Abstract
The geochemical characteristics of fluorine in coal obtained from 534 borehole exploration data and mining test data from the Xiangning mining area were studied, and fluorine emissions were calculated. Results reveal that the fluorine content in Carboniferous-Permian coal in the mining area ranges from 2.00 to 911.00 mg/kg, and the Carboniferous coal seam contents are generally higher than those of the Permian system. The calculated average fluorine contents in Nos. 5 and 12 coal seams of the Carboniferous system are 203.42 and 308.00 mg/kg, respectively, and therefore, both are high-fluorine coal. In addition, the calculated mean fluorine contents of Nos. 2 and 10 coal seams are 168.58 and 187.02 mg/kg, respectively, which are higher than average value of Chinese and global coal values. The highest fluorine content of 896.00 mg/kg is found in medium- to high-fluorine coal. Fluorine in coal mainly exists in an inorganic bound state in forms such as fluorapatite and calcium fluoride. The Xiangning Coal Mine predominantly produces coal for coking and power, and the release of fluorine during coal combustion is harmful to the environment. According to the information obtained in relation to coal resource reserves and development planning, the amount of fluorine released from combustion is estimated at 2258.1 t/year and is estimated to reach 4862.9 t/year by 2020. Coal washing is an effective way of reducing fluorine emissions, and flotation experiments have shown that this can reduce the amount of fluorine by up to 30–47%. Different engineering measures, such as denitrification and desulfurization, can also significantly reduce the amount of fluorine released during coal combustion and can thus be employed to reduce the associated environmental impacts.
Introduction
Fluorine is one of the most active elements cycled in nature, and it is pervasive within almost all areas of the natural environment, such as water bodies, the soil, the atmosphere, and in rocks and organisms (Hong et al., 2018). Fluorine in coal is a harmful element (Ren et al., 1999) as it enters the environment in various forms during the process of coal mining, stacking, storage and combustion, thus causing pollution and affecting human health (Finkelman and Tian, 2017; Liu et al., 2007a). Coal-burning fluorosis is an endemic disease in the areas of Guizhou, north-eastern Yunnan, southern Hunan and southern Shanxi in China (Wang et al., 2018).
The fluorine content in Chinese coal ranges from 20 to 300 mg/kg, and the average value has been calculated as 122 mg/kg (Liu et al., 2007b) and 130 mg/kg (Dai and Ren, 2006). Qi et al. (2006) considered that the fluorine content of coal follows an approximately logarithmic normal distribution and ranges from 17 to 3088 mg/kg, where 73% of coal ranges from 50 to 300 mg/kg and the average value is 202 mg/kg (Qi et al., 2006). Wu (2004) compared the fluorine content of coal globally with that in China and concluded that the content within most (90%) of the coal in China was within a range of 47–347 mg/kg and the average content was 136 mg/kg. However, Luo et al. (2004) studied 300 coal samples of different ages from different regions and found that the fluorine content in Chinese coal ranged from 20 to 300 mg/kg, with an average content of 82 mg/kg, which is close to the world’s average of 88 mg/kg (Ketris and Yudovich, 2009).
The content of fluorine in coal has a close relationship with sulphur and it has a strong non-polar affinity (mainly silicate–aluminate). Fluorine also exists in minerals such as kaolinite, montmorillonite and mica (Eskenazy et al., 2013; Finkelman, 1995; Gurdal, 2011). Liu (2009) discovered a poor correlation between the fluorine content and chlorine in Guizhou coal but an evident correlation between the fluorine content and organic sulphur, and the fluorine in coal was found to occur mainly as mineral fluorine in an inorganic state. Wu et al. (2005) also considered that fluorine in coal mainly occurs in an inorganic form and stated that there is no definite relationship between the fluorine content and the degree of metamorphism. Furthermore, the studies of Dai et al. (2008) and Wang et al. (2011) determined that boehmite may be a form of fluorine in coal and that a small amount of fluorine exists in aluminophosphate.
Although the fluorine content in China's coal is relatively low, but the country uses coal as its main energy source, therefore, the annual fluorine emissions from coal combustion are approximately 1.62 × 105 t (Chen et al., 2013). Coal-fired hydrogen fluoride (HF) is a fluorine pollutant that has a serious and harmful effect on people, animals and plants (Wang et al., 2018). The toxicity of HF to plants is 20–100 times greater than that of sulphur dioxide, and its harmful effect on the human body is 20 times (Demir et al., 1998) greater. Fluorine pollution in the form of coal smoke is monitored in China, and corresponding pollution emission standards have been stipulated (Wang, 2016). Therefore, studying the distribution characteristics of fluorine in coal is important to determine effective ways of reducing and controlling environmental hazards caused by coal combustion.
Based on test data of 534 borehole seams from previous exploration stages within the Xiangning mining area (XMA), and test data from six main mining coal seams, the distribution of F, Cl, As, Cr and Pb contents and those of other environmental sensitive elements in coal were analysed within the mining area (Figure 1). Abnormal F contents were found in the coal of the XMA; therefore, this paper aims to determine the spatial distribution characteristics of F in Nos. 2 and 10 coal seams of the main mining area. In addition, the geochemical genesis is studied, and the environmental effects of F in coal during mining and utilization are evaluated. Through the study of the distribution characteristics of fluorine in the main coal mining layer in the XMA, the fluorine emission from coal combustion can be further calculated, which can draw the attention of relevant departments to detect the fluorine release during coal combustion in the mining area and take corresponding measures to reduce the fluorine emission.
Coal sampling at the study area was conducted according to the method in the Chinese Standard for Coal Seam Sample Adoption (GB/T 482-2008). The grooving sampling method was used for the No. 2 coal seam according to the Chinese Standard method GB/T 212-2008, total sulphur was determined in accordance with Chinese Standard method GB/T 214-2007 and Mad, Aad, Vdaf, St, d and F were determined in accordance with GB/T4633-2014.
Geological background of study area
The XMA is located in the south of Hedong Coalfield in Shanxi Province. The mining area has a length from north to south of approximately 100 km and a width from east to west of around 55 km, and it covers an area of approximately 4203 km2. It is rich in coal resources and is one of three famous coking production bases in China. The strata in the mining area are almost horizontal, and the coal seam structure is a generally simple monoclinic stratum that gently inclines to the west. According to surface exposure and borehole evidence, the strata in this area are from old to new as follows: the lower Majiagou Formation of the Ordovician; the Shangmajiagou Formation, Fengfeng Formation, Benxi Formation of the middle Carboniferous; the upper Taiyuan Formation, the Shanxi Formation of the lower Permian; the lower Shihezi Formation of the middle Paleozoic; upper Shihezi Formation of the upper Paleozoic and Quaternary Pleistocene. Coal-bearing strata include the Benxi Formation of the middle Carboniferous, the Taiyuan Formation of the upper Carboniferous and the Shanxi Formation of lower Permian.
The Shanxi Formation contains the main recoverable coal seam (No. 2), which has a buried depth of between 50 and 1500 m (approximately) and a thickness of 0.91–8.18 m with an average of 5.34 m. The entire area is stable and recoverable. The coal in No. 2 seam is lean coal or meagre lean coal with a low ash and extra low sulphur content. It also has a high calorific value and is weakly bonded. The Taiyuan Group contains No. 10 coal seam, which is the main recoverable coal seam that has a thickness of 0.54–7.70 m (with an average thickness of 2.67 m). The entire area is also stable and recoverable. The coal in No. 10 coal seam is weakly bonded lean coal and lean-lean coal, which can be used for coking, coal blending and power, and has a low ash and high sulphur content and a high calorific value.
Spatial distribution characteristics of fluorine in coal of XMA
To systematically study and analyse the distribution characteristics of fluorine in coal of the XMA, the fluorine content of 661 coal seam samples was statistically analysed. According to the China coal standard for fluorine content classification in coal (MT/T 966-2005), the Permo-Carboniferous coal seam in the XMA is medium- to high-fluorine coal, and the fluorine content in coal seams ranges from 2.00 to 911.00 mg/kg. The standard deviation indicates that the dispersion degree of fluorine content is large. The minimum, maximum and average values of fluorine in coal within various areas of the XMA are presented in Table 1.
Distribution of fluorine content in coal within the XMA.
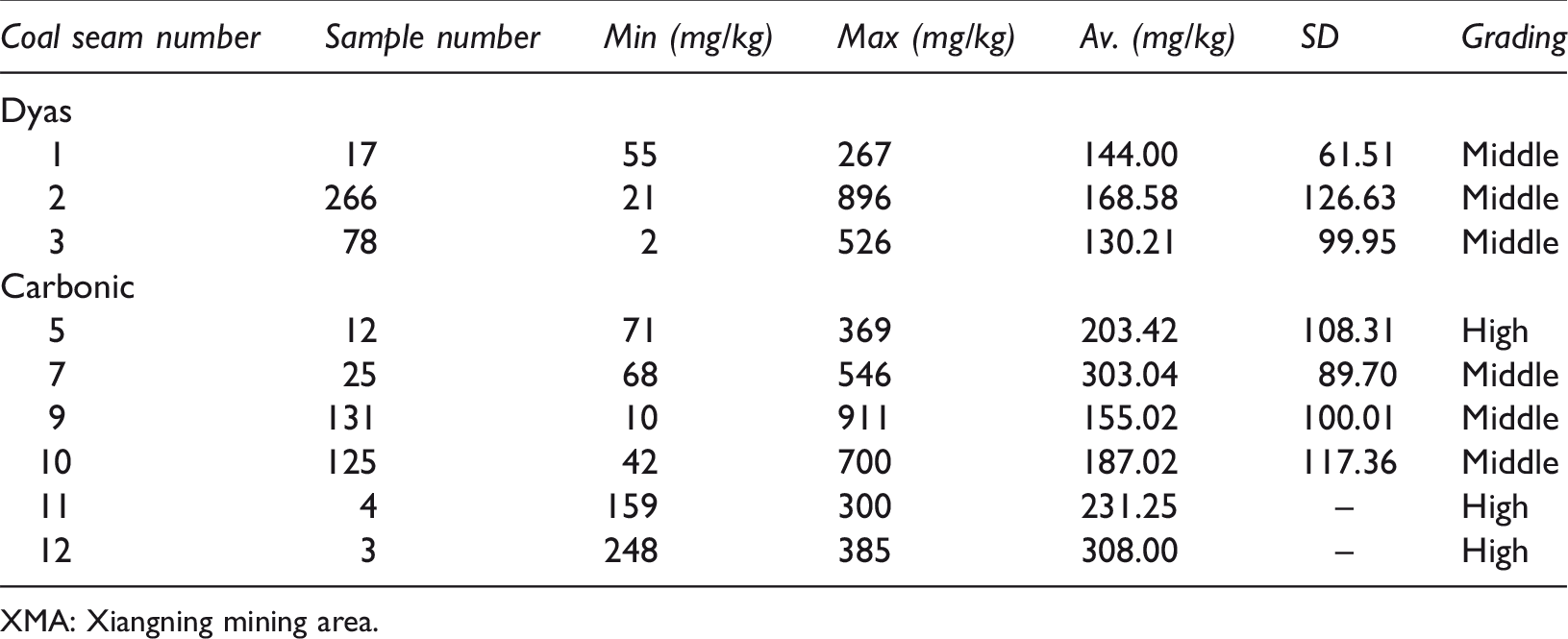
XMA: Xiangning mining area.
Statistics relating to fluorine contents of within major coal mining areas of Shanxi province.
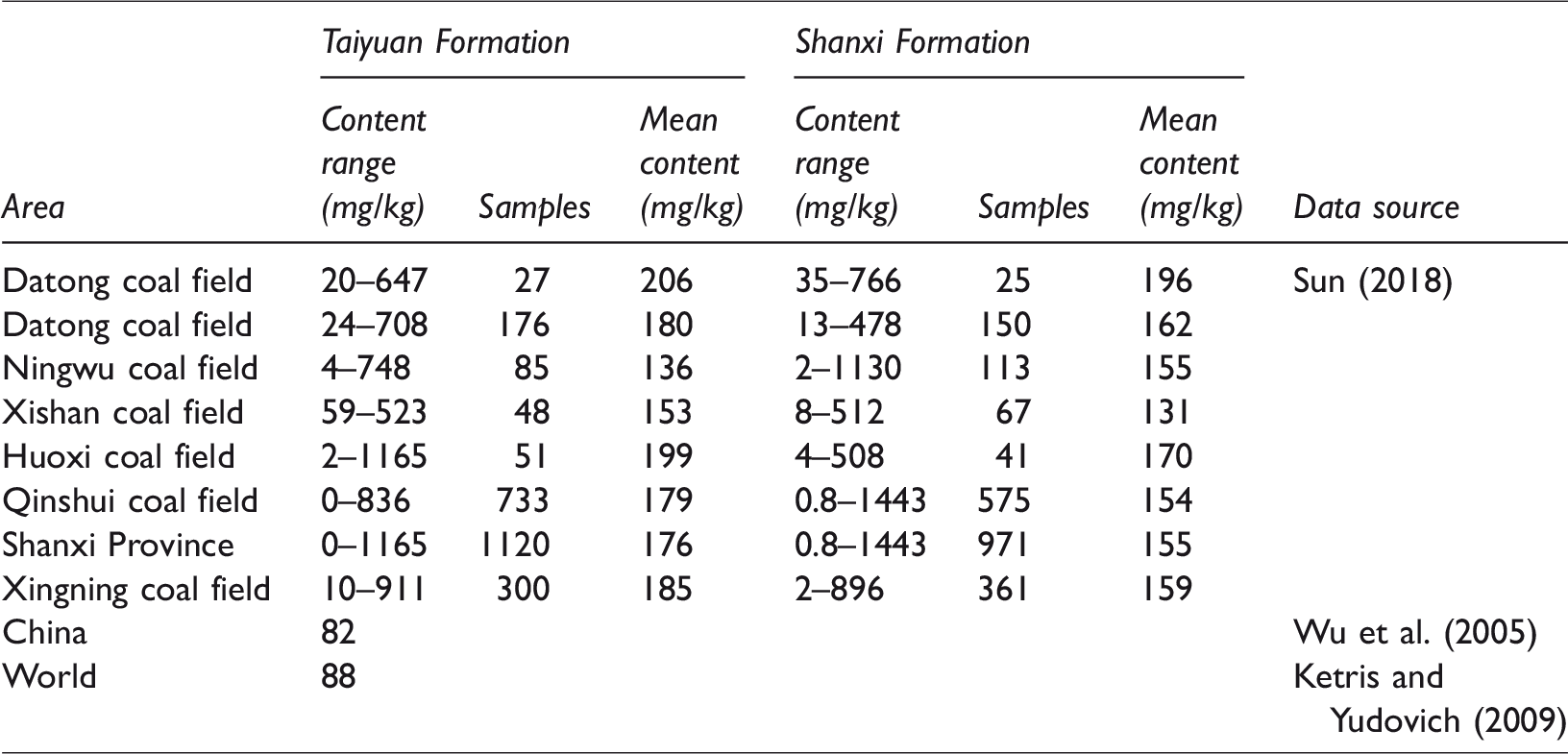
The content of fluorine in the coal varies greatly in different regions of China. Lu (1996) considered that the fluorine content in coal-accumulating areas in South China is higher than that in north and north-eastern China. It has been shown that the fluorine contents of coal in the Taiyuan and Shanxi Formations in the XMA are 185 and 159 mg/kg, respectively, which is consistent with that of Taiyuan and Shanxi Formations in Shanxi Province. However, the calculated average fluorine content of coal in the XMA is 170.80 mg/kg, which is higher than the average fluorine content of other coal in China and throughout the world of 82 mg/kg (Wu, 2004) and 88 mg/kg, respectively (Liu et al., 2008) (Table 2).
Distribution characteristics of fluorine in No. 2 coal seam
No. 2 coal seam is currently the main coal seam in the XMA and other coal mining areas in eastern China. Based on 266 coal samples obtained from No. 2 coal seam in the XMA, the distribution of fluorine in No. 2 coal seam was analysed and is shown in Figure 2. These results show that the fluorine content in No. 2 coal seam in the XMA has typical characteristics: it is higher in the northern and southern parts and lower in the middle. A high-fluorine coal area is found in the northwest part of the mining area near Xuecun in Puxian County, where the fluorine content exceeds 300 g/kg and the highest value is 896.00 mg/kg. In addition, a high-fluorine coal area reaching 650.0 mg/kg is found in No. 2 coal seam in the Tanping exploration area in the south of the XMA. High-fluorine content (>200 mg/kg) coal in No. 2 coal seam accounts for approximately 20% of the mined seam area, and medium-fluorine coal and high-fluorine coal (>130 mg/kg) account for approximately 70% of the total seam area.
Occurrence and distribution of coal resources in XMA.
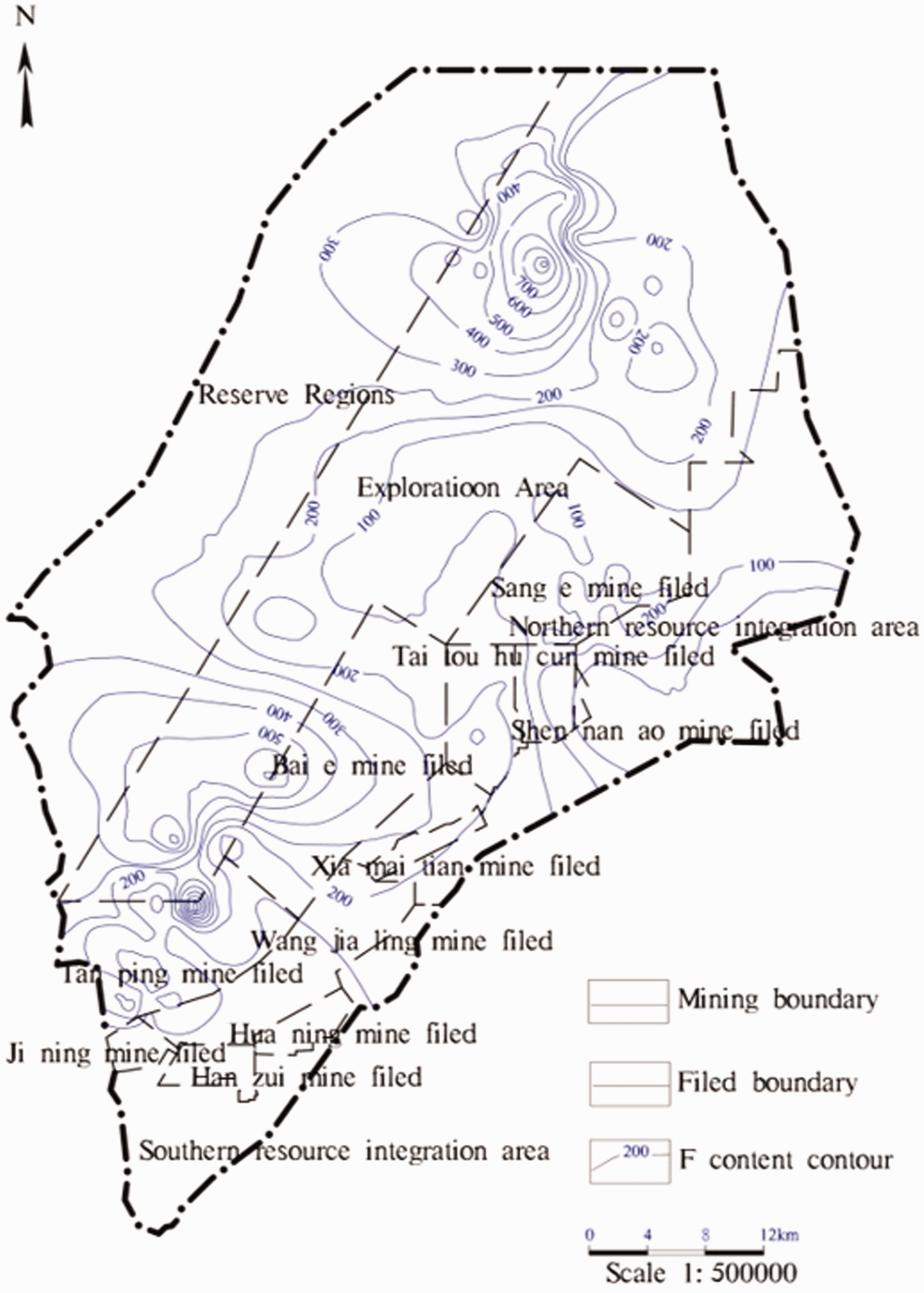
Contour map showing fluorine content in No. 2 coal seam within XMA.
Geochemical characteristics of fluorine in coal
Analysis of correlation between fluorine content of coal and other components (ash and volatile matter contents)
Fluorine is a ubiquitous element that occurs in coal in both organic and inorganic forms. However, it has been shown that fluorine in most coal exists in the form of inorganic minerals, including fluorapatite and calcium fluoride (Wu et al., 2005). Fluorine in coal can be adsorbed onto ions in clay minerals such as montmorillonite and kaolin (Liu et al., 2008; Ning et al., 2017), and organic-bound fluorine in coal mainly combines with organic matter (such as organosilicate) through complexation, chelation or adsorption (Wang et al., 2011).
The ash and fluorine contents of coal from the Nos. 2, 9 and 10 coal seams in the XMA were investigated, and the correlation between contents of fluorine, ash and volatile matter was analysed (Table 3). The statistical results show a significant positive correlation between fluorine and ash in coal, but no significant correlation between fluorine and volatile matter. The results also show that fluorine mainly exists in the form of inorganic minerals, including fluorine-containing minerals (fluorapatite) or inorganic salt minerals. These minerals remain in the residue after burning and form coal ash; therefore, the fluorine content in this kind of coal is significantly correlated with the ash content (Zhang et al., 2018). Wu et al. (2005) studied the content and distribution characteristics of fluorine in Chinese coal, and the same results were obtained.
Correlation between contents of fluorine, ash and volatile matter.
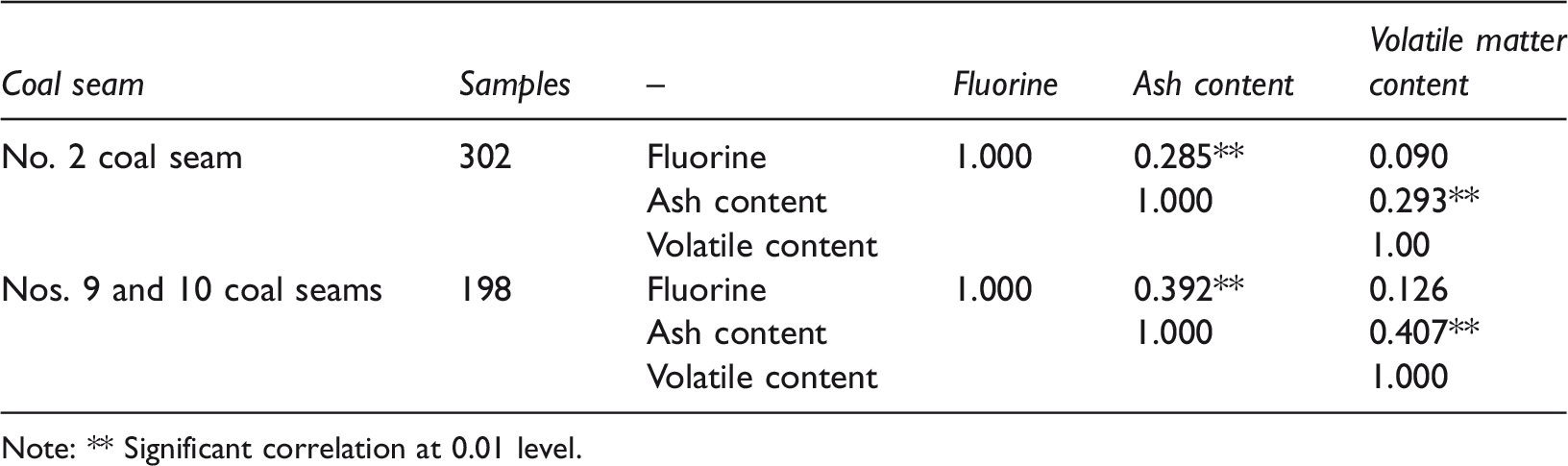
Note: ** Significant correlation at 0.01 level.
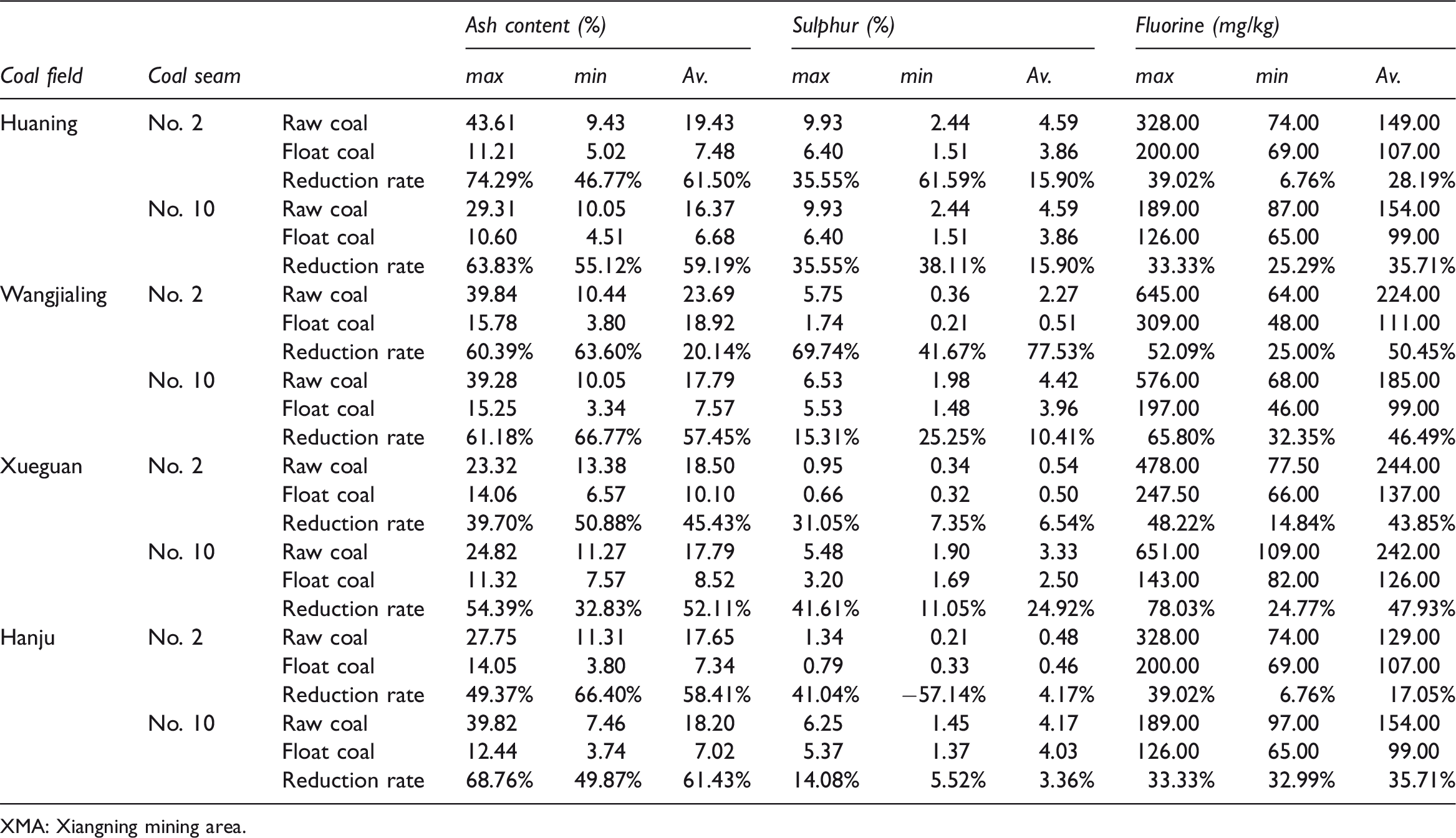
Analysis of ash, fluorine and sulphur contents after raw coal washing in XMA.
XMA: Xiangning mining area.
XRD and SEM analysis of coal sample showed that minerals such as quartz, kaolinite, muscovite and pyrite were found in No. 2 coal seam (Figures 3 and 4). Therefore, it was speculated that fluorine in No. 2 coal seam in XMA might be adsorbed in clay minerals such as muscovite and kaolinite in ionic state.
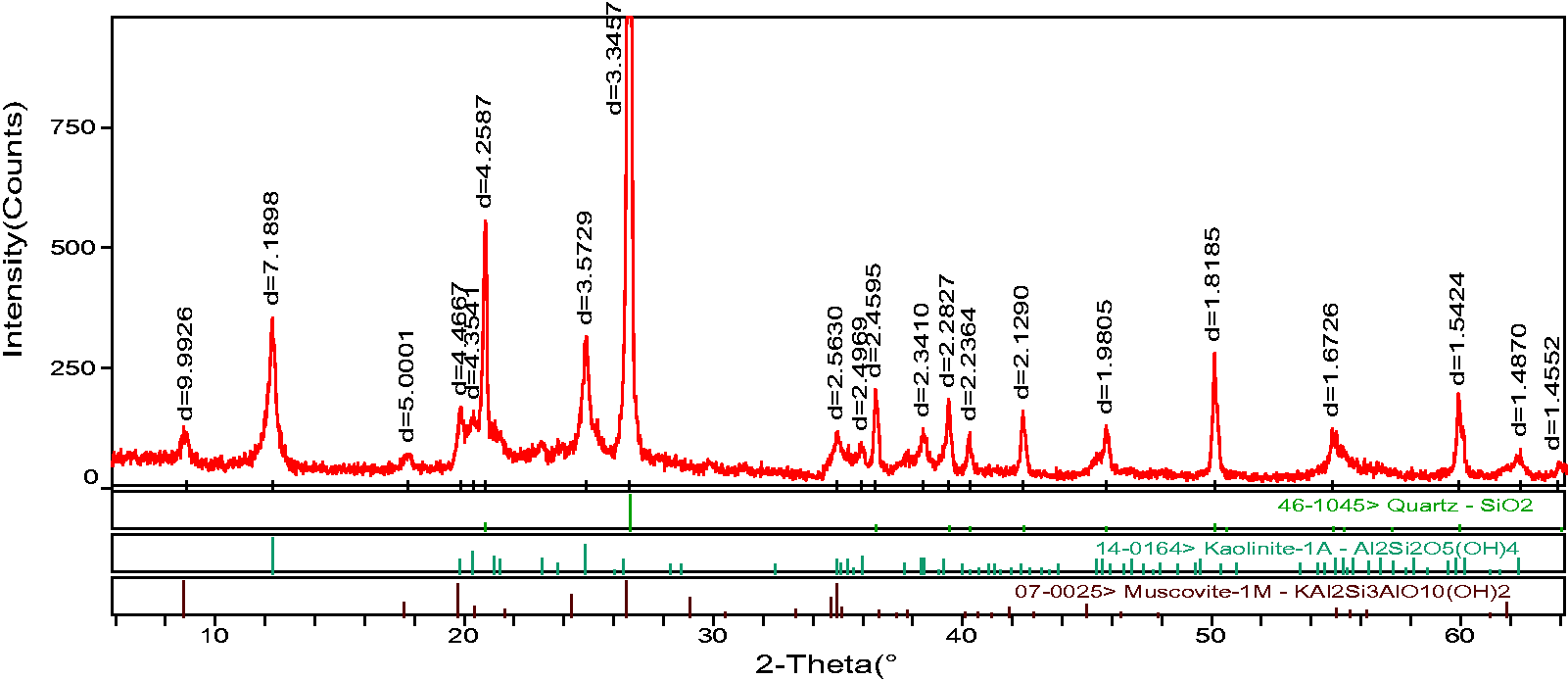
XRD pattern of No. 2 coal seam.

SEM image of No. 2 coal seam ×350.
Figures 5 to 7 show the relationship between contents of ash, volatile matter and fluorine in coal of the main mining seam in the XMA. From the graph, it can be seen that fluorine is positively correlated with the ash content, and the fluorine content increases with an increase in the content of ash. However, the relationship between fluorine and volatile matter is not obvious, and fluorine in coal mainly exists in an inorganic binding state. In addition, in the same ash range, the fluorine content in some of the samples is relatively high but the volatile matter content is relatively low, which further illustrates that most of the fluorides in the coal samples occur in an inorganic bound form. The relationship between calcium oxide and fluorine in coal ash can be clearly seen: coal samples with a higher fluorine content generally have a higher calcium oxide content. These results show that in the XMA, the high fluorine in the coal mainly occurs in the form of fluorapatite and calcium fluoride.
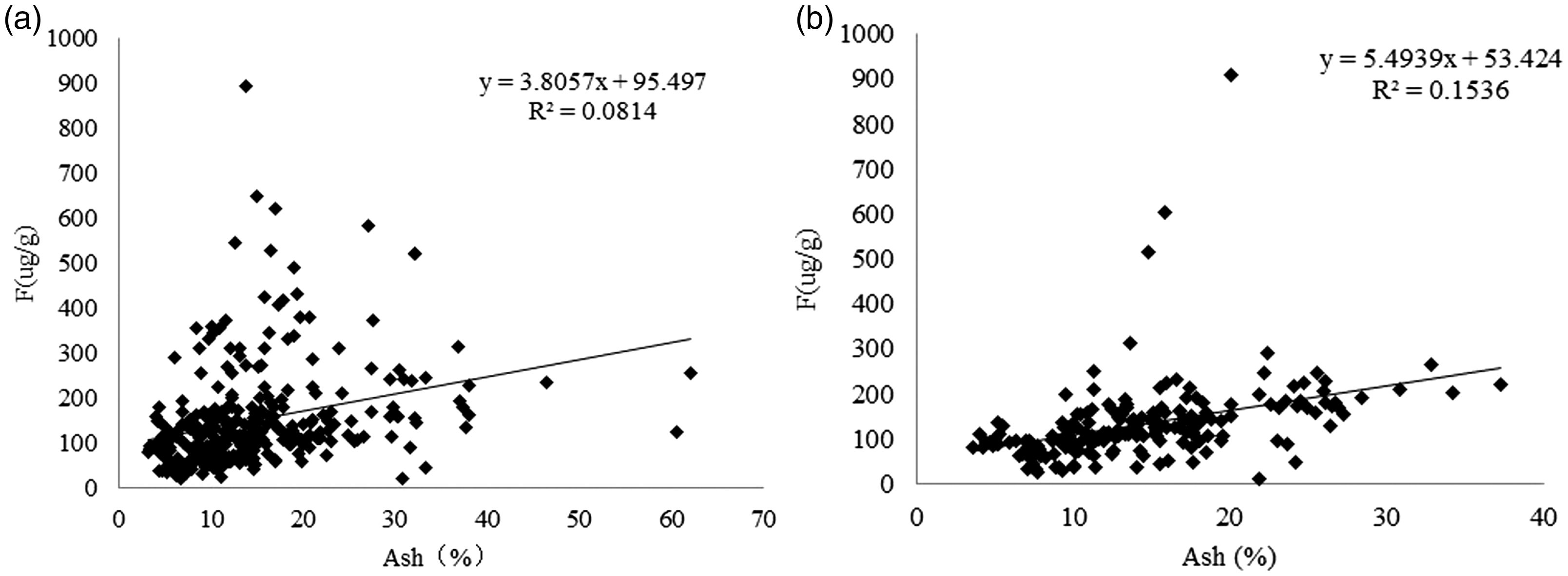
Relationship between ash and fluorine contents of coal. (a) No. 2 coal seam and (b) No. 9 + 10 coal seams.
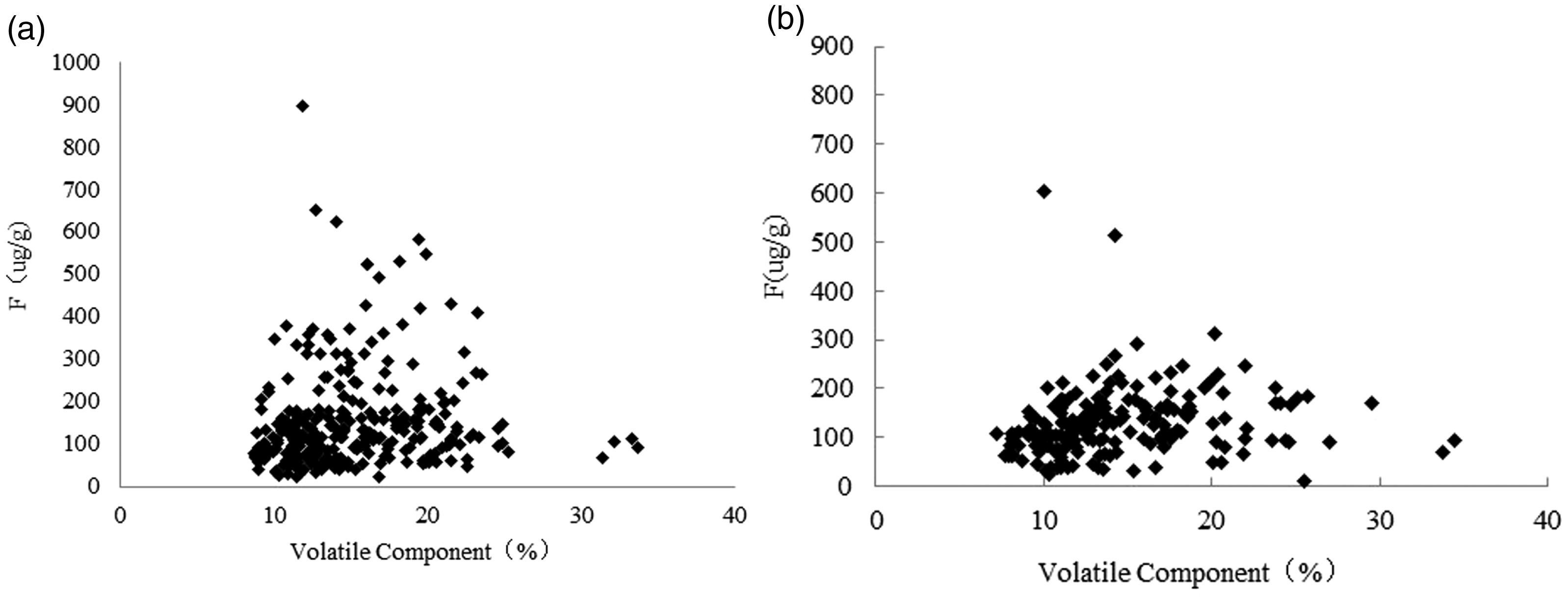
Relationship between volatile matter and fluorine contents of coal. (a) No. 2 coal seam and (b) No. 9 + 10 coal seams.
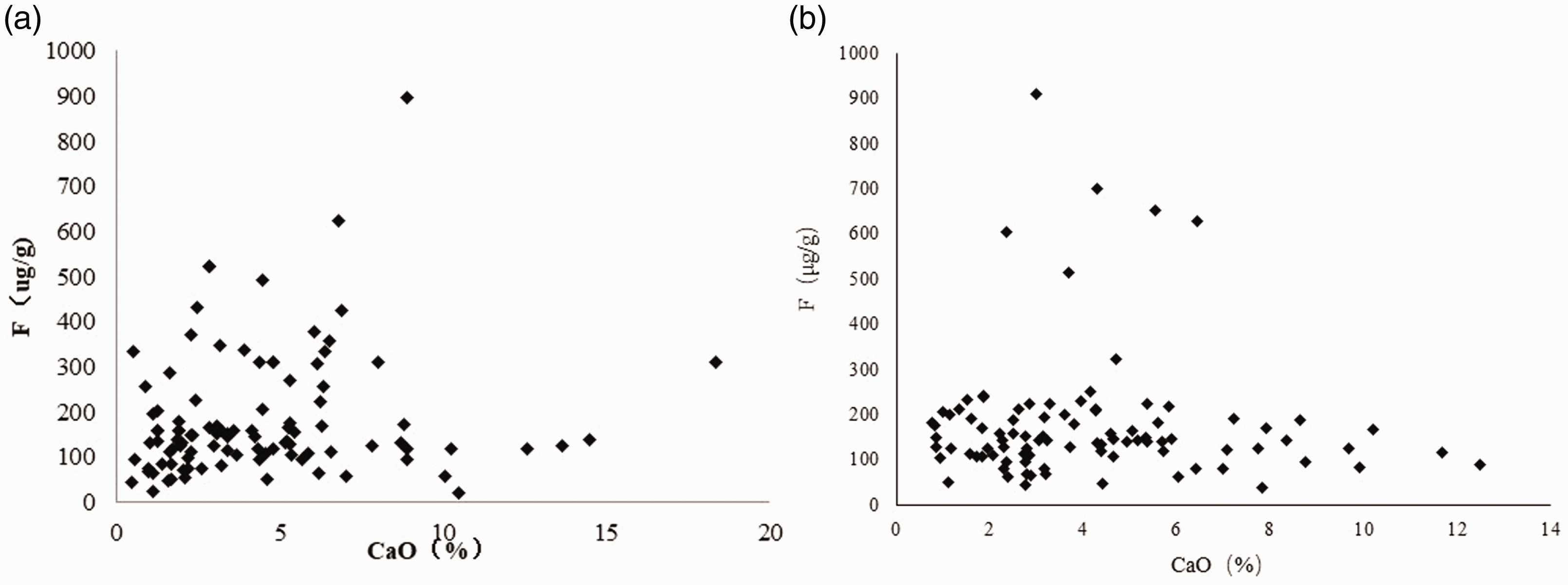
Relationship between CaO and fluorine contents of coal. (a) No. 2 coal seam and (b) No. 9 + 10 coal seams.
Analysis of geochemical origin of fluorine
Trace element enrichment in coal is controlled by many factors and multi-stage actions and is often the result of multiple factors. In the stage of coal-forming peat, the main controlling factors are the parent rock properties, sedimentary environment, coal-forming plant types, microbial actions, climate and hydrogeological conditions within the terrestrial source area (Ren et al., 1999). At the stage of coalification, coal seam roof sedimentation and diagenesis, microbial action, tectonics, magmatic hydrothermal activity and groundwater activities are the main controlling factors (Dai et al., 2012). When the coal-bearing basin is reformed later, the coal seam enters the supergene stage, and wind oxidation can also enrich or leach trace elements from coal (Dai et al., 2014a; Dai et al., 2014b).
The coal-bearing strata in the XMA mainly belong to the Benxi Formation of the middle Carboniferous, the upper Taiyuan Formation and the Lower Permian Shanxi Formation. The middle Carboniferous Benxi Formation comprises a high-level system tract and a transgressive system tract, which were mainly developed in a tidal flat system, mud flat, peat swamp and sand flat. The upper Carboniferous Taiyuan Formation mainly developed within a tidal flat system, tidal channel, peat swamp and sand flat, and the Shanxi Formation of Lower Permian belongs to continental marsh deposits (Tan, 2017; Zhu et al., 2010). It is thus evident that the sedimentary environment is one of the causes of fluorine enrichment within the coal (Dai et al., 2014b, 2012). Marine transgression has a great impact on peat marshes, not only because the fluorine content in seawater is higher than that in fresh water, which can provide more abundant material sources, but also because seawater changes the pH and Eh values of peat marshes and produces a specific geochemical barrier that is conducive to the enrichment of fluorine (Liu and Cao, 1987). In addition, for the continental marsh sedimentary environment of the Shanxi Formation of the Lower Permian, the fluorine in coal is likely also related to the parent rock of the terrestrial source area, which resulted in fluorine enrichment in the ancient soil of the peat swamp: the coal-forming plants and peat swamp media were thus enriched with fluorine. Therefore, the geochemical origin of fluorine in the coal within the XMA is considered to be related to both terrestrial enrichment and sedimentary-biological enrichment type.
Emissions of fluorine and associated prevention
Coal preparation and emission reduction
Coal washing can effectively reduce the content of impurities such as ash, gangue and sulphur; it also improves the quality and utilization efficiency of coal and reduces pollutants emitted during coal combustion. The composition of raw coal and floated coal in some of the coalfields in the XMA was analysed, and results show that coal preparation can greatly reduce the ash, sulphur and fluorine content in coal. The ash reduction rate in prepared samples is mostly over 40% and the ash reduction rate of coal in the Huaning coalfield reaches 74.29%. The sulphur reduction rate varies, but the volatilization of sulphur in coal is controlled to some extent by preparation, and the sulphur reduction rate in the Huaning minefield reaches 61.55%. The reduction rate of fluorine in prepared coal reaches between 17.05 and 50.45%. Therefore, coal preparation is shown to be a direct and effective way of reducing fluorine emissions from coal (Table 4).
Coal combustion and emission reduction
Solid fluoride in coal is basically decomposed into gaseous fluoride and discharged into the atmosphere during combustion. Only a very small amount of fluoride (<5%) remains in the ash in a solid state form (Luo et al., 2002). In the process of coal combustion, the release rate of fluorine in coal increases with an increase in temperature, and when the temperature reaches 1100–1200°C, the release rate is close to 100% (Finkelman et al., 1999). The annual output of coal in the XMA is currently approximately 2100 t, and there is a total planned construction scale of 45 million t/year. As the average fluorine calculation value of coal in the mining area is 170.80 mg/kg, it is estimated that the annual output of fluorine in the mining area is 3586.8 t. The total planned construction scale in 2020 will reach 7686 t/a of fluorine after the production. Research results show that the fluorine release rate of industrial coal from power plants is basically concentrated between 80 and 99% (Luo et al., 2001; Qi et al., 2005), and coal from the XMA is calculated to have a fluorine release rate of 90%. Current coal preparation rate in China is 77.8% and the reduction rate of fluorine after preparation reaches 38.17%. It is predicted that the current annual release of fluorine from coal combustion of 2258.1 t in the mining area will reach 4862.9 t after 2020. In the process of coal combustion, most of the fluoride in coal decomposes and forms HF and a small amount of SiF4, CF4 and other gaseous pollutants are released into the atmosphere that are harmful to plants, animals, the human body and the ecological environment. In some areas, fluoride released after combustion enters the human body through the food chain and the respiratory tract, resulting in the occurrence of fluorosis in people. Therefore, it is necessary to focus on the release of fluorine in coal and adopt effective technical methods to reduce fluorine emissions from coal and to achieve clean and efficient utilization of coal resources.
Defluorination technology and emission reduction
Defluorination treatment technology can currently be divided into four types: defluorination before combustion (coal treatment and purification technology used prior to combustion), defluorination during combustion (coal purification technology employed during combustion), flue gas defluorination after combustion (coal purification technology used after combustion) and coal conversion technology (Yang, 2015). Prior to burning, defluorination is the first step used to remove or reduce some of the fluorine and ash impurities in raw coal. Low-grade coal and pulverized coal are then manufactured into coal products that have a certain shape or granularity; these briquettes produced in relation to lowering the impurities and fluorine content of coal (Yang, 2015). Defluorination technology used during combustion involves mixing a defluorinating agent or calcium-based fluoride fixing agent into the coal prior to combustion and then spraying a defluorinating agent into the combustion chamber to remove fluoride during the combustion process. This is an effective method of ameliorating coal-fired fluorine pollution (Yu, 2007). Flue gas desulfurization technology used after combustion employs wet and dry methods. With wet flue gas defluorination technology, the entire desulfurization system is located at the end of the flue and after the dust collector. Application of the defluorination agent (CaCO3), use of the defluorination process, creation of the reaction by-products and their regeneration and treatment are all conducted in a wet state. The flue gas after defluorination is emitted from smokestacks after reheating (Guo, 2015). A dry defluorination processes is used to spray dry absorbent directly into the air flow of the boiler furnace, and the heat in the furnace calcines the absorbent into active CaO particles. The surfaces of these particles then react with HF in the flue gas to form CaF2, and these reaction products are captured with fly ash by dust removal equipment, such as electrostatic precipitator or bag filter (Yang, 2015).
Conclusion
The fluorine content of coal in the XMA ranges from 2.00 to 911.00 mg/kg with an average content of 170.80 mg/kg, which is higher than that in other coal samples from China and throughout the world. The coal from the Carboniferous-Permian seam in the Xiangning Coal Mine has a medium–high fluorine content, and the calculated average fluorine contents of coal from Nos. 2 and 10 coal seams, which are the main mining seams, are 168.58 and 187.02 mg/kg, respectively; this coal is classified as having a medium-fluorine content. The fluorine content of coal in the No. 2 coal seam is higher at its northern and southern ends and lower in the middle part. These two high-fluorine coal areas are located in the northwest and southwest of the mining area, respectively.
There is a positive correlation between the fluorine and ash contents in coal within the main coal seam of the XMA, but the relationship between fluorine and volatile matter is not obvious, and the fluorine in coal mainly exists in an inorganic binding state. The relationship between calcium oxide and fluorine in coal ash was analysed, and results show that the high-fluorine content in coal within the XMA mainly occurs in the form of fluorapatite and calcium fluoride, and might be adsorbed in clay minerals such as muscovite and kaolinite in ionic state. Furthermore, it is considered that the geochemical origin of fluorine in coal within the XMA is related to both terrestrial enrichment and sedimentary-biological enrichment.
It is predicted that 2258.1 t of fluorine are currently released during coal combustion each year in the XMA, and an amount of 4862.9 t will be released every year after 2020. Flotation experiments show that coal preparation can greatly reduce the fluorine content of coal, and preparation is a direct and effective way of reducing fluorine emissions in coal. The release of fluorine from coal is harmful to plants, animals, human beings and the environment. It is thus necessary to reduce fluorine emissions from coal using effective technical methods to achieve the clean and efficient use of coal resources and ultimately protect the ecological environment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by the National Natural Science Foundation of China (No. 41572218) and China Geological Survey Geological Survey Fund (DD20160187).
