Abstract
The pyrolysis kinetics of oil-palm solid waste was investigated by performing experiments on its individual components, including empty fruit bunch, fibre, shell, as well as the blends by using a simultaneous thermogravimetric analyser at a heating rate of 10°C/min under nitrogen atmosphere and setting up from initial temperature of 30°C to a final temperature of 550°C. The results revealed that the activation energy and frequency factor values of empty fruit bunch, fibre, and shell are 7.58–63.25 kJ/mol and 8.045E-02–4.054E + 04 s−1, 10.45–50.76 kJ/mol and 3.639E-01–5.129E + 03 s−1, 9.46–55.64 kJ/mol and 2.753E-01–9.268E + 03, respectively. Whereas, the corresponding values for empty fruit bunch–fibre, empty fruit bunch–shell, fibre–shell, empty fruit bunch–fibre–shell are 2.97–38.35 kJ/mol and 1.123E-02–1.326E + 02 s−1, 7.95–40.12 kJ/mol and 9.26E-02–2.101E + 02 s−1, 9.14–50.17 kJ/mol and 1.249E-01–2.25E + 03 s−1, 8.35–45.69 kJ/mol and 1.344E + 01–4.23E + 05 s−1, respectively. It was found that the activation energy and frequency factor values of the blends were dominantly due to the role of the components with a synergistic effect occurred during pyrolysis.
Introduction
Nowadays, renewable energy sources utilisation has been increasingly demanding due to the pressures on the global environment. Biomass is one of the most common forms that is widely used due to its abundant availability in agricultural and plantation residues. Currently, large scales of oil-palm (Elaeis guineensis Jacg.) plantations have been intentionally expanded for palm oil production in order to fulfil world’s increasing palm oil demand. Consequently, the quantity of oil-palm solid waste, including empty fruit bunch (EFB), fibre, shell, and kernel will certainly be abundant. It can be quantitatively explained that for every ton of palm oil produced from fresh fruit bunches, approximately 1 t of EFB, 0.7 t of palm fibres, 0.3 t of palm kernels, and 0.3 t of palm shells remained (Chang, 2014). By this approximation, it can be estimated that for world’s oil-palm production in 2017 as 66.86 million metric tons (USDA, 2017), the total biomass generated from the palm oil industry would be around 153.778 million metric tons. Hence, the oil-palm solid waste deserves appropriate concerns and handling for gaining great worth and minimising environmental impacts.
Due to the volume and environmental constraints, pyrolysis techniques have received much attention in recent years because they provide an attractive way to utilise waste. Recently, a number of investigations have been devoted on the pyrolysis characteristics and kinetic analysis of different rank coals blend with a variety of lignocellulosic biomass, including hazelnut shell (Haykiri-Acma and Yaman, 2010), giant reedgrass (Guan et al., 2015), plastic (Cai et al., 2008), printed circuit board scraps (Hao et al., 2014), corn stover and switchgrass (Bhagavatula et al., 2016), lignocellulosic components (Wu et al., 2014), and raw/torrefied wood (Lu et al., 2013). The findings of the above investigations revealed that pyrolysis is a promising, economic, and environmental-friendly technology for both energy production and waste handling. However, there is no report on the pyrolysis behaviours of both individual components and blends of oil-palm solid waste. Therefore, it is challenging to investigate the pyrolysis characteristics and kinetics of oil-palm solid waste, not only for their individual components but also their blends. The knowledge and understanding on kinetics of thermal decomposition of fuels or fuels blends is crucial to properly design and establish efficient conversion systems.
In this research, pyrolysis behaviours of both individual components and blends of oil-palm solid waste were investigated under inert atmosphere using a thermogravimetric (TG) analyser to obtain an overall understanding of their pyrolysis process. The Coats–Redfern method was applied to gain their kinetic parameters, for it had been successfully applied in the study of lignocellulosic waste (Fernandez et al., 2016).
Material and method
Material
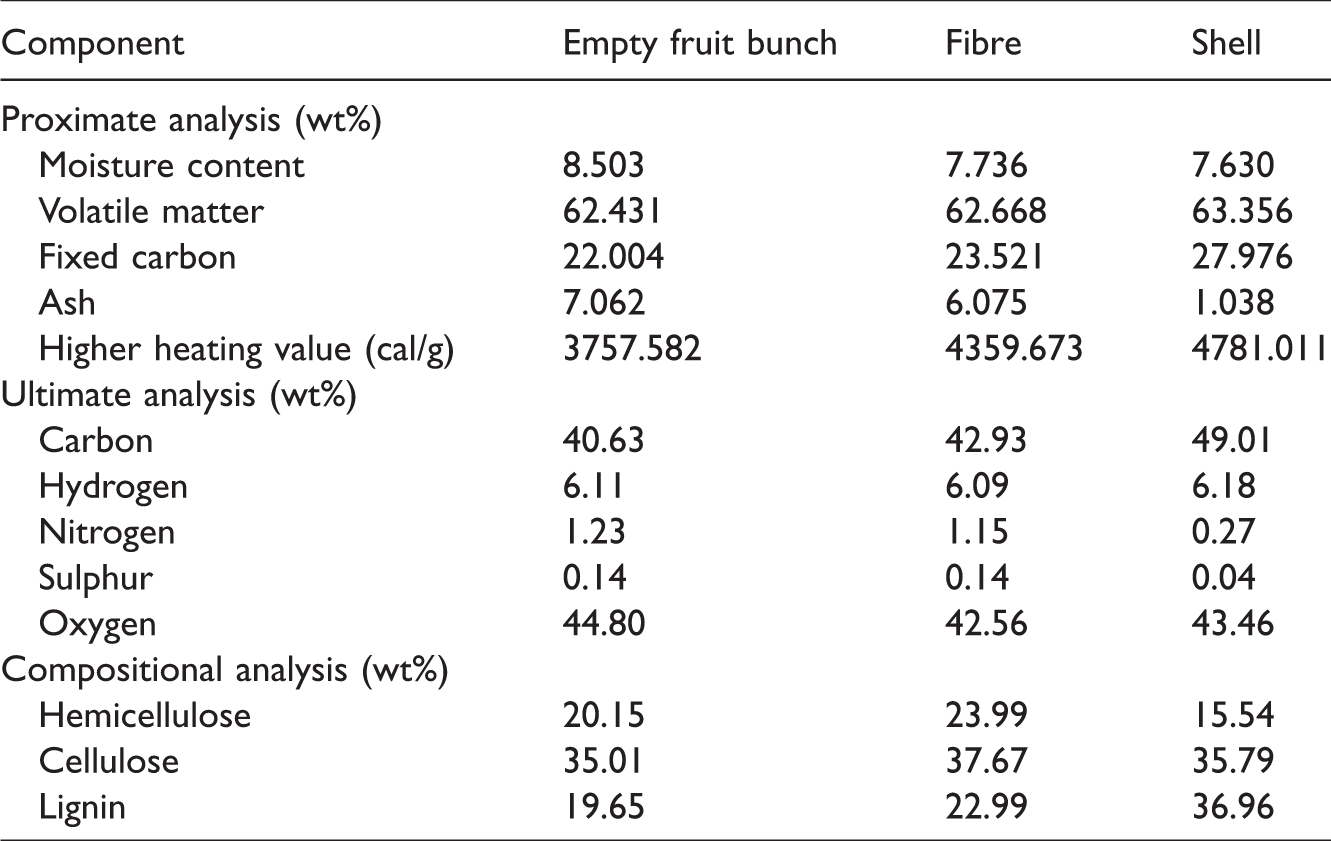
Oil-palm solid waste, including EFB, fibre, and shell, used in this study were obtained from a palm oil mill in West Sumatra Province, Indonesia. Prior to experiments conducted, these materials were crushed into small particles and sieved into particles sized of 0.21 mm. Proximate, ultimate, and compositional analysis were done to these samples presented in Table 1.
Proximate, ultimate, and compositional analysis of oil-palm solid waste.
TG experiment
An automatic simultaneous thermal analyser, which provides simultaneous TG and differential thermal analysis (DTA) on a single sample (Shimadzu, DTG-60, Japan, with temperature accuracy of 0.1 K, DTA sensitivity of 0.1 µV and TG sensitivity of 1 µg) was used to conduct pyrolysis tests. For every run, about 8 mg sample with particles size of 0.21 mm was used and 15 ml/min nitrogen was flowed to provide an inert atmosphere (Abu-Bakar and Moinuddin, 2012). Heating rate of 10°C/min, initial temperature of 30°C, and final temperature of 550°C were applied. The instrument provided continuous recording of TG and DTA curves and data that were used to calculate the kinetic parameters. Each experiment was repeated at least twice to ensure its reproducibility.
Kinetics theory
To perform the kinetic analysis of the reaction conversion curve, it is necessary to employ an integral analysis method. In the literature, different integral methods have been proposed for the determination of activation energy. The integral methods have been obtained from the corresponding approximations for the temperature integral, which does not have an exact analytical solution. Among these integral methods, the most commonly used in the kinetic analysis of thermal decomposition of coal and biomass is the integral method proposed by Coats and Redfern. The Coats and Redfern method for the determination of the activation energy has been extensively used in the kinetic analysis of thermal decomposition of coal, plastic, and biomass (Cai et al., 2008; Lu et al., 2013; Oyedun et al., 2014). Non-isothermal solid decomposition rate is mathematically expressed as (Ebrahimi-Kahrizsangi and Abbasi, 2008)



Function f (α) can be written as

Substitution of equations (3) and (4) into equation (1) results in

By using constant heating rate

Equation (5) can be arranged as

Solution of equation (7) results in
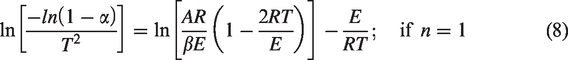
or
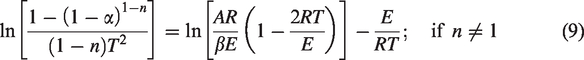
By using asymptotic approximation, where 2RT/E ≪ 1, so
Results and discussion
Pyrolysis characteristics and kinetics of the components
Figure 1 shows the thermogravimetric (TG) and derivative thermogravimetric (DTG) curves of EFB, fibre, and shell, at a heating rate of 10°C/min. Pyrolysis process commonly comprises moisture evaporation, active pyrolysis (main devolatilisation), and passive pyrolysis (Fernandez et al., 2016). These stages also happened at EFB, fibre, and shell. As shown in Figure 1, EFB underwent moisture evaporation from initial temperature to 199°C with maximum mass loss rate of 2.45%/min at 137°C. This stage was also experienced by fibre and shell from initial temperature to 167 and 200°C with maximum loss rate of 1.6%/min at 116°C and 1.94%/min at 119°C, respectively.
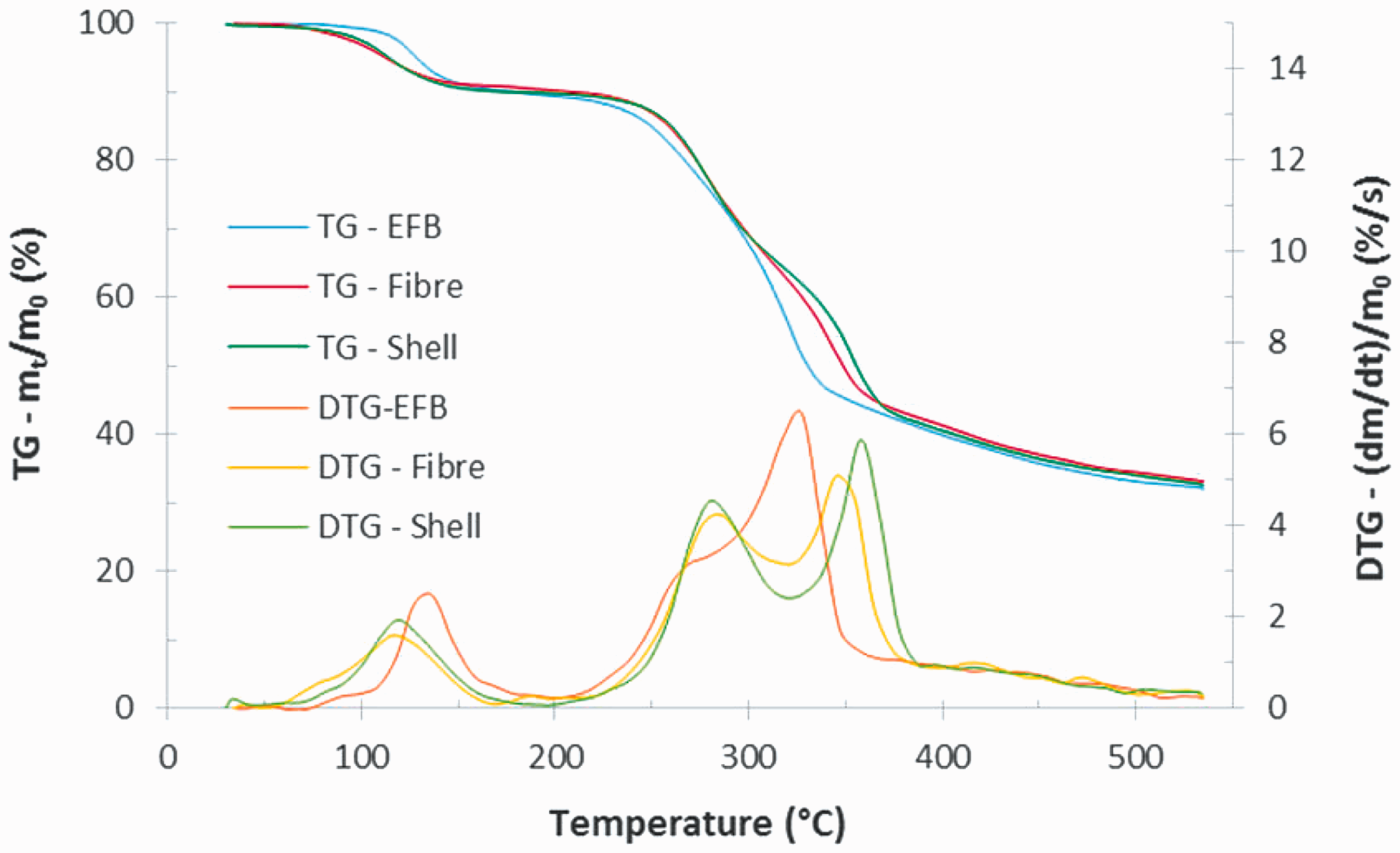
TG/DTG curves of EFB, fibre, and shell at a heating rate of 10°C/min. DTG: derivative thermogravimetric; EFB: empty fruit bunch; TG: thermogravimetric.
Within the active pyrolysis stage in the DTG curves, two peaks of mass loss rate appear for fibre and shell. The first ones located on 286°C (4.24%/min) and 280°C (4.52%/min) while the later ones located on 345°C (5.09%/min) and 358°C (5.88%/min), for fibre and shell, respectively. It indicates that that hemicellulose and cellulose greatly took a rule in this stage. Meanwhile, only one mass loss peak appears in the devolatilisation of EFB, at about 328°C (6.44%/min) with a small shoulder located at the lower temperature side. This indicates that at least there was one major reaction scheme taking place during the pyrolysis process. The maximum peak of the DTG curve is probably contributed by the decomposition of the heavier fraction (cellulose) and the small shoulder corresponds to the lighter fraction (hemicellulose). The one peak of DTG data with small shoulder was also found for the pyrolysis of poplar wood as reported by Slopiecka et al. (2012).
As can be seen in Table 2, the values of activation energy and frequency factor for EFB vary from 7.58 to 63.25 kJ/mol and from 8.045E-02 to 4.054E + 04 s−1, respectively. The lowest value of activation energy took place in the temperature range between 347 and 405°C, whereas the highest one took place in the temperature range between 308 and 327°C. Different from EFB, the values of activation energy and frequency factor for fibre, as can be seen in Table 2, vary from 10.45 to 50.76 kJ/mol and from 3.639E-01 to 5.129E + 03 s−1, respectively. The lowest value of activation energy took place in the temperature range between 364 and 520°C, whereas the highest one took place in the temperature range between 266 and 285°C. Being compared to fibre, shell has similar trend with fibre, where the activation energies and frequency factors for shell, as can be seen in Table 2, vary from 9.46 to 55.64 kJ/mol and from 2.753E-01 to 9.268E + 03 s−1, respectively. The lowest value of activation energy took place in the temperature range between 377 and 533°C, whereas the highest one took place in the temperature range between 338 and 377°C.
The activation energy and frequency factor values of oil-palm solid waste components at different temperature ranges.
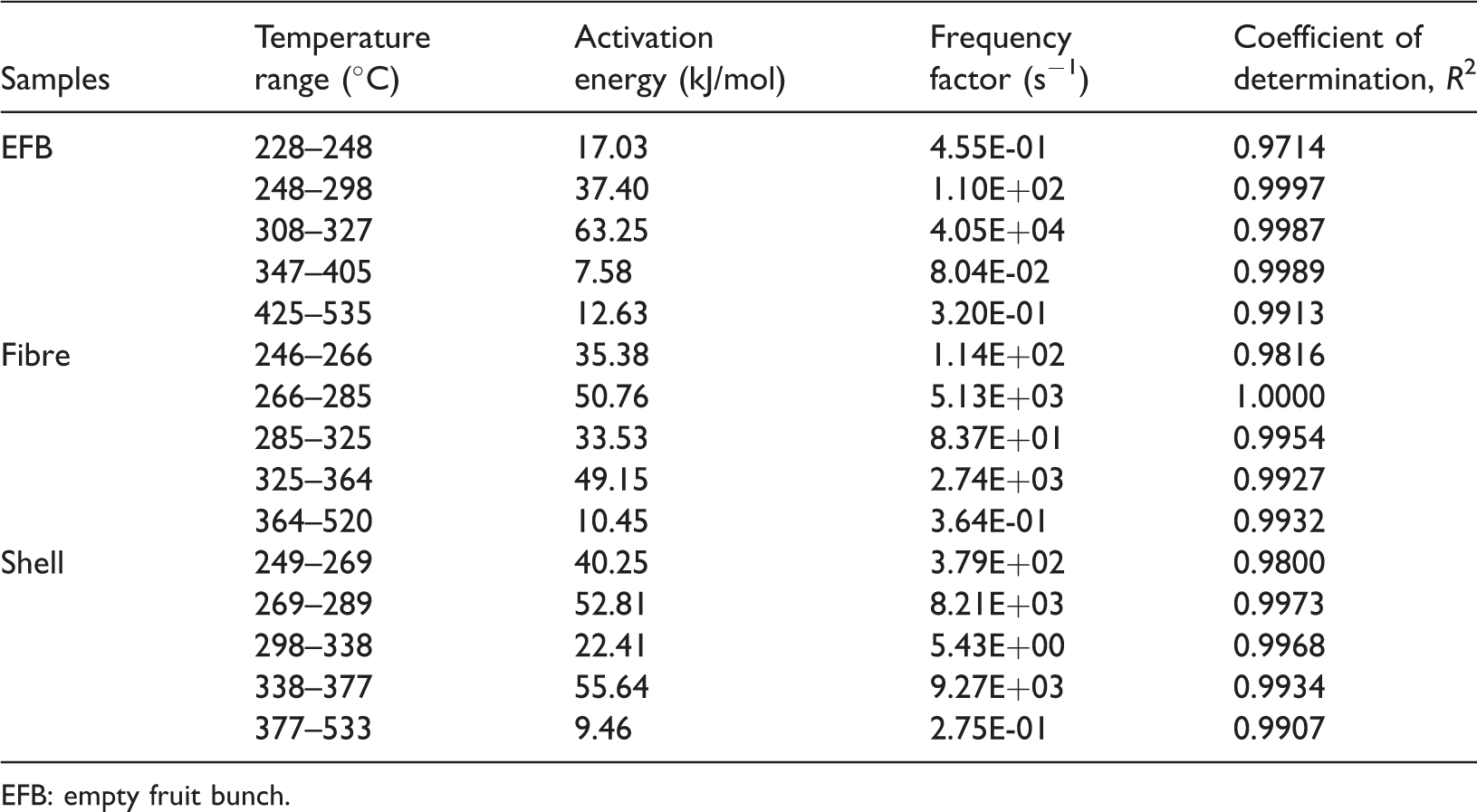
EFB: empty fruit bunch.
Based on the compositional analysis, these three components of oil-palm solid waste are mainly constituted of hemicellulose, cellulose, and lignin. As presented in Table 1, the proportions of cellulose, hemicellulose, and lignin in the shell are 15.54, 35.79, and 36.96 wt%, respectively, indicating that the lignin contents are high and the cellulose contents are low. Inversely, the cellulose contents are high and lignin contents are low in EFB and fibre. In more detail, the proportions of those constituents in EFB are 20.15, 35.01, and 19.65 wt%, whereas the corresponding contents in the fibre are 23.99, 37.67, and 22.99 wt%. These composition analysis results are similar with the previous studies (Shibata et al., 2008). Hemicellulose and cellulose are highly reactive and completely pyrogenic in the temperature range of 200–400°C, while lignin decomposes at lower rate in a broader temperature range. Therefore, the second stage was dominantly generated by the decomposition of hemicellulose and part of lignin. The third stage reflected the decomposition of cellulose and remaining lignin. This would appear to indicate that the curves of EFB, fibre, and shell are greatly influenced by the amount of cellulose, hemicellulose, and lignin in each (Orfao et al., 1999). So the pyrolysis process of them was essentially the thermal decompositions of the three constituents.
Pyrolysis characteristics and kinetics of the blends
Figure 2 shows the TG and DTG curves of equal proportion–blends: EFB–fibre (50%–50%), EFB–shell (50%–50%), fibre–shell (50%–50%), and EFB–fibre–shell (33.33%–33.33%–33%) at a heating rate of 10°C/min. It can be seen that the influence of the high cellulose content in EFB and fibre as well as the high lignin content in shell plays a significant role in the thermal decomposition process on their blends.
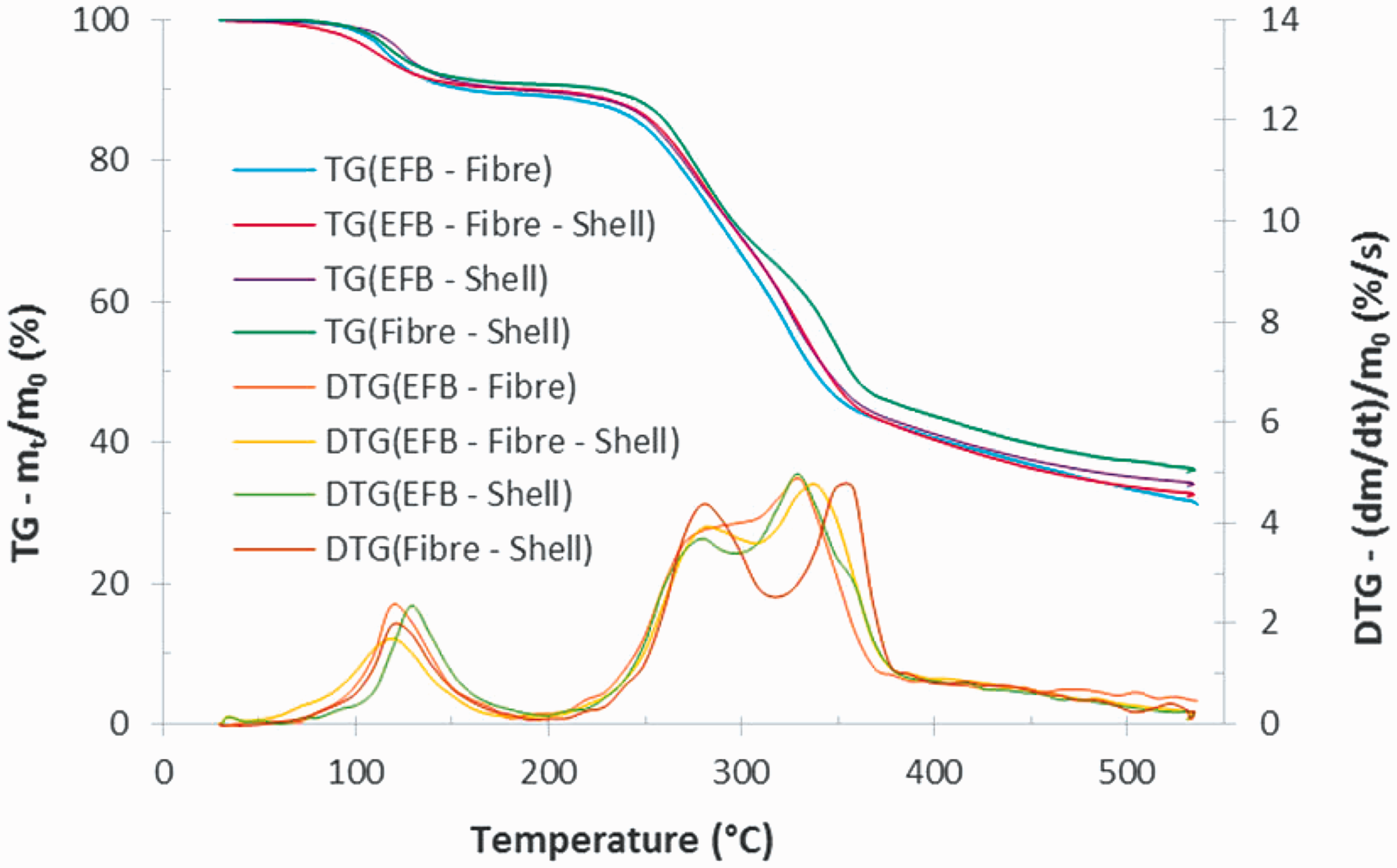
TG/DTG curves of blends at a heating rate of 10°C/min. DTG: derivative thermogravimetric; EFB: empty fruit bunch; TG: thermogravimetric.
In more detail, it appears in Figure 3(a) that the DTG curve of EFB–fibre blend has a similar shape as that of EFB, in which both have only one mass loss peak appearing in the devolatilisation of EFB, with a small shoulder located at the lower temperature side. This may be due to the influence of high cellulose content in EFB and fibre. On the contrary, the DTG curve of EFB–shell blend, as shown by Figure 3(b), could not form only one mass loss peak with a small shoulder, anymore, because of the significant involvement of lignin content in shell. Likewise, two mass loss peaks are found in Figure 3(c) and (d), even though it can be observed that the valley of the EFB–fibre–shell blend is shallower than that of fibre–shell blend, due to the mixing role of the three constituents. Overall, it can be seen in Figure 3(a) to (d) that the curves of all blends with equal component composition are nearly the mean of the mass loss rate of its components. However, the curves of the experimental blends and the theoretical ones are nearly coincident.
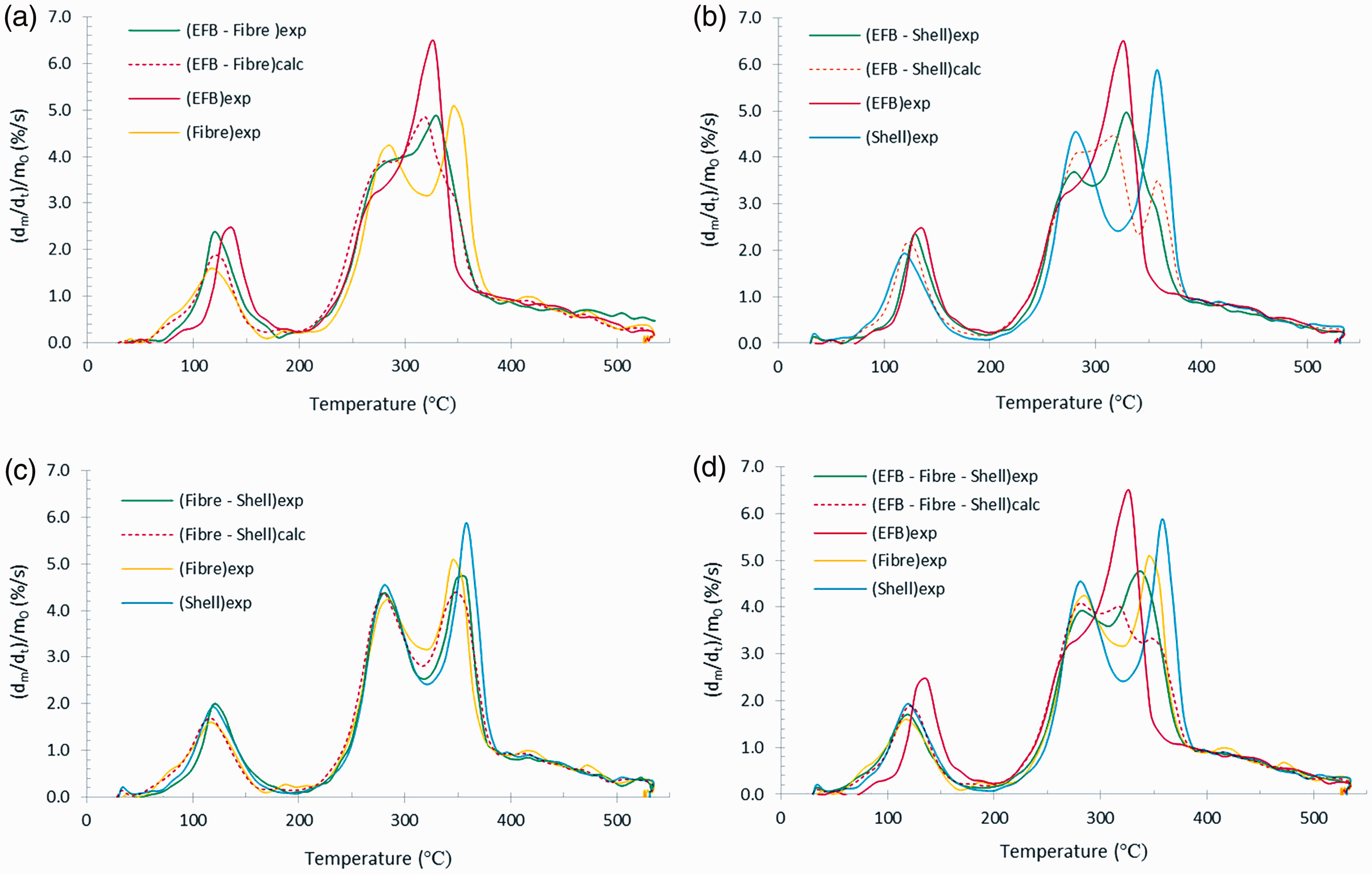
Comparison of DTG curves of blends with their components at a heating rate of 10°C/min. (a) Blend of EFB–fibre, (b) blend of EFB–shell, (c) blend of fibre–shell, and (d) blend of EFB–fibre–shell. EFB: empty fruit bunch.
To further investigate the synergistic effect among the oil-palm solid waste, the difference of weight loss, ΔW = Wblend −
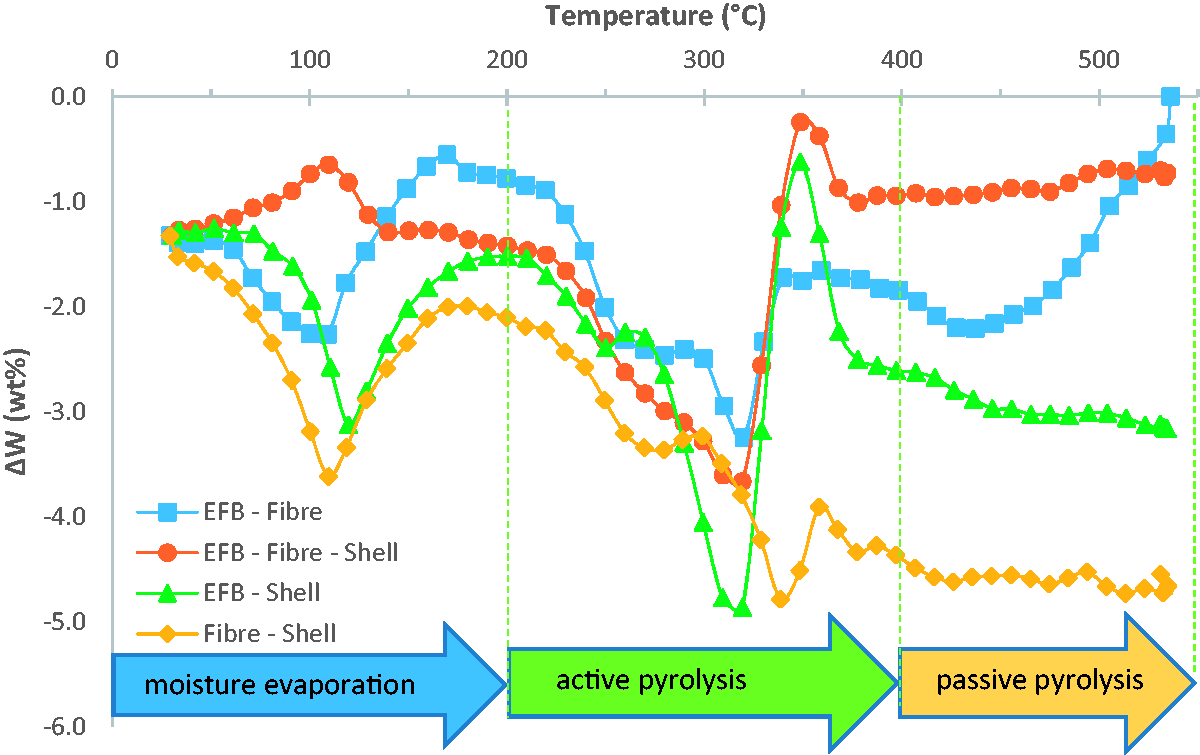
Variation of ΔW for the oil-palm solid waste blends. EFB: empty fruit bunch.
The maximum and minimum ΔW values for all mixtures.
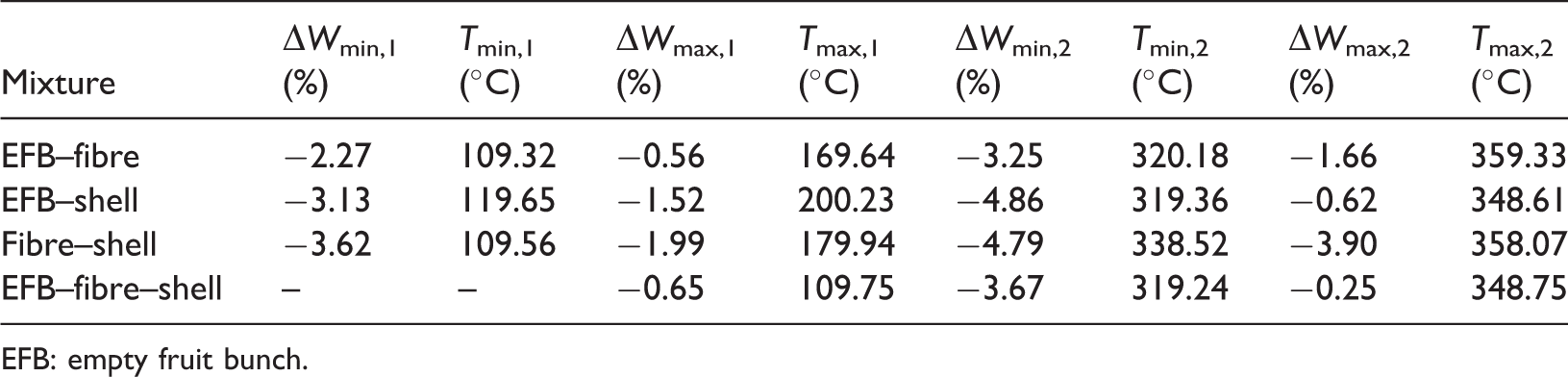
EFB: empty fruit bunch.
Furthermore, it has been known from previous research findings that cellulose, hemicellulose, and lignin decomposed at different temperature ranges (Dorez et al., 2014). It was found that lignin started to decompose at low rate and low temperature of about 200°C and continued until 600°C. Thus, it decomposed at both active and passive pyrolysis stage, whereas hemicellulose decomposed between 200 and 350°C. Then, cellulose is the last component that decomposed at higher temperature range of 280–400°C. By considering the decomposition temperature ranges of those three components, two peaks of the DTG curves in both active and passive pyrolysis stage for the blends in Figure 3 can be related to the decomposition of hemicellulose, cellulose, and lignin. The first peak probably involved hemicellulose and lignin decomposition, while the second peak was formed of cellulose and lignin decomposition. Cellulose is a semi-crystalline material, while lignin and hemicellulose are non-crystalline so the pyrolysis of cellulose must first destroy the lattice of crystals that needs energy, leading to higher activation energy (Mae et al., 2000).
As can be seen in Table 4, the activation energy and frequency factor values for blend of EFB–fibre vary from 2.97 to 38.35 kJ/mol and from 1.123E-02 to 1.326E + 02 s−1, respectively. The lowest value of activation energy took place in the temperature range between 359 and 417°C, whereas the highest one took place in the temperature range between 249 and 269°C. Still in the second temperature range, about 249–349°C, EFB–fibre blend had the high activation energy values, but then it had the low activation energy in the three higher temperature ranges, about 359–533°C. This could be possibly caused by the role of high content of cellulose in both EFB and fibre.
The activation energy and frequency factor values of (a) EFB–fibre, (b) EFB–shell, (c) fibre–shell, and (d) EFB–fibre–shell at different temperature ranges.
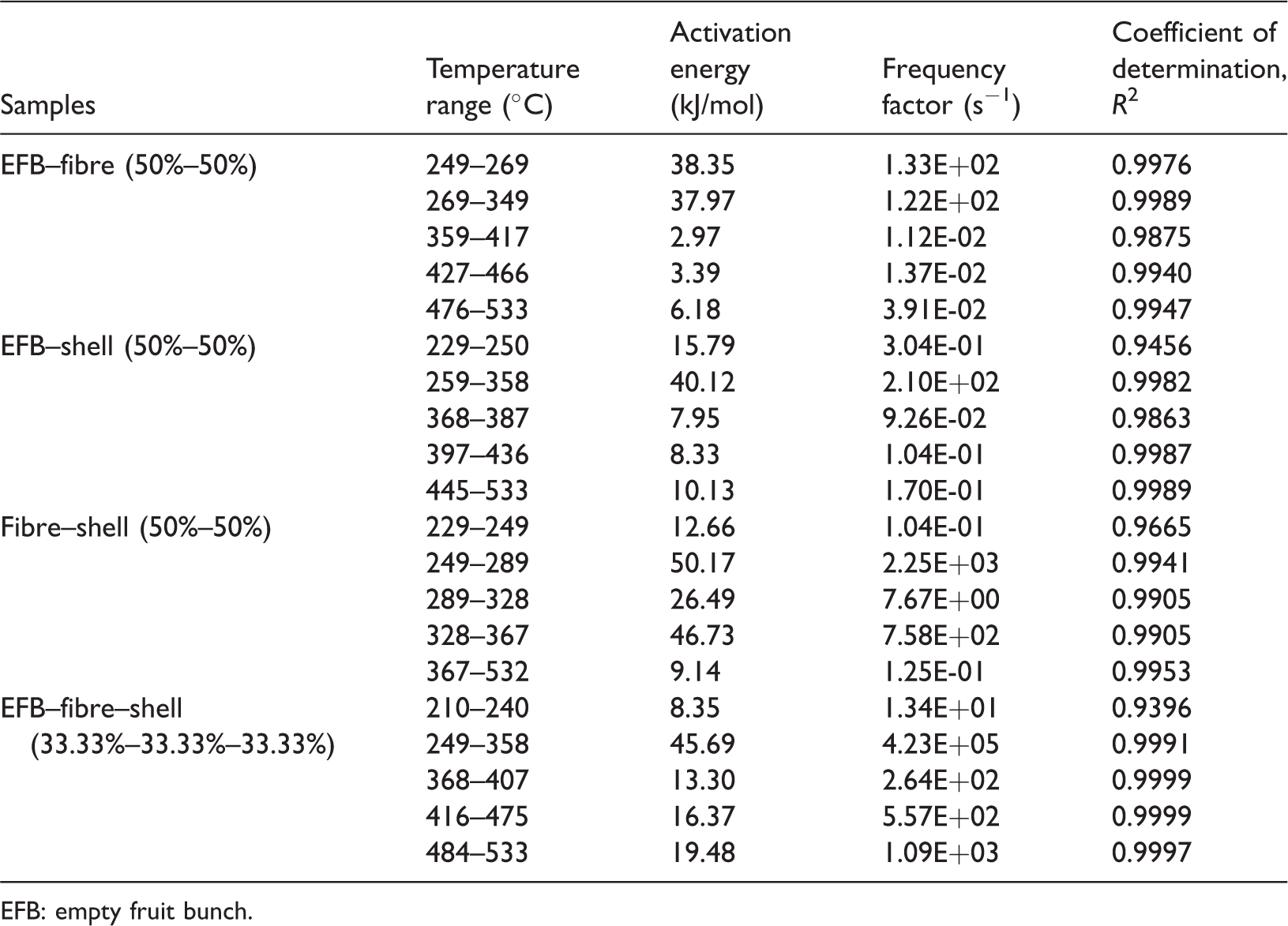
EFB: empty fruit bunch.
Different from the blend of EFB–fibre, the activation energies and frequency factors for blend of EFB–shell vary from 7.95 to 40.12 kJ/mol and from 9.260E-02 to 2.101E + 02 s−1, respectively. The lowest value of activation energy took place in the temperature range between 368 and 387°C, whereas the highest one took place in the temperature range between 259 and 358°C.
Meanwhile, fibre–shell blend had high activation energy in its second, third, and fourth temperature ranges. The activation energy and frequency factor values for fibre–shell blend vary from 9.14 to 50.17 kJ/mol and from 1.249E-01 to 2.250E + 03 s−1, respectively. The lowest value of activation energy took place in the temperature range between 367 and 532°C, whereas the highest one took place in the temperature range between 249 and 289°C.
Lastly, the activation energies and frequency factors for blend of EFB–fibre–shell vary from 8.35 to 45.69 kJ/mol and from 1.344E + 01 to 4.230E + 05 s−1, respectively. The lowest value of activation energy took place in the temperature range between 210 and 240°C, whereas the highest one took place in the temperature range between 249 and 358°C.
It is obvious that every blend has its own temperature ranges with nearly the same range. In addition, the activation energy and frequency factor values of the blends are different from those of each component. This ensures that the pyrolysis mechanism of blends is different from that of each component.
It needs to be noted that the coefficient of determination (R2) is employed to obtain the activation energy in various methods. When applying Coats–Redfern method, the coefficient of determination is a measure of how well the regression line represents the data and reflects the validity of the obtained activation energy for the method. It is clear that the obtained activation energy and coefficient of determination for the reactions from the Coats–Redfern method is substantially determined by the selection of temperature regions corresponding to the main mass loss stages varied with the heating rate. The different ranges of temperature were results of selecting adjacent points (in the plot
Conclusions
Summing up the results, it can be concluded that the curves of EFB, fibre, and shell were influenced by the content of cellulose, hemicellulose, and lignin. So, the pyrolysis process of them was essentially the thermal decompositions of these three constituents. The curves of all blends with equal component composition reflect nearly the average value of the mass loss rate of its components. Moreover, the curves of the experimental blends and the theoretical ones are nearly coincident. This would seem to suggest that the activation energy and frequency factor values of the blends were dominantly due to the role of the components with a synergistic effect (ΔW values ranges from −3.25 to −4.86%) occurred during pyrolysis.
The Coats–Redfern method has been successfully applied to determine the kinetic parameters, including activation energy and frequency factor, of the pyrolysis oil-palm solid waste by selecting the adjacent points in the plot of
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
