Abstract
With the expansion of the scale of coal mining, the safety problems caused by the reburning of coal are becoming more and more serious. In this paper, the pyrolysis characteristics of gas coal and the exothermic characteristics of reoxidation of residues were studied by using a synchronous thermal analyzer. The functional groups of pyrolysis residues were tested, and the group content and characteristic structural parameters were calculated based on quantum chemistry method. The results show that with the increase of pyrolysis temperature, Volatile maximum separation rate (Vmax) and the change in the residual weight of the coal sample (ΔWvp) increase. The increase of temperature will lead to the decrease of hydroxyl and aliphatic hydrocarbon content in coal, and the increase of aromatic hydrocarbon. With the deepening of pyrolysis, the ignition point temperature of coal samples decreases first and then rises, the combustion intensity and combustion concentration are strengthened. The pyrolysis results show that 462.8°C is the critical temperature for the transition during pyrolysis. The ignition point of the residue is less affected by the pyrolysis conditions, and the ignition temperature of the raw coal and the pyrolysis residue varies within 330.57°C–334.98°C.
Introduction
Coal field fire burned a large amount of coal resources (Cheng et al., 2021; Shen and Zeng, 2021; Zhang et al., 2020d; Zheng et al., 2020b), indirectly caused tens of times of the stagnant resources unexploited, produced a large number of greenhouse gases and toxic and harmful substances seriously harm the local ecological environment and groundwater resources (Zhang et al., 2004, 2020c; Zhong and Hu, 2008, Zhong et al., 2008).
Ningwu Coalfield is one of the six largest coal fields in Shanxi Province (Yang et al., 2016). It is located in the northern part of Shanxi Province. It has four mining areas: Pingshuo, Shuonan, Xuangang, and Lanxian. The coal field covers an area of approximately 2760 square kilometers. It has large coal reserves and a high degree of exploration (Liu et al., 2020a; Sun et al., 2013; Yang et al., 2016). Gas coal is the main coal type in Pingshuo mining area. It is very easy to ignite spontaneously. During the strip mining process, the coal seam reburns and ignites seriously. In the Pingshuo mining area, a large amount of coal oxidation and combustion is caused by serious air leakage in abandoned roadways and gobs of small coal kilns. The coal in a low-oxygen state is affected by high temperature and undergoes pyrolysis to varying degrees, producing a large amount of pyrolysis residual coal. Pyrolysis breaks the macromolecular structure in coal, which produces more active small molecules and flammable substances. These substances will make the pyrolysis residual coal more likely to be oxidized and reburned than the original coal. With the progress of strip mining, the oxidation area has been continuously exposed, and a large amount of pyrolysis residual coal has been exposed to air and reburning, resulting in a large area of oxidation high temperature zone and open flame (Figure 1), which not only destroys the surrounding ecology environment, but also waste a lot of coal resources. This caused huge disasters to safety production. In order to accurately monitor and prevent the occurrence of coal field fires in Pingshuo mining area, it is urgent to study the reburning characteristics of Pingshuo gas coal pyrolysis residues.

Small coal mines reburning after the surface was uncovered.
At present, researchers mostly use thermogravimetric analysis to study the reaction characteristics of coal pyrolysis and combustion (Hameed et al., 2020; Jayaraman et al., 2017; Kanca, 2020; Lin et al., 2014; Liu et al., 2020b; Seo et al., 2011). The changes of groups during coal pyrolysis and combustion were analyzed by in-situ Fourier transform infrared spectroscopy (Chen et al., 2020; Dou et al., 2014; Feng et al., 2006; Liu et al., 2018; Niu et al., 2016; Qi et al., 2019; Zhang et al., 2021; Zhao et al., 2020). The TGA-DSC was used to study the effect of metal oxides on the catalytic combustion of flame coal (Zhang et al., 2020a). The effects of heating rate and coal type on the yield of functional coal-tar components using GC-MS (Okumura, 2017). The importance of aromatic structures in tar to the destruction of tar itself during the volatile-char interactions was studied (Song et al., 2015). Some researcher made the char samples burned in a thermogravinietric analyzer to study the intrinsic reactivity of char samples (Chen et al., 2004). Zhang found that with the increase of the pyrolysis temperature, the extension of the pyrolysis time and the decrease of the coal particle size, the combustion reactivity of the semi-coke decreases (Zhang et al., 2019). Wang. used thermogravimetry to study the pyrolysis characteristics of bituminous coal, and he found that the formation of CH4 in bituminous coal mainly occurs in the depolymerizing stage. The formation of H2 mainly occurs in the condensation polymerization stage, and its formation requires higher temperatures (Wang et al., 2020). The used thermogravimetric analysis to study the pyrolysis kinetics of ultrafine coal, providing a new idea for explaining the effect of particle size on coal pyrolysis characteristics (Liu et al., 2017; Zhang et al., 2020b). Yu investigated the coking coal sample solution-extraction rate, chemical component and major functional group changes which based on the chromatography and fourier transform infrared spectroscopy (Yu et al., 2018). Zheng conducted in-situ FTIR and high-temperature oxidation experiments on raw coal samples, and studied the changing trend of functional groups and the corresponding gas generation rules (Zheng et al., 2020a). Qi used in-situ Fourier transform infrared spectroscopy to study the real-time changes of aliphatic hydrocarbons and oxygen-containing groups of lignite in the temperature range of 30°C–220°C (Qi et al., 2014). Xu used in-situ infrared spectroscopy to study the effect of pyrolysis temperature on the structure and functional groups of lignite (Lin et al., 2014). Cui used a Fourier transform infrared instrument to study the effect of temperature on the types and contents of groups in Datong coal, and explained the evolution mechanism of oxygen-containing functional groups during coal pyrolysis (Cui et al., 2017). Niu used in-situ Fourier transform infrared spectroscopy to study the evolution of the six main functional groups of the two coals during the pyrolysis process (Niu et al., 2018). According to the evolution of the functional groups during the pyrolysis process, the pyrolysis stage characteristics were proposed. Although, there are not much researches on the residual structure and reburning characteristics of coal pyrolysis (Xin et al., 2020), these researches provided the feasible experimental methods.
Based on previous studies, in view of the problem of reburning the residual structure of gas coal in the fire area of Pingshuo coalfield, according to the temperature distribution characteristics of the fire area, this paper uses the TA-Q600 synchronous thermal analyzer to conduct a constant temperature pyrolysis experiment on the coal in the fire area. The TG-DTG curve of gas coal at 350°C–500°C is obtained. The quality characteristics of coal samples at different pyrolysis temperatures are analyzed. The group distribution of gas coal pyrolysis residues were researched using in-situ Fourier transform infrared spectrometer at different pyrolysis temperatures. The changes of functional groups and characteristic structural parameters were analyzed. The oxidation characteristics of pyrolysis residues were the key factors affecting the reburning of coal. The oxidation experiments of gas coal pyrolysis residue were carried out, and the heat release characteristics during the reburning process were analyzed. The research results illustrated the process factors of the reburning of the Pingshuo coalfield fire. It is great significance for the safety of strip mining and control of the Pingshuo coalfield fire.
Experimental section
Coal sample
Coal sample from the East strip mine of Ningwu (DLT) coalfield in Shuozhou of Shanxi Province in China, it was collected and wrapped with plastic wrap in a vacuum vessel to the laboratory. The coal samples were ground in the vacuum glove box, and in the range of 200–300 mesh were selected as the experimental samples. The results of the coal proximate and ultimate analyses were listed in Table 1.
Ages, mines, origins, vitrinite reflectance, rank, proximate and ultimate analyses for DLT gas coal 200–300 mesh in size.
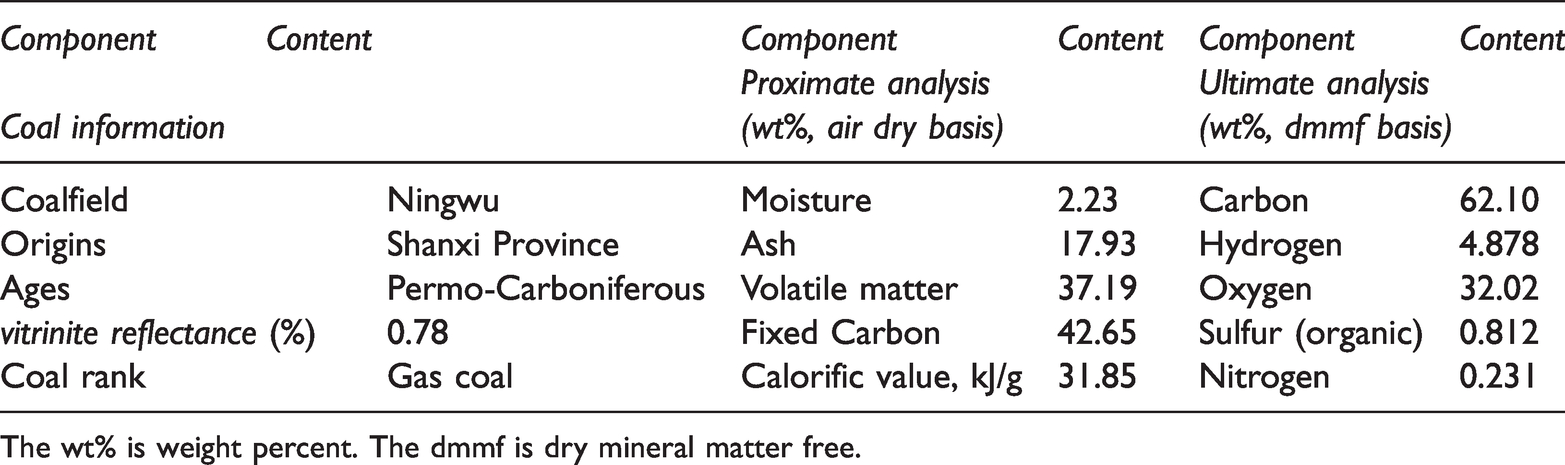
The wt% is weight percent. The dmmf is dry mineral matter free.
Testing facility
In this paper, Differential thermal analysis (TG, DTG and DSC) was performed the TA-Q600 simultaneous thermal analyzer (TA Instruments, Unit States), the instrument has been equipped with an extremely accurate AST10 series mass flow controller. The molecular structure and functional group content of the pyrolysis residue were analyzed by Nicolet 6700 infrared spectrometer.
Experimental procedures
In order to analyze the macroscopic mass change and pyrolysis characteristic parameter characteristics of gas coal at different pyrolysis temperatures. Take 10 mg of 200–300 mesh coal sample and put it into the crucible to start the experiment. Pyrolysis was carried out in a nitrogen atmosphere (100 ml/min) and the heating rate is 10°C/min. The coal sample was heated to 350°C, 400°C, 450°C and 500°C respectively, then kept constant temperature for 30 min to obtain pyrolysis residue.
In order to analyze the influence of pyrolysis temperature on ignition point and exothermic characteristics of gas coal pyrolysis residue in the reburning process. In a nitrogen environment, the pyrolysis residue was cooled to room temperature, then the reoxidation experiment of the pyrolysis residue was carried out at 30°C–900°C with an airflow rate of 100 ml/min and a temperature rise rate of 5°C/min. As a contrast, raw coal was oxidized from 30°C to 900°C under the same conditions.
In order to analyze the structural changes of gas coal at different pyrolysis temperatures and reveal the evolution mechanism of functional groups during gas coal pyrolysis, FTIR experiments were carried out. Nicolet 6700 was used to analyze pyrolysis residue microscopic characteristics. The wave number is from 650 cm−1 to 4000 cm−1, with a resolution of 4 cm−1 and scanning number of 64 scans. Pure ground KBr was used to obtain the reference spectrum before testing the coal sample. About 8 mg of the powdered coal sample, was packed into the sample holder, and then placed in the diffuse reflectance chamber.
Discussion and results
Analysis of functional groups in pyrolysis residue
Based on the experiments of coal pyrolysis, Obtain coal pyrolysis residues samples at different pyrolysis temperatures. This section carries out infrared spectrum analysis. Obtaining changes in the content of functional groups in the coal sample under different pyrolysis reaction temperatures, thus analyzed the structural characteristics of gas coal at different pyrolysis temperatures, and revealed the evolution mechanism of the functional groups in the pyrolysis stage of DLT gas coal. The infrared spectra of raw coal and pyrolysis residue are shown in Figure 2.
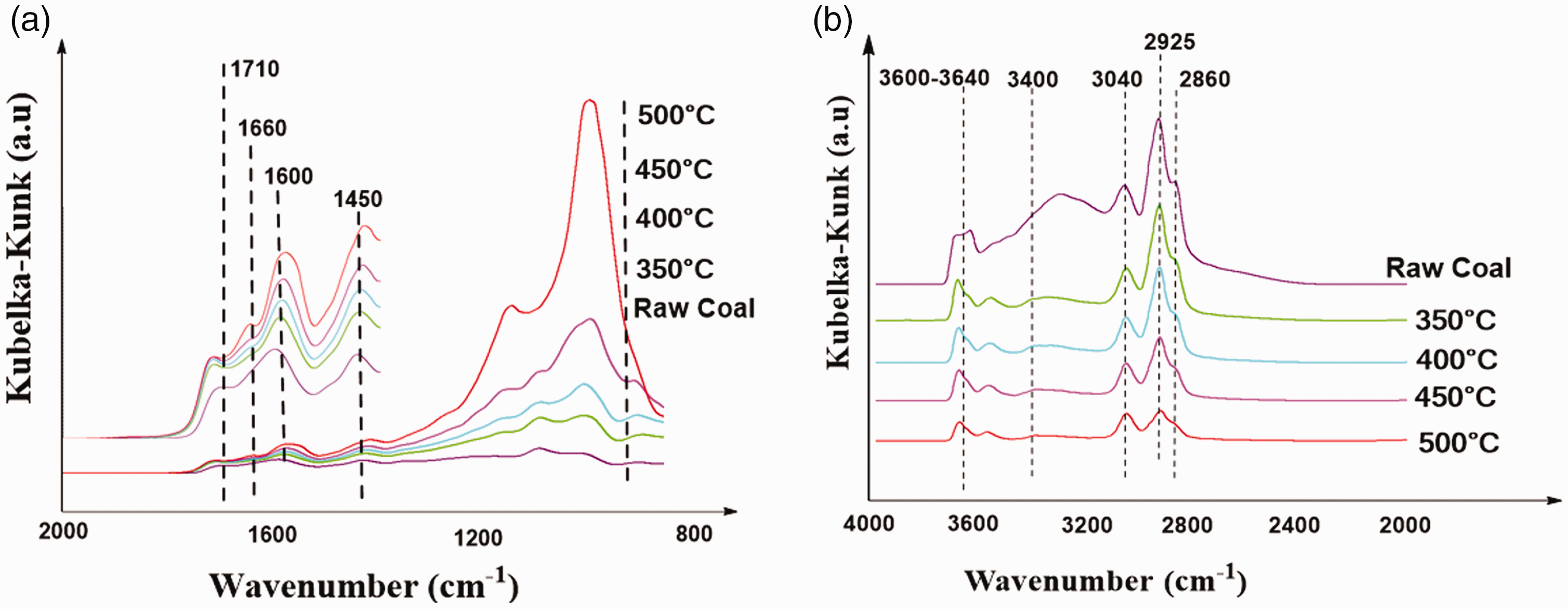
Infrared spectrogram of residual structure and raw coal, (a) the wave number ranges from 2000 to 800 cm−1. (b) the wave number ranges from 4000 to 2000 cm−1.
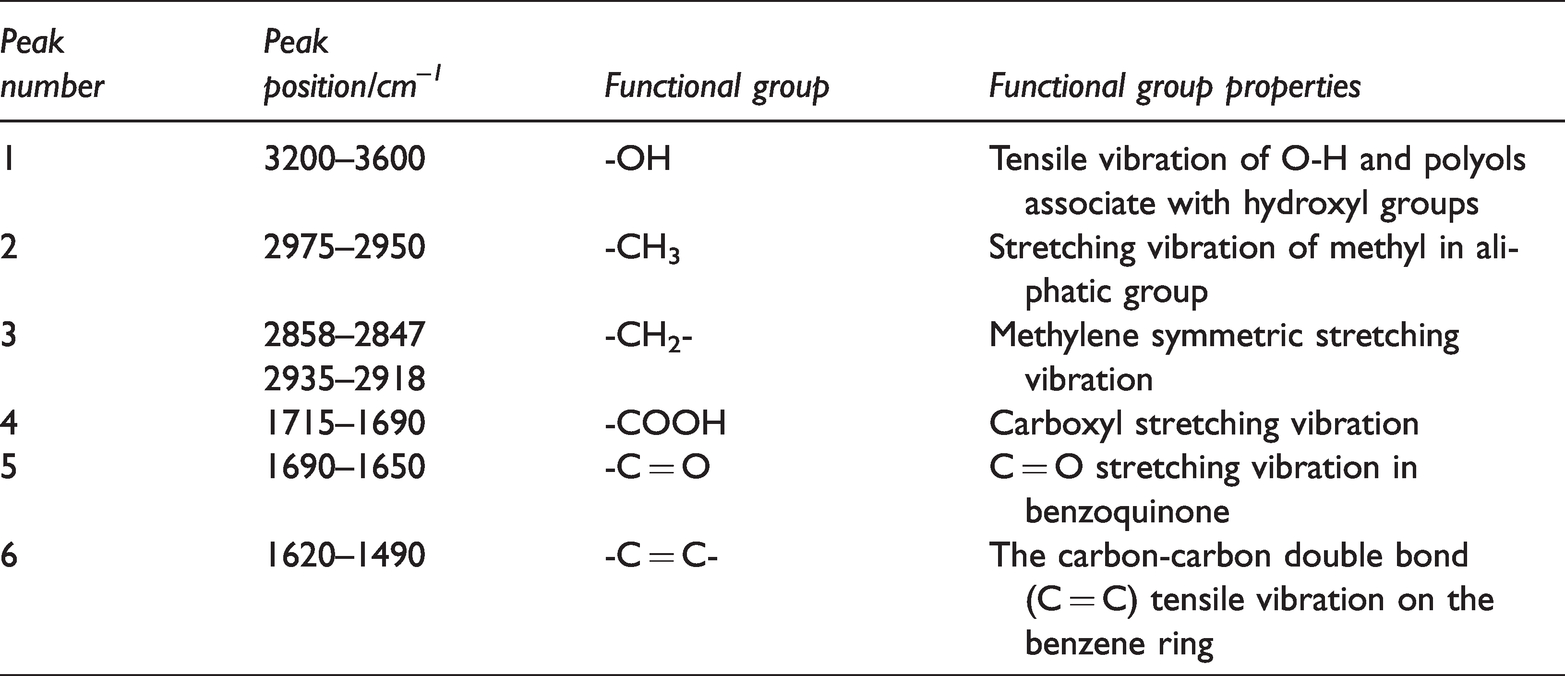
According to the position of the peak, the functional groups of the pyrolysis residue were determined. Table 2 lists the peak positions corresponding to the functional groups.
FTIR peaks of the main functional groups of coal samples.
According to the theory of reflection spectrum, the quantitative analysis result of the infrared spectrum curve of coal is shown as Kubelka-Munk peak area value. There is a quantitative relationship between Kubelka-Munk and group concentration as shown in (1), the concentration ratio of different groups is the ratio of KM peak area to extinction coefficient of each group. According to Schrodinger's tunneling effect based on quantum mechanics, the absorbance of a group can be determined by the probability flow density of the reflected wave. Therefore, the independent vibration intensity f calculated by quantum chemistry of the group is approximately equal to the extinction coefficient ε of the group, as shown in equation (2). According to the KM peak area, the accurate distribution characteristics and corresponding structural parameters of each group in coal can be obtained
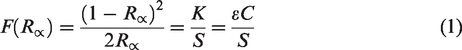
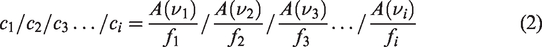
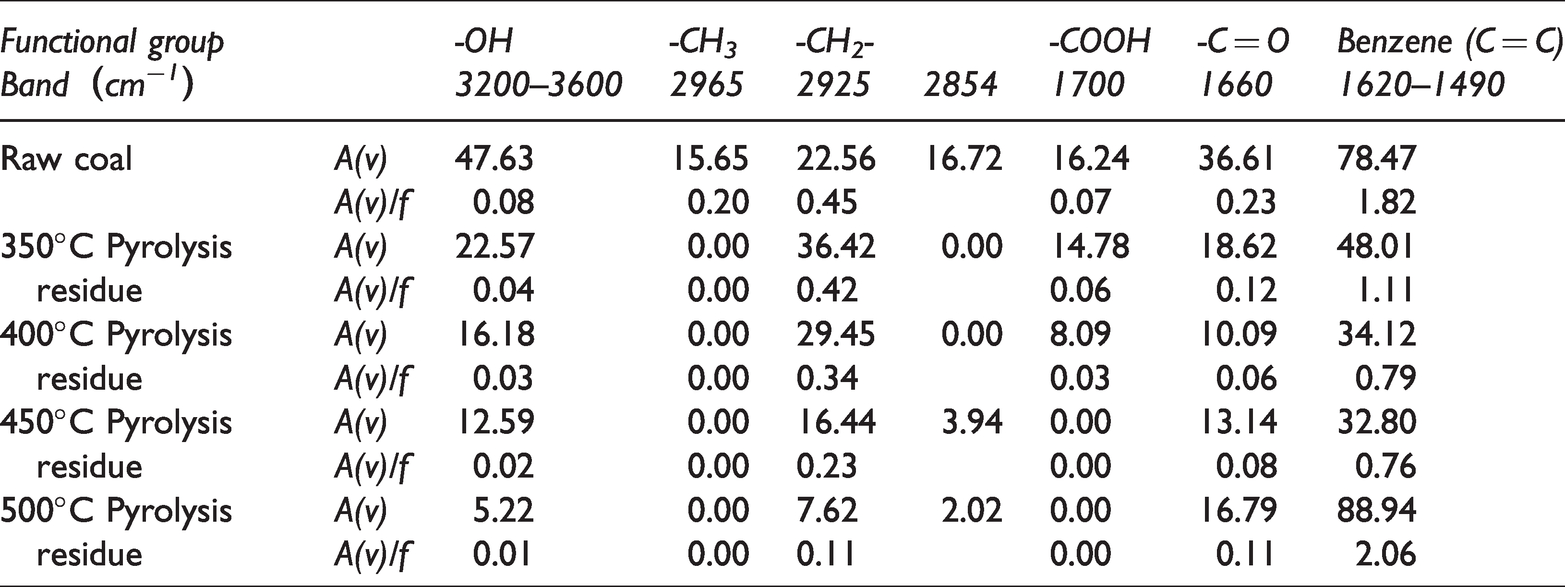
In the spectrogram, for independent absorption peaks, the value can be measured after baseline correction. For overlapping absorption peaks, the reproducibility is good after separation by curve fitting. By integrating the separated absorption peaks, the original peak area A(v) is quantitatively obtained, the vibration intensity f of different groups has been obtained by quantum chemistry calculation. Table 3 lists the peak area A(v) and A(v)/f of different functional groups.
Original peak areas and corrected values of functional groups in coal.
The vibration intensity f of the functional group is different, so the A(v) and A(v)/f of each functional group are quite different. The influence of absorption coefficient was eliminated by quantum chemistry method, and the contents of various functional groups in raw coal and pyrolysis residues were calculated accurately as shown in Table 4.
The contents of functional groups in different pyrolytes.
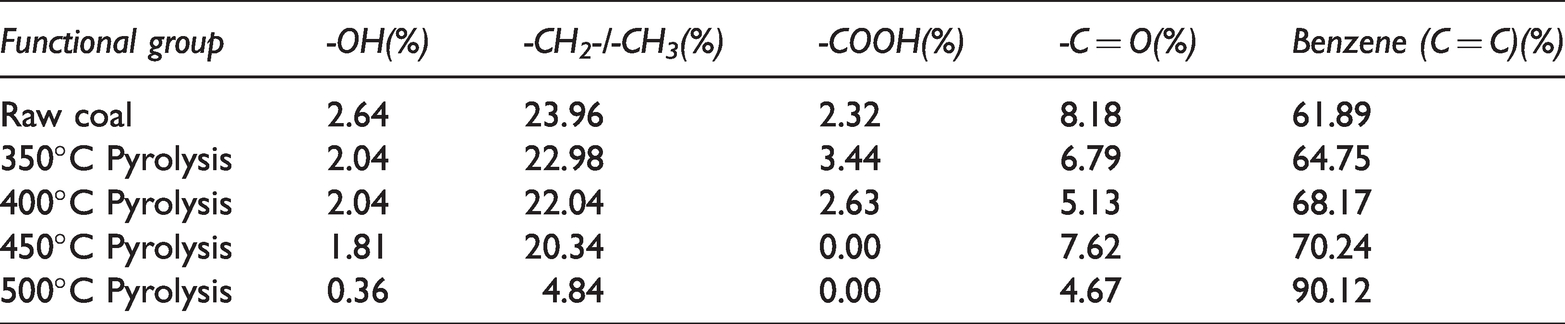
It can be seen from the table, aromatic and aliphatic hydrocarbons have the highest content in raw coal, reaching 61.89% and 23.96% respectively. The content of aromatic hydrocarbons is the highest in different pyrolysis residues, exceeding 60%.
The content of hydroxyl in raw coal is low, only 2.64%, which is related to the water content of gas coal. With the pyrolysis temperature rise, the content of hydroxyl groups generally decreases. At 350°C and 400°C, the pyrolysis residue has the same hydroxyl content, because the moisture is almost completely dried at 350°C, the content of water-associated hydroxyl groups is extremely small, the hydroxyl groups mainly exist polyols and phenols in free and associated forms. At 450°C, the part of polyphenol associating hydroxyl group began to decompose. When the pyrolysis temperature reaches 500°C, the content of hydroxyl decreases to 0.36%, it mainly exist in a free state, this is consistent with previous findings that hydroxyl groups persist until 700°C.
The aliphatic hydrocarbon content is 23.96% in raw coal. With the increase of pyrolysis temperature, aliphatic hydrocarbons show a trend of continuous decrease, In the range of 350°C to 400°C, the aliphatic hydrocarbon changes very little, which is related to the precipitation of volatile matter in the early stage of thermal decomposition. After 400°C, aliphatic hydrocarbon decreased greatly, and its content decreased to 4.84%. At 500°C, the rapid decomposition of colloids at high temperatures, and begin to condense to form semi-coke, weak chemical bonds are broken, resulting in a sharp decrease in aliphatic hydrocarbon content.
We found that aromatic hydrocarbons have different changing trends from other functional groups. With the increases of pyrolysis temperature, its content continues to increase. When the temperature rises from 400°C to 450°C, the increase of aromatics is not obvious. At 500°C, its content increases rapidly and reached 90.12%. The rapid increase of aromatics means that the pyrolysis has entered the later stage, the molecules begin to condense to form semi-coke, the degree of aromaticity of the molecular skeleton becomes larger, and the content of aromatics increases.
The contents of carboxyl and carbonyl groups are relatively small in raw coal, 2.32%, and 8.18% respectively. With the increase of pyrolysis temperature, the content of the carboxyl group decreases gradually. At 450°C, it has gone down to 0, the carbonyl group shows a fluctuating change, and the overall trend is decreasing.
Structural parameters changing in coal pyrolysis
The characteristic structural parameters of group distribution in coal can provide the structural information of coal. In this paper, the calculation of the structural parameters based on the infrared characteristics of functional groups after correction using quantum chemistry. Aromatic peak areas in the infrared fingerprint region cannot be determined accurately, because of the overlap in peak positions of several vibrations. We select C = C in the benzene ring at 1490–1620 cm−1 to represent coal aromaticity rather than the C-H flexural vibrations at 700–900 cm−1.
The benzene ring C = C was selected to represent the number of aromatic rings in coal, and the C-H content and structural characteristic parameters Aar/Aal were calculated, as shown in equation (3)
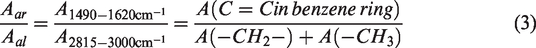
Hal/H is the fraction of total hydrogen present as aliphatic hydrogen and Hal/Cal is the hydrogen-carbon atomic ratio for aliphatic groups, which is assumed to be 1.8 for coals. The ratio Hal/H can be obtained from equation (4)
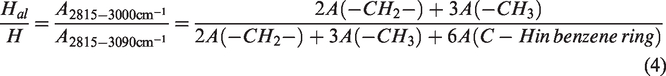
Aar/Aal and Hal/H were calculated according to the functional group content, and the line chart was drawn.
The shows the Hal/H and Aar/Aal parameters change of different temperature pyrolysis residues in Figure 3. It can be seen that Aar/Aal increases with pyrolysis temperature. It rises rapidly after 450°C, and the value of 500°C is 5.7 times that at 450°C. The value of Hal/H decreased from 0.13 to 0.02 after 400°C. It is indicated that the content of aliphatic hydrocarbons in the pyrolysis residual structure decreases with the increase of pyrolysis temperature, and the aromatic hydrocarbon content increases and the degree of change is greatest after pyrolysis at 500°C.
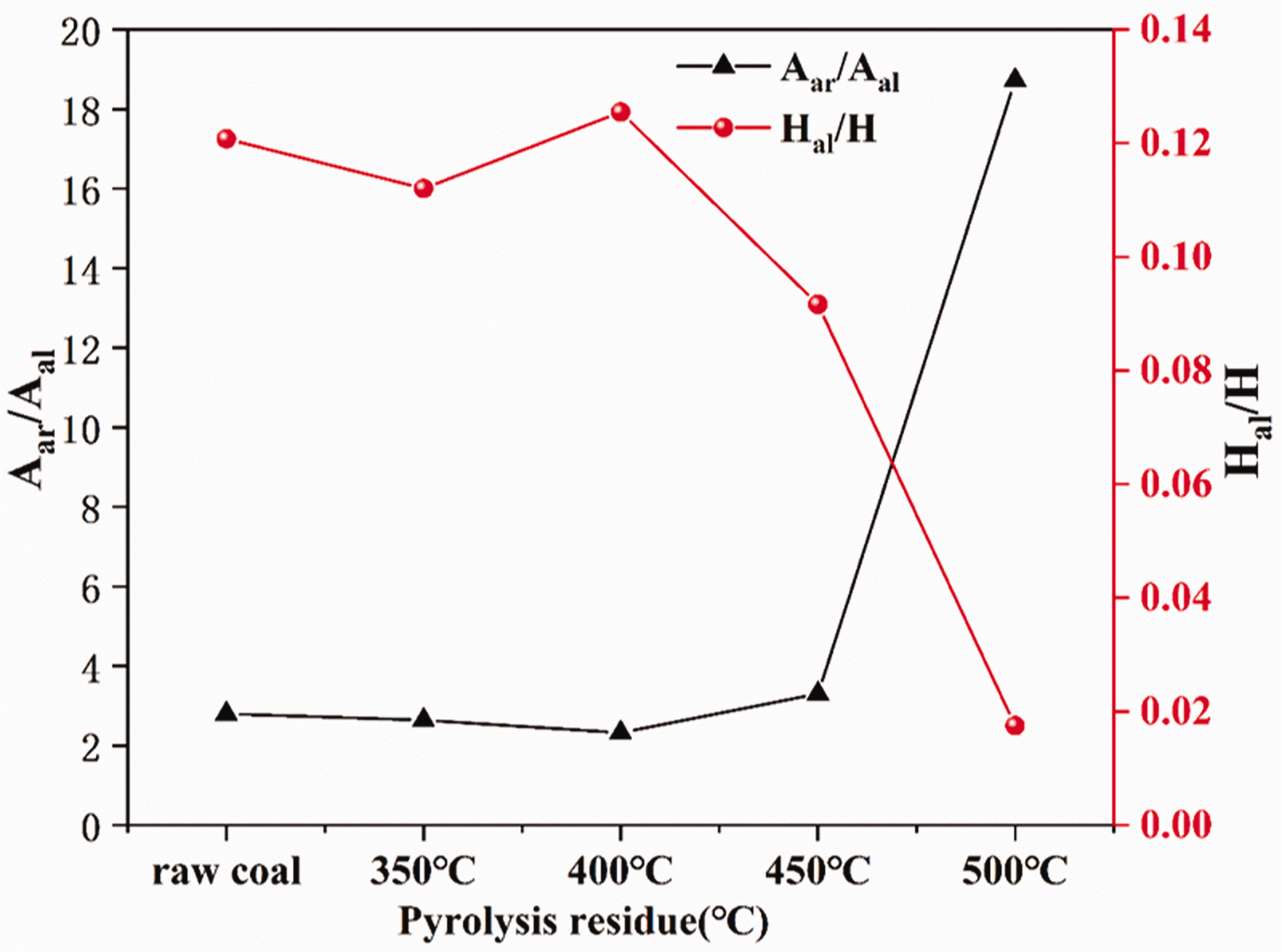
Structural parameters curve of pyrolysis residues.
Pyrolysis characteristics and residual parameters
The programmed temperature experiment of DLT gas coal was carried out. The quality characteristics of the residue were measured in real time, and the influence of pyrolysis temperature on the quality and structure characteristics of the residue was analyzed. Because the thermogravimetric curves overlap each other, it is difficult to analyze the influence of pyrolysis conditions on coal pyrolysis characteristics. The characteristic parameters of coal pyrolysis were obtained by analyzing the thermogravimetric curve of gas coal. The results are shown in Table 5.
Pyrolysis characteristics of coal at different temperatures.
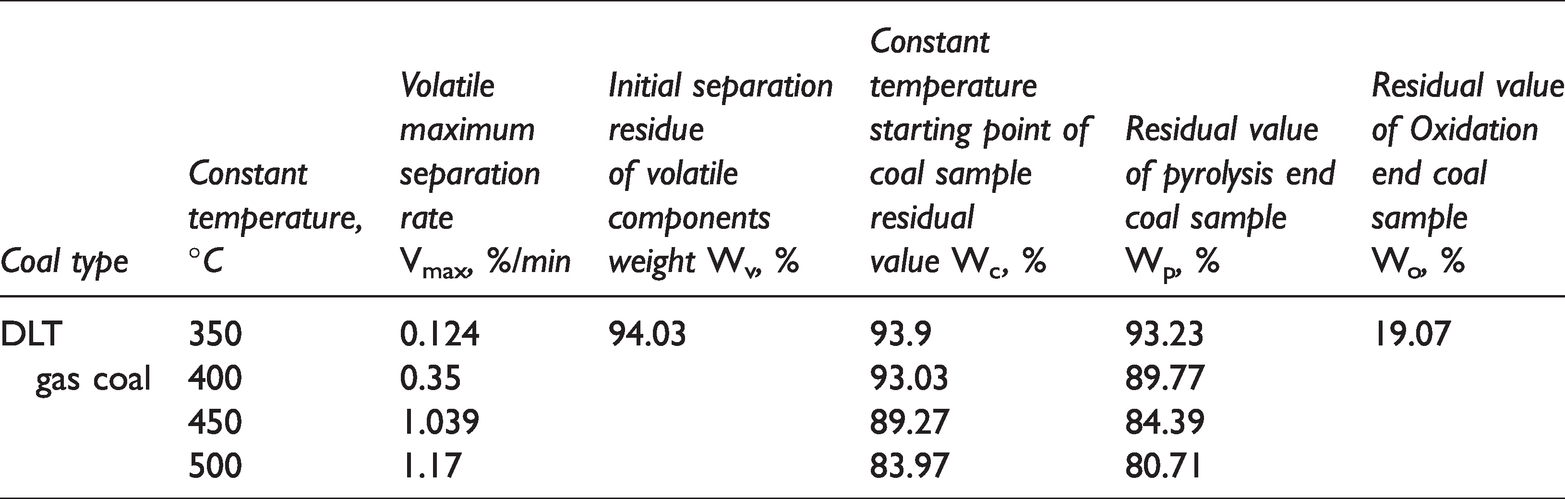
Vmax is the Volatile maximum separation rate, and its value is the peak value of the DTG curve, which represents the strength of the volatiles released. WV represents the initial separation residue weight of volatile matter, which is quality of coal sample at the beginning of weightlessness. WC represents the residual weight of coal at the beginning of pyrolysis. Wp represents the residual value of the end of pyrolysis, the residual weight of the coal after pyrolysis for 30 min at constant temperature. Wo mean residual value of oxidation end, the residual ash and other non-combustible substances after the reoxidation of coal.
From Table 5, the DLT gas coal maximum release rate of volatile Vmax is increased gradually with the increase of pyrolysis temperature. It is reached the maximum at 500°C. According to the growth trend, DLT gas coal has entered the pre-thermal decomposition stage at 350°C–400°C, and the decomposition rate is slower. The Vmax value between 400°C–450°C becomes larger, indicating that it has entered the rapid decomposition stage in the early stage of thermal decomposition. At this time, the thermal decomposition reaction proceeds rapidly, and volatiles such as tar and gas are rapidly formed. Although Vmax still does not reach the maximum at 450°C, it has a small difference from the Vmax at 500°C, indicating that it has approached the maximum Vmax, and the reaction spans thermal decomposition early stage in the range of 450°C–500°C. At 500°C, it has entered the late stage of thermal decomposition, and the colloidal body begins to solidify and becomes semi-coke. Therefore, the thermal decomposition critical temperature of DLT gas coal is 500°C. The maximum precipitation temperature of volatile matter is 479.83°C.
Three new pyrolysis characteristic parameters were defined: ΔWvp, ΔWcp, and ΔWpo. To observe the changes of the four characteristic parameters, a line graph was drawn. ΔWvp represents the change in the residual weight of the coal sample (the content of volatile matter in the coal), and the calculation formula is ΔWvp=Wv–Wp. Wcp is the change of residual weight, which is the change of coal sample under constant temperature pyrolysis, the calculation formula is ΔWcp=Wc–Wp. ΔWpo is the pyrolysis residual combustible component weight, ΔWpo=Wp–Wo, the amount of combustibles left in the residue after thermostatic pyrolysis.
The shows pyrolysis parameters change rule of the DLT gas coal in 350°C, 400°C, 450°C, 500°C in Figures 4 and 5.
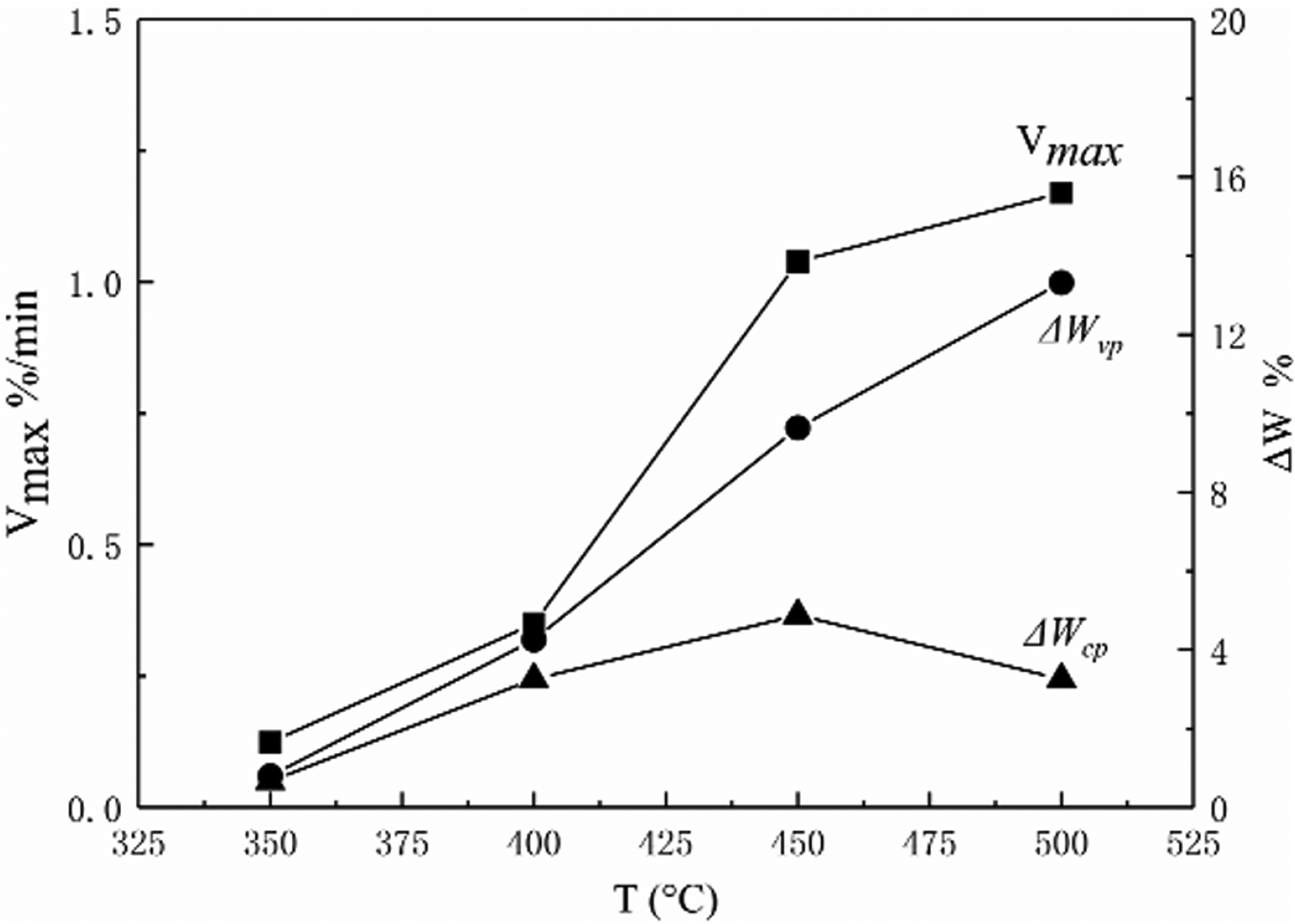
The tendency of Vmax, ΔWvp and ΔWcp to change with pyrolysis temperature.
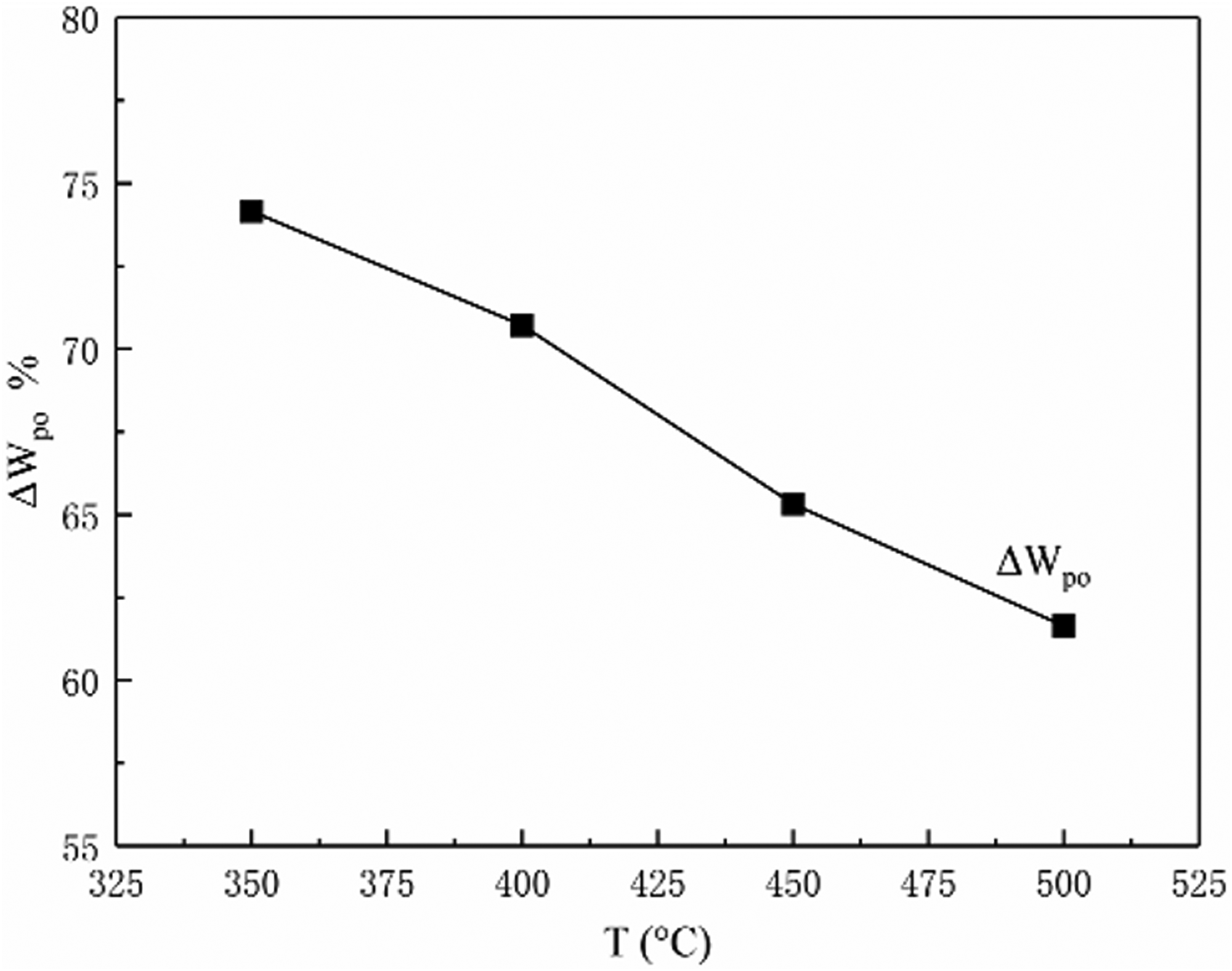
The variation trend of ΔWpo with pyrolysis temperature.
Taking the Vmax increase rate as the boundary, the pyrolysis process is divided into two stages: the early stage and the late stage. The early stage starts at the initial precipitation point of the volatiles and ends at the fastest precipitation point, and the late stage starts at the fastest precipitation point until the end of thermal decomposition. It can be seen from Figure 4 that Vmax increases with the increase of pyrolysis temperature. At 350°C–400°C, the Vmax is small and the growth rate is slow, indicating that the coal in the slow thermal decomposition stage. At 400°C–450°C, the Vmax value began to increase rapidly, indicating that the coal sample entered the rapid thermal decomposition stage. At 450°C–500°C, the Vmax increased slowly, from 1.039 to 1.17, indicating the coal sample entered the later stage of thermal decomposition. The critical temperature is the maximum precipitation of 462.8°C at 500°C pyrolysis.
Between 450°C–500°C, the growth rate of ΔWvp value is slightly lower than that of 400°C–450°C, but it still maintains a faster rate of increase overall. ΔWcp increase first and then decreased, and peaked at 450°C. However, the difference between the ΔWcp value at 450°C and the ΔWcp value at 500°C is small, from 4.88 to 3.26. This shows that although between 450°C–500°C, the coal sample has entered the late stage from the early stage of thermal decomposition, although there are many macromolecular structure involved in the reaction at 450°C, but it has not reached the maximum. Therefore, the ΔWcp value is still very high at 500°C.
It can be seen from Figure 5, at 350°C–450°C, the coal sample is in the early stage of thermal decomposition, and the ΔWpo dropped by 8.84%. The reason is that gas coal has a short branch chain, and it is easy to precipitate volatiles to form colloidal coal. At 450°C–500°C, the coal sample entered the late stage of thermal decomposition, and ΔWpo decreased by 3.68%, the colloid formed in the early stage began to decompose to form semi-coke. At 450°C–500°C, the quality of combustibles decreases more slowly than before 450°C, The combustibles content of pyrolysis residue as high as 61.64% in 500°C, which is related to the nature of gas coal. In general, the trend of the four characteristic parameters changed at 450°C.
Reoxidation characteristics of coal pyrolysis residues
The shows the results of TG-DTG of DLT raw coal and pyrolysis residual structure at different temperatures in Figure 6.
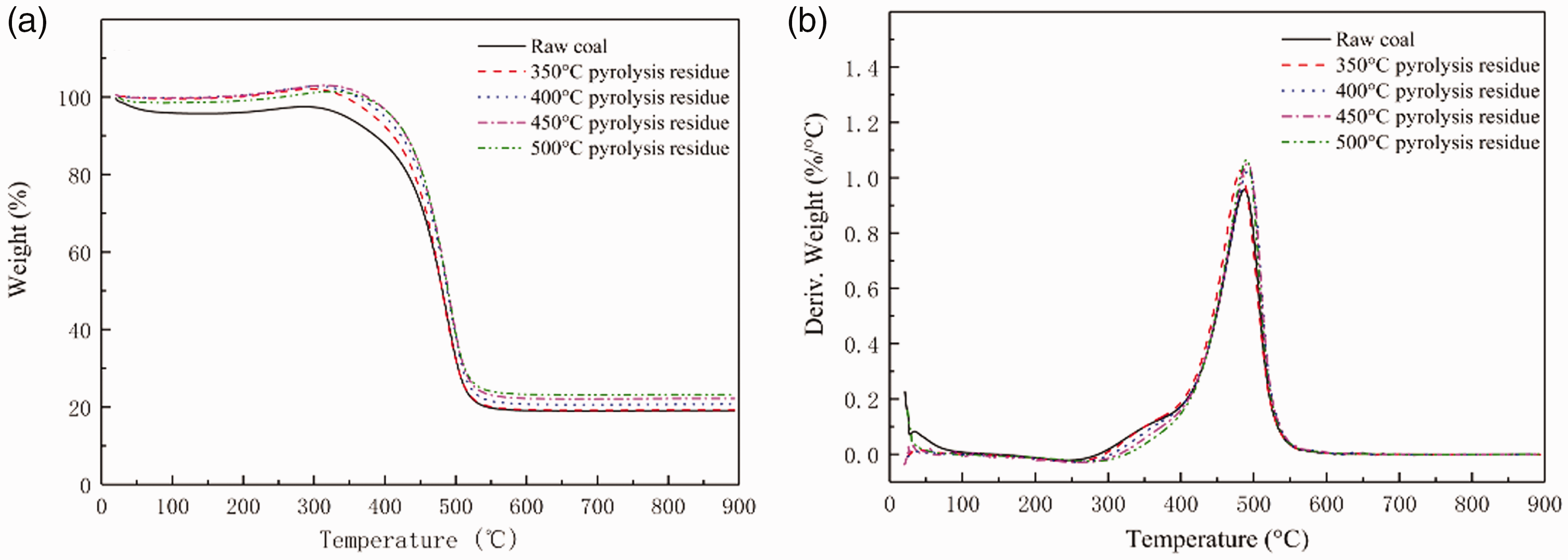
Thermogravimetric analysis (a) and Derivative thermogravimetric Analysis (b) at Different Pyrolysis Temperates.
As can be seen in Figure 6, DLT gas coal appear an oxygen absorbing process in the whole oxidation reaction, but the weight gain is small. In general, the changing trend of the reoxidation curve of the pyrolysis residue is similar. As the temperature increases, the oxygen absorption of the residue first increases and then decreases. The oxygen absorption of the residue at 450°C reaches the maximum. At 500°C, the colloid solidifies into semi-coke, and the oxygen absorption becomes smaller. Obviously, pyrolysis changes the structure of coal, the reoxidation characteristics of pyrolysis residues are stronger than raw coal. The reoxidation curves overlap each other, it is difficult to analyze the reoxidation characteristics of the pyrolysis residue. Therefore, the four characteristic points of ignition temperature Ti, burnout temperature Th, the maximum rate of weight loss dWmax, and combustion half peak width
Ignition point Ti, the lowest temperature when the coal sample burning. In the DTG curve, Ti is the temperature when the ordinate changes from 0 to positive. Burnout temperature Th
,
the temperature when combustible components are burned out, remaining incombustible components such as ash, meaning the end of the entire combustion process. The maximum rate of weight loss dWmax, the most violent moment in the combustion process, it is the peak of the DTG curve. At this moment, the residual weight loss rate is the largest and the combustion intensity is the strongest. Combustion half peak width
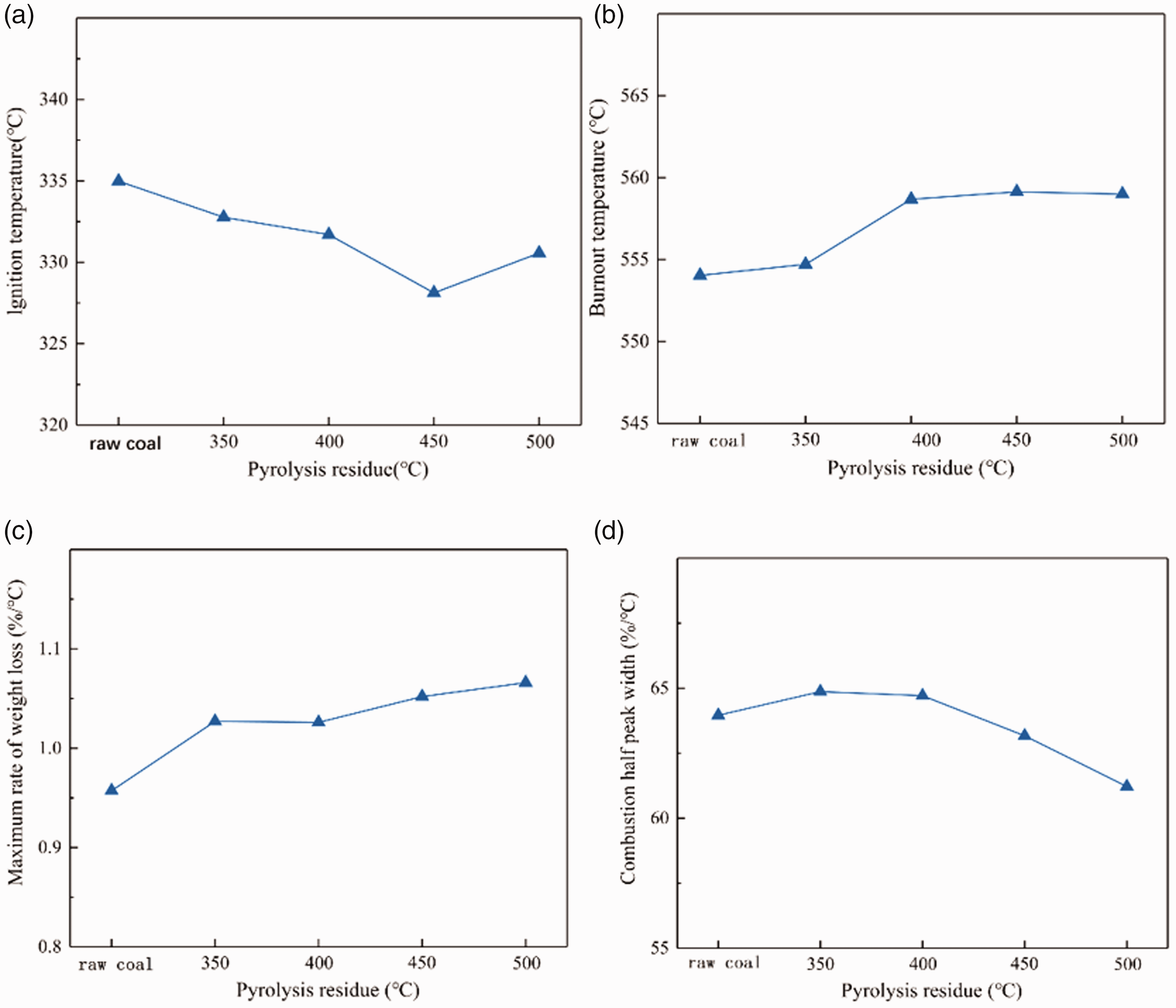
The trend of Ti (a), Th (b), dWmax (c) and
The ignition point has decreasing trend before 450°C, and it drops to minimum at 450°C. The ignition temperature of the pyrolysis residue at 400°C and 500°C is similar. The ignition point of the residue is less affected by the pyrolysis conditions, and the ignition temperature of the raw coal and the pyrolysis residue varies within 330.57°C–334.98°C. Due to the short side chain of gas-coal, thermal decomposition can produce small molecular substances, which are easy to volatilization from coal. Therefore, the ignition point is less affected by pyrolysis.
Before 400°C, the burnout temperature of the residue slowly increases with pyrolysis temperature. After 450°C, the volatile matter has been separated, and incombustible components in the residue is small.
The maximum weight loss rate of pyrolysis residues shows stepwise upward trend with the increase of pyrolysis temperature. In the range of 350°C–400°C, the formation of small molecules is relatively slow, and the maximum mass loss rate remains unchanged. After 400°C, with the temperature rises, the formation of small molecules such as tar accelerates, and the combustion intensity increases, the maximum weight loss rate increases.
The combustion half peak width of the pyrolysis residue is almost unchanged before 400°C. Gas coal has higher aromaticity and less affected by pyrolysis, small molecules are produced less. So it is little difference in the combustion concentration of pyrolysis residues and raw coal. After 400°C, the thermal decomposition reaction speed increases, and the burning of the pyrolysis residue becomes vigorously.
The exothermic characteristics of pyrolysis residual structure reoxidation
The influence of pyrolysis reaction on the oxidation characteristics of coal samples includes not only the change of the quality characteristics of the combustion process but also the heat release. The change between the raw coal and the pyrolysis residual structure cannot be clearly seen on the TG-DTG curve. Therefore, the DSC curve is used to investigate the law of coal oxidation and heat release. Due to different structural characteristics and small molecular structures of residuals, heat release not the same. As shown in Figure 8, the DSC curve of the raw coal of DLT gas coal and the different pyrolysis residual structures in the oxidative combustion process are compared and analyzed to obtain the influence of pyrolysis reaction on the heat release of coal.
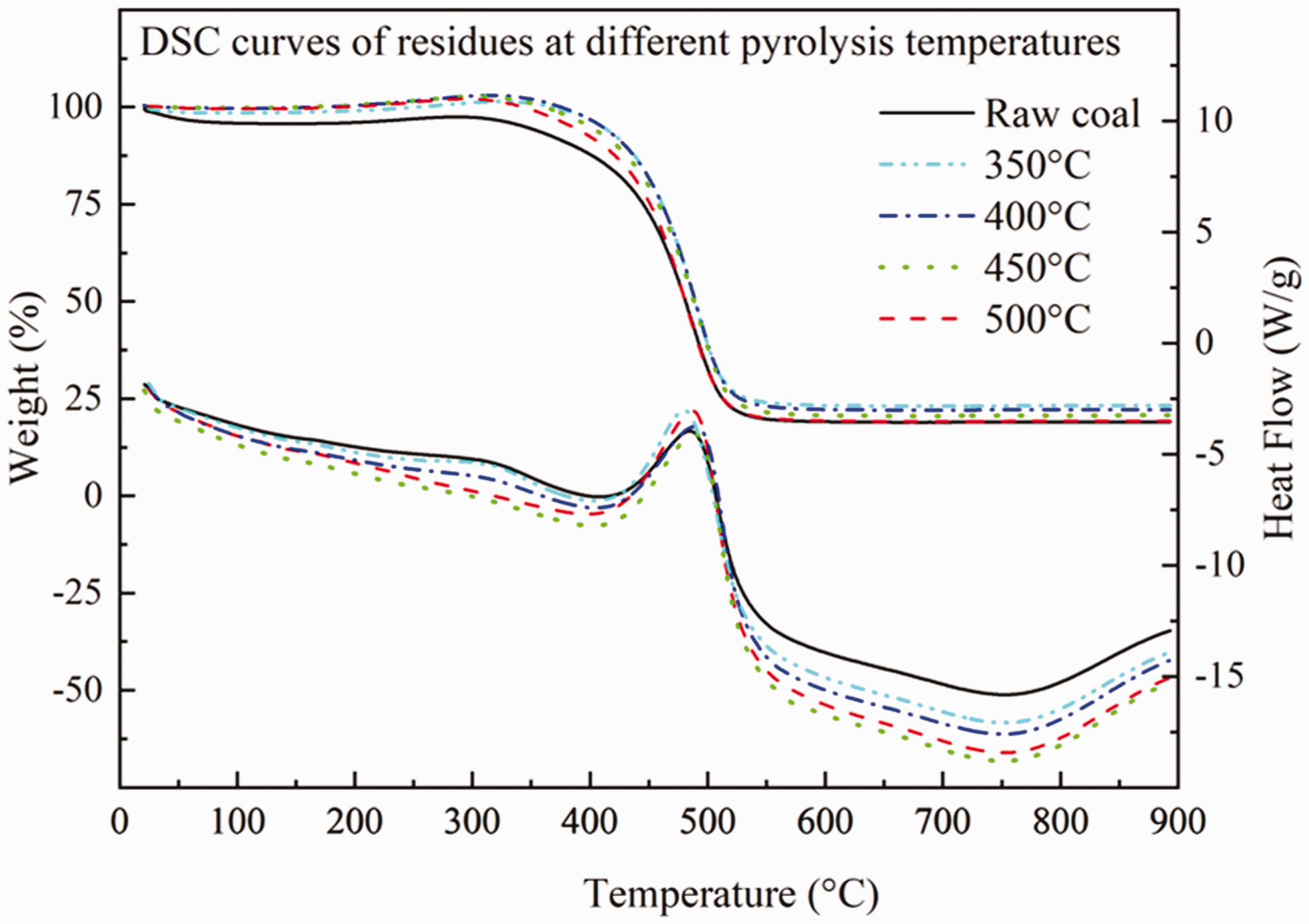
The DSC curve of coal oxidation.
As can be seen from Figure 9, there is no significant difference in the initial heat release temperature point between raw coal and pyrolysis residual structure by different temperatures, which indicates that pyrolysis reaction has no obvious influence on the initial heat release temperature of gas coal. The initial heat release temperature of the DLT gas coal is around 450°C. The trend of heat release cannot be directly seen in the DSC curve. In Figure 9 the heat release peaks of different coal samples of DLT gas coal are integrated to obtain the trend of heat release. It can be seen that the heat release of gas coal increases with the increase of pyrolysis temperature. From raw coal to pyrolysis residues of 500°C, the heat release of gas coal increased by 1.45 times that of raw coal.
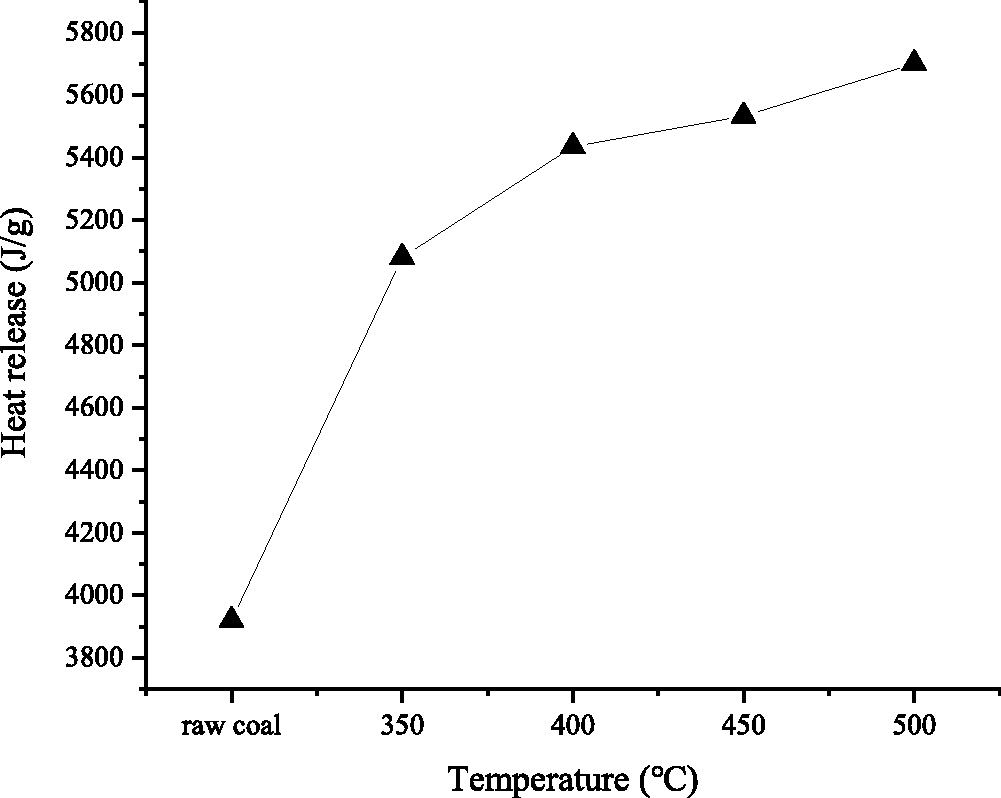
The variation tendency of the heat release.
The gas coal heat release has an inflection point at 350°C, and the slow increase of heat release after 350°C is mainly related to its initial exothermic temperature point. The initial exothermic temperature of gas-fired oxidation is around 450°C, which causes the coal pyrolysis residue at 350°C to undergo an oxidation warming effect between 350°C and 400°C before reaching the initial exothermic temperature point. The high temperature effect leads to the further polymerization of the 350°C coal pyrolysis residual structure, which increases the degree of coal aromatization. Therefore, when it reaches the initial exothermic temperature point of 400°C, the aromaticity of the skeleton increases, and the amount of heat release increases. The residual structure of coal pyrolysis at 400°C is not affected by the temperature rise of the initial exothermic temperature, so the degree of aromatization is unchanged at the beginning of the oxidation exotherm, This results in a decrease in the difference between the aromaticity of the 350°C and the 400°C pyrolysis residue at the beginning of the oxidative exotherm, which directly leads to a decrease in the increase in the amount of heat release.
In summary, the oxidative heat release of gas coal increases with the increase of pyrolysis temperature, which is mainly due to the change of the aromatic structure of skeleton. The content of aromatic hydrocarbons in gas coal increases with the degree of pyrolysis. Therefore, although the overall combustible composition becomes smaller and smaller as the degree of pyrolysis becomes deeper, the amount of heat released is increasing.
Conclusions
This paper studied the pyrolysis characteristics of DLT gas coal at 350°C, 400°C, 450°C and 500°C, and analyzed the heat release rule in the reoxidation process of the pyrolysis residue. The effect of extinction coefficient on quantitative analysis of infrared spectrum is eliminated by quantum chemistry method. The influence of pyrolysis temperature on functional groups of gas coal was analyzed. It provides theoretical support for the prevention and control of coal field fire, It is great significance for the safety of strip mining and control of the Pingshuo coalfield fire.
Raw coal and its residues at different pyrolysis temperatures were analyzed by infrared spectroscopy. The distribution characteristics of functional groups in coal were obtained. The main components of gas coal are aromatic hydrocarbon and aliphatic hydrocarbon, which are 61.89% and 23.96%, respectively. With the increase of pyrolysis temperature, Aar/Aal increased and Hal/H decreased from 0.13 to 002 after 400°C. This indicates that pyrolysis will reduce the content of aliphatic hydrocarbons and increase the degree of aromatization of coal. Taking Vmax increase rate as the boundary, pyrolysis can be divided into two periods. With the increase of pyrolysis temperature, Vmax and ΔWvp, gradually increased, while ΔWcp first increased and then decreased. Different characteristic parameters showed a turning point at 462.83°C. The oxidation results of coal pyrolysis residue show that the Ignition point Ti decreases with the increase of pyrolysis temperature, the content of combustible components decreases, the Burnout temperature Th increases, and the combustion intensity increases. The heat released by oxidation gradually increases, which is related to the increase of aromatic hydrocarbon content in coal. In other words, coal loses a large number of combustible components in the pyrolysis process, but its oxidation performance is improved, and coal samples are more prone to spontaneous combustion.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the following financial support for the research, authorship, and publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities (2017QNA01).
