more complex pores, uneven surface, high looseness degree, large specific area, and large adsorption constant promoted the adsorption of H2S in coal seams, causing high-concentration anomaly of adsorbed-state H2S. Besides, it was found that the adsorption of H2S positively correlated with the content of total sulfur, so that the latter could be used for roughly evaluating the adsorbing capability of H2S in the research area.
Liang et al. (2016) analyzed the adsorbed H2S characteristics of coal surfaces via quantum chemical analysis. They established the molecular model of H2S-containing coal surface in Tiexin, Shanxi, to assess the adsorption energy of H2S and CH4 by coal surface molecules (Bertoncini et al., 2000; Yang et al., 2002). It was found that under the co-existence condition of H2S and CH4, the adsorption energy values of H2S and CH4 by coal were 2.230 and 94.861 kJ/mol, respectively. Therefore, the coal seam's adsorption of CH4 exceeded that of H2S, so that the former process inhibited the latter one. Through calculation, the adsorption energy of the mixed gas exceeded the sum of the individual adsorption energy values of the single gas with the same numbers and kinds, suggesting that coal's adsorption capability of the H2S/CH4 mixed gas far exceeded the adsorption of a single gas. Therefore, H2S promoted the adsorption of CH4 by the coal seam.
Based on the occurrence characteristics of adsorbed-stated H2S in the coal seam, some active measures such as advanced detection, coal seam pressure-difference pre-drainage H2S, and spraying alkaline liquid for the neutralization of H2S can be applied. Since active prevention and treatment should always consider the coal seams’ occurrence condition and the mining technologies, they are expensive and problematic. The Gaojiabao Coal Mine was used as a case study by several researchers (Dai et al., 2002; Wu et al., 2016; Xu, 2020); the concentration of H2S in the No. 4 coal seam was ∼50 ppm. The adsorbed-state H2S in coal seams could be easily disturbed and spread into the air. The recommended measures for controlling the adsorbed-state H2S in the coal seams were reduced to drilling the coal seam surface for advanced detection of H2S before mining, followed by the injection of alkaline liquid in the H2S anomaly regions of the coal seam for the neutralization of H2S. Moreover, to enhance the treatment performance, the corresponding alkaline injection amount could be assessed by the distribution characteristics of H2S content in the coal seams (Zhang et al., 2020). Given the neutralization capability, treatment efficiency, cost, and equipment structure, NaHCO3 was a lucrative alkali-injection solute. The analysis of physical properties of coal seams in the Gaojiabao Coal Mine, operation safety, the requirement on drilling equipment, and the final hole sealing difficulty and degree revealed that a drilling hole with a 65 mm diameter provided the optimal solution. When the overall drilling length exceeded the coal seam width, the overflow induced by excessive alkaline liquid amount could be effectively avoided. Based on the working face length, the drilling depth was set at 80 m. Before injecting the alkaline liquid, the airflow's H2S concentrations in the working face and the air return ways reached 30 and 40 ppm, respectively. After the injection of alkaline liquid, they were reduced to 11 and 22 ppm, i.e., by 60% and 50%, respectively, which implied a good governance effect.
Water-soluble H2S
Liu (2014) pointed out that the burned areas in the coal mines at the southeast margin of the Junggar Basin included well-developed fractures: the surface water seeped through burnt rocks forming underground water storage units. Under the spontaneous combustion of coal seams, the sulfur in the coal was partly oxidized to SO2; the latter was dissolved in water, forming sulfate ions, which were then decomposed to produce H2S under TDS and TSR mechanisms. The generated H2S gas could be dissolved in the coal seams of burnt rocks or driven away by the underground water, thereby forming water-soluble anomalous H2S enrichment areas in the regions with poorly developed burnt rocks or favorable water-resisting layers (Cai et al., 2009; Su et al., 2017). Deng et al. (2017, 2018, 2020) analyzed mineral (underground) water characteristics at the southeast margin of the Junggar Basin and H2S genesis. They revealed the co-existence of large content of H2S with water in the coal seams. Moreover, the H2S content positively correlated with the CO content and pressure value. No correlation between the H2S content and the coal seam burial depth was detected, as shown in Figures 7 and 8.
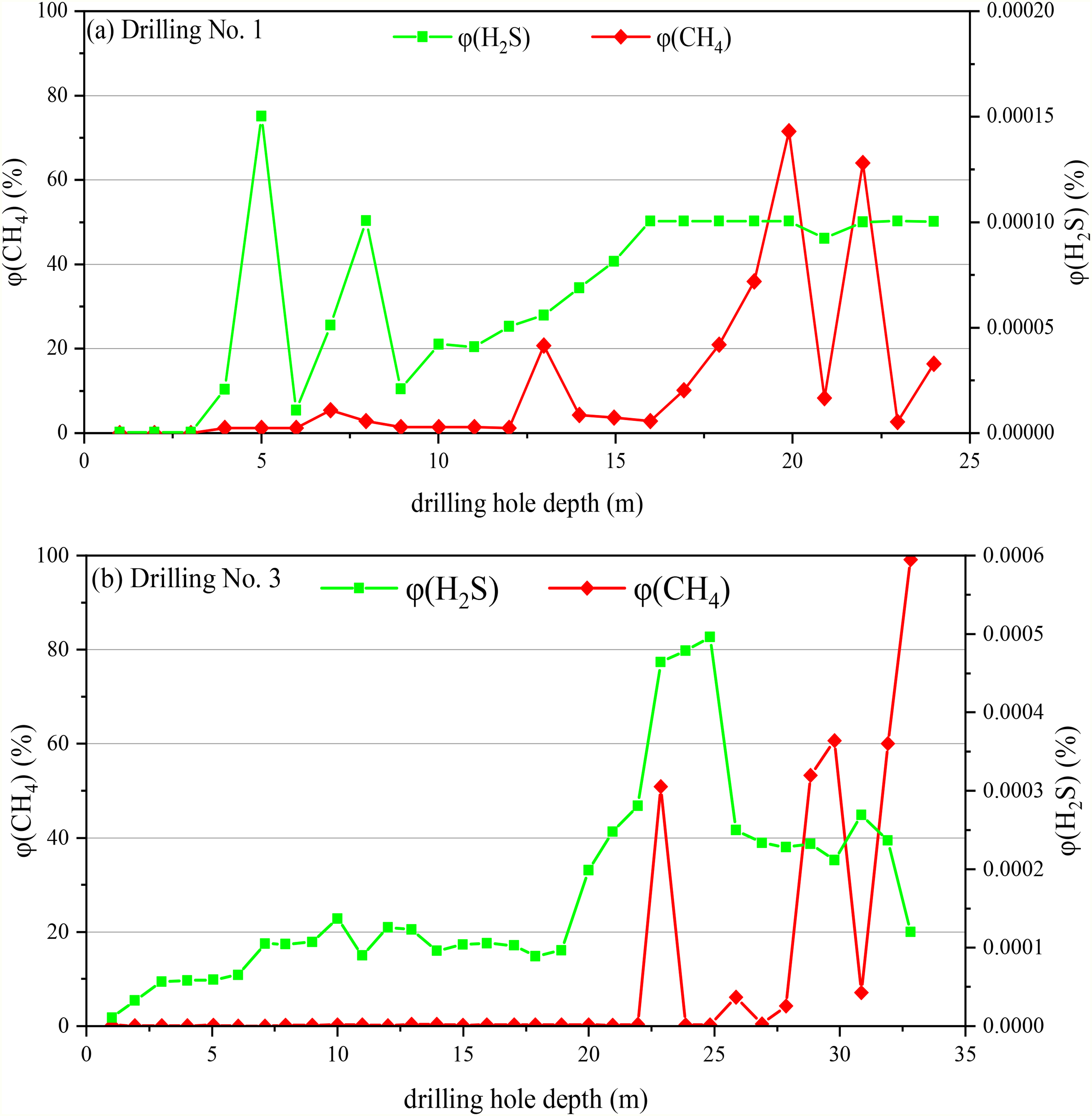
Hydrogen sulfide (H2S) and CH4 contents versus drilling hole depth in Xinjiang Xishan Coal Mine (Deng et al., 2018).
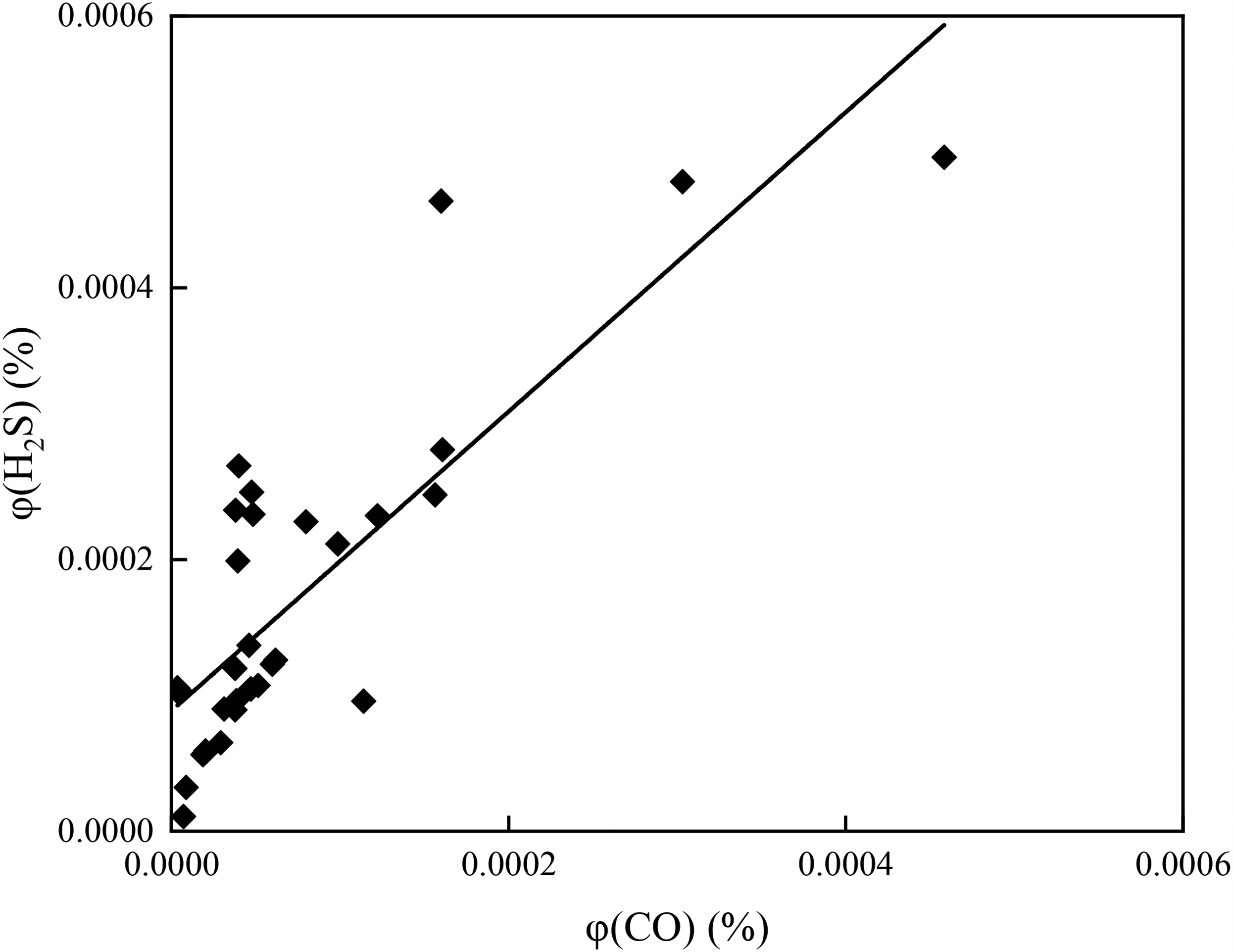
Hydrogen sulfide (H2S) content versus CO content in the drilling hole of the Xinjiang Xishan Coal Mine (Data from (Deng et al., 2018)).
Generally, CH4 in water has a form of a water-soluble gas with quite stable properties. When water is abundant in SRB, the latter can use CH4 in water as a sulfate for reductive dissolution of a hydrogen donor, producing H2S under dissimilation and promoting the reaction between CO2 in water and soluble Ca2+ ions to form calcium carbonate crystals. BSR reaction is a kind of exothermic reaction. High temperature can suppress the forward reaction; therefore, the decline in water temperature is favorable for the production of H2S, and simultaneously more CH4 can be consumed (Song et al., 2017). Consider a particular case: the southern margin of the Junggar Basin contained coal with a great amount of carbonate, sulfate, and abundant organic matter. The No. 4 spring showed declining temperature year by year, leading to constant consumption of CH4 and increasing H2S and CO2 contents. Moreover, the content of positive Ca2+ ions in confined water at deep coal mines dropped, and burn-in regional coal rocks mostly occurred in the shallow part. A limited amount of H2S was produced under TDS and TSR. These hydrochemical characteristics strongly indicated that active BSR action was the principal cause of the anomaly concentration of water-soluble H2S in this region (Deng et al., 2018).
Water-soluble H2S can generally be governed via sealing, dredging drainage, and spraying lime powder into the water gushing port. The following analysis was performed by taking the Huayingshan Mine and the Xishan Coal Mine in Urumqi as examples. On account of the karst landscape and complexly developed underground rivers, the maximum water inflow in the Huayingshan Mine reached up to 200,000 m3/h (Lei et al., 2011). Moreover, H2S escaped from fractures and could be dissolved in water. The aquifer in coal seams was close to the coal seam, triggering water inrush accidents and bringing a great threat to safety production. Some sealing and dredging drainage measures were recommended to prevent the potential hazard caused by the prevention of water-soluble H2S. Multi-component composite grouts (including barite powder, bentonite, and sodium carboxymethyl cellulose binder) were the most lucrative for sealing pores and fractures in the coal seams, isolating water, and achieve the goal of sealing water-soluble H2S. Next, water-soluble H2S could be dredged and discharged to the specified positions, and lime or alkaline liquid would be sprayed for neutralization. Insofar as the Xishan Coal Mine had a moderate water inrush of about 924.43 m3/h and high content of H2S in the water of 38 ppm (Fu et al., 2015), it was recommended that lime or alkaline liquid should be periodically sprayed into water gushing port for prevention. The tunnel should also maintain regular water discharge to prevent gas dissolution in water and reduce the hazard risks.
Prospects
Due to the limitations of available experimental and calculation techniques, the main research efforts have been focused on the occurrence state of adsorbed-state H2S in coal seams. However, the occurrence states of free-state and water-soluble H2S are also topical, insofar as free-state and water-soluble H2S is more inclined to rush and leak, causing enormous potential safety hazards to safety production. Scholars can improve the sampling techniques and the current methods and explore innovative theories in the future to reduce the potential risks. Firstly, the sampling schemes of free-state and water-soluble H2S can be enhanced in terms of occurrence to explore more targeted sampling techniques. Secondly, the existing dissolved quantity measurement methods should be refined. For example, physical extraction or reactive precipitation method can be considered for enhancing measurement accuracy. Thirdly, some portable devices for non-contact rapid on-site measurement and the analysis of H2S content of collected water and gas samples should be developed to acquire first-hand information at the site under the premise of ensuring personnel safety. Fourthly, the physical properties of coal seams on the storage and migration of H2S-containing coal-water should be investigated in depth. Fifthly, the effects of hydrodynamic and fire-burning conditions on acidic gas generation and storage mechanism should be clarified. Finally, the difference in sulfur utilization capability of different ranks of coals by SRB under the same hydrodynamic conditions should be examined in detail. However, most available active or passive H2S prevention techniques can only be regarded as local prevention measures. There is still a large gap between safety in a coal mine and high-efficiency mining. In future studies aiming to establish comprehensive and efficient H2S prevention techniques, more fundamental research efforts should be made to develop economical, safe, and high-efficiency H2S absorbents for particular site conditions.
Conclusions
High-concentration H2S coal seams in China's coal seams are mainly found in the Carboniferous, Permian, and Jurassic series, located in Xinjiang, Shandon, Hebei, Henan, Shanxi, Hunan, Sichuan, Shannxi, Ningxia, and Inner Mongolia. BSR genesis plays a dominant role in H2S anomalies (mainly medium- and low-concentration H2S anomalies). H2S anomaly mainly exists in adsorbed states, while H2S anomalies in water-soluble and free states are relatively scarce.
Free-state H2S generally exists in the coal seams with developed fractures and weak hydrodynamic conditions. Both roofs and floors, as the overlying layers, show compact lithological characteristics. The burial depth and coal quality can significantly affect the enrichment of free-state H2S in coal seams.
The Langmuir curves adequately describe the adsorption characteristics of adsorbed-state H2S. The maximum adsorption capacity of H2S by the coal seams positively correlates with the number of transition pores and micropores, being negatively correlated with the number of medium and large pores. In terms of the effects on anomaly enrichment of adsorbed-state H2S, the reducibility index ranks the first, followed by total sulfur content, the adsorption constant, the thermal evolution temperature, the BET specific area, and the burial depth.
In coal seams, the reduction of water temperature can promote BSR reaction for the production of H2S, and the coal fire area in the shallow seam is conductive to TDS or TSR reaction. Anomaly enrichment areas of water-soluble H2S are easily formed in the regions with poorly developed burnt rocks or favorable water-resisting layers.
Some measures can be adopted for the prevention of H2S in coal mines. In terms of free-state H2S, such measures include: changing the ventilation mode to a more effective one, increasing the exhaust air rate, spraying alkaline fog into the air for neutralization, and wearing anti-H2S masks to reduce the hazard. In terms of adsorbed-state H2S, the most effective would be the advanced detection, pressure-difference pre-pumping H2S, and spraying alkaline liquid for neutralization. In terms of water-soluble H2S, the above measures can be reduced to sealing, dredging, and spraying lime powder at the water gushing port for governance.
More research efforts should be focused on free-state and water-soluble H2S, the improvement of sampling techniques, experimental methods, and the development of relevant innovative theories to reduce the H2S-related safety risks.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by the National Natural Science Foundation of China (grant nos. 41902171 and U1903303), the Natural Science Foundation of Xinjiang Uygur Autonomous Region (2018D01C050), the Scientific Research Program of the Higher Education Institution of Xinjiang (XJEDU2018Y014), and the 2017 PhD Research Startup Foundation of Xinjiang University (BS180256).
