Abstract
Coal spontaneous combustion is one of the main potential hazards in the process of mining. To study the spatial and temporal variations of higher-temperature area and indicator gases, an adiabatic oxidation testing system was developed to simulate the whole process of coal spontaneous combustion. The experimental results show that the entire process of coal spontaneous combustion could be divided into three stages: slow-oxidation, accelerated-oxidation and combustion stages. In the slow-oxidation stage, the higher-temperature area shifted slowly from the bottom to the top and then stayed at the top until accelerated-oxidation stage was reached;
Introduction
Coal is one of the main energy sources in the world (Ren et al., 2019; Zhang et al., 2018a). In China, coal fire is one of the main hazards in coal mines, most of which were caused by coal spontaneous combustion. This indicates that on one hand, a large amount of coal resources are wasted annually by coal fire; on the other hand, coal spontaneous combustion also influences the coal production greatly. So far, it is well-known that coal spontaneous combustion contains not only physical reactions but also chemical (coal-oxygen) reactions (Cheng et al., 2017; Fan et al., 2020a; Ge et al., 2019; Ren et al., 1999; Zhang et al., 2018b, 2020).
In the past decades, international and domestic academics have conducted extensive researches on coal spontaneous combustion aiming at exploring the mechanism of this phenomenon through experimental tests (Beamish et al., 2003; Benfell et al., 1997; Peng et al., 2020; Yang et al., 2018) and numerical simulations (Fan et al., 2020b; Jiang et al., 2008; Song et al., 2015). Obviously, in comparison with numerical simulations, the experimental tests are more realistic and suitable for in situ cases, which is primarily because many assumptions (contradictory with the fact) are directly simplified in numerical simulations. Therefore, more attention has been paid to the physical modeling of coal spontaneous combustion. At present, better understanding of this topic has been successfully achieved. These results show that coal spontaneous combustion is impacted by a couple of factors, primarily consisting of particle size, oxygen concentration, air supply rate and temperature. Temperature is the most significant influence factor on coal spontaneous combustion. The increase in coal temperature could result in the formation of indicator gases (such as CO, C2H4, CO2). Thus, to predict coal spontaneous combustion, it is necessary to analyze the precursors (e.g., indicator gases, temperature) which would occur during the combustion process. Zhou et al. (2010) analyzed the gas (CO, C2H4, CO2) generation laws at different oxygen concentration. Liu et al. (2013) put forward the energy migration theory of spontaneous combustion and obtained the empirical equation of the higher temperature area in the gob and of the minimum safety mining velocity. Wang et al. (2018) proved that the liberations of CO, H2, CO2 are positively related to the oxygen consumption. Luo et al. (2019) pointed out that the liberations of hydrocarbons (CO) first rise and then descend with the decrease in particle size. However, the above-mentioned experiments were on relatively small scales. Although these results could provide convincing explanations in some aspects, it is not completely suitable for the real coal mines on account that some parameters can not be directly detected (Yang et al., 2018).
Therefore, large-scale tests seem to be a better stepping-stone for exploring the true physics of coal spontaneous combustion. Zhou et al. (2009) employed a large-scale device to investigate the liberation of indicator gases. Deng et al. (2015) studied the variations of temperature and indicator gases with a 15 tons testing system. However, it is found that due to the limitation of the experimental device, the spatial and temporal variation of the higher-temperature area and indicator gases have not been fully understood. In fact, dynamic variation of high-temperature area reflects the development of the interaction of heat accumulation and migration, which is the basis of mechanism and significant for the prediction of coal spontaneous combustion. In this work, the aim is to simulate the whole process of spontaneous combustion using an adiabatic oxidation testing system, and to study the variations of higher-temperature area and indicator gases. The results from this work can provide an improved understanding of precursors which appears in the combustion process.
Testing sample and scheme
The coal sample was collected from Tashan mine, which is located in the city of Datong, Shanxi province, China. The coal was first crushed and then sieved out for the prescribed particle size. To avoid oxidation reaction and keep the coal sample as fresh as possible, it was wrapped with aluminium foil and then was put into the fridge. The corresponding parameters for the coal are listed in Table 1.
Coal parameters used in the experiment.

The 2 m adiabatic self-heating device was adopted for this issue as shown in Figure 1. Testing system mainly consists of the experimental furnace, temperature switcher, signal controller, computer system, gas compressor, etc. First the coal sample is placed in the furnace (the top thermocouple must be covered). Subsequently, the thermocouples are inserted into the coal through the holes on the furnace wall as shown in Figure 1. Gastightness is examined before testing. Finally, the test starts with the initial temperature and preset air supply rate. During the test, the temperatures for two positions are monitored: one is at the center and the other is on the furnace wall at the corresponding height. To reduce the loss of the heat generated by oxidation reaction, both temperatures are always kept close to each other using a heater. The temperature was automatically recorded every 10 minutes and the maximum value in one day was chosen for analysis. The gas sample at different heights was collected through the holes on the furnace wall to analyze the gas composition and concentration with Odalog 6000 (Figure 2). 2 hours (fixed time) on each day were spent on monitoring the concentration of each indicator gas and the average was determined for analysis.
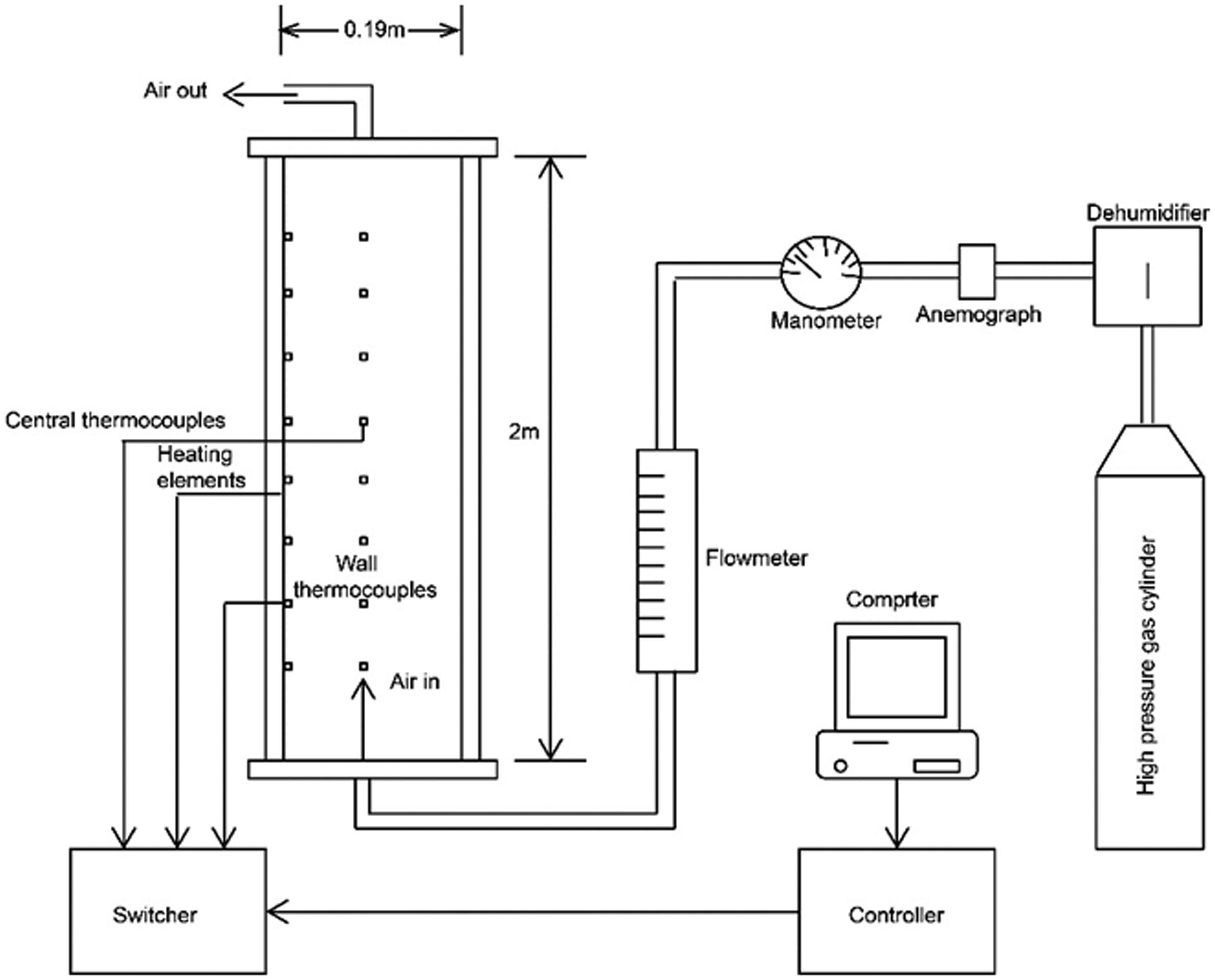
The testing system.

The gas detector.
Testing results
Spatial and temporal variation of higher-temperature area
The important indicator that directly reflects the degree of coal spontaneous combustion is the temperature of the coal. As time went on, the temperatures of the coal at different heights continuously varied during coal spontaneous combustion. The variations of each monitored point in temperature are shown in Figure 3. The
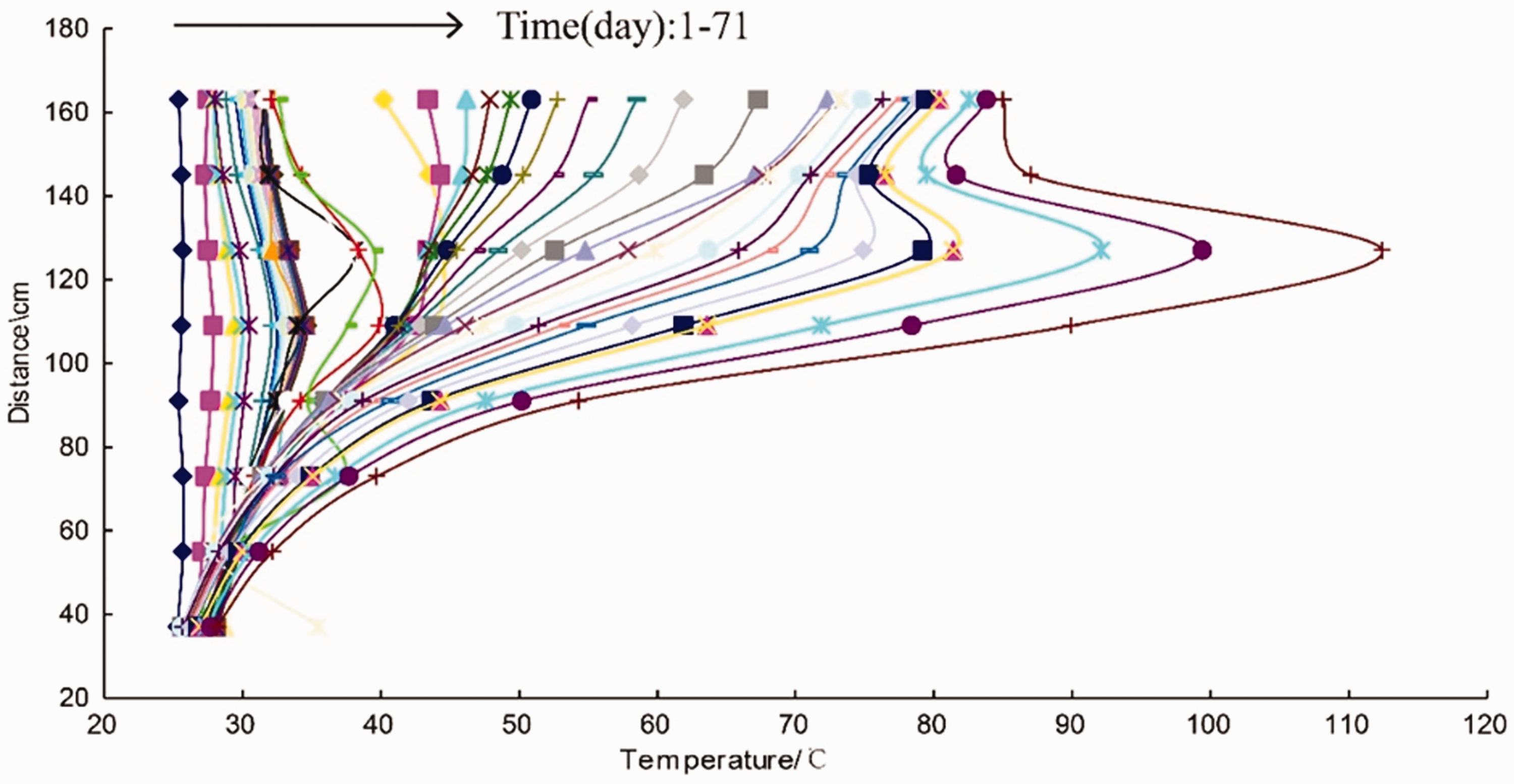
The dynamic and spatial variation of the temperature along the height of the furnace.
Figure 3 shows that the temperature in different positions and the higher-temperature area were in the dynamic change. As can be seen from Figure 3, there was no significant difference in the temperature in the first day. However, from the second day, the temperature of each point increased at different heating rates and a relative higher-temperature area gradually appears. First the higher-temperature area came out at 109
In general, the oxygen consumption during coal spontaneous combustion are mainly attributed to the physical adsorption, chemical adsorption and chemical reaction. In the initial stage, the oxygen consumption of physical adsorption accounted for the largest proportion (Ma et al., 2006). However, the amount of released heat from physical adsorption was small. Therefore, the heat accumulated very slowly and there was no significant difference in the temperature of coal body. But as time went on, the higher-temperature area was firstly generated in the central area (approximately 109
On another aspect, it is apparent that the heating rate of the upper coal is higher than the lower. As time passed,
Indicator gas
In general, the main features of indicator gases are high sensitivity, stability and detectability (Shao and Dai, 1996). Among these various gases,
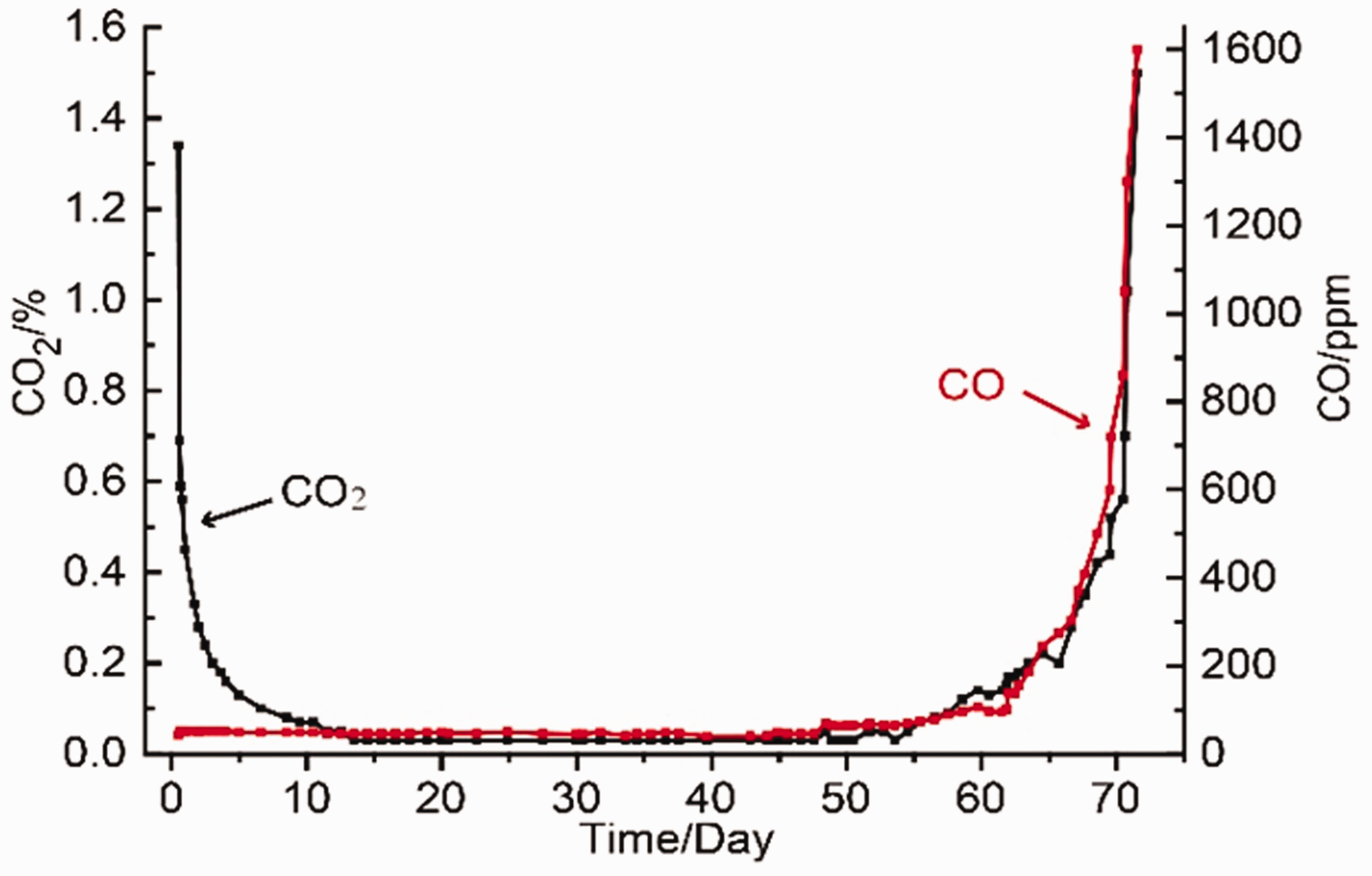
The progressive variation of gas (
As seen in Figure 4, both
The obtained results indicate that during the process of coal spontaneous combustion, production rate of indicator gas differs as the temperature rises. The higher the temperature, the larger the production rate. Therefore, the production rates for different areas varies due to the difference in temperature. For instance, as shown in Figure 3, in 65th day the higher-temperature area was located at 127
Spatial and temporal variation of oxygen concentration
Oxygen consumption is generally proportional to the production of indicator gases (
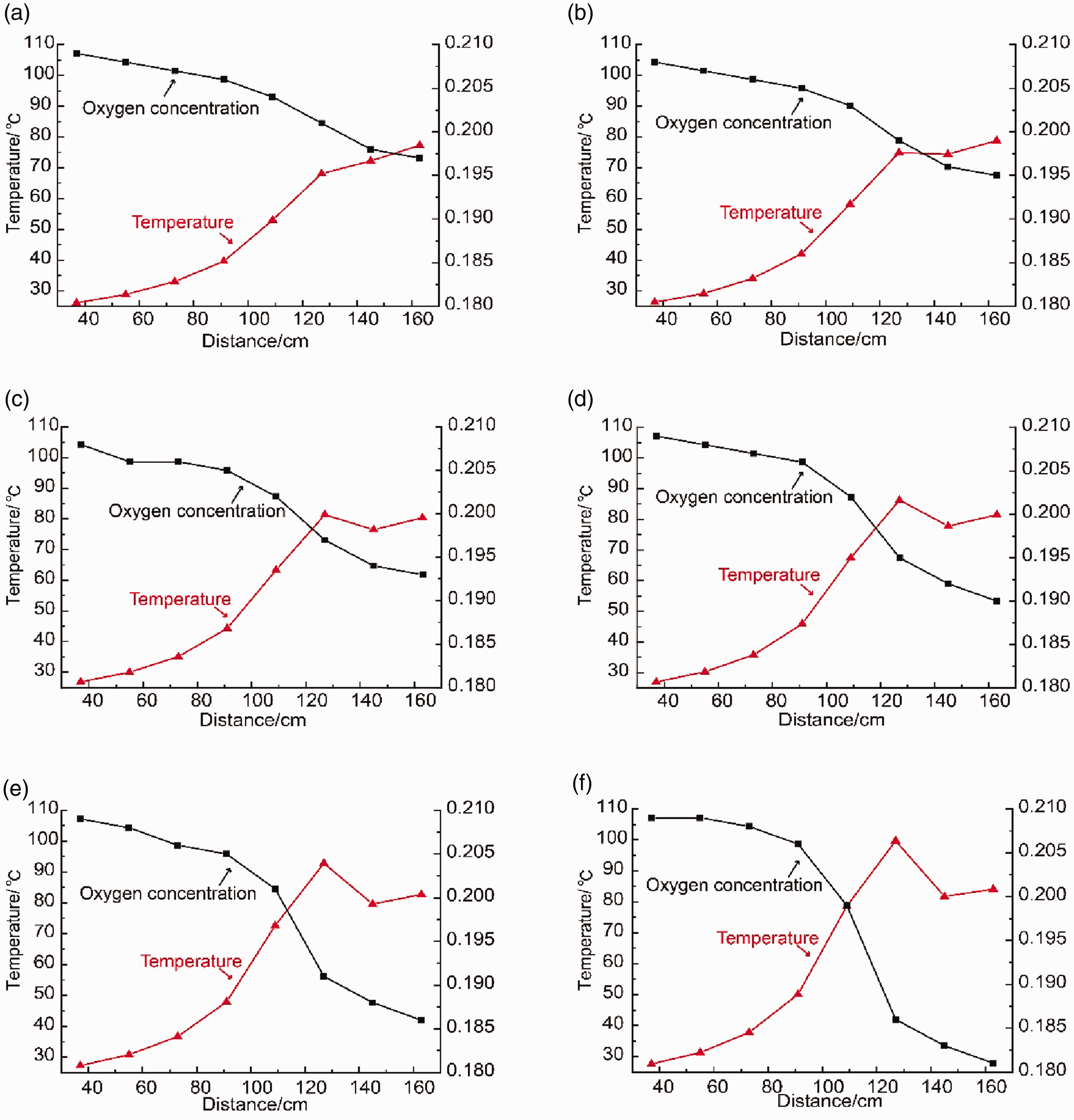
The dynamic variations in oxygen concentration and temperature along the height of the furnace in the last stage. (a) The 64th day (b) The 66th day (c) The 68th day (d) The 69th day (e) The 70th day (f) The 71th day.
Spatial and temporal variation of oxygen consumption rate
The oxygen consumption rate is the crucial characteristic parameter in coal spontaneous combustion as well (Kuenzer et al., 2012). Based on the experimental conditions (Nordon et al., 1979), some assumptions are made for quantitatively calculating the oxygen consumption rate: (1) the air flows uniformly along the vertical direction (
Generally, oxygen consumption rate is proportional to the oxygen concentration. Therefore, it read:

Substituting equation (1) into equation (2), equation (3) is obtained:

Integrating both the sides of equation (3), equation (4) can be rearranged as:
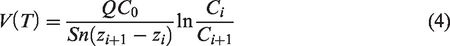
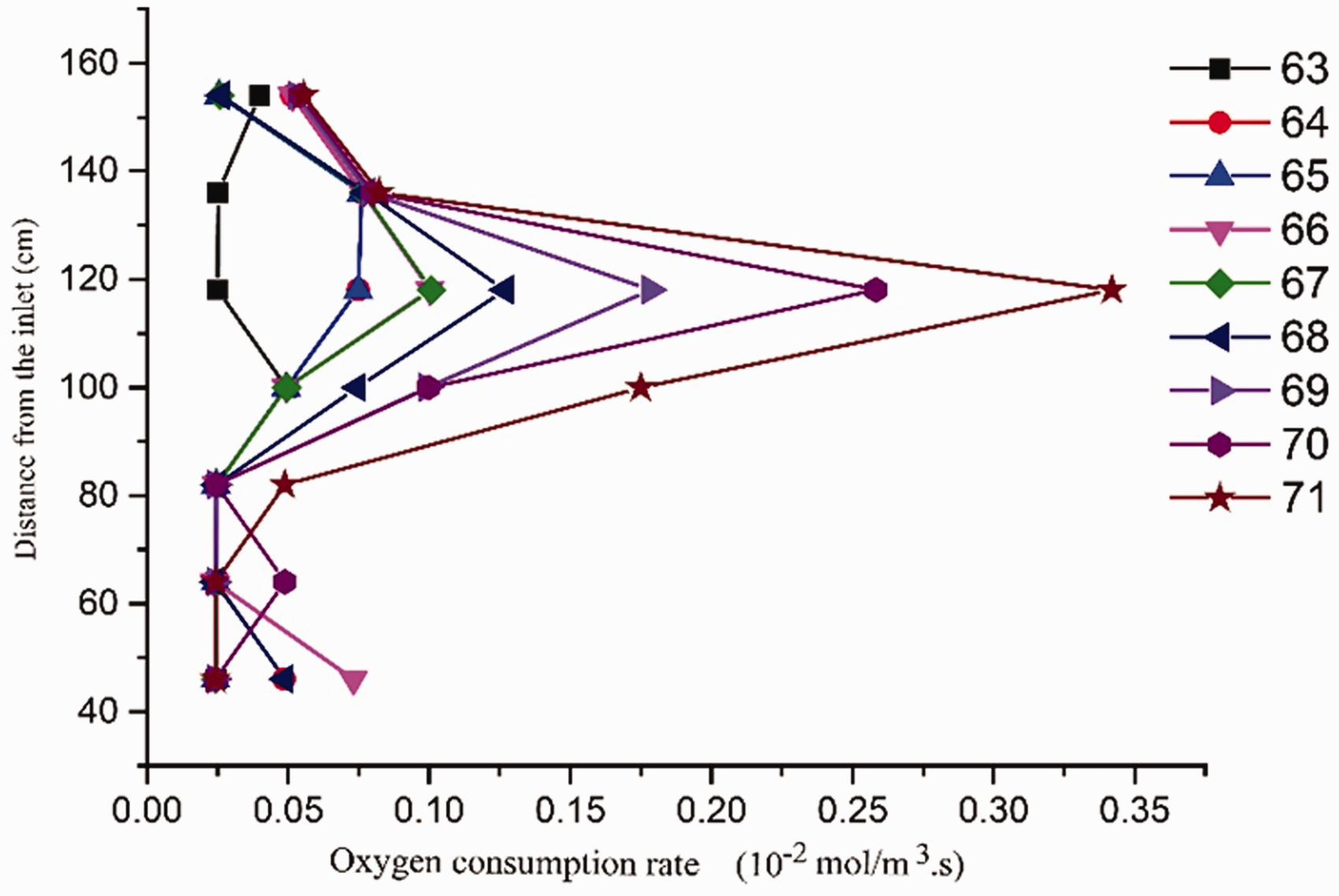
The oxygen consumption rates at different monitoring points in the last stage.
The effect of air supply rate
Generally, increasing the air supply rate could provide a higher level of oxygen concentration, but at the same time more heat generated are also taken away. Therefore, it is necessary to investigate the effect of air supply rate and then determine the appropriate level that meets the requirement of providing insufficient oxygen and meanwhile of taking heat generated away as more as possible. As a result, two additional experiments were also conducted to investigate how the air supply rate affect coal spontaneous combustion. Two different air supply rates are imposed: one was 0.8
The properties of coal sample.

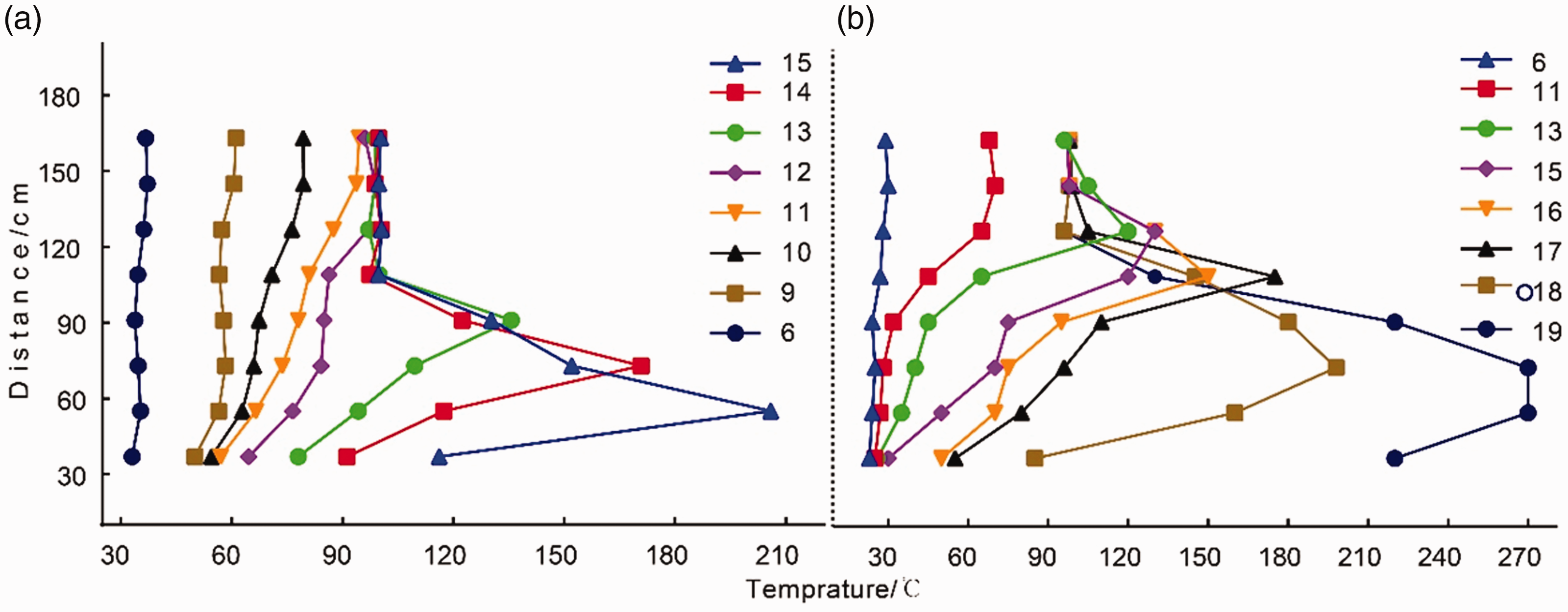
The temperature variations for different air supply rate: (a) 0.8
It depicts that on the whole, the higher-temperature area first appeared to be at the top of the coal, then moved towards the bottom and eventually burnt at approximately 73
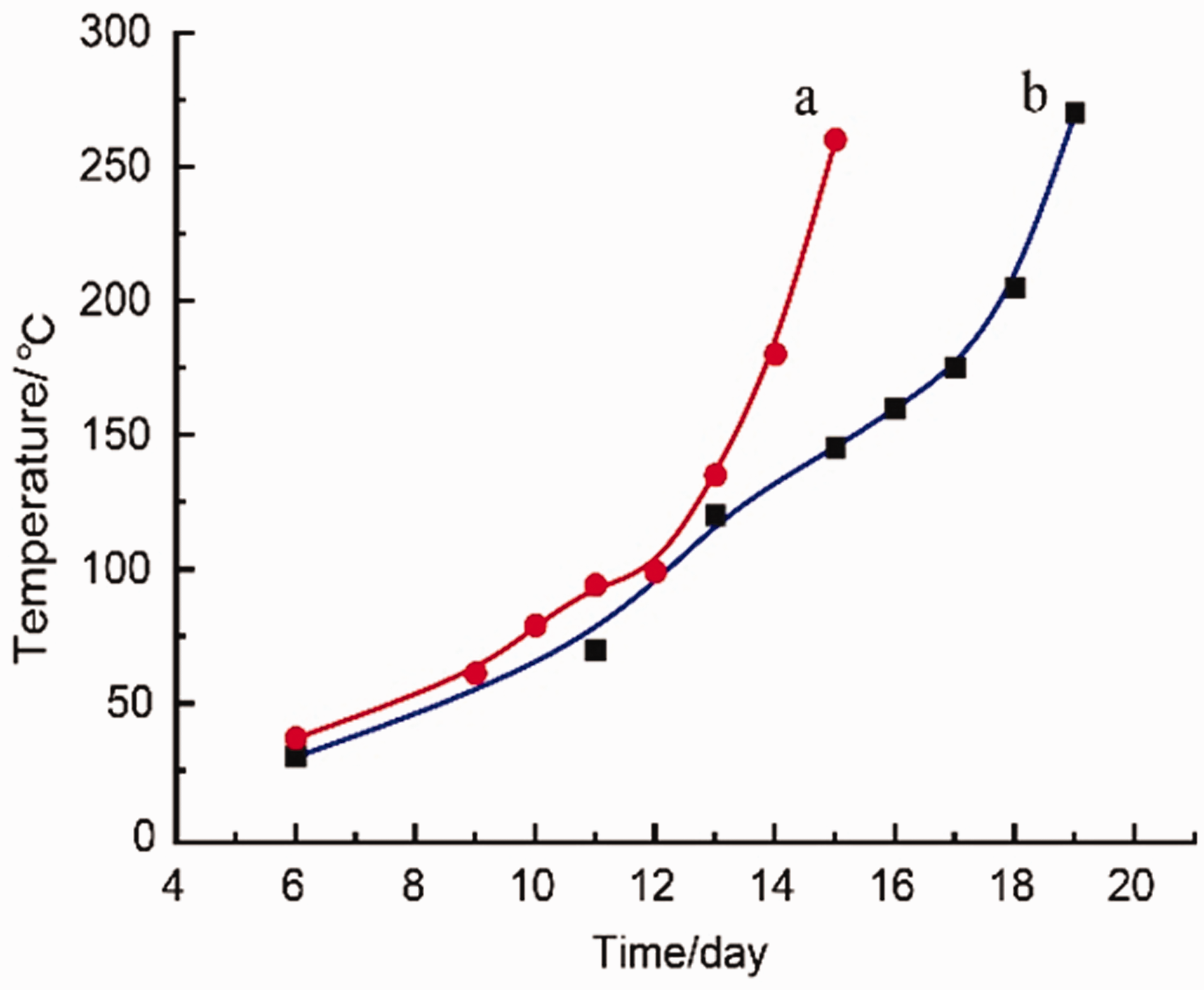
The temperature comparison for different air supply rate: (a) 0.8
To quantitatively identify the relationship between the temperature and time (Figure 8), the numerous trials have led to the following simple functions:


Conclusion
The aim of this work is to provide a simple estimation of spatial and temporal variation of higher-temperature area during the coal spontaneous combustion. For this purpose, an adiabatic oxidation testing system was established to simulate the natural temperature rise. The obtained results show a strong dependence of the coal temperature on time. The coal mainly experienced three stages during coal spontaneous combustion: (i) slow-oxidation stage; (ii) accelerated-oxidation stage; (iii) combustion stage. The higher-temperature area first moved from the bottom to the top slowly, then stayed at the top until accelerated-oxidation stage is reached and eventually moved towards to the bottom rapidly. Higher air supply rate could shorten spontaneous combustion period and there exists a hyperbolic relationship between the temperature and time.
With regard to indicator gases, plenty of
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China (No.2017YFC0804202), National Science and Technology Major Project (2016ZX05045001-005).
