Abstract
In this study, to ascertain the carbon and hydrogen isotope fractionation of oil cracking gas (secondary gas) in hydrothermal conditions, non-isothermal pyrolysis of oil with and without water was carried out by a gold-tube system. By determination of the yields of individual gas products, it is found that the presence of water enhanced the yields of hydrocarbon gases. However, kinetic calculations indicate that Ea for the generation of methane and C2–5 in pyrolysis in hydrothermal conditions are essentially identical with those in anhydrous pyrolysis. The yields of carbon dioxide (CO2) and alkene gases in pyrolysis in hydrothermal conditions are evidently higher than those in anhydrous pyrolysis. It is reasonable that water–hydrocarbon reactions occurred and contributed to the generation of secondary gas in hydrothermal conditions. Meanwhile, the presence of water resulted in a slight depletion of 13C for methane and an evident depletion of 13C for CO2. Thermodynamic calculations suggest that water–hydrocarbon reactions in non-isothermal pyrolysis are dominated by free radical mechanism rather than ionic mechanism. Moreover, δ2H values of methane are apparently different in pyrolysis involving water with different δ2H. This result demonstrates that water provided hydrogen for hydrocarbon gas generation. Finally, we established mathematical models based on isotope fractionation to quantitatively determine the contribution of water–hydrocarbon reactions for gas generation in both experimental and geological conditions.
Keywords
Introduction
Water was suggested to affect and even participate in the petroleum generation from organic matters (Helgeson et al., 1993; Lewan et al., 1979; Lewan, 1997; Seewald, 2003, Seewald et al., 2000). Till now, numerous hydrous or hydrothermal experiments have been conducted to understand the effects of water on oil and gas generation (Behar et al., 2003; Gao et al., 2014; He et al., 2018a; Lewan et al., 1979; Lewan, 1997; Lewan and Roy, 2011; Pan et al., 2009; Wang et al., 2017). These works observed that water can provide hydrogen and oxygen for hydrocarbon generation, and alter the features of individual products as well as acting as a medium for petroleum expulsion from source rocks (Hoering, 1984; Lewan, 1997; Schimmelmann et al., 2001). Essentially, the hydrogenation by water promoted the yields of particular products including non-hydrocarbon gas, nitrogen-sulfur-oxygen bearing compounds (NSOs) compounds and specific biomarkers by inhibiting the cross-linking reactions (Behar et al., 2003; Koopmans et al., 1998; Lewan, 1997; Lewan and Roy, 2011). On consideration of the additional hydrogen source from water, Seewald (2003) implied a greater gas potential in sedimentary basins. However, most hydrous pyrolysis of organic matters did not generate higher yields of hydrocarbon gases compared with anhydrous pyrolysis (Cai et al., 2017; Gao et al., 2014; Lewan et al., 1997). Although water–hydrocarbon reactions were observed at elevated temperature (Helgeson et al., 1993; Leif and Simoneit, 2000; Seewald, 2001), the effect of water on the generation of oil cracking gas (secondary gas) remains controversial (He et al., 2011, 2018a; Hesp and Rigby, 1973; Shuai et al., 2012).
Previous works have observed D or 2H transfer from deuterated water (D2O or 2H2O) to organic matters and oils by hydrous pyrolysis (Hoering, 1984; Lewan, 1997; Schimmelmann et al., 2001). To interpret the incorporation of water-derived hydrogen, two possible reaction pathways via radical and ionic mechanisms were both proposed by former studies (Hoering, 1984; Leif and Simoneit, 2000; Lewan, 1997; Seewald, 2003). Lewan (1997) has suggested that water–organic molecules interactions should proceed via free radical reactions rather than the ionic mechanism. In this opinion, additional hydrogen, which was produced by reactions between water and carbonyl groups, can quench the free-radical sites and subsequently inhibit cross-linking process. Besides, hydrogenation by water can also occur according to direct reaction between water and hydrocarbon free radicals (He et al., 2018a; Lewan, 1997). As alternative pathways, ionic reaction involving transient alkenes and H+ derived from dissociation of water were demonstrated to be critical for the incorporation of deuterium (2H) from 2H2O to hydrocarbons (Leif and Simoneit, 2000; Schimmelmann et al., 2001). In addition, there is general agreement that the direct hydration of alkenes by water can occur and result in the generation of CO2 and H2 via alcohols, ketones and carboxylic acids at elevated temperature (Leif and Simoneit, 2000; Seewald, 2001, 2003). On this basis, researchers proposed that organic–inorganic interactions involving water may be responsible for hydrogen isotope rollover of natural gases with extremely high dryness (Gao et al., 2014; Tang and Xia, 2011; Zhang et al., 2018). Meanwhile, pyrolysis experiments found that carbon isotope ratios of methane derived from organic matters or hydrocarbons were somehow different in anhydrous and hydrothermal conditions (He et al., 2011, 2018a; Wang et al., 2008). These facts implied that both carbon and hydrogen isotopic compositions of natural gases are affected by the occurrence of water–organic molecules reactions (He et al., 2018a). Unfortunately, the isotope fractionation of hydrocarbon gases in water–hydrocarbon reactions are little understood, which makes it hard to be identified in the subsurface (He et al., 2018a). Most recently, He et al. (2018a) and Zhang et al. (2018) have established a mathematical model to address the hydrogen isotopic ratios of methane derived from water–hydrocarbon reactions by thermodynamic calculations. This makes it possible to determine the contribution of hydrogenation by water for gas generation based on hydrogen isotope of methane.
Gold tube, which has excellent flexibility and chemical inertness, is usually considered as an ideal material for hydrothermal pyrolysis (Seewald, 2001). In this study, non-isothermal pyrolysis of a crude oil in anhydrous and hydrothermal conditions with deionized or sea water were conducted, respectively. Combined with experimental results and kinetic calculations, the effects of water on the generation and isotopic compositions of oil cracking gas (secondary gas) were understood. By associating thermodynamic and kinetic calculations, the C and H isotope fractionation of secondary gas in anhydrous and hydrothermal conditions was addressed. On this basis, we tried to establish mathematical models to quantitatively determine the contribution of water–hydrocarbon reactions for gas generation in the subsurface.
Experimental
Sample and reagents
The crude oil was produced from a carbonate reservoir with depth of 5805 m in well Yingmaili 201 (YM201), which is of Ordovician age and located in Tabei uplift in the Tarim Basin, NW China. The oil has an American Petroleum Institute gravity of 33.0°, 4.37 wt.% wax and 0.84 wt.% sulfur. The detailed chemical properties of YM201 oil are shown in Table 1. Water used included deionized water with hydrogen isotopic ratios (δ2H) of –51.1‰, sea water with δ2H of –6.8‰ and pure deuterated water 2H2O (2H % = 99.9 mol%).
The essential chemical properties of YM201 oil.
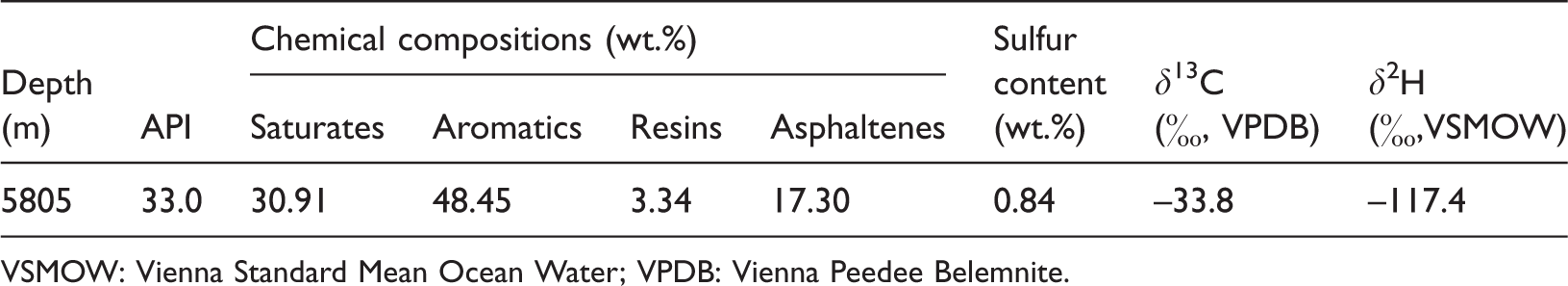
VSMOW: Vienna Standard Mean Ocean Water; VPDB: Vienna Peedee Belemnite.
Gold-tube pyrolysis
The non-isothermal pyrolysis including anhydrous pyrolysis and pyrolysis with water (pyrolysis in hydrothermal conditions) in this study were carried out by a gold-tube pyrolysis system (He et al., 2014). The length, inner diameter and thickness of gold tubes were 50, 4.0 and 0.50 mm, respectively. Firstly, one end of the tube was sealed by an argon arc welder, and certain amounts of samples (i.e. 30 mg oil and 10 mg water) were loaded into the tube. Secondly, the tube with sample loaded was flushed by argon gas for more than 10 minutes. Then, the other end of the tube was sealed in a liquid nitrogen environment (He et al., 2018a). Finally, the tubes with sample loaded were put into autoclaves, where desired liquid pressure and temperature program were conducted. Non-isothermal pyrolysis was conducted at heating rates of 2 and 20°C/h with pressure of 50 MPa (online Supplementary Table S1). When desired reaction temperature reached and the pressure was released, the tubes were taken out from autoclave.
Determination of the yields and isotopic compositions of gas products
The gas products in gold tubes after pyrolysis were collected in a special apparatus, which was connected with a vacuum pump (Zhang et al., 2013). In a typical procedure, the tube was firstly set at a particular position in the apparatus, which was then sealed. After the pressure in the apparatus was pumped to less than 0.1 kPa, the tube was pieced and certain pressure (P) of gas products in the tube can be collected. The molar yields of total gas products were determined by PV= nRT (He et al., 2014).
The compositions of gas products were determined by Agilent 7890 Series Gas Chromatograph (GC), which was custom-configured by Wasson-ECE instrumentation (For Collins, CO). The instrument has two capillary and six packed analytical columns, a flame ionization detector (FID) and two thermal conductivity detectors (TCD). High-pure N2 and He were used as the carrier gas for FID and TCD, respectively. The temperature program of the GC oven was: heating from initial 68°C (held for 7 min) to 90°C (held for 1.5 min) at 10°C/min, then to 175°C (held for 5 min) at 15°C/min. Meanwhile, a standard gas with known gas compositions was applied to calibration in determination of the molar content of individual gas components.
Thermo Delta V Advantage isotope ratio mass spectrometry (IRMS) was applied to analyze the stable carbon isotope of hydrocarbon gases and carbon dioxide. Thermo Mat253 mass spectrometer, which includes Agilent 6890N GC and Mat253 IRMS was used to measure the stable hydrogen isotope ratios of methane. Carbon (δ13C) and hydrogen isotope (δ2H) ratios are referenced to the standards of VPDB (Vienna Peedee Belemnite) and VSMOW (Vienna Standard Mean Ocean Water), respectively, and expressed as follows (Coplen, 2011)
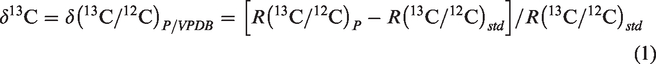
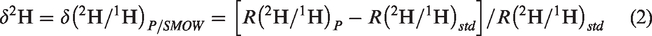
An external methane with known δ13C (–34.18‰) and δ2H (–185.1‰) was used as reference for calibration. During isotope analysis, the reference gas was detected at five sample intervals. The precision is ±0.5‰ for δ13C and ±5.0‰ for δ2H, respectively (He et al., 2018b).
Results
Effects of water on secondary gas generation
The detailed experimental conditions, yields and isotopic ratios of gas products for pyrolysis of YM201 oil with and without water are shown in online Supplementary Table S1. In anhydrous pyrolysis, methane yield gradually increases from about 0.001 to 27.84 mmol/g oil with Easy%Ro from 0.54 to 4.68%. The presence of water resulted in a slight increase of methane yield (Figure 1(a)). For instance, methane yield at temperature of 650°C or Easy%Ro of 4.68% in pyrolysis with water is 29.14 mmol/g oil (652.73 ml/g oil), which is about 1.297 mmol/g oil (29.06 ml/g oil) higher than that in anhydrous pyrolysis (online Supplementary Table S1). Similarly, the yields of heavy hydrocarbon gases (C2–5) in pyrolysis with water are also higher than those in anhydrous pyrolysis (Figure 1(b)). The maximum yield of C2–5 in non-isothermal pyrolysis without and with water is 7.44 and 8.71 mmol/g oil, respectively. The decrease of C2–5 yields at Easy%Ro higher than about 2.45% should be attributed to the cracking of C2–5 during pyrolysis in confined system (Hill et al., 2003). Although previous work have implied greater gas potential on consideration of the hydrogen source of water (Seewald, 2003), the presence of water did not result in intensive increase of the yields of hydrocarbon gases in non-isothermal pyrolysis. This result may be attributed to the relative low amounts of water introduced and the fast heating rate applied in our experiments, where water–hydrocarbon reactions did not play a dominant role for secondary gas generation. Indeed, isothermal pyrolysis of oil/hydrocarbons with higher contents of aqueous solutions at 330–420°C for 48–936 h has observed 0.5–3.0 times increase in hydrocarbon gas yields compared with anhydrous pyrolysis (He et al., 2018a; Shuai et al., 2012).
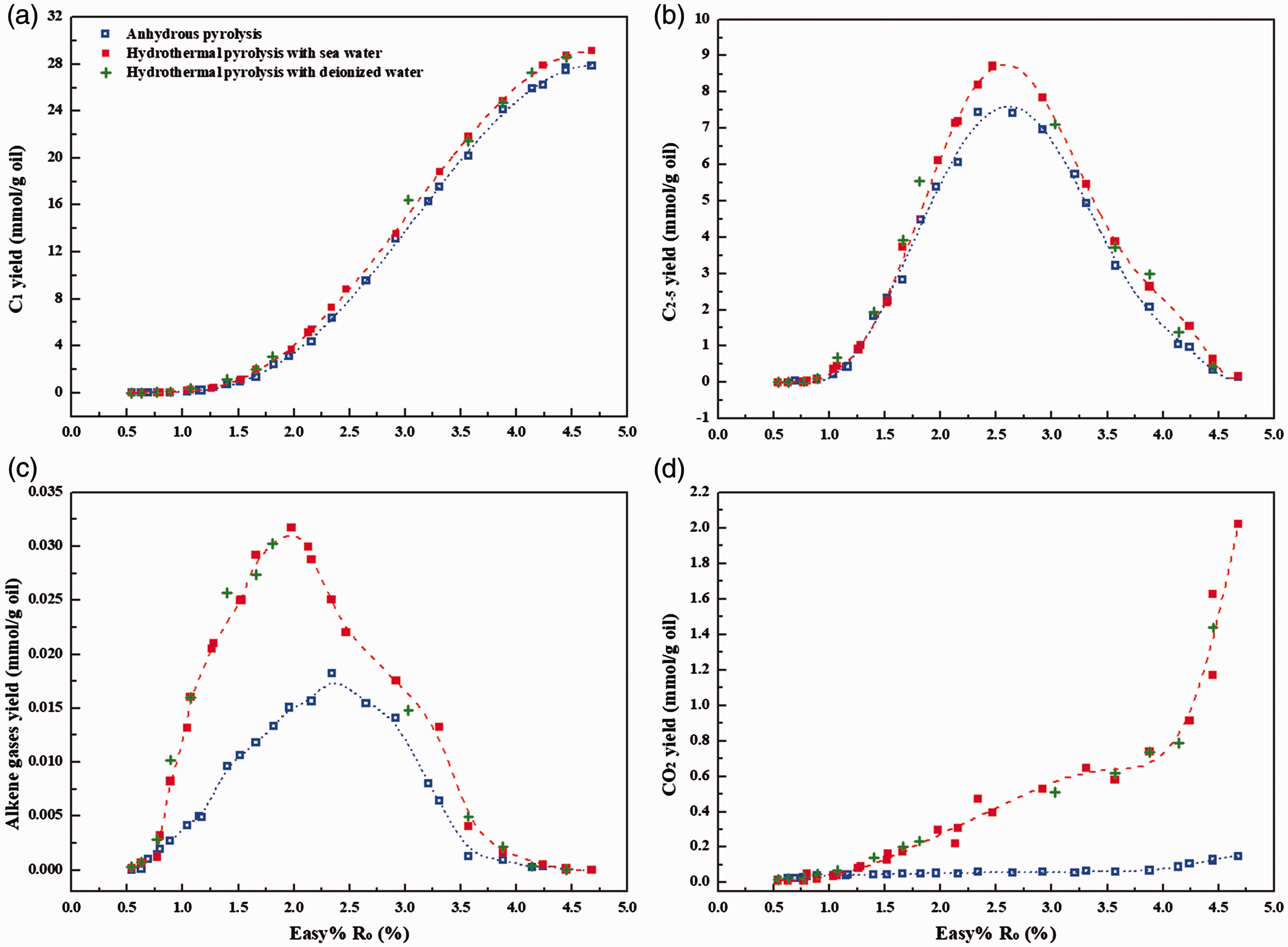
Gas yields from non-isothermal pyrolysis of YM201 oil in anhydrous and hydrothermal conditions at 50 MPa: (a) methane; (b) C2–5 hydrocarbon gases; (c) alkene gases; (d) CO2. The equivalent vitrinite reflectance value (Easy%Ro) for each heated sample was calculated based on the kinetic parameters from Sweeney and Burnham (1990) and their corresponding temperature conditions (online Supplementary Table S1).
The yields of alkene gases generated in pyrolysis with water are obviously higher than those in anhydrous pyrolysis (Figure 1(c)). This is consistent with the experimental results in hydrous pyrolysis involving organic matters or hydrocarbons (He et al., 2018a; Leif and Simoneit, 2000). As it is known, alkenes are usually considered as important intermediates for water–hydrocarbon reactions at elevated temperature (Leif and Simoneit, 2000). The evident increase of alkene yield implies that water may react with oil and affect the generation of gas products in pyrolysis with water (He et al., 2018a; Seewald, 2003). Moreover, the presence of water led to the generation of certain amounts of carbon dioxide (CO2) (Figure 1(d)). For instance, the yield of CO2 at Easy%Ro of 4.68% in pyrolysis with sea water is 2.023 mmol/g oil (45.31 ml/g oil), which is almost 13.7 times of that in anhydrous pyrolysis (0.148 mmol/g oil). In addition, the yields of hydrogen gas (H2) and hydrogen sulfide were more or less enhanced by the presence of water. In anhydrous pyrolysis, H2 may be derived from the cracking of oil or hydrocarbons. The increase in the yields of H2 and CO2 in hydrothermal conditions should be attributed to the water–hydrocarbon reactions (Lewan, 1997; Seewald, 2003). These results demonstrate that water should provide O and H for gas generation in our pyrolysis (He et al., 2018a; Leif and Simoneit, 2000; Seewald, 2003; Wang et al., 2017).
Effects of water on carbon isotopic compositions of hydrocarbon gases
In anhydrous pyrolysis, the δ13C values of methane (δ13C1) decrease from –49.1‰ to –51.1‰ with Easy%Ro from 0.63 to 1.04%, and then increase to –36.3‰ with Easy%Ro increasing to 4.68% (Figure 2(a)). Similar evolution trends can be also observed for δ13C2 (Figure 4(b)) and δ13C3 (Figure 4(c)). The evolution of δ13C of hydrocarbon gases with Easy%Ro in pyrolysis with water is essentially identical with that in anhydrous pyrolysis (Figure 2(a) to (c)). The carbon isotopic compositions of hydrocarbon gases in terms of Easy%Ro do not seem to be apparently affected by the presence of water. However, the δ13C values of CO2 (δ13CCO2) generated from pyrolysis in hydrothermal conditions are much lower than those in anhydrous pyrolysis (Figure 2(d)). It is demonstrated that the generation of CO2 during pyrolysis with water is mainly attributed to water–hydrocarbon reactions but the cracking of organic oxygenated components in oil (He et al., 2018a).
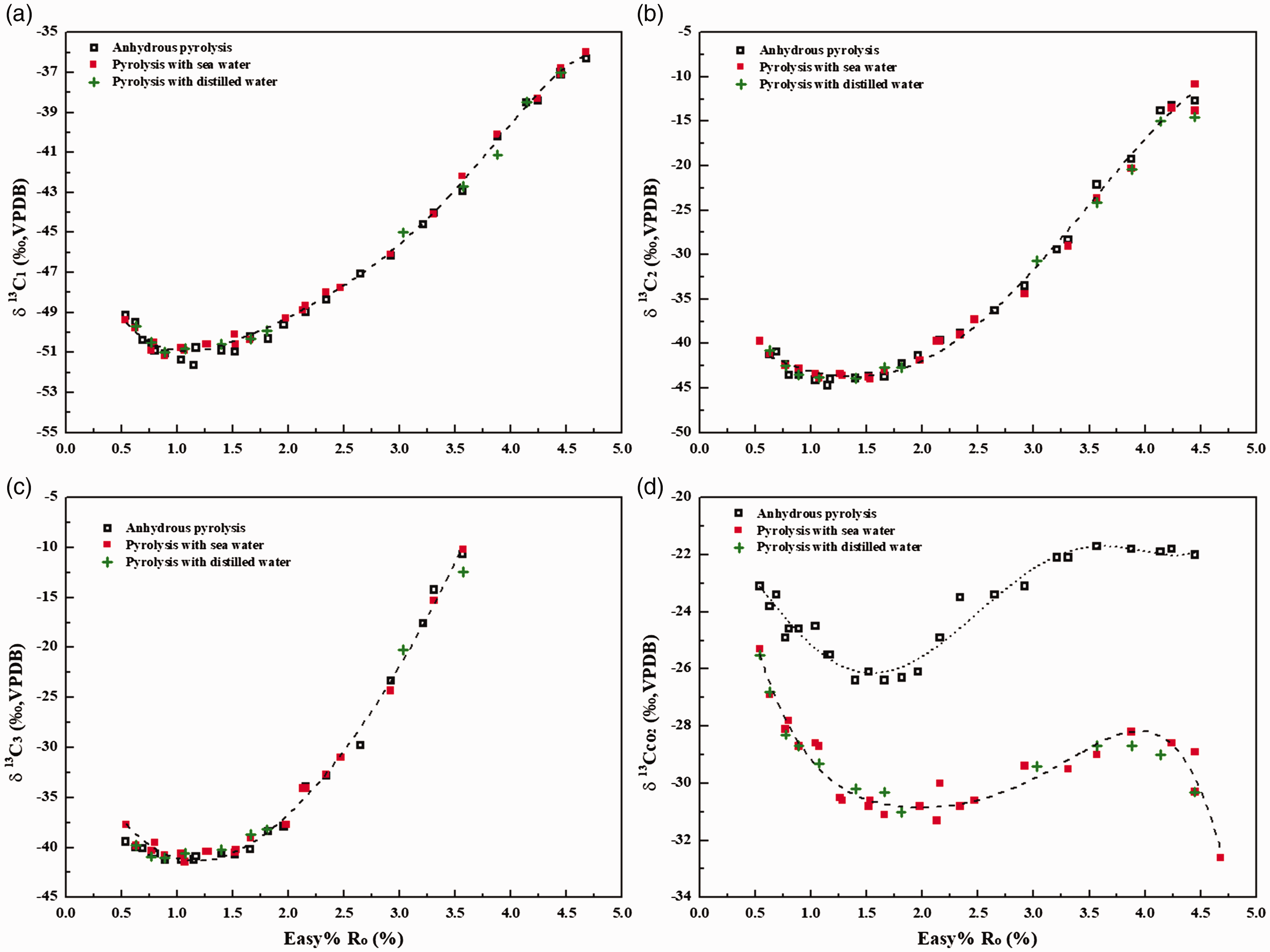
Evolution of δ 13C values of methane (a), ethane (b), propane (c) and carbon dioxide (d) with Easy%Ro during non-isothermal pyrolysis of YM201 oil with and without water.
Effect of water on hydrogen isotopic compositions of methane
There is a positive correlation between the δ2H values of methane (δ2H1) and methane yield (Figure 3(a)). In anhydrous pyrolysis, δ2H1 increases from –320.6‰ to –91.4‰ with methane yield increasing from 0.004 to 27.843 mmol/g oil. 2H isotope of methane generated in pyrolysis with deionized water (δ2H = –51.1‰) is evidently depleted compared with that in anhydrous pyrolysis and pyrolysis with sea water (δ2H = –6.8‰) (Figure 3(b)). These results indicate that the δ2H of methane derived from oil decomposition in hydrothermal conditions was somehow affected by the δ2H values of water (He et al., 2018a). Meanwhile, it can be found that the presence of heavy water (δ2H = 3328‰) resulted in the apparent enrichment of 2H for methane during pyrolysis of oil (Figure 3(b)). This result further demonstrates that water provided 1H or 2H for methane generation via water–hydrocarbon reactions in our non-isothermal pyrolysis.
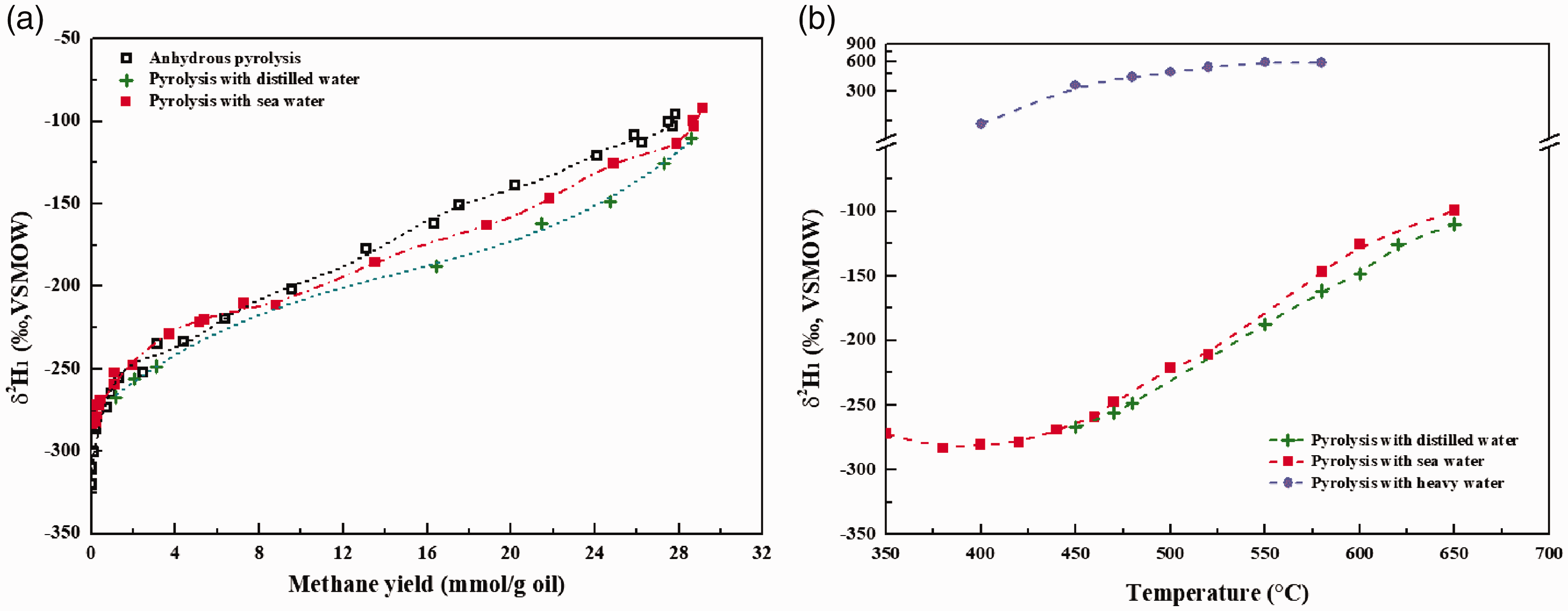
Evolutions of δ2H values of methane with Easy%Ro in anhydrous pyrolysis and pyrolysis with water (a) and with temperature in non-isothermal pyrolysis at 20°C/h with different water (b). Heavy water was prepared by mixing of deionized water (δ2H=–51.1‰) and pure 2H2O (2H%=99.9 mol%) in volume of 1900:1.
Discussion
Kinetics for hydrocarbon gas generation and cracking
To learn about the effects of water on the generation of secondary gas, we addressed the kinetic parameters for methane generation and the generation and cracking of C2–5 hydrocarbon gases. Till now, mathematical models were well established to study the kinetic characteristics for hydrocarbon gas generation from oil cracking (Behar et al., 2008; Dieckmann et al., 1998; Tsuzuki et al., 1999; Vandenbroucke et al., 1999). Generally, gas generation during pyrolysis of oil in a confined system should be attributed to complex chemical processes including the cracking of various liquid components and C2–5 hydrocarbon gases (Behar et al., 2008; Tsuzuki et al., 1999; Vandenbroucke et al., 1999). Accordingly, methane generation is attributed to a set of n parallel first-order reactions. For instance, the concentration of methane (ci) generated from reaction i at a certain time (t) during pyrolysis can be expressed as (equation (3))


The total conversion (x) of methane at temperature T (K) can be calculated by
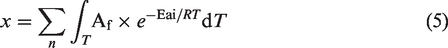
By fitting the experimental data during non-isothermal pyrolysis at two heating rates, the kinetic parameters for the generation of hydrocarbon gases can be obtained (Pan et al., 2009). In this study, Weibull distributions of Ea for methane generation from pyrolysis in anhydrous and hydrothermal conditions were addressed as indicated in Figure 4(a). For comparison, Af for methane generation from pyrolysis in two conditions were selected as same value of 1.0 × 1014 s−1. The comparison of the methane conversions obtained from kinetic calculations and experimental data was also shown in Figure 4(b). The Ea for methane generation is mainly distributed in the range between 52 and 73 kcal/mol (Figure 4(a)). This broad distribution should be attributed to the cracking of different components with diverse thermal stabilities, which include C14+ NSOs, C14+ aromatics and saturates, C6–14 hydrocarbons and C2–5 hydrocarbon gases (Behar et al., 2008; Tsuzuki et al., 1999; Vandenbroucke et al., 1999; Zhao et al., 2017). Although the yield of methane in pyrolysis with water is slightly higher than that in anhydrous pyrolysis, the conversions of methane at certain temperature in two conditions are essentially identical (Figure 4(b)). The average Ea for methane generation from pyrolysis in anhydrous and hydrothermal conditions were calculated to be 65.99 and 65.93 kcal/mol, respectively.
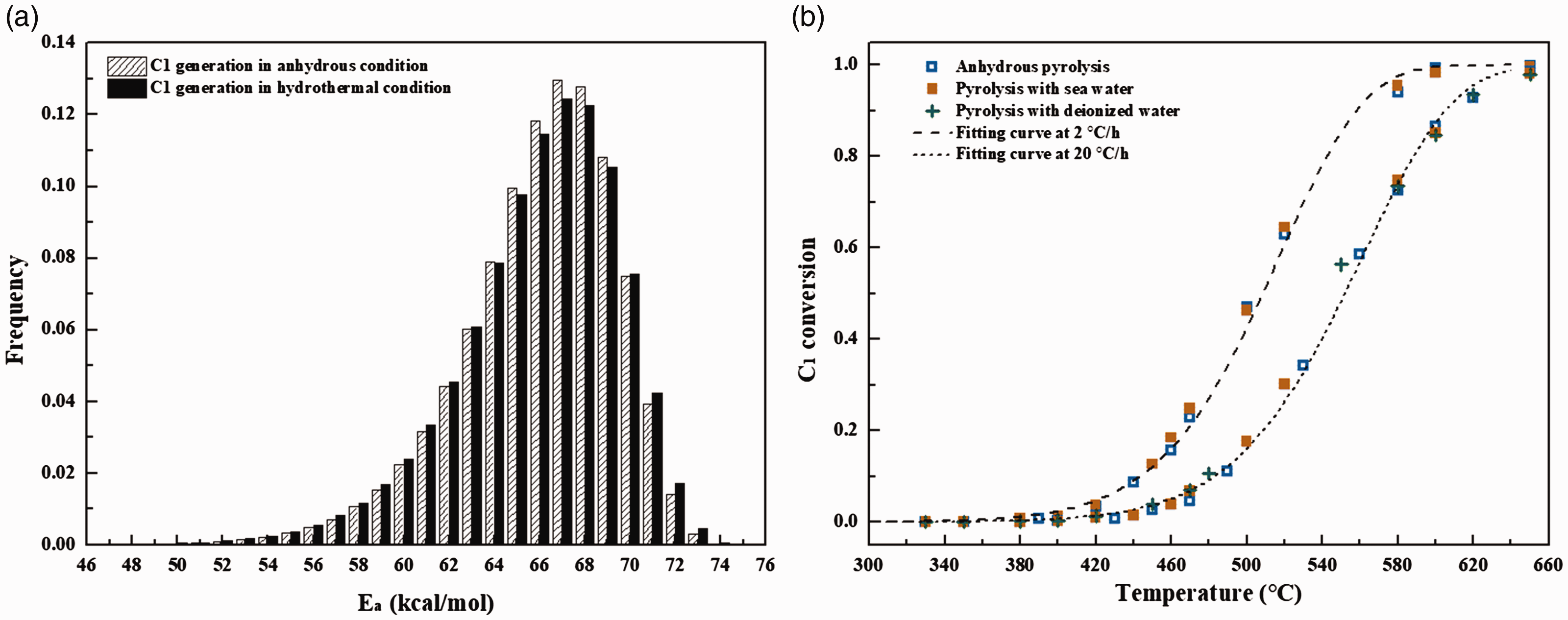
Weibull distribution of Ea for methane generation (a) and the comparison between the calculated conversion of methane and experimental data from pyrolysis at 2°C/h and 20°C/h in anhydrous and hydrothermal conditions (b). The value of Af is 1.0 × 1014 s−1. The maximum yields of methane used in conversion calculation for anhydrous and hydrothermal pyrolysis were 27.85 and 29.20 mmol/g oil, respectively.
For C2–5 hydrocarbon gases, their conversion should be collectively dominated by the generation and cracking processes during pyrolysis in a confined system (He et al., 2011; Shuai et al., 2006). According to the kinetic model proposed by Shuai et al. (2006), the Ea distribution for the generation and cracking of C2–5 were addressed as indicated in Figure 5. It can be found that Ea distributions for C2–5 generation and cracking in two conditions are also identical. The average Ea for C2–5 generation from pyrolysis in anhydrous and hydrothermal conditions were calculated to be 59.83 and 59.99 kcal/mol, respectively. The average Ea for C2–5 cracking in pyrolysis in anhydrous and hydrothermal conditions were calculated to be 69.14 and 68.74 kcal/mol, respectively. That is, the presence of water did not apparently affect the generation rate of hydrocarbon gases in non-isothermal pyrolysis of oil. Two possible reasons are responsible for this result: (1) Water–hydrocarbon reactions were not intensive enough to affect the generation of hydrocarbon gases due to the fast heating rate and less content of water in our pyrolysis experiments and (2) water–hydrocarbon reactions in our pyrolysis mainly proceeded via free radical mechanism, where radicals prior generated from the cleavage of organic compounds were captured by water or water-derived hydrogen (Lewan et al., 1997). For such a process, the rate determining reaction is the homo-cleavage of hydrocarbons (He et al., 2018a), which is same with oil cracking in anhydrous condition (Kissin, 1987). Actually, there is no evident increase of the relative contents of isomeric hydrocarbons (i-C4 and i-C5) during pyrolysis with water compared with those in anhydrous pyrolysis (online Supplementary Table S1). This result demonstrates that the generation of hydrocarbon gases in our pyrolysis in hydrothermal conditions should be attributed to free radical reactions (Kissin, 1987).
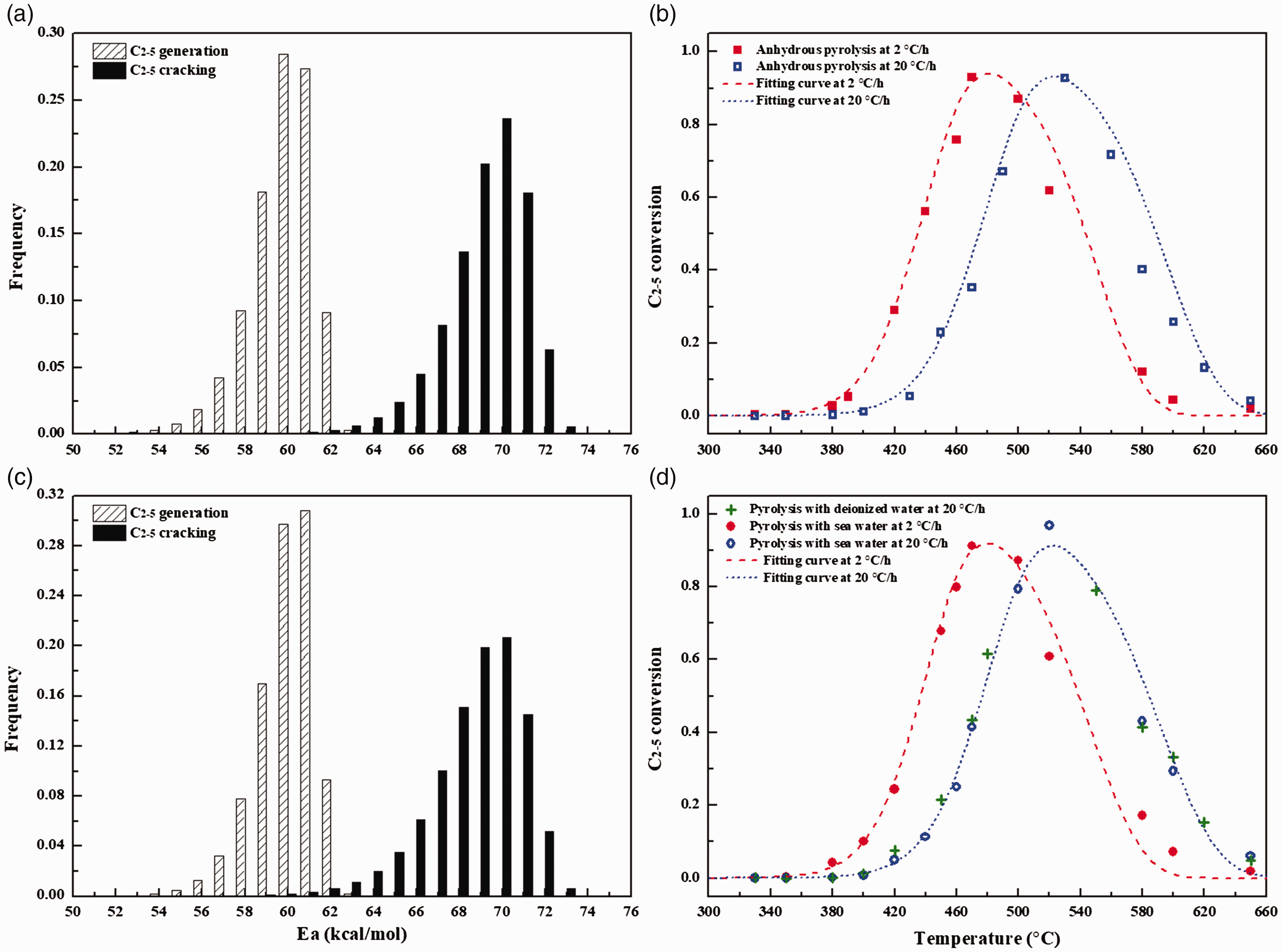
Weibull distributions of Ea for C2–5 generation and cracking and the comparison between the calculated conversion of C2–5 and experimental data with experimental data from pyrolysis in anhydrous (a,b) and hydrothermal (c,d) conditions at 2°C/h and 20°C/h. The value of Af is 1.0 × 1014 s−1. The maximum yields of C2–5 used in conversion calculation for pyrolysis in anhydrous and hydrothermal conditions were 8.0 and 9.0 mmol/g oil, respectively.
Carbon isotope fractionation kinetics for methane generation
As known, the thermal cracking of oils or hydrocarbons in anhydrous condition is dominated by free radical mechanism (Kissin, 1987). In this process, methane generation is initiated by the release of methyl radical (CH3·), which can be later terminated by capture of H. In anhydrous conditions, the isotope ratios of methane with thermo-origin are governed by the kinetic fractionation between the generation of 12CH3· and 13CH3· (Cramer et al., 1998; Tang et al., 2000)
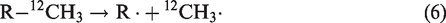
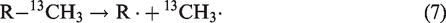
The isotope fractionation factor α (k*/k), which is the ratio between the generation rate of 12CH3· (k) and 13CH3· (k*) dominates the δ13C values of methane. Previous works based on theoretical calculations calculated the kinteic parameters (ΔEa, Af*/Af, k*/k) for the generation of alkyl radicals with and without one 13C substituted by the cleavage of different precursors (He et al., 2018b; Tang et al., 2000). It is addressed that Af*/Af for 13C frationation of CH3· ranges from 1.0 to 1.04 with an average value of 1.02. Meanwhile, the difference in bond dissociation energy (ΔΔH) between 13CH3· and 12CH3· clevage (ΔEa) is positive correlated with ΔΔH. On this basis, Tang et al. (2000) established a mathematical model to obtain the activation energy (Ea*) for 13CH4 generation from the cracking of precursor with particular Ea

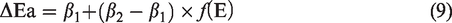
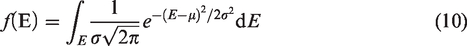
Combined with this model, the Weibull distribution for Ea (Figure 4(a)) and the experimental results (online Supplementary Table S1), the values of ΔEa and Ea* in terms of different Ea can be figured out. Then, the conversion of 13CH4 and δ13C1 values at different temperature can be addressed. Here, Af*/Af and the initial carbon isotope values (δ13C1,i) were selcted as 1.02 and –37.0‰. Table 2 shows the fitted parameters for carbon isotope fractionation of methane generated in anhydrous pyrolysis (Table 2). It can be found that the calculation curve for the evolution of δ13C1 with temperature fitted well with the experimental results (Figure 6(a)). The values of β1, β2, σ and μ are 45 cal/mol, 70 cal/mol, 4.0 and 54.5 kcal/mol, respectively. Meanwhile, the evolution of the cumulative and instantaneous δ13C1 at different geological temperatures at the heating rates of 1 and 10°C/Ma was also calculated as shown in Figure 6(b).
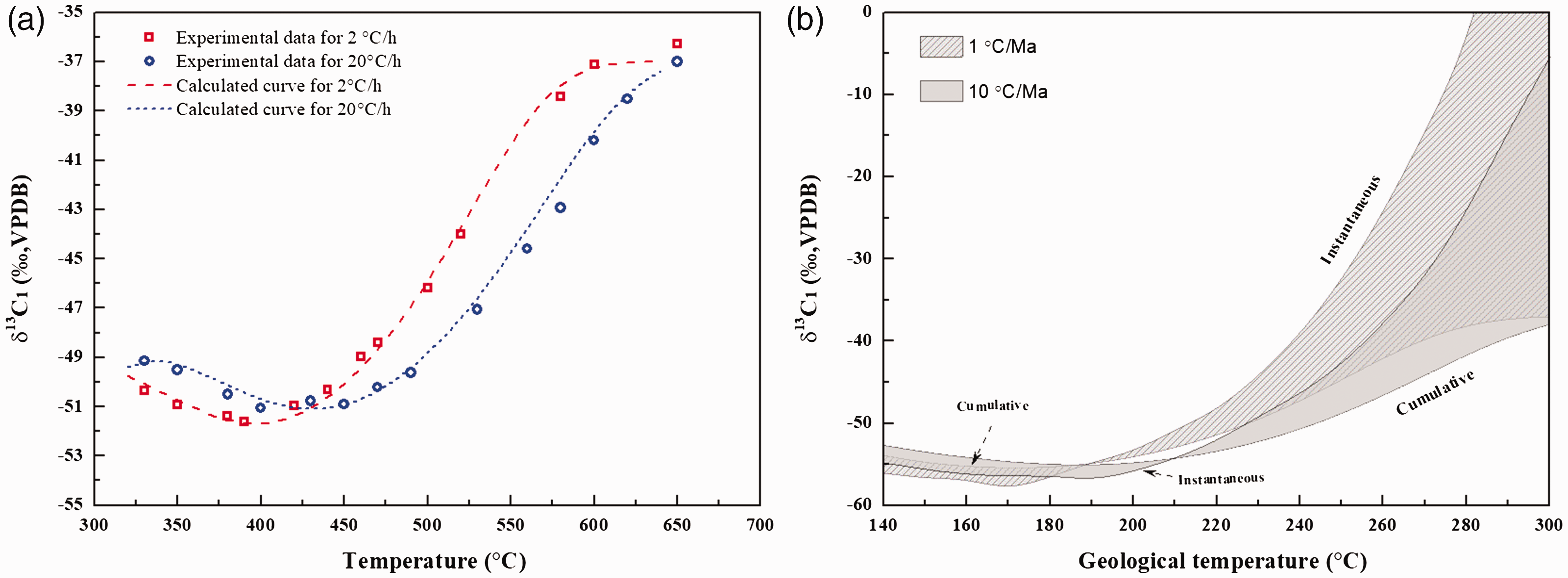
The fitting curve for carbon isotope fractionation (a) and the calculated cumulative and instantaneous δ13C1 at different geological temperatures at the heating rates of 1 and 10°C/Ma (b).
The kinetic parameters for carbon and hydrogen isotope fractionation of methane generated in anhydrous pyrolysis.

δ13Ci or δ2Hi refers to the initial carbon or hydrogen isotopic ratios for the precursor of methane.
In hydrothermal conditions, the generation of methane is collectively attributed to oil cracking and water–hydrocarbon reactions. Thermodynamic calculations demonstrated that the carbon isotope fractionations for methane generation from water–hydrocarbon reactions via two mechanisms (i.e. ionic and free radical mechanisms) are totally different (He et al., 2018a). If methane was generated from water–hydrocarbon reactions via free radical mechanism, secondary isotope fractionation will occur according to Reactions (11) and (12) (He et al., 2018a)
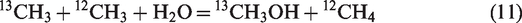
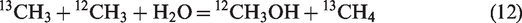
The ratio of equilibrium constant (K) between Reactions (11) and (12) at 25–600°C and 50 MPa is in the range of 1.035–1.012 (He et al., 2018a). That is, Reaction (11) is more thermodynamically feasible than Reaction (12), methyl radical substituted with 13C prefers to form alcohol rather than methane. The k*/k for methane generation from water–hydrocarbon reactions via free radical mechanism is mainly distributed in the range of 0.91–0.98, which is apparently lower than those from oil cracking, whereas the k*/k values for methane generation from water–hydrocarbon reaction via ionic mechanism are essentially same with those of α (k*/k) from oil cracking (He et al., 2018a). Although free radical mechanism seems to dominate the water–hydrocarbon reactions in our hydrothermal experiments, the presence of water did not evidently affect δ13C vales of hydrocarbon gases (Figure 2). Actually, when we look into the evolution of δ13C1 with methane yield, a slight 13C depletion at Easy%Ro higher than 3.0% (within 1.0‰ for Δδ13C1) can be observed (Figure 7). However, such a secondary fractionation induced by free radical reactions involving water and alkyl radicals is not intensive enough to govern the carbon isotopic compositions of hydrocarbon gases in non-isothermal pyrolysis.
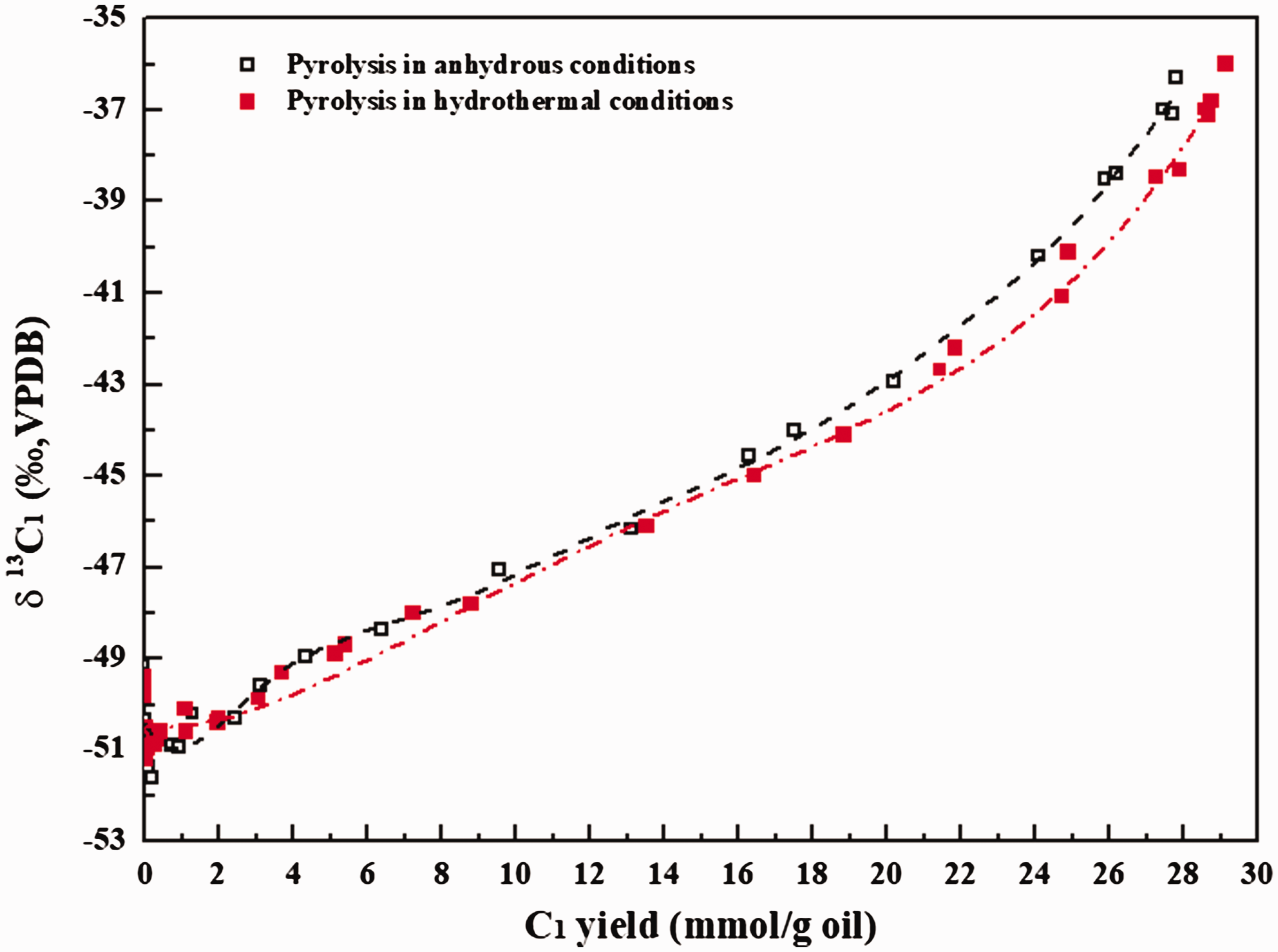
Evolution of δ13C1 values with methane yield during non-isothermal pyrolysis of YM201 oil in anhydrous and hydrothermal conditions.
Evaluation of the contribution of water-derived H for methane in geological conditions
We also addressed the kinetic parameters for hydrogen isotope fractionation of methane generated in anhydrous pyrolysis of oil (Table 2, Figure 8(a)) based on Tang’s model. Previous theoretical calculations indicate that Af*/Af for CH22H cleavage from organic chemical groups is mainly in the range of 1.05–1.30 with temperature from 25 to 650°C (He et al., 2018b; Ni et al., 2011; Tang et al., 2005). Here, Af*/Af was selected as the average value of 1.20 for hydrogen isotope kinectic fitting. The values of β1, β2, σ and μ are fitted as 830 cal/mol, 550 cal/mol, 3.5 and 65.0 kcal/mol, respectively. It is different with carbon isotope fractionation that β1 is higher than β2 for hydrogen isotope fractionation. This result can be reasonably interpreted by that the values of ΔEa for CH22H cleavage is negative correlated with bond dissociation energy ΔΔH (He et al., 2018b). That is, ΔEa for methane generation from the cracking of precursors with high Ea should be lower than that with relatively low Ea. Meanwhile, the evolution of the cumulative and instantaneous δ2H1 at different geological temperatures at the heating rates of 1 and 10°C/Ma were also calculated as shown in Figure 8(b).
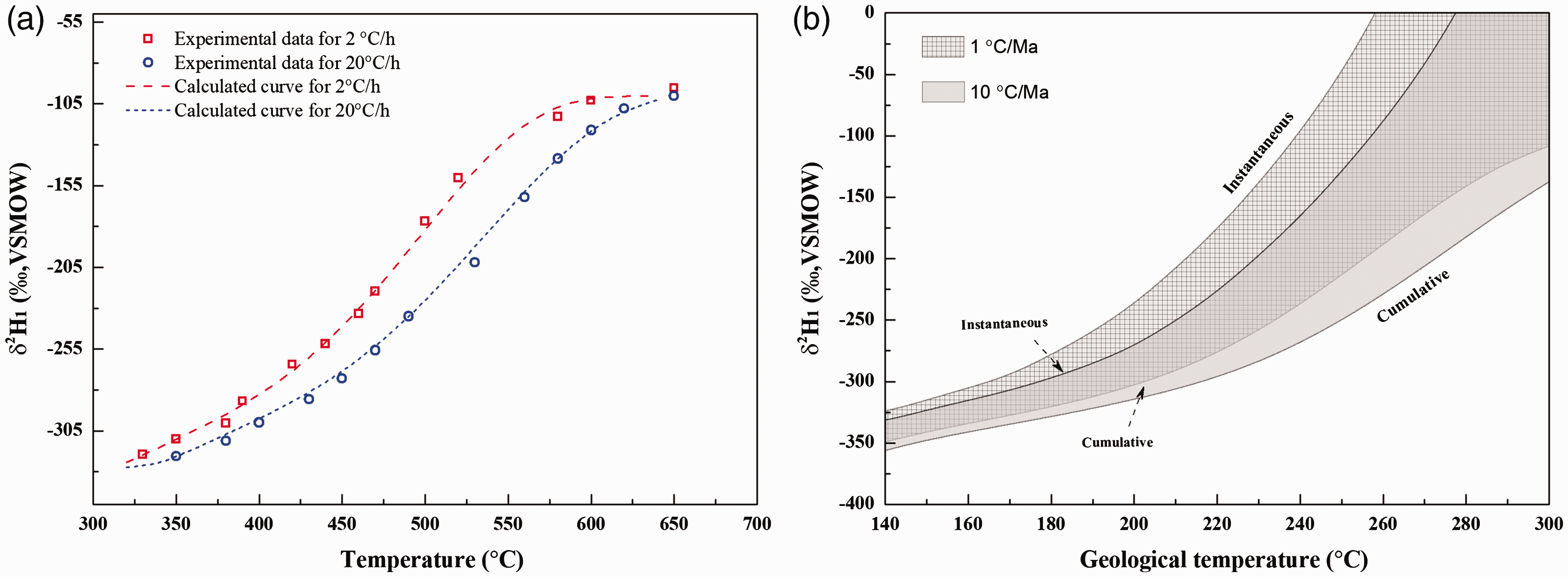
The fitting curve for hydrogen isotope fractionation (a) and the calculated cumulative and instantaneous δ2H1 at different geological temperatures at the heating rates of 1 and 10°C/Ma (b).
As we mentioned above, water provided 1H or 2H for methane generation during pyrolysis in hydrothermal conditions by water–hydrocarbon reactions via free radical mechanism (Reactions (13) and (14)). In this case, the supplement of hydrogen from water for methane (Reaction (15)) is responsible for 2H isotope enrichment or depletion of methane in hydrothermal experiments (He et al., 2018a)


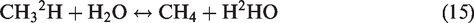
The difference of Gibbs energies (ΔΔG) and the equilibrium constant (Keq) in Reaction (9) can be calculated by equations (16) and (17), respectively
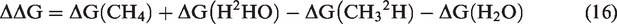

The values of ΔΔG and Keq were addressed as –394.91 to –437.72 cal/mol and 2.07–1.30 with temperature from 0 to 600°C (Zhang et al., 2018). On this basis, we can obtain the hydrogen isotopic ratios (δ2H1,w) of CH4 derived from water–hydrocarbon reactions by equations (18) and (19)

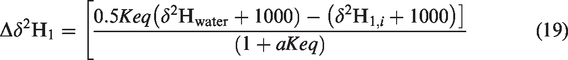
Apparently, δ2H1,
w
and Δδ2H1 values are governed by the initial δ2H values of precursors (or δ2H1,i) and water (δ2Hwater) as well as the molar ratio of methane versus water (He et al., 2018a). As mentioned above, the generation of methane in hydrothermal experiments should be attributed to two pathways, including water–hydrocarbon reactions and thermal cracking. If the percent contributions for methane by two pathways are x and 1-x, the total hydrogen isotopic ratio of methane (δ2H1,T) generated in hydrothermal conditions should be
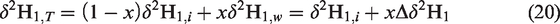
Hence, the δ2H1,T values can be obtained with certain x and Δδ2H1 values, which can be calculated with given a, δ2H1,i and δ2Hwater based on equation (20). As indicated above, the δ13C1 generated from oil pyrolysis in anhydrous and hydrothermal conditions are essentially same. Then, we can established the model for the evolution of δ2H1,T with δ13C1 in terms of different x (Figure 9). Figure 9(a) shows the evolution of δ2H1,T with δ13C1 in terms of different x in experimental conditions. The experimental data from anhydrous pyrolysis and pyrolysis with distilled water at 20°C/h were also plotted in this model. It can be found that the water–hydrocarbon reactions contributed to within 10% percent of methane and also resulted in the depletion of 2H for methane in hydrothermal experiments with distilled water. Field studies have extensively observed the rollover of δ2H1 (i.e. δ2H1 becomes abnormal lower at extremely high maturity) for natural gas in sedimentary basins (Burruss and Laughrey, 2010; Milesi et al., 2016; Tang and Xia, 2011; Zhang et al., 2018; Zumberge et al., 2012). Most researchers attributed the potential hydrogenation by water-derived H to this rollover for natural gas (Burruss and Laughrey, 2010; Milesi et al., 2016; Zhang et al., 2018; Zumberge et al., 2012). Previous pyrolysis experiments found that water-derived hydrogen can be transferred to organic matters or hydrocarbons (Hoering, 1984; Lewan, 1997; Reeves et al., 2012; Schimmelmann et al., 2001). Thermodynamic calculations also demonstrated that the H exchange equilibrium between water and methane can result in the depletion of 2H for methane in geological conditions (He et al., 2018a; Zhang et al., 2018). Here, we firstly established a model to quantitatively determine the contribution of water-derived hydrogen for methane generation in the subsurface. From Figure 9(b), it can be concluded that this contribution (x) for shale gas ranges from 0 to 50%. Such a different contribution should also be responsible for the hydrogen isotope rollover for both carbonate reservoirs gases and shale gas at extremely high maturity. As known, the potential and timing for hydrocarbon gas generation are mainly governed by the hydrogen content of organic matters or oil (Tissot and Welte, 1984). On consideration of the significant contribution of the hydrogen source from water, the gas potential especially at high maturity may be much higher and should be re-evaluated (Seewald, 2003). It is implied that there are greater exploration potential for shale gas and carbonate reservoir gas in deep formations. Surely, further studies should be conducted in future to determine the contribution of hydrogen from water for gas accumulation in different geological conditions.
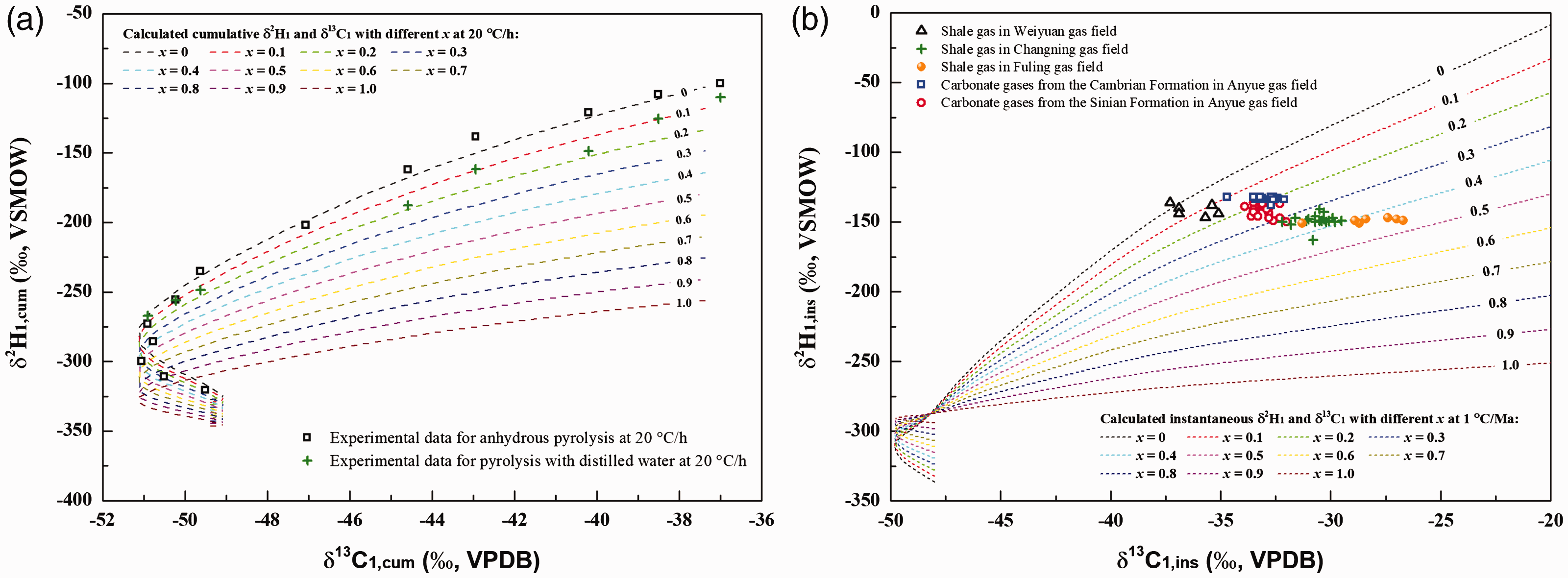
The models for the evolution of δ2H1,T with δ13C1 in terms of different contribution (x) of water–hydrocarbon reactions to methane in experimental conditions (a) and geological conditions (b). The a (C1/H2O), Keq, δ2H1,i, δ2Hwater, δ13C1,i and heating rate for experimental conditions were given as 0.5, 1.35, –100‰, –51.1‰, –37‰ and 20°C/h, respectively. The a (C1/H2O), Keq, δ2H1,i, δ2Hwater, δ13C1,i and heating rate for geological conditions were given as 0.1, 1.50, –100‰, –50‰, –30‰ and 1°C/Ma, respectively. The geological data for carbonate reservoir gases and shale gas were obtained from previous literature (Zhang et al., 2018; Zhao et al., 2019).
Conclusions
The conclusions in this study are summarized as:
The presence of water resulted in a slight increase of yields of both methane (29.06 ml/g oil) and C2–5 in non-isothermal pyrolysis of oil. Meanwhile, the yields of alkene gases and CO2 in pyrolysis with water are apparently higher than those in anhydrous pyrolysis. Kinetic calculations indicate that the presence of water does not seem to affect activation energies for the generation and cracking of hydrocarbon gases in non-isothermal pyrolysis of oil. Water–hydrocarbon reactions occurred during pyrolysis of oil with water and affected the carbon isotopic compositions of gas products. The δ13C values of CO2 generated during pyrolysis with water are much lower than those in anhydrous pyrolysis. Meanwhile, water–hydrocarbon reactions via free radical mechanism should be responsible for the depletion of 13C of methane in our non-isothermal pyrolysis with water. The hydrogenation by water via water–hydrocarbon reactions affected the hydrogen isotope ratios of methane (δ2H1) in pyrolysis of oil with water. The presence of deionized water (δ2H = –51.1‰) and heavy water (δ2H = 3328‰) leads to evident depletion and enrichment of 2H isotope for methane, respectively, while the presence of sea water (δ2H = –6.8‰) does not seem to apparently affect δ2H1 values compared with anhydrous pyrolysis. Hence, the effect of water–hydrocarbon reactions on δ2H of methane is governed by δ2H values of water. Mathematical models based on isotope fractionation were established to quantitatively determine the contribution of water–hydrocarbon reactions for gas generation in both experimental and geological conditions. It is suggested that water-derived hydrogen played a significant role for the generation of both carbonate reservoir gas and shale gas in the subsurface.
Supplemental Material
Supplemental material for Carbon and hydrogen isotope fractionation for methane from non-isothermal pyrolysis of oil in anhydrous and hydrothermal conditions
Supplemental Material for Carbon and hydrogen isotope fractionation for methane from non-isothermal pyrolysis of oil in anhydrous and hydrothermal conditions by Kun He, Shuichang Zhang, Jingkui Mi, Yu Fang and Wenlong Zhang in Energy Exploration & Exploitation
Footnotes
Acknowledgments
Careful reviews and constructive suggestions of the manuscript by three anonymous reviewers are greatly appreciated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the supports from National Key R&D Program of China (2017YFC0603102), Strategic Priority Research Program of the Chinese Academy of Sciences (XDA14010101) and the Scientific Research Project from RIPED (2018ycq01).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
