Abstract
Major, trace, rare-earth elements, and noble metal have been analyzed from the brown coal of the Amur region (Russian Federation) and coal combustion products to understand the provenance and depositional environment. It was discussed that weathering crusts of ore deposits and host rocks located in the mountain-folded frame of the Zeya–Bureya coal-bearing sedimentary basin were the main source of mineral substance and chemical elements in brown coals. The distribution of minerals and elements inside the forming coal seams was influenced by the type of vegetation incorporated into coal-forming peat mires bog by catastrophic floods along the existed hydraulic network, the physicochemical parameters of plant debris transformation during their conversion to coal, as well as the sorption properties of inclusions and interlayer of bitumen and humic acids in coal beds. An experimental technological complex was created to study the microelements transfer process in the dust–gas stream and the distribution of rare earth and trace elements separately in varieties of coal combustion products (slag, fly ash, dust-like part of the dust–gas flow, and technogenic cleaning water). The resulting combustion products of coal have been studied by inductively coupled plasma (ICP)-spectrometry methods (ICP-mass spectrometry and ICP-atomic emission spectroscopy). Sample preparation technique for analysis was improved to determine the elemental composition. It was established that more than 60% of trace and rare earth elements were concentrated in ash and sludge. Data on these elements’ concentration in the wash water sediment demonstrated a sufficiently high efficiency of the dust–gas flow cleaning. The use of a wet scrubber, of course, solves the problem of collecting the dust and gas flow cleaning from harmful and toxic elements of coal smoke, which should be a part of the technological chain of a coal combustion process in industrial boilers. Rare earth elements, scandium, and yttrium are considered economically valuable components in the coal combustion products of the Yerkovetskoe brown coal deposit in the Amur Region.
Keywords
Introduction
Consumption of rare earth elements (REEs) is growing in a wide range of commercial and defense applications, in particular, in the communications and wind energy industry, where REEs containing magnets are used in generators. In this regard, the study of the coal combustion products (CCP) as a potential source for the extraction of REEs, as well as some precious metals and trace elements, is considered a priority subject for research by scientists from different countries (Orem and Finkelman, 2003; Stuckman et al., 2018; Vejahati et al., 2010). Currently, technological solutions for the extraction of metals from ash and collecting mineralized coal-dust associated flue gas are under development. The Chinese company Shenhua Group has built a pilot plant producing 10,000 tons of aluminum and concentrates containing Ga and Si from coal ash. The British company RockTron is developing processes for extracting REEs from enriched coal ash fractions. Latrobe Magnesium is building a plant in Australia for extracting Mg from coal ash (Sorokin et al., 2018). An Israeli company built a plant using a wet venture scrubber to clean coal flue gases from Hg and its compounds (Free Pollution Online Newsletter, 2017). There are forecasts for the production of coal waste (slag and ash) in the next 20 years at the current level.
The main source of rare earth and trace elements in coal deposits of the Amur region are products of the physical disintegration and chemical decomposition of rocks and numerous ore veins, located in the rocky frame of the Zeya-Bureya coal-bearing sedimentary Basin, and their distribution within the emerging strata of coal, influenced the type of vegetation and plant debris, transported by streams and catastrophic floods to depositional sites on the territory of coal-forming peat mires, physicochemical parameters of vegetation maturing during their conversion (diagenetic alteration) into brown coal, as well as the sorption properties of inclusions and interlayers of organic pitch, bitumen, and humic acids in peat and coal beds. Accessory minerals in the brown coal deposits of the Basin are concentrators of economically valuable metals and elements, the amount of which is correlated also with content of ash in coal, which ranges from 9 to 20 wt.% (Sorokin et al., 2013).
It is believed that during the combustion of coal, REE + Y + Sc mostly remain in the solid residue (slag and ash) and are not transferred by the gas phase, as in the case with other more volatile elements (Ratafia-Brown, 1994). At the same time, in recent years, it has been established that a large amount of trace elements and REEs is carried away with the dust–gas flow in a flue gas. One of the facts confirming the transfer of them with flue gas during coal combustion is the formation of contrasting geochemical anomalies of REEs in snow cover around large urban thermal power plants with enrichment in solid snow sediment with light REEs relative to heavy ones (Radomskaya et al., 2018).
Increased metal concentration is peculiar to brown coal from many deposits of the world (Seredin et al., 2013; Shpirt et al., 1999; Singh et al., 2016; Sorokin et al., 2013; Sun et al., 2010), therefore, the CCP are considered by many scientists as potential additional sources of noble metals and rare earth and trace elements (Kolkera et al., 2017; Sorokin and Konyushok, 2018).
Geochemical background
The authors studied the mineral composition and chemical elements concentration in brown coal from most deposits of the Zeya–Bureya Basin (Figure 1). The largest amount of data has been accumulated for deposits in the southern part of the Basin (Arkharo-Boguchanskoe, Raichikhinskoe, Sergeevskoe, and Svobodnoe deposits), and particularly for Yerkovetskoe brown coal deposit. The border of the Russian Federation and the People’s Republic of China passes along the Amur River.
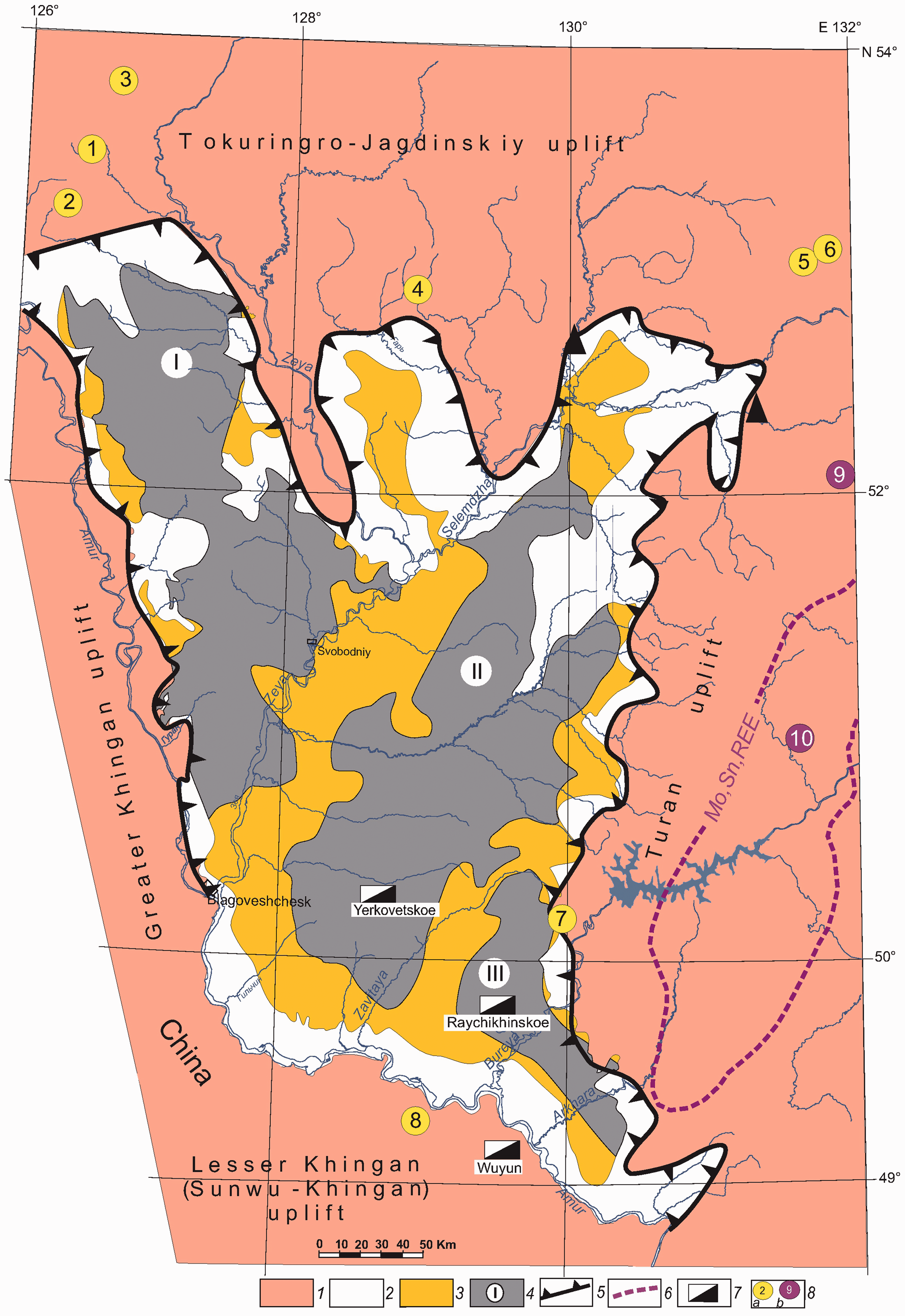
Legend: 1—mountain-folded basin frame; 2—denudation plain; 3—alluvial plain; 4—coal-bearing areas (I: Pikansko-Sergeyevskaya, II: Selemdzhinsko-Yerkovetskaya, and III: Zavitinsko-Arkharinskaya); 5—Zeya–Bureya Basin boundary; 6—Melgin-Nimansky metallogenic belt; 7—brown coal deposits; 8a—gold deposits (1: Pioneer, 2: Pokrovskoe, 3: Borgulikanskoe, 4: Inkanskoe, 5: Malomyr, 6: Quartsevoe, 7: Prognoznoe, and 8: Wulaga); 8b—rare metal deposits; 9—Metrekskoe; and 10—Chergilenskoe.
There is some evidence to suggest that rock complexes of the northern part of the Bureya-Jiamusi super terrain, usually allocated as the Turan uplift (Krasniy, 1999), were major sources of metal-containing minerals for the above-mentioned coal deposits.
The main part of the uplift is occupied by the Melgin-Nimansky metallogenic belt, which includes uranium–rare-earth deposits (Chergilenskoe is the most famous), Mo–porphyry deposits (for example, Metrekskoe deposit with W, Sn, and Bi), and several rare-earth ore occurrences (Khanchuk, 2006). The river channels, mainly of latitudinal direction, drain the Turan uplift, almost all the way from the northern margin of Basin and contain sands with mineral dispersion halos of monazite (Ce, La, and Th) and fergusonite (Nb and Y), with a permanent presence Hg, Sn, and W minerals, and rare metals—Cu, Pb, Zn, Mo, and Bi in the rock samples from the rivers.
In addition to these mineral associations, river sands in the junction zone of the Turan uplift and the Zeya–Bureya Basin are enriched with noble metals, primarily Au. Gold ore deposits (Kvartsevoe, Malomyrskoe, and Prognoznoe; Eirish, 2002) and a number of Au placers are found here.
The discovered Au placers on the right bank of the Amur River (China), within the coal-bearing Basin formed at the same time as the Zeya–Bureya Basin, are associated with a number of well-known gold ore deposits of the Sunwu–Khingan uplift adjacent to the Turan uplift. The most famous of them, the epithermal gold ore deposit, Wulaga, is localized, as well as the Prognoznoe deposit, in the subvolcanic granodiorite porphyry rocks of the early Cretaceous. In addition to Au, rocks with ore mineralization contain higher concentrations of REE + Y in total from 103.30 to 117.40 mg/kg. The studied samples are enriched more with light REE compared with heavy ones. The nature of the distribution of REEs suggests that amphiboles from the host rocks of the Lesser Khingan (Sunwu-Khingan) uplift may be their source (Wang et al., 2016).
The ratio of the contents of light REE over the heavy is used for genetic interpretations of the sources of terrigenous material transported to the area of coal-forming peats in the Paleocene (Sorokin et al., 2019). For coal and the host rocks of the Yerkovetskoe deposit, it is in the range of 7.64–9.9, which is determined by the scale of development of effusive and granitoids in the sources of demolition. The ratios of the normalized contents of individual rare-earth elements were also calculated. In the lower part of the coal seam in the Yerkovetskoe deposit, there are higher (≥16) ratios of the LaN/YbN ratio, low contents of yttrium (not more than 7.0) and other trace elements, which indicates the possible predominance of acidic igneous rocks in the Turan and Lesser Khingan uplifts in the Cenozoic in the areas of nutrition paleo coal-forming peats. This led to the dominance of mineral substance associated with the weathering of granites and quartz veins with gold mineralization. At the final stage of the formation of paleo peats, the composition of the introduced microcomponents was dominated by mineral substance from volcanic rocks and, to a lesser extent, granitoids. The ratio of the normalized contents of LaN/YbN is on average no more than 12.0. These figures confirm the presence of an effusive source of trace elements deposited in coal-bearing seams at this time.
In the Yerkovetskoe deposit, the average total concentration of REEs in the brown coal beds and host rocks is about 150.00 mg/kg, and the average content for the coal beds is about 180.00 mg/kg. In the bottom part of the coal bed, lower REE concentration is noted, and the coal between the tuff tonsteins inside the coal strata and in the roof host rocks, there is a maximum concentration of REEs (up to 250.00 mg/kg). The Y content in the coal beds varies unevenly (from 7.00 to 70.00 mg/kg), but with a general tendency for growth in the upper part of the beds. In the coals of the Raichikhinskoe deposit adjacent to the Yerkovetskoe deposit, the average total concentration of REEs also increases from the floor of the bed to the roof, but does not reach as high as in the coals of the Yerkovetskoe deposit. The content of REE + Y averages no more than 70.00 mg/kg.
Higher concentrations of trace elements are usually observed in the brown coals in the patterns of tuff, kaolinite, montmorillonite, illite, smectite, as well as in accessory minerals—chlorite, carbonates, and feldspars. Among them, there are higher concentrations of Li, Cs, and Ga in the minerals of the lower parts of the coal beds, and Y and Zr—mainly in the upper ones.
The Wuyun (Paleocene) brown coals located on the right bank of Amur river (in China) is comparable to that of the Yerkovetskoe and Raichikhinskoe deposits and contain elevated REEs concentrations, as well as Y and Sc, mainly in phosphates and organic matter.
In the present investigation, distribution of selected elements in CCP from brown coal of Yerkovetskoe deposit is undertaken, and their geochemistry is discussed especially in light of potential possibility to determine an economically feasible source for the extraction of REEs.
Materials and methods
For more than two decades, the Amur Scientific Center of the Far East Branch of the Russian Academy of Sciences has continued to systematize the metal concentrations within Zeya–Bureya Basin in the Amur Region and to develop the technology of separate extraction and research of CCP (slag, ash, and sludge). At present, such a complex study of metal-bearing coals in the Far East is being conducted for the first time. We believe that it is necessary to conduct scientific research not only for the development of technologies for the extraction of chemical elements from CCP, but also experimenting with technologies for the combustion of coal on an industrial scale.
Coals are “difficult” objects for analyzing the composition of chemical elements, the results of which are significantly influenced by the physical and chemical characteristics of coal samples and the chosen method of chemical sample preparation. Therefore, in this work, due attention is paid to the method of improving the accuracy of analytical determinations made at the analytical laboratory of the Far Eastern Geological Institute (Vladivostok).
Experimental works on separate extraction of brown CCP
The discovering of small concentrations of chemical elements in the halo of dispersion from dust–gas flow (flue gas) in soils around industrial funnels led many researchers to the assumption that metals from burning coal were carried away into the atmosphere within this flow in the form of minerals and oxides, volatile chemical compounds, or in the form of nanoparticles. Recently, it was experimentally proved that during the combustion of coal, up to 70% of Au is carried away in the dust and gas flow (Kuz’minykh and Sorokin, 2004). To study the process of distribution of Au and other metals in coal and their transfer with CCP (slag, fly ash, and flue gas), an experimental technological complex was created in 2017 at the Amur Scientific Center (Sorokin and Ageev, 2018). The complex, called “Amur” (Figure 2), provides the possibility of separate collection of all types of CCP, including furnace slag, fly ash, and a residue after refining flue gas in wet scrubber with water.
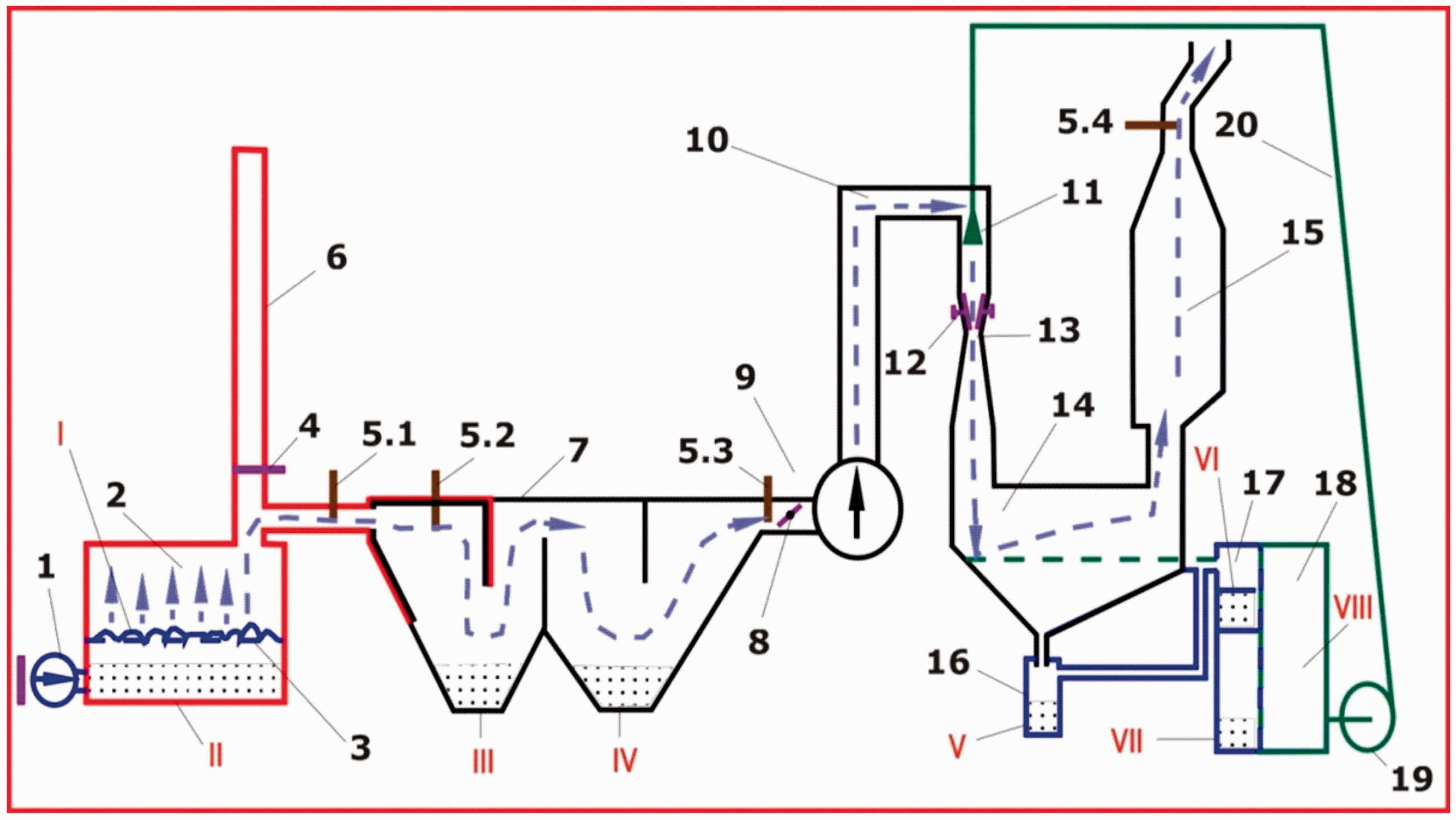
Components of the technological complex: 1—blower fan; 2—combustion chamber; 3—grate; 4—damper; 5.1–5.4—thermocouples; 6—air intake; 7—ash collector; 8—regulating damper; 9—exhauster; 10—chimney; 11—sprinkler system; 12—air duct reducer unit; 13—adjustable neck; 14—scrubber; 15—centrifugal moisture column; 16—precipitation can; 17—precipitation column; 18—tank for cleaning water; 19—water pump; and 20—the water pipeline.
Combustion products of the bulk sample of brown coal (I) from Yerkovetskoe deposit were concentrated in different units of the complex (Figure 1)—furnace slag in the form of a non-combustible residue with heavy mineral impurities (II), heavy (III) and light (IV) fraction of ash, sludge from precipitation can (V), sediment from a scrubber filter (VI), slurry (residue) from a sprayed cleaning water (VII), and purified cleaning water sediment (VIII).
Coals and their combustion products elemental composition determination using plasma spectrometry methods (ICP-AES and ICP-MS)
Inductively coupled plasma atomic emission spectroscopy (ICP-AES) and inductively coupled plasma mass spectrometry (ICP-MS) methods were used in this research to determine the elemental composition of coals and their combustion products after transferring the initial samples to the solution. The most acceptable method of sample preparation for inductively coupled plasma (ICP) analysis in this case, to measure the maximum possible number of elements, is preliminary ashing of coal samples and subsequent decomposition of obtained ash by fusing with lithium borates (GOST R 54237-2010 (ASTM D 6349-2008), 2012; Oleinikova et al., 2015). However, this method of sample preparation excludes the correct determination of Ge, Se, Te, As, and Sb, forming compounds that become volatile.
Chemical sample preparation
Ashing of coal samples was performed at a temperature of 500 ± 10°С. Samples of coal ash weighing 0.05 g were melted in platinum crucibles with lithium metaborate in the ratio 1:3 at a temperature of 1050°С for 15 min. Then melts were dissolved in 5% nitric acid, evaporated to a minimum volume, treated with Hf, followed by heating to remove Si and B in the form of volatile fluoride compounds to wet salts. Further processing was carried out by evaporation with concentrated HNO3 to wet salts and subsequent addition of 10 ml of 20% HNO3 with heating until the salts dissolved. The resulting solutions were transferred to 50 cm3 volumetric polypropylene flasks and brought to the mark with deionized water (type I) with the addition of traces of Hf to prevent possible hydrolysis and polymerization of highly charged ions, Zr, Nb, Hf, Ta, and Mo and W. The final solutions are stable in time.
The chosen method is most appropriate in the case, since removing the silicate matrix from the sample reduces the salt load on the ICP instrument and provides the lowest trace elements detection limits. All reagents used were of high purity quality. Laboratory-grade nitric acid was distilled at the MileStone distillation unit without boiling, hydrofluoric acid and lithium metaborate were suprapure grade (Merck). Deionized water with a specific resistance of 18.2 MΩ/cm was obtained using the MiliQ water purification system (Millipore).
The instrumental parameters
The major elements’ composition (Al, Ti, Fe, Ca, Mg, K, Na, and P in terms of oxides) was determined by ICP-AES in the mode of radial plasma observation on the iCAP 7600 Duo spectrometer (Thermo Scientific). The operating parameters of the device are listed in Table 1; 10 mg/kg Cd solution was used as an internal standard.
Instrumental analysis conditions for the ICP-AES iCAP 7600 Duo.

Analytical wavelengths of the determined elements.

A solution of lithium metaborate, prepared by melting 1.5 g of reagent in a platinum crucible at a temperature of 1050°С for 15 min, was used to prepare the calibration solutions from ICP multi-element standard solution IV (Al, Ca, Fe, Mn, Mg, K, and Na), Merck; Phosphorus (P), High-Purity Standards; Titanium (Ti), High-Purity Standards. The obtained melts were dissolved in 4% nitric acid by heating, transferred to a polypropylene volumetric flask with a capacity of 500 cm3 and brought to the mark with 4% nitric acid. Use of lithium metaborate prepared in a similar manner for dilution of basic standard solutions provided a matrix composition of calibration standards similar to the analyzed samples. The concentrations of the components to be determined in the calibration solutions ranged from 0.01 to 100 mg/kg. Characteristics of the analytical wavelengths of the determined elements are presented in Table 2.
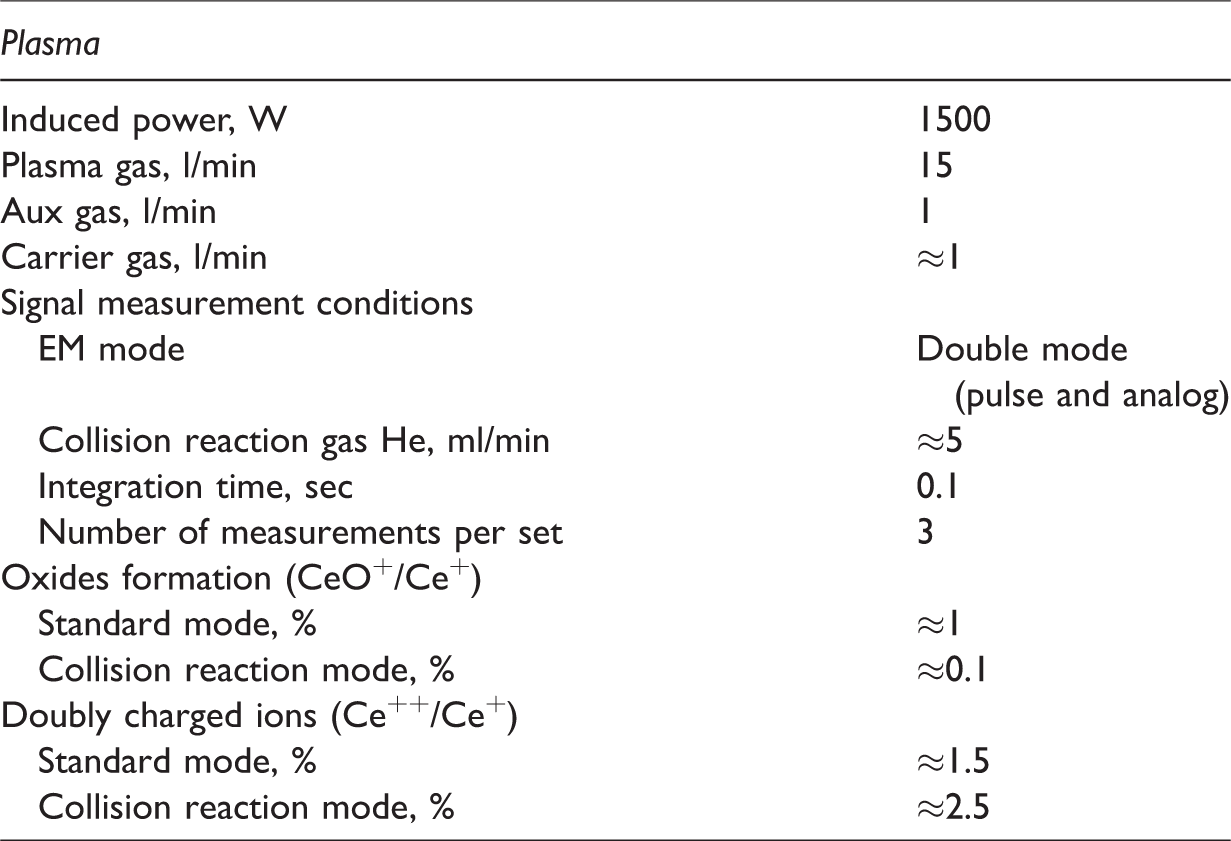
Sample solutions were diluted with 2% HNO3 five-fold immediately before measurements by ICP-MS to reduce the salt background; 5 cm3 of the obtained solutions were introduced by indium as internal standard at a final concentration of 10 mg/g, and analysis was performed on Agilent 7500c quadrupole ICP-MS (Agilent Technologies). The operating parameters of the device are shown in Table 3.
Operating parameters of Agilent 7500c ICP-MS.
A series of calibration solutions were prepared by the gravimetric method from multi-element standard solutions. Multi-element Calibration Standard 1 (No. 8500–6944): Ce, Dy, Er, Eu, Gd, Ho, La, Lu, Nd, Pr, Sc, Sm, Tb, Th, Tm, Y and Yb; Multi-element Calibration Standard 2A (No. 8500–6940): As, Ba, Be, Cd, Co, Cr, Cs, Cu, Ga, Li, Ni, Pb, Rb, Sr, Tl, U, V, and Zn; Multi-element Calibration Standard 3 (No. 8500–6948): Hf; and Multi-element Calibration Standard 4 (No. 8500–6942): Mo, Nb, Ta, W, and Zr (Agilent Technologies) were used. The concentrations of calibration solutions ranged from 0.1 to 1000 mg/g.
To conduct research with experimental technological complex, 221.0 kg of coal from the Yerkovetskoe deposit with ash content of 9.41 wt. % was burned. The combustion temperature in the burner chamber was 600–1000°С, the temperature in the afterburner chamber—500–750°С, at the outlet of the ash collector—180–240°С. The duration of the combustion process is 16.5 h with loading capacity for coal 13.40 kg/h. Combustion process produced 23.56 kg of CCP: bottom slag 20.00 kg (84.89%), heavy fraction of fly ash 2.40 kg (10.19%), light fraction of fly ash 0.90 kg (3.82%), sludge (combined sludge from precipitation can (V), sediment from a scrubber (VI), and slurry (residue) from a sprayed cleaning water) (VII)—0.26 kg (1.10%).
Slag, after grinding to a particle size of 1.00 mm, the ash was passed through a sieve with a cell of 0.04 mm. Fractions greater than 0.04 mm were studied under a binocular. Both size fractions of slag and ash were then divided into heavy and light fractions. Grains of native gold, magnetite balls, hematite, martite, ilmenite, zircon, native copper, and lead were found in the heavy nonmagnetic fraction of slag. Of nonmetallic minerals, feldspar, quartz, amphibole, and corundum predominate. Light non-magnetic fraction of slag and ash contained quartz as well as silicates of silicon, aluminum, iron, and calcium. Magnetic and vitreous microtectites, zircon and quartz and single particles of pyrite, galena, native copper, and lead were found in the heavy fraction of fly ash.
The slurry after precipitation was washed in bromoform, divided into magnetic, electromagnetic, non-magnetic, heavy, and non-magnetic light into fractions. Measurements were carried out separately for the heavy and light fraction of fly ash, as well as for large (+0.04 mm) and small (−0.04 mm) particles of the main combustion products (slag and ash).
Results and discussion
Distribution of trace elements in CCP
Table 4 presents the results of ICP-AES and ICP-MS determination of trace elements content in slag, heavy and light fractions of fly ash, and the combined sample of precipitates from can and filters after the scrubber. To control of the degree purification of flue gas in the scrubber and determination of metal concentrations in the sediment from the cleaning water were conducted. Table 4 shows the content of metals in the initial sample of coal and in combustion products.
Content of selected minor and trace elements in CCP, obtained at the experimental technological complex, mg/kg.
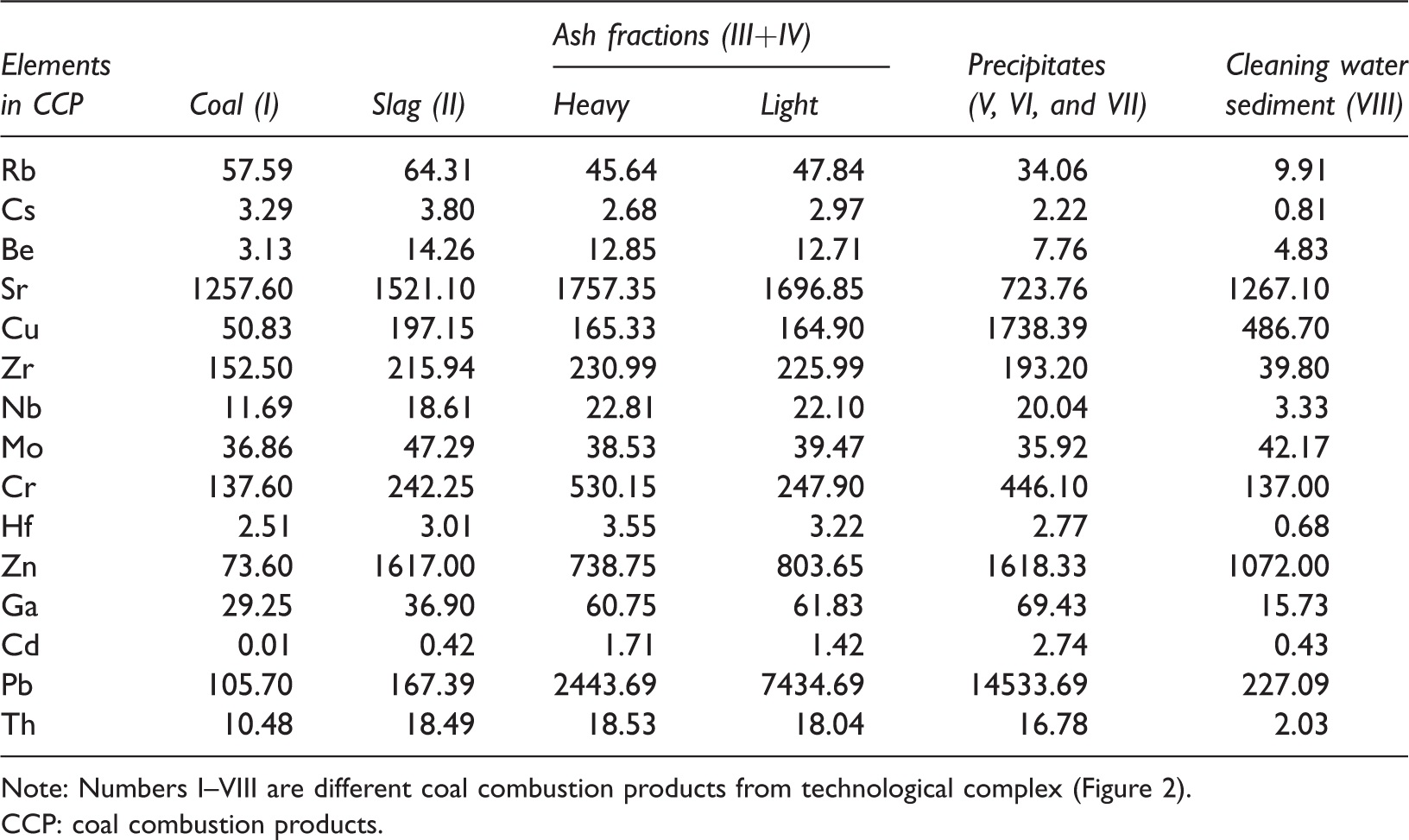
Note: Numbers I–VIII are different coal combustion products from technological complex (Figure 2).
CCP: coal combustion products.
The content of minerals and concentration of minor and trace elements in coal to a greater extent corresponds to the geochemical environment of sedimentation in peat and the physicochemical conditions of brown coal diagenesis (Orem and Finkelman, 2003; Singh et al., 2016). Rb and Cs are mainly associated with clay minerals—kaolinite and montmorillonite, which are autogenic, and other most common minerals in coal. In the process of burning coal, these minerals are sintered and remain in the slag (Table 4). Be and Sr are associated with organic matter and carbonates. When coal is burned, these compounds are transferred and accumulated by a dust–gas stream in all CCP and are partially transferred into environment. In the slurry from sprayed cleaning water, these elements are almost not fixed.
Zr, Nb, Cr, and Hf are incorporated into coal as a part of chemically insoluble minerals—such as rutile, zircon, and rare-earth phosphates. Under pressurized air and high temperature, these minerals, released from the coal matrix, are removed from the combustion zone and are evenly distributed between different combustion products—slag, ash, and dust and gas flow. Fe-Mn minerals—pyroxene, amphibole, and olivine, are very rare in coal; feldspar is unusual. These minerals are unstable under conditions of low pH values (about 2–4) in the peat and, apparently, were dissolved in the early stages of coalification.
Table 4 draws attention to the accumulation of chalcophile elements Cu, Zn, and Pb in the final products of combustion. These elements in coal were mainly contained in sulfides, which did not melt at the specified temperatures of combustion, but in the presence of oxygen, they formed oxides, and then in the presence of sulfur in the coal, they formed sulfates. Oxides and sulfates at high temperatures have a pulverized state and are carried with dust–gas stream components into the atmosphere. But in the experiment, these minerals under the influence of cleaning water spray in the scrubber have remained in the sludge and on filters. The content of trace elements in the slag and ash is approximately the same, but higher than the Clarke of concentrations in the continental crust (Rudnick and Gao, 2004).
A comparative analysis of data on distribution of minor and trace elements in slag and ash of Russian brown coal (Yerkovetskoe deposit) with the same combustion products of five Indian power plants (Bhangare et al., 2011) was conducted. It is established that a concentration of Pb, Cr, Co, and Zn in the ash and a concentration of V, Co, Cu, Zn, Mo, Cd, and Pb in the slag of the Yerkovetskoe deposit are higher than in the ash and slag from brown coal from India. There were elements in coal from India that are completely carried away by the dust and gas flow into the atmosphere. The content of most other trace elements in fly ash from coals of the Yerkovetskoe deposit is comparable to the content in fly ash from coals of India deposits (Table 5).
Comparison of the contents of the minor and trace elements in the slag and fly ash from Indian deposits and Yerkovetskoe deposits in Russia, mg/kg.
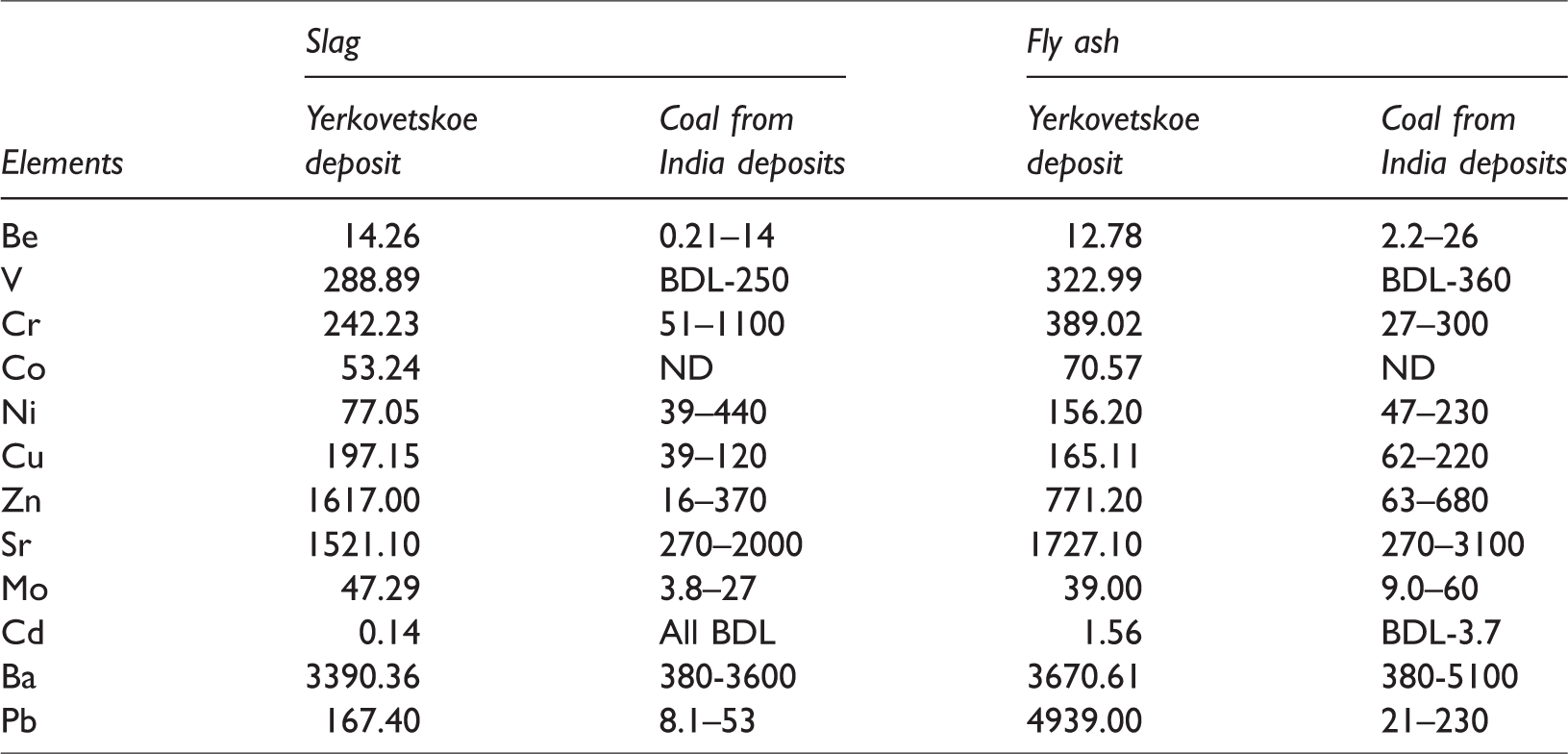
It should be noted the predominance of chalcophile elements (Co, Cu, Zn, Mo, Cd, and Pb) in the coals of the Amur region. Obviously, this is due to the widespread sulfide mineralization in the rocks of the eastern flank of the Zeya–Bureya Basin (Eirish, 2002). L.V. Eirish identified the Turan gold-bearing zone in this area, including the Buyankovskoye, Koykoye, and Prognoznoe hydrothermal quartz ore veins and deposits. The first two occurrences contain chalcopyrite, pyrite, bismuthine, galena, and sphalerite. The Prognoznoe deposit has been studied in more detail and acanthite, opal, barite, and amphibole are also distinguished among the main minerals. Accessory minerals are represented by ilmenite, epidote, sphalerite, arsenopyrite, chalcopyrite, scheelite, and rutile. Slag from local coal has significant, sometimes two orders of magnitude higher, content of Cu, Zn, Mo, Cd, and Pb. Slag from Yerkovetskoe brown coal has higher concentration of selected trace elements than fly ash.
Distribution of Sc, Y, and REEs in CCP
Content of REE + Sc + Y in the combustion products of coal from the Yerkovetskoe deposit (Table 6) shows that the REEs are mainly distributed evenly between the slag, fly ash, and scrubber sludge. This is confirmed by the close LaN/YbN, LaN/SmN, and GdN/YbN ratios, normalized to chondrite according data of Evensen et al. (1978), in coal and in separately obtained combustion products. The ratio of LaN/YbN in coal is 7.94, in the slag—6.91–7.59, ash—7.38–7.66, and sludge—8.52–10.87; LaN/SmN—3.40, 3.30–3.82, 3.58–4.52, and 3.41–4.27, respectively; and GdN/YbN—1.57, 1.50–1.64, 1.62–1.71, and 1.60–1.87, respectively.
Distribution of REEs, Y, and Sc in coal combustion products, obtained at the experimental technological complex, mg/kg.
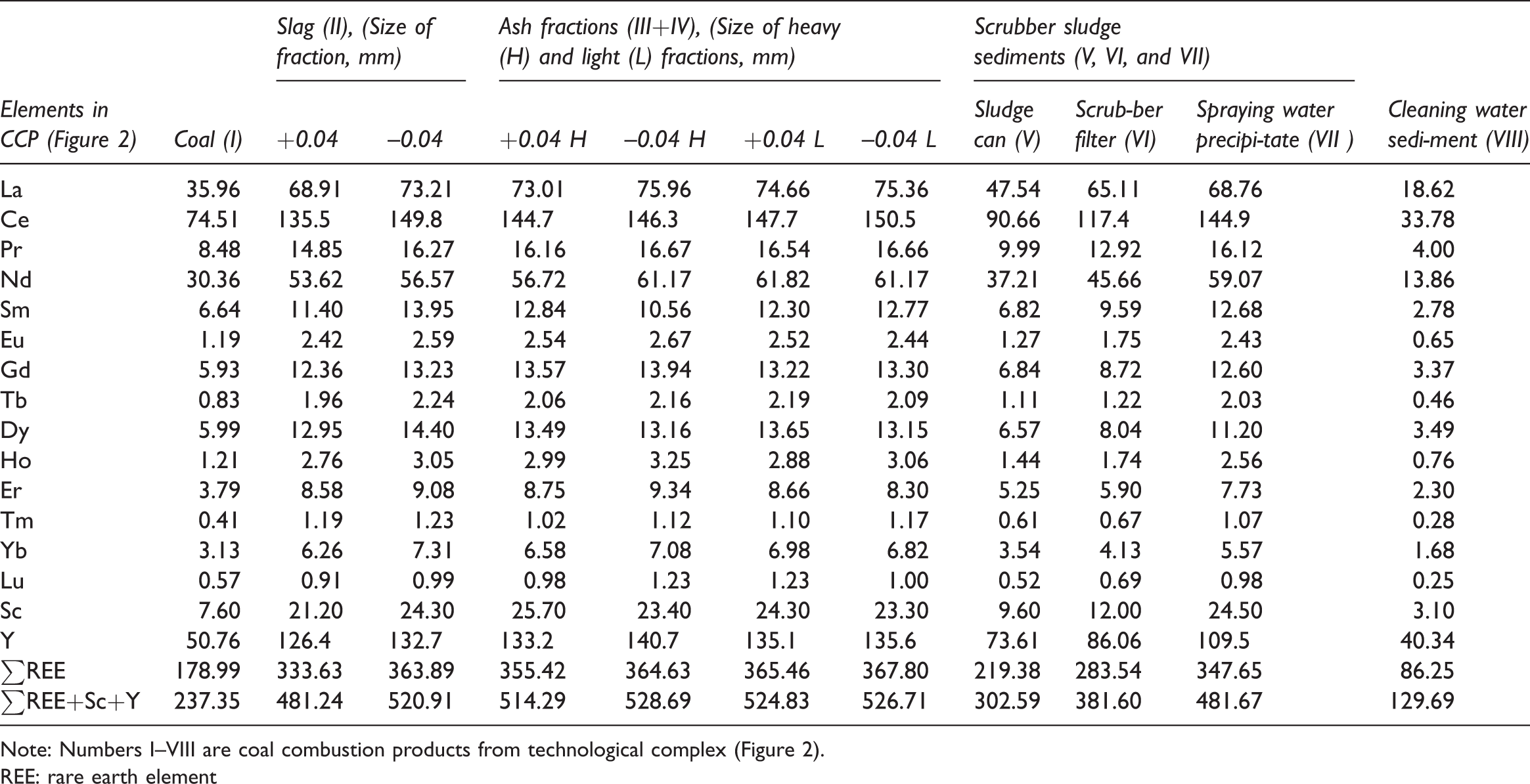
Note: Numbers I–VIII are coal combustion products from technological complex (Figure 2).
REE: rare earth element
Data on the content of REEs in the sediment from cleaning water show that the cleaning of the dust–gas flow in the experimental complex occurs with a perfect efficiency. The use of a scrubber solves the problem of cleaning the dust and gas flow from harmful and toxic elements of coal smoke, which should be part of the technological chain of combustion of coal in industrial combustors.
Chondrite-normalized REE concentrations (Evensen et al., 1978) in CCP from experimental complex (Figure 3) clearly shows the same fractionation pattern of these elements in the slag, ash, and sludge. The Eu minimum is preserved in the CCP, which was determined by the authors for natural coals of the Yerkovetskoe deposit.
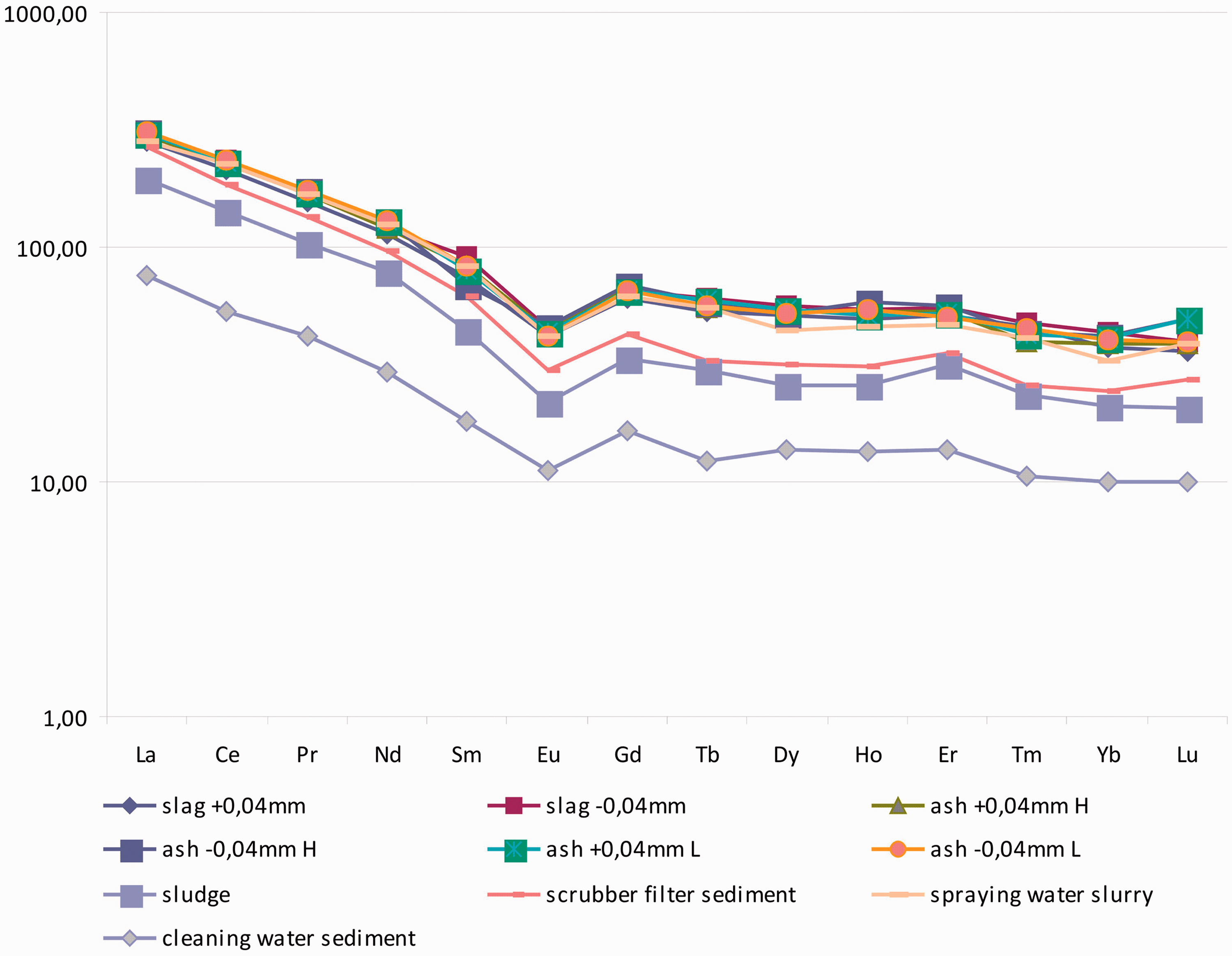
Chondrite-normalized REE concentrations in CCP from experimental complex.
On the basis of the data obtained, it is difficult to designate one of the three CCP, promising for subsequent feasible work on the extraction of REEs. Further research will focus on the composition of fly ash and scrubbing sludge (CCP V, VI, and VII). Considering the fact that the physical state of the slag after combustion of lower rank brown coal 2B differs from fly ash only by a higher content of quartz, the concentrate consisting of slag purified from quartz, ash, and sludge will also be considered as a semi-finished product for REE + Y + Sc.
Conclusions
In brown coal beds of the Yerkovetskoe deposit, the most valuable trace elements (Sr, Be, Zr, Rb, and Nb) are unevenly distributed, and rare-earth elements have elevated concentrations in the roof rocks and in the coal layer between tuff tonsteins (up to 250 mg/kg total). The metallogenic structures of the Turan and Lesser Khingan uplifts, composed mainly of volcanics and intrusive rocks of different ages, served as the main source of REE and trace elements transported into the Cenozoic coal-forming peatlands. The release of trace and rare-earth elements came from the weathering crusts of the chemical type by river tributaries flows, which conducted drainage of eroding areas at the uplifts. The experimental and technological complex “Amur” provided the possibility to separate production of all types of CCP, including furnace bottom slag, heavy and light fractions of fly ash, sludge, and a residue after refining flue gas in wet scrubber with water (sediment and slurry). The coals of the Erkovetskoe deposit and its combustion products were studied by plasma spectrometry (ICP-AES and ICP-MS), which ensured reliable analysis results from the highest to trace concentrations. At the same time, chemical sample preparation was significantly improved, which included the ashing of samples of coal at a temperature of 500 ± 10°С, fusion in crucibles, dissolution in nitric acid, evaporation, and treatment with acid. Studies of concentrations of rare earth and selected trace elements in the combustion products of coal from the Yerkovetskoe deposit indicate that they are almost evenly distributed in furnace slag, fly ash, and sludge. This changes the common notion of non-volatility of the above elements (Ratafia-Brown, 1994), which considers that when burning coal, they are stored in the solid phase of the combustion products. The authors consider the REE + Sc + Y as economically valuable components of the CCP from a studied deposit. At the same time, toxic elements (Cu, Pb, Zn, and others), that are common in the combustion products of thermal and power plants of Russia and foreign countries, were found in these coals. The use of a wet scrubber as a part of the technological complex for the purification of coal smoke can solve the problem of cleaning the dust–gas flow from harmful and toxic elements and become part of the technological chain of coal combustion in industrial boilers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Russian Academy of Science (RAS) (0280–2020-0010) and the Integrated Program of Scientific Research of Far Eastern Branch of RAS (18–2-019).
