Abstract
Minerals in coal are of great significance in determining coal properties, washing, combustion, gasification, and liquefaction. The content of alkali metals in coal is an important factor determining corrosion and/or erosion in combustion boilers. Eleven coal samples were taken from the No. 6 coal seam of the Fukang mining area, Zhunnan coalfield, Xinjiang province, China. X-ray fluorescence, inductively coupled plasma-mass spectrometry, low-temperature ashing-X-ray diffraction, and electron probe microanalyzer were used for analyzing the minerals, major elements, and trace elements of the coal. The results indicated that the minerals mainly consist of dawsonite, dolomite, albite, calcite, kaolinite, quartz. The average content of Na2O in the ash of the No. 6 coal seam samples was 10.91%. Na mainly occurs in the form of organic compounds in the No. 6 coal seam, then in the form of inorganic minerals, such as dawsonite and albite. Na in surface and underlying strata was brought into the coal by water and combined with organic matter, resulting in relatively high Na contents in the upper and bottom parts of the coal seam and relatively low Na contents in the middle part.
Introduction
The inorganic constituents in medium- or high-rank coal mainly occur in the form of minerals (Ward, 2016). The content and occurrence of minerals in coal are important factors determining the coal’s quality. Minerals in coal are of great significance to coal properties, washing, combustion, gasification, and liquefaction (Bryers, 1996; Schatzel and Stewart, 2012); in some cases, minerals in coal are the major carriers of valuable elements that can be industrially extracted, such as Al, Ga, and rare earth elements (Dai et al., 2016; Hower et al., 1999; Prachiti et al., 2011; Qin et al., 2015; Rajak et al., 2018; Singh et al., 2016; Sun et al., 2015, 2016). The content of alkali metals and alkaline earth metals in coal is an important indicator for evaluating the severity of problems occurring in coal combustion, such as ash accumulation, slagging, and equipment corrosion (Guo et al., 2017; Wang et al., 2015). Theoretically, some alkali metals could also serve as indicators of coal rank and mineral matter origin in coal (Dai, 2015a; Vassilev et al., 1996). When the coal enriched in sodium and calcium contents is burned, these alkali metals would be released under the high temperature ambience. Alkali metals released from combustion would impact the boiler’s normal operation (Wang et al., 2014; Xu et al., 2014; Zhou et al., 2015).
Currently, coal is the main fossil energy resource in the energy consumption of China (Dai, 2015a). The contents and sources of alkali and alkaline earth metals in coals, as well as the method(s) to reduce the effects of these metals on the combustion equipment, have attracted the attention of many researchers (Zhou et al., 2010). Coal in the Junggar Basin contains high-alkali and high-alkaline earth metal elements (Zhou et al., 2010; Li et al., 2018). The Junggar Basin includes three major coalfields (Figure 1), namely, the Zhunbei coalfield, Zhundong coalfield, and Zhunnan coalfield. The Zhundong coalfield located in the eastern edge of the Junggar Basin is a great energy base of China, which has a huge reserve of 164 Gt (Li et al., 2012a). According to current coal exploration in China, the vast reserves of coal offer a long-term supply, up to 100 years of power generation in China (Song et al., 2018).
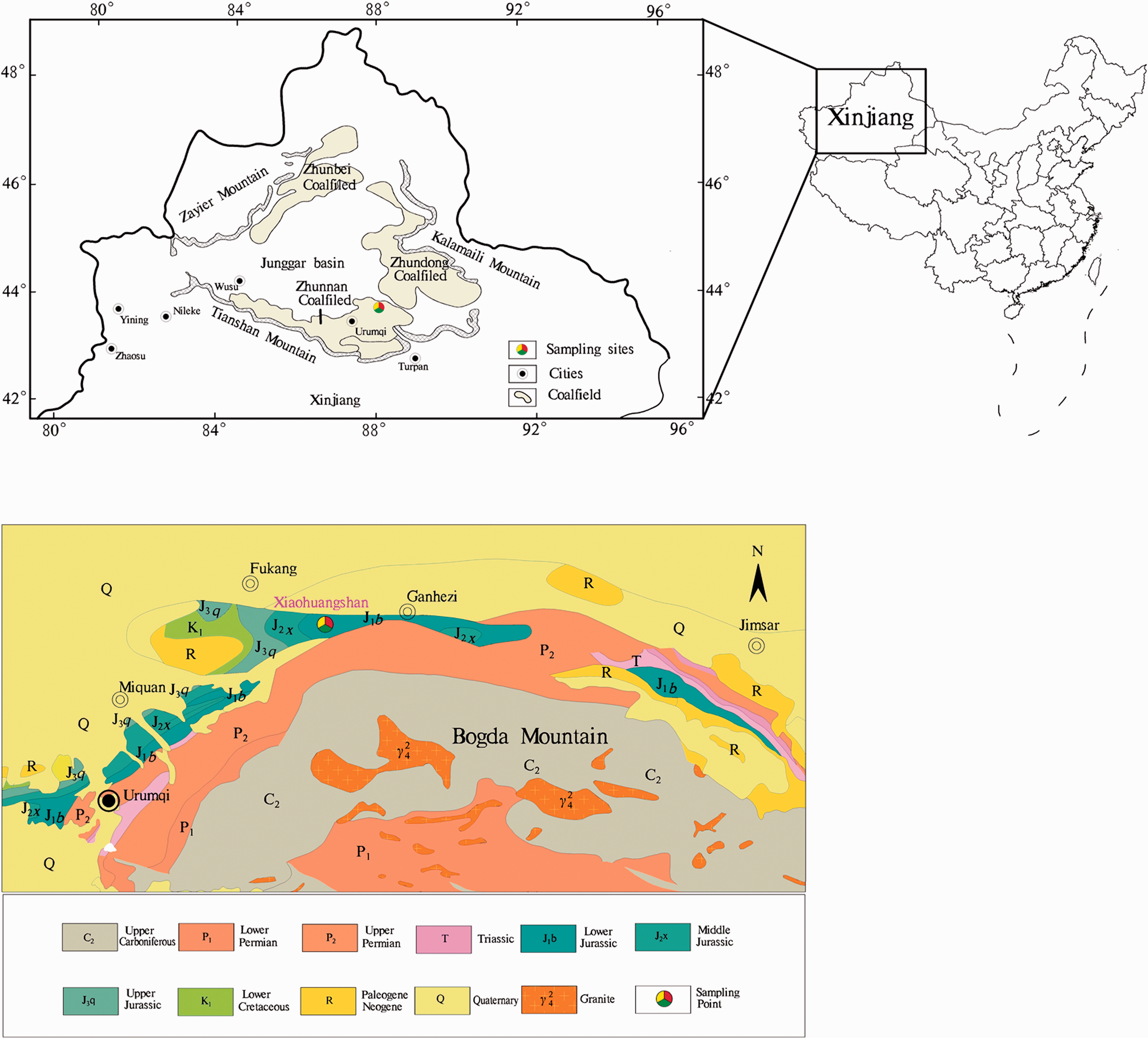
Distribution of Jurassic coal fields in Junggar Basin, and the geological map of Fukang area (Zhou et al., 2010).
Therefore, some studies have focused on the Na and K concentrations of coals from the Zhundong coalfield. Those studies took into consideration the migration and transformation processes during coal combustion (Guo et al., 2017; Li et al., 2012; 2015; Song et al., 2017; Wang et al., 2015; Zhu et al., 2013, 2017). Compared with the huge reserves of the Zhundong coalfield, the coal reserves of the Zhunnan coalfield are only 30 Gt (Cheng et al., 2016). The occurrence, contents, and sources of minerals and alkali, and alkaline earth metals have been rarely reported in the coals from Zhunnan coalfield. As an important coal resource in China, it is of high significance to study the content of minerals and alkaline earth metal in the Zhunnan coalfield. In this study, the mineral composition and elemental geochemical characteristics were reported. The contents and sources of Na in the coal samples were also discussed.
Geological setting
The Fukang mining area is located in the eastern part of the Zhunnan coalfield (Figure 1). It has a total area of approximately 2000 km2. The altitude of the mining area is lower in the north and higher in the south (Li, 2014). The region is rich in natural resources such as coal, oil, natural gas, and coalbed methane (Chen et al., 2016). Among these, the reserve of coal and coalbed methane are 8.5 billion tons and 3.7 ×1011 m3, respectively (Cheng et al., 2016; Fu et al., 2015; Wang et al., 2018a).
The regional strata encapsulate mainly the upper Paleozoic, Carboniferous, and Permian, followed by the Mesozoic, Triassic, Jurassic, and Cretaceous, and finally the Cenozoic, Paleogene, Neogene, and Quaternary (Figure 1). Since the Early Permian to present, the Junggar Basin has undergone the Hercynian, Indosinian, Yanshanian, and Himalayan tectonic movements. The igneous rocks broadly distribute in the front belt of Bogda Mountain, which consist of alkaline granites and granodiorite (Ma et al., 2015). The Jurassic stratum is extensively exposed and partially covered by the Quaternary loess. The coal-bearing strata in the area are the lower Jurassic Badaowan Formation (J1
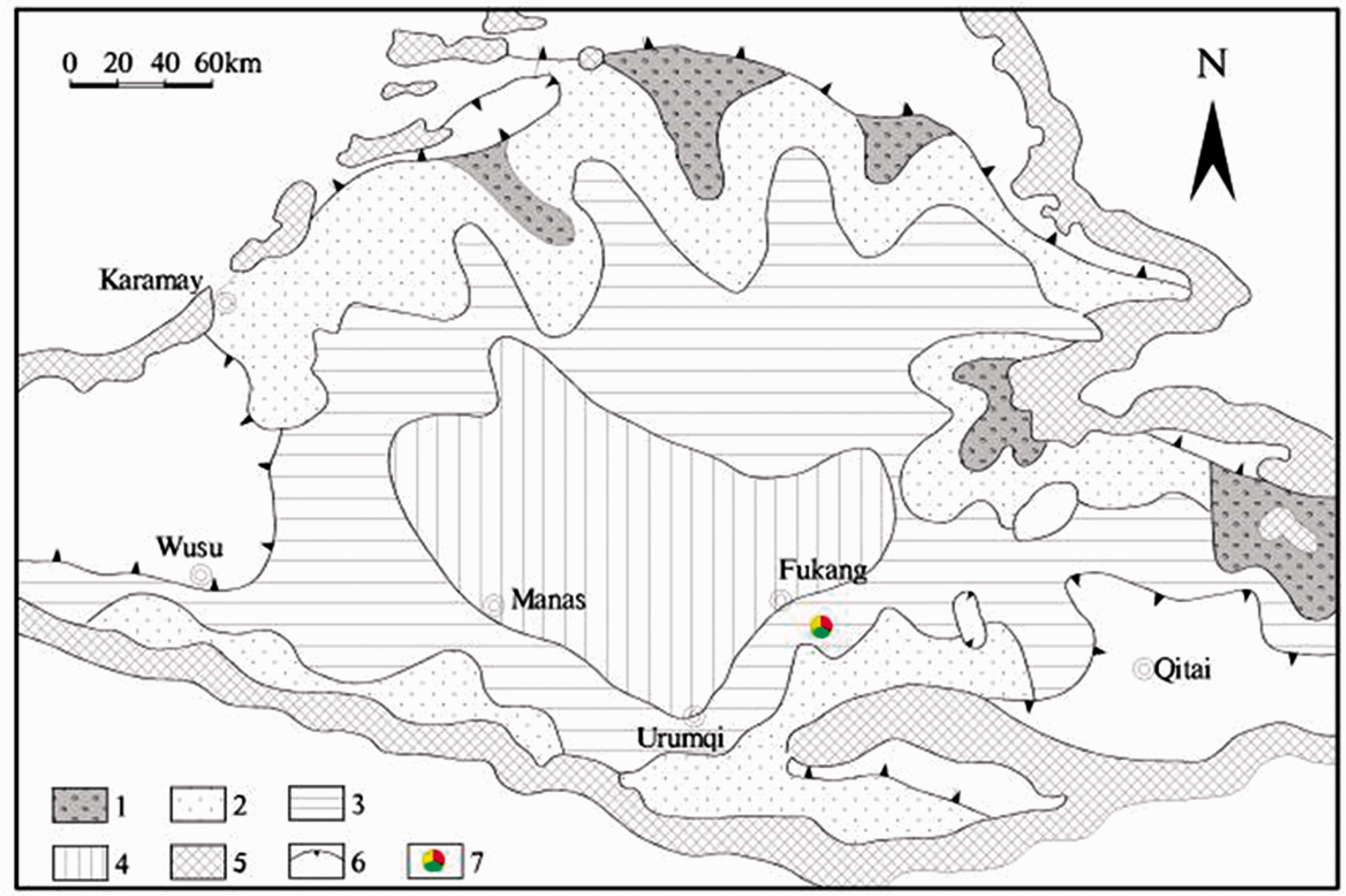
The lithofacies palaeogeography map of the Badaowan Formation in Junggar Basin (Zhang, 1998). 1—alluvial fan facies; 2—fluvial facies; 3—lakefront facies; 4—shallow lagoon facies;
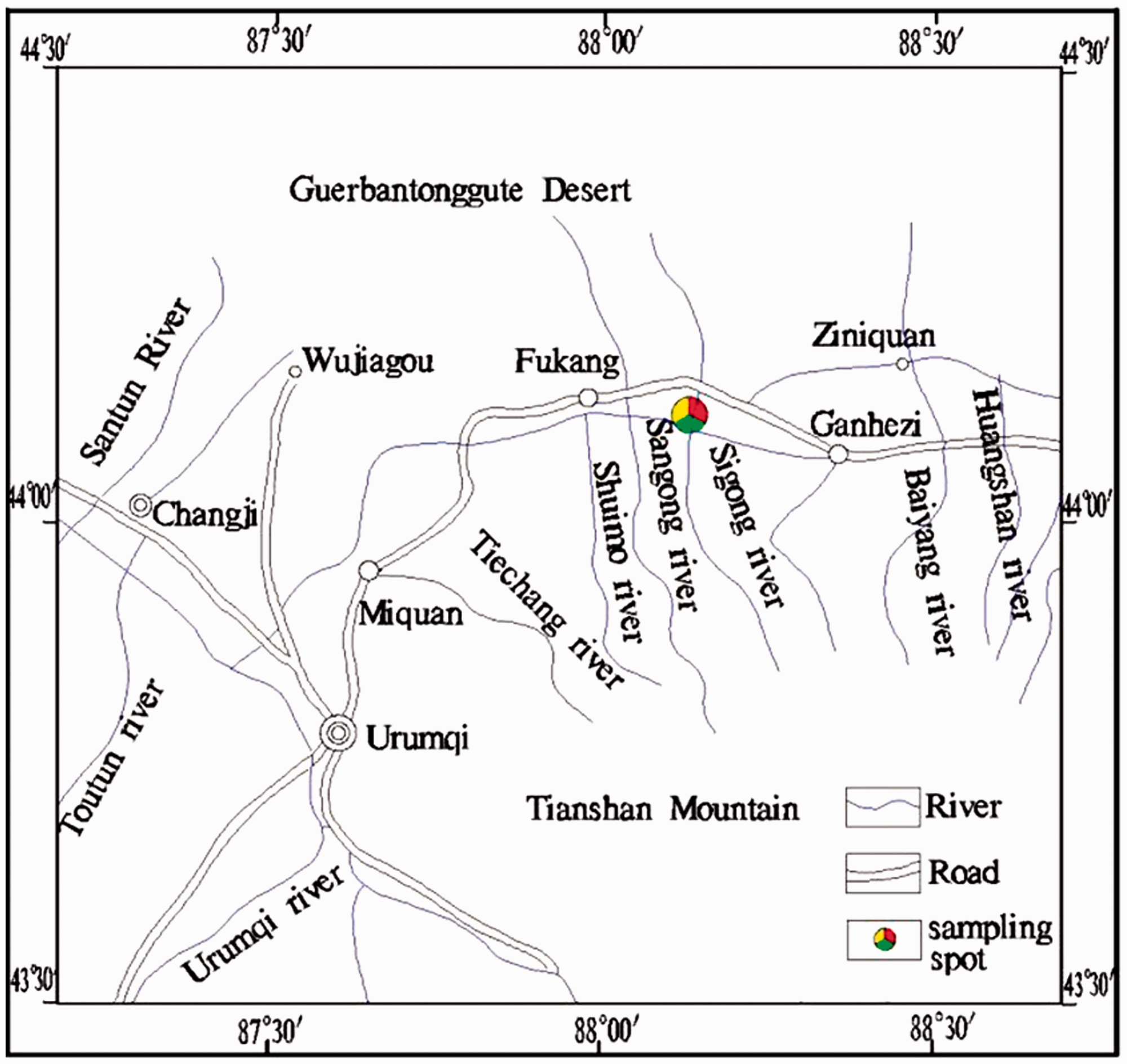
The distribution of rivers in Fukang area.
Samples and methods
In the present work, 11 bench coal samples from the bottom to the top of the No. 6 coal seam were collected from the Xiaohuangshan coal mine, Zhunnan coalfield. Every coal bench sample was cut over an area 10 cm wide and 10 cm deep into the coal face to represent a section of the coal and were named as 6-1 to 6-11 (Figure 4). The samples were sealed to avoid efflorescence and cross-contamination.

Stratigraphic column of the Sourthern Junggar Basin (modified from Tian et al., 2011).
The collected samples were dried and crushed under atmospheric conditions. All the samples were ground to 18 mesh and 70 mesh for the mineral and chemical analyses. For petrographical investigations, the 18-mesh coal samples were mounted in epoxy resin and prepared as polished sections, to measure their vitrinite reflectance, then the polished blocks were coated with carbon for electron probe microanalysis (EPMA, JXA 8230, JEOL, Japan).
The random vitrinite reflectance (Rr) were measured using a Leica DM2500P reflected light microscope and fitted with a halogen lamp (oil 32/0.65, 548 nm, 3 × 3 mm, EMI 9592 S-11; glass standard Leitz, Ro = 0.889%).
The mineralogical composition was determined by powder X-ray diffraction (XRD). Low-temperature ashing of coal was used for the 200-mesh samples and performed on an EMITECHK1050 plasma asher (Quorum, Ashford, UK), with the temperature maintained below 150°C. XRD analysis of the low-temperature ashes was performed on a D/max-2500/PC powder diffractometer (Rigaku, Tokyo, Japan), with Ni-filtered, Cu-Kа radiation and a scintillation detector. The XRD pattern was recorded over a 2θ interval of 3–70°, with a step size of 0.01° (Yuan et al., 2018).
All the samples were ashed at a temperature of 815°C. The oxides of major elements for the coal ashes were carried out by an X-ray fluorescence spectrometry (XRF, Thermo ADVANT XP+). The XRF setup is characterized by an Rh-anode end-window X-ray tube, a vacuum light channel, a rotation angle (2θ) of 5°–150°, and an analysis voltage and current of 30 kV and 80 mA, respectively (Zhao et al., 2014).
The measurement of the trace elements was carried out with an inductively coupled plasma-mass spectrometry (ICP-MASS, mode: X-II). The samples intended for trace element quantification were microwave acid-digested. Samples of approximately 200 mg were weighed into polytetrafluoroethylene (PTFE) vessels. The basic load for the PTFE vessels was composed of 2 ml HF (50%) + 5 ml HNO3 (65%) + 2 ml H2O2 (30%). Microwave digestion time was fixed at 1 h, with a temperature of 210°C. Then, the digestion liquid was evaporated at 90°C under vacuum, which took approximately 1 h. The residue was then dissolved in 1 ml HCl (32%) + 8 ml deionized water +1 ml HNO3 (65%). This solution was then transferred into 125 ml fluorinated ethylene propylene (FEP) bottles which were filled up to 100 g with deionized water (Xiao et al., 2016; Zhao et al., 2017).
Results
Proximate analyses
The proximate analysis, various sulfur forms and vitrinite random reflectance of the No. 6 seam samples are shown in Table 1.
Proximate, forms of sulfur (%), and vitrinite reflectance of the No. 6 coals from Xiaohuangshan coal mine, Xinjiang, China.
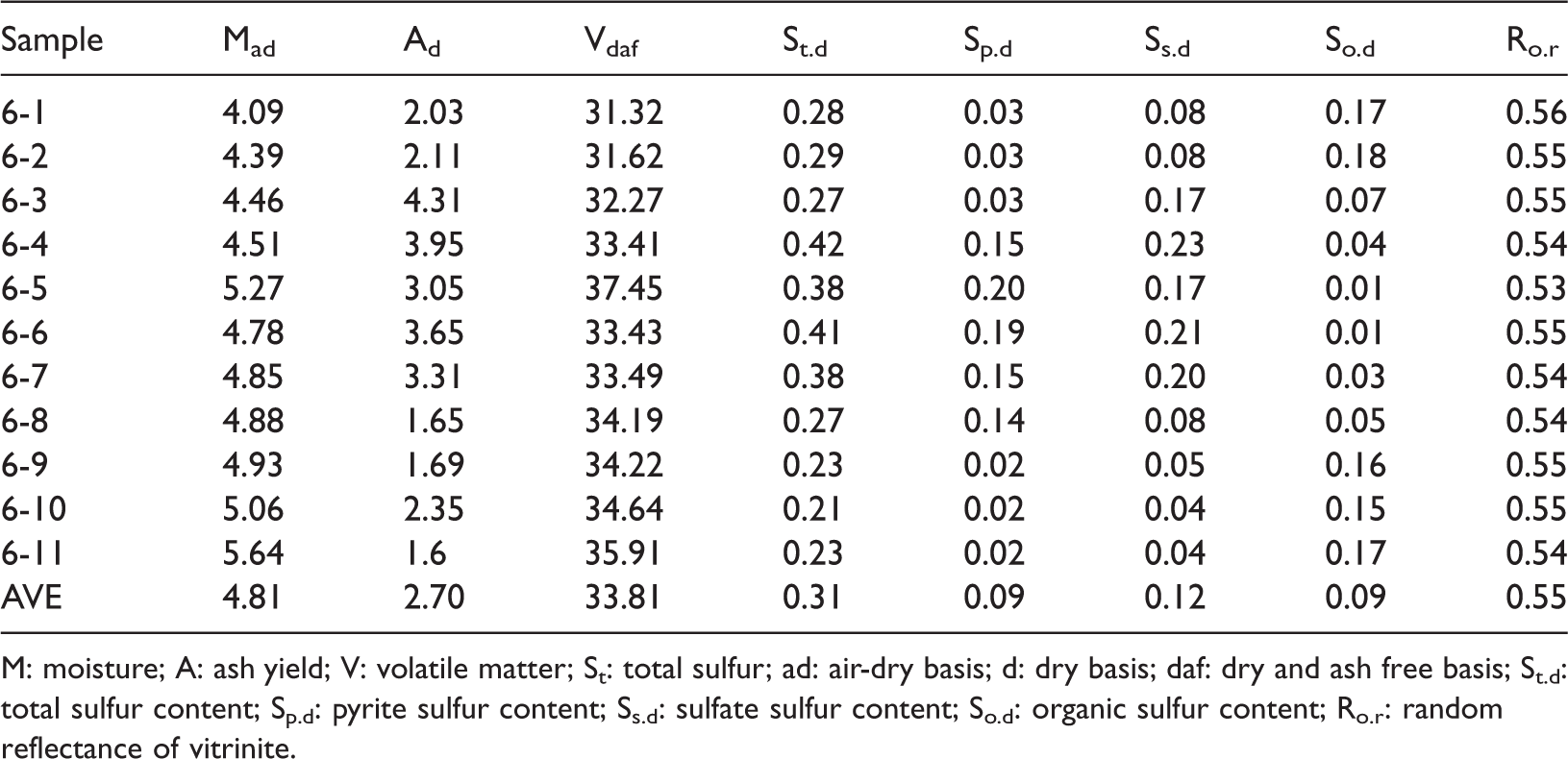
M: moisture; A: ash yield; V: volatile matter; St: total sulfur; ad: air-dry basis; d: dry basis; daf: dry and ash free basis; St.d: total sulfur content; Sp.d: pyrite sulfur content; Ss.d: sulfate sulfur content; So.d: organic sulfur content; Ro.r: random reflectance of vitrinite.
The random vitrinite reflectance of the No. 6 coal seam ranged from 0.53% to 0.56%, with an average of 0.55%, indicating that this coal is a medium-rank coal (Alpern and Lemos de Sousa, 2002). The moisture content of No. 6 coals ranges from 4.09% to 5.64%, with an average of 4.72%, showing that No. 6 coals are low moisture coals, in accordance with the Chinese Standard (MT/T 850-2000, <5% for low-moisture coal, 5% to 15% for medium-moisture coal, and >15% for high-moisture coal).
The volatile matter contents of the No. 6 coal seam ranged from 31.32% to 35.91%, with an average of 33.26%, indicating that this coal is a medium-high-volatile coal, according to the Chinese Standard (MT/T 849-2000, 20.01–28.00% for medium-volatile coal, 28.01–37.00% for medium-high-volatile coal, 37.01–50.00% for high-volatile coal, and >50.01% for super high-volatile coal).
The ash yield ranged from 1.6% to 4.31%, with an average of 2.71%, suggesting that this coal is a low-ash coal, according to Chinese National Standard (GB/T15224.1-2010, 2011, 10.0–20.00% for low-ash coal, 20.0–30.00% for medium-ash coal, and 30.0–40.00% for high-ash coal).
Regarding the total sulfur contents, they ranged from 0.21% to 0.42%, with an average of 0.31%. This coal is an ultralow-sulfur coal, according to the Chinese National Standard (GB/T 15224.2-2010, 2011) (<0.5% for ultralow-sulfur coal, 0.51–0.9% for low-sulfur coal, and 0.9–1.50% for medium-sulfur coal). Total sulfur consists of inorganic sulfur (including pyritic sulfur, 0.02–0.2%, 0.09% on average; sulfate sulfur, 0.04–0.23%, 0.12% on average) and organic sulfur (0.01–0.18%, 0.09% on average). In summary, the No. 6 coals of the Xiaohuangshan mine are characterized by low-moisture, medium-high volatiles, low-ash, and low-sulfur coals.
Major elements, minerals, and the contents of trace elements in the coal
For the ash yield of the No. 6 coal seam samples of the Xiaohuangshan mine, the oxide composition of the major elements is shown in Table 2. The Na2O contents ranged from 7.41% to 15.21%, with an average of 10.91%. Since the ash yields are low (average value 2.71%), the converted Na content in coal is 0.19%, which is slightly higher than the average Na2O in Chinese coal (0.16%) (Dai et al., 2012). The K2O contents in coal ash ranged from 0.20% to 1.32%, with an average of 0.71%.
Concentration of major oxides in coal ashes from No. 6 coal seam of Xiaohuangshan Mine (%).
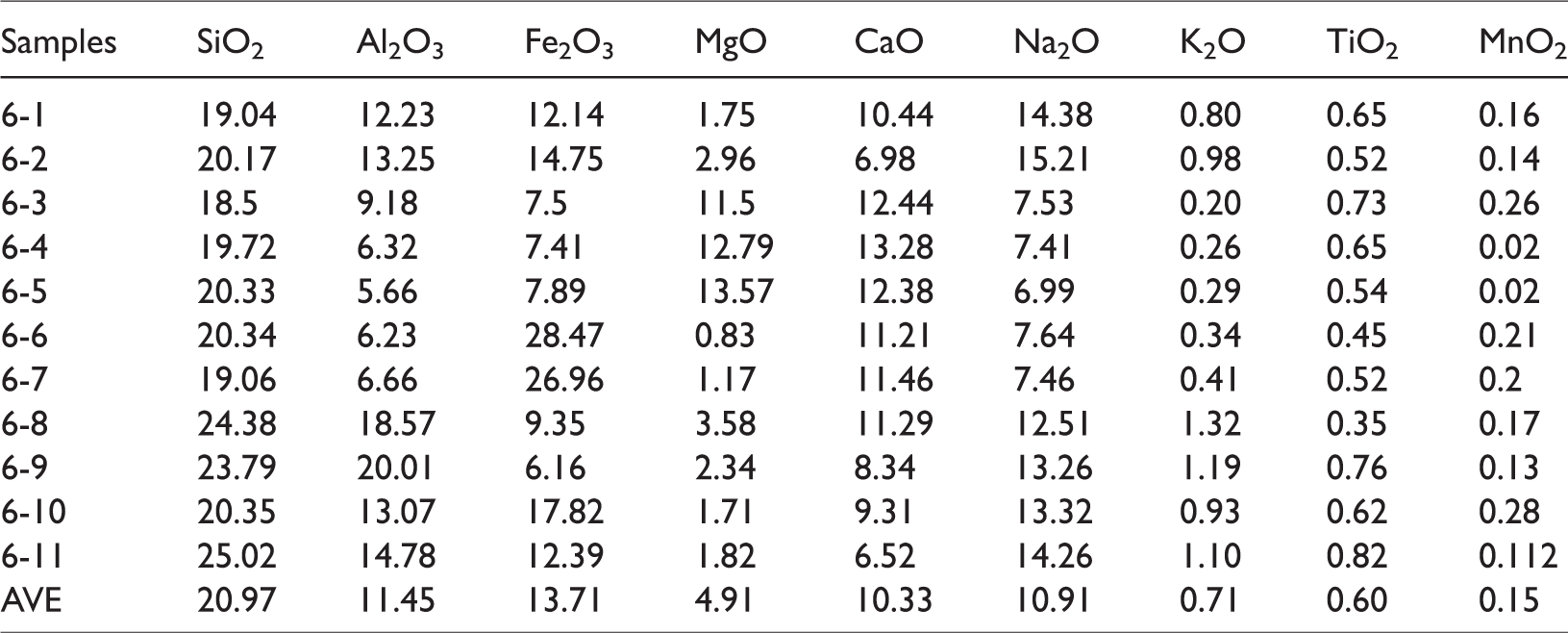
According to the Chinese grading standard for Na and K contents MT/T 1074-2008 (<0.1 for alkaline coal, 0.1–0.3 for low-alkaline coal, 0.3–0.5 for medium-alkaline coal, >0.5 for high-alkaline coal), the No. 6 coal seam samples are low-alkaline coal. In the ash of steam coal from China, the content of Na2O is lower than 1% (Li et al., 2018; Zhang et al., 2013). Therefore, many researchers have considered that ash with an Na2O content higher than 2% should be classified as high-sodium coal (Li et al., 2015, 2018; Song et al., 2017). For the ash samples from the Xiaohuangshan coal mine, the Na2O contents are much higher than 2%, so they are high-sodium coal, according to the aforementioned standard. The average CaO content in ash is 10.33%, corresponding to a Ca value of 0.21% in coals, which is lower than the average CaO content (1.23%) of Chinese coal (Dai et al., 2012).
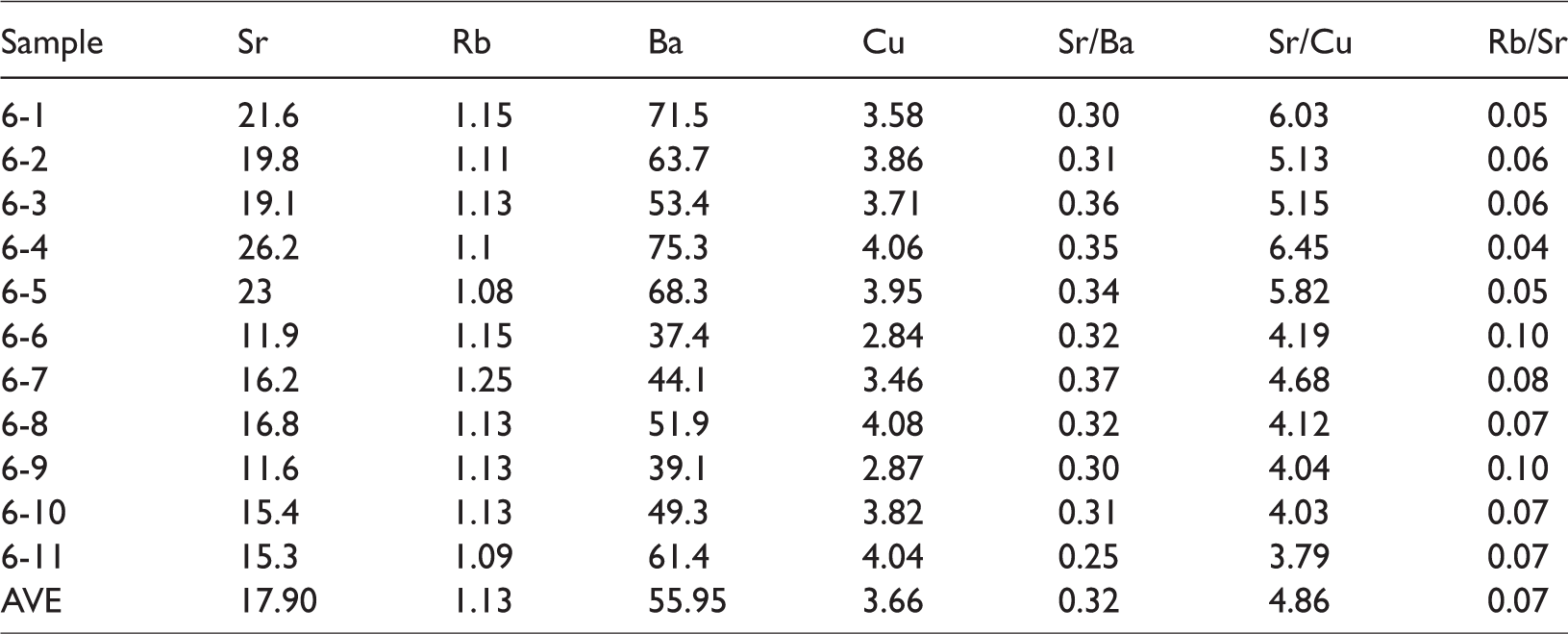
Some elements in sediments are sensitive indicators for paleoenvironment conditions, such as Sr, Rb, Ba, and Cu. So as to investigate the evolution of the sedimentary environment and paleoclimate during coal accumulation in the Badaowan formation, the geochemical parameters including Sr/Ba, Sr/Cu, and Rb/Sr were presented. The contents of trace elements and its ratios in the No. 6 coal samples are listed in Table 3. From Table 3, the average content of Sr, Rb, Ba, Cu are 17.9 µg/g, 1.13 µg/g, 55.95 µg/g, 3.66 µg/g, respectively. The Sr/Cu ranged from 3.79 to 6.45, with an average of 4.86; the Rb/Sr ranged from 0.04 to 0.1, with an average of 0.07.
Concentration of trace elements (µg/g) and geochemical indicates of trace elements of Xiaohuangshan No. 6 coal seam.
Minerals in the Xiaohuangshan No. 6 coal seam are mainly composed of carbonate minerals (dawsonite, dolomite, calcite), clay minerals (kaolinite, illite), albite, quartz, and a trace of apatite, goethite (Figures 5 and 6). The semiquantitative content of dolomite, dawsonite, quartz, albite, clay minerals, and calcite in No. 6 coals are 45.57%, 24.16%, 12.26%, 7.67%, 7.43%, and 2.91%, respectively.
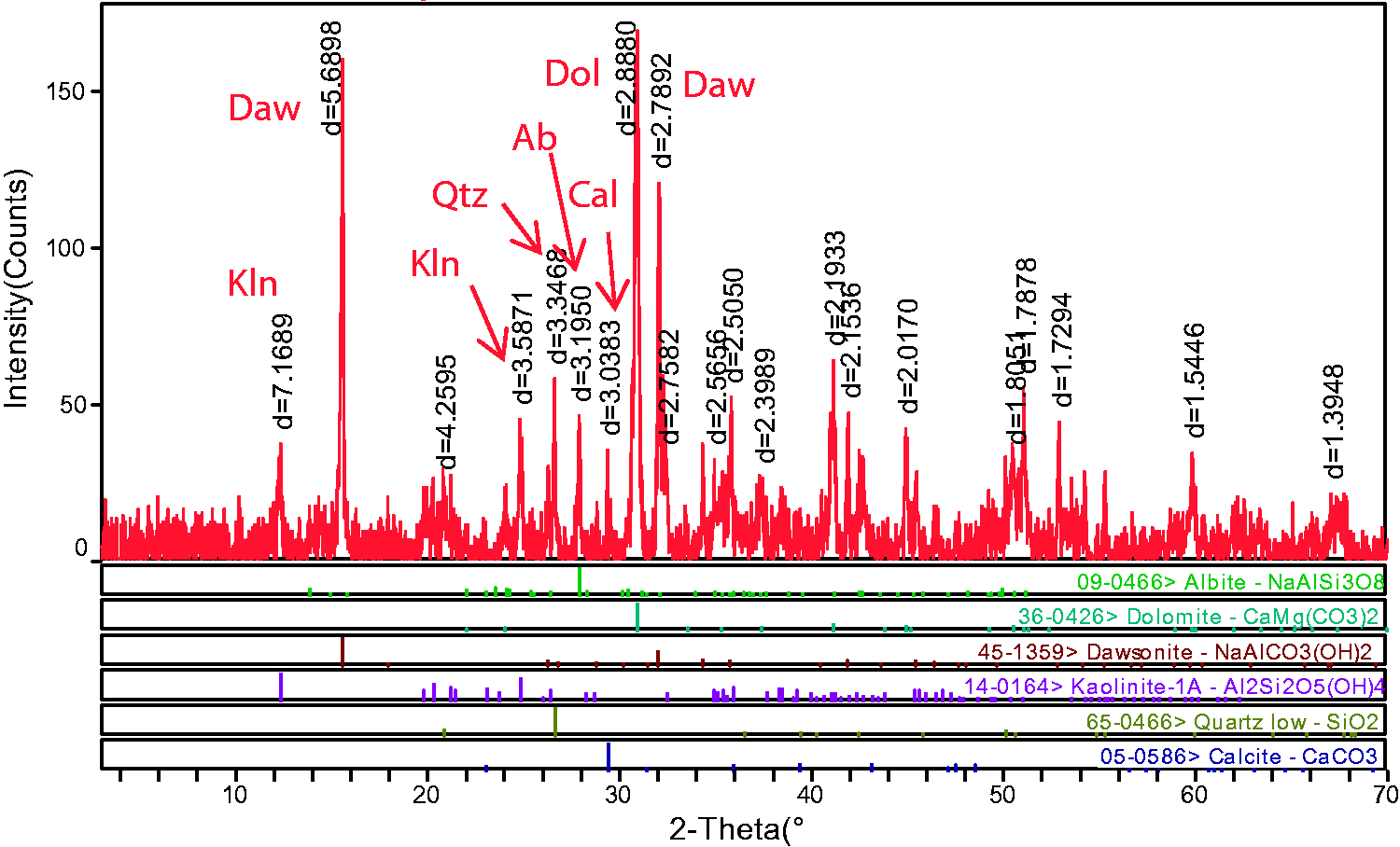
X-ray diffraction pattern of LTA ash of Xiaohuangshan coal.
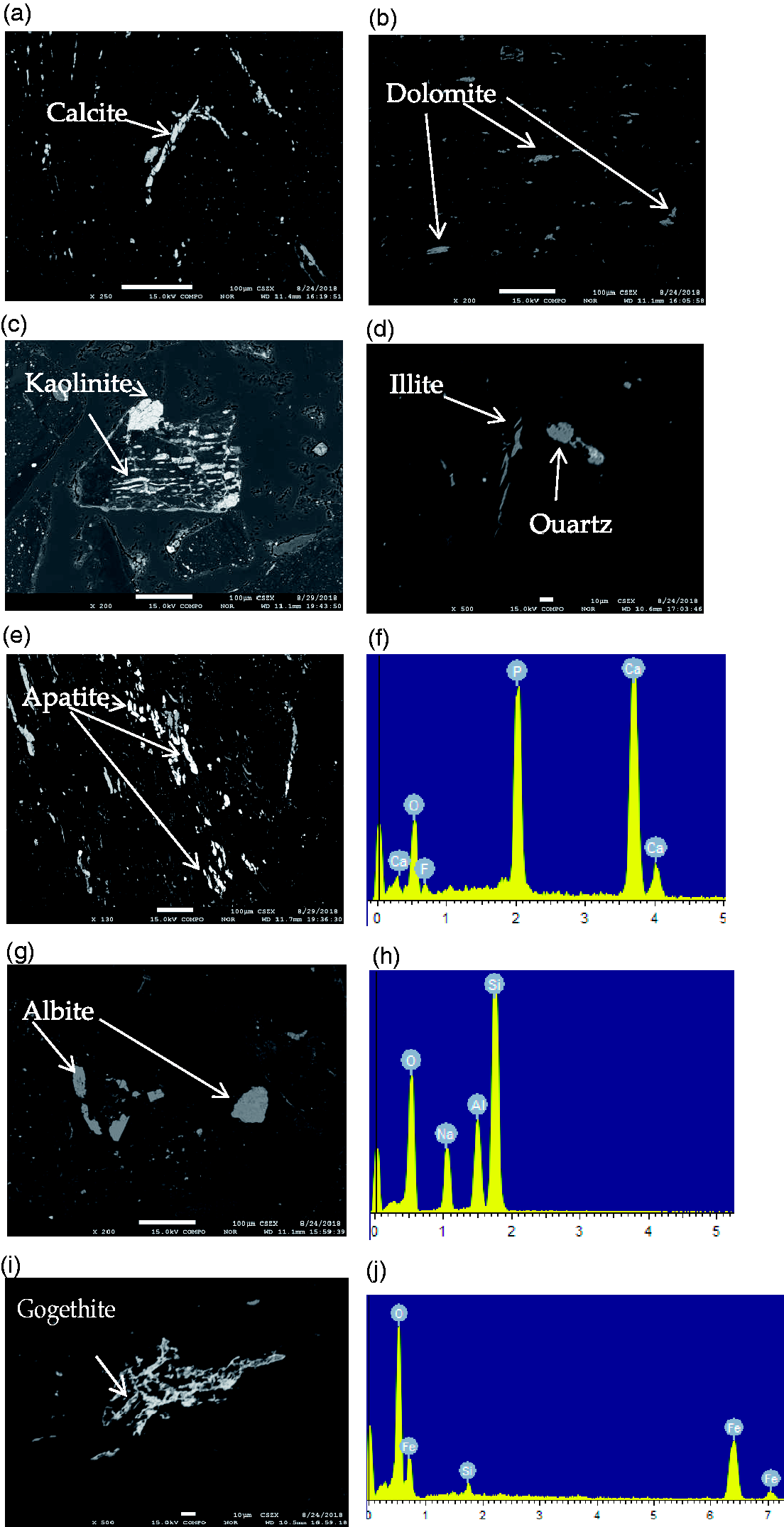
EPMA back-scattered electron images of minerals in No. 6 coal seam from Xiaohuangshan coal mine. (a) calcite; (b) dolomite; (c) kaolinite; (d) illite and quartz; (e) apatite; (f) The EDS (Energy Dispersive X-ray Spectrometry) date for apatite; (g) albite; (h) The EDS date for albite; (i) goethite; (j) The EDS date for gogethite.
Discussion
Genesis of minerals in No. 6 coal
Carbonate minerals, such as calcite, dolomite, ankerite, and siderite, are common cleat infilling materials in coal seams (Ward, 2016). As discussed further by Ward (2002), carbonate minerals in coal are formed from CO and CO2, which are produced from coalification process, reacting with the fluid in coal. As shown in Figure 5, dawsonite, dolomite, and calcite are the main carbonate mineral in No. 6 coal seam. Figure 6(a) and (b) shows that dolomite and calcite in the A6 coal seam mainly occur as lumps and infillings of fracture, indicating they are authigenic minerals.
Although dawsonite was found in coal cores from Australia in 1972 (Loughnan and Goldberg, 1972), dawsonite is rarely reported in coal. However, the content of dawsonite in A6 coal seam is 24.16%, second only to dolomite (45.57%). Dawsonite forms in alkaline fluids and high concentrations of CO2; as a type of authigenic mineral, it commonly enriched in clastic rocks, volcaniclastic rocks, and coal (Baker et al., 1995; Stevenson and Stevenson, 1977). The CO2 that leads to dawsonite formation may be derived from many sources, such as magma activity, the products of pyrolysis of carbonate rocks, and kerogen in hydrocarbon source rock (Gao et al., 2005; Hutcheon et al., 2016; Ming et al., 2017; Zhou et al., 2014). In addition, sanidine can interact with alkaline hydrothermal fluid rich in sodium, aluminum, and carbonate ions to form dawsonite (Dai et al., 2008). Many scholars’ research shows that when CO2 is injected into a stratum rich in aluminosilicate minerals, it will react with these minerals to form dawsonite (Dai et al., 2008; Mohd Amin et al., 2014). The formation of dawsonite can be represented by reactions in Table 4. As mentioned before, the Fukang coal field is rich in coalbed methane, which formed from the coalification process. Meanwhile, geological data show that quartz albitophyre intrusive bodies, which have been formed in the Carboniferous period (Cao et al., 2017; Gong et al., 2017), located in the Tianshan piedmont, are distributed in the south and southeast of the coal field. Moreover, albite has been found in the No. 6 coal seam and occurs as angular particles, indicating a terrigenous origin (Figure 6(g)). Therefore, it is speculated that during the peat depositional period, feldspar minerals were carried to the peat swamp by water and reacted with CO2 released from the coalification process to form dawsonite. Meanwhile, the pH of surface water and phreatic water are 8.9 and 8.5, respectively, showing alkalescent water in this area (Cao et al., 2017; Li et al., 2013). In addition, the dissolution of aluminosilicate minerals will provide the Na+ and Al3+ needed for the precipitation of dawsonite (Mohd Amin et al., 2014). The formation of Na+ and Al3+ can be represented by reactions in Table 4. All these would provide formation for dawsonite.
The chemical reactions considered in this study (Mohd Amin et al., 2014).
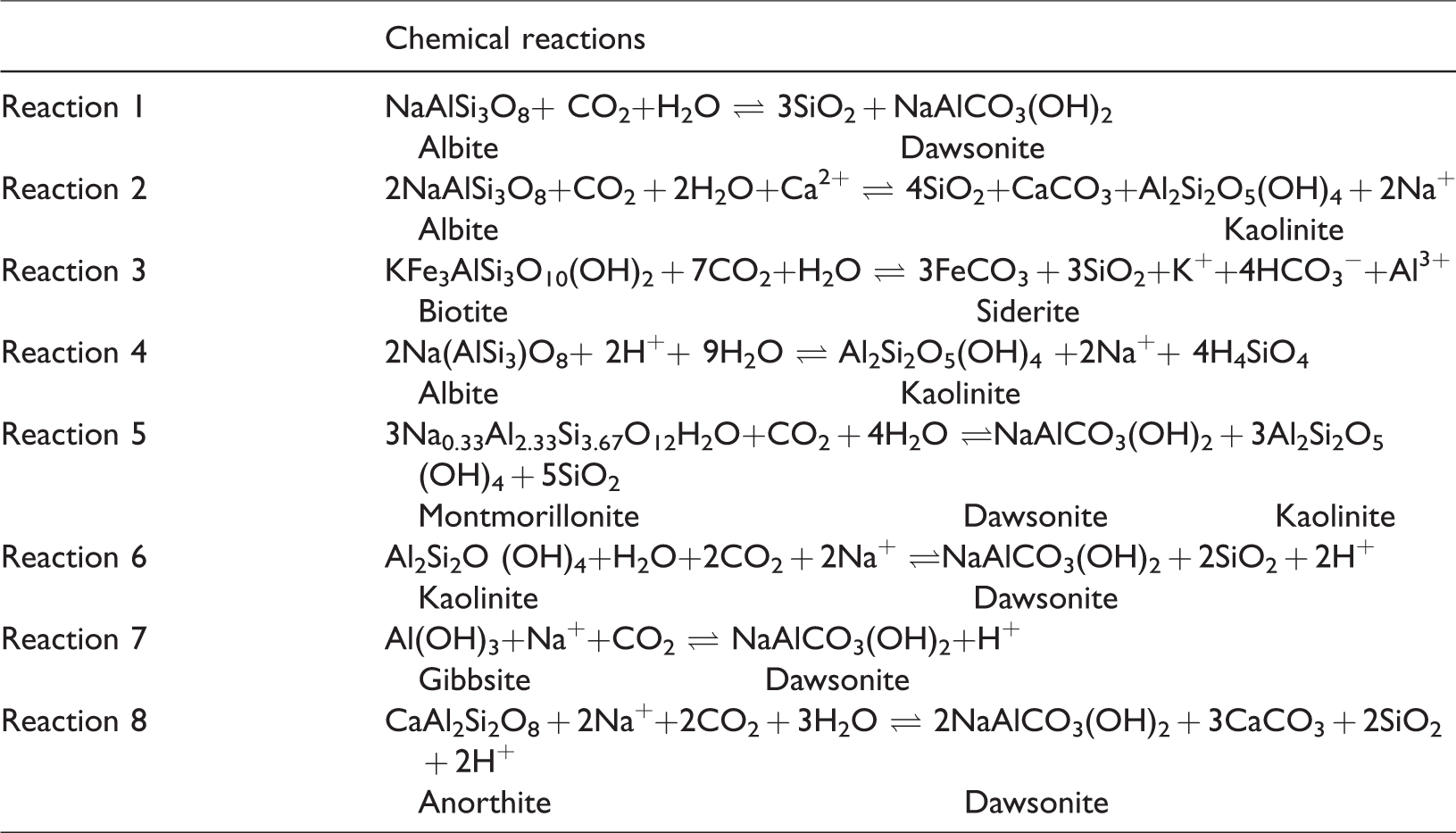
The saturation index range of calcite and dolomite in ground water ranged from –2.33 to 1.06, from –4.25 to 2.00, respectively, showing that the solubility of the two minerals in water is up to the point of limitation (Li, 2014). Therefore, it was speculated that abundant carbon dioxide from the coalification, and the presence of feldspar minerals, and alkalescent water offered favorable conditions for the formation of the carbonate minerals.
Kaolinite and illite are the most commonly occurring clay minerals in coals (Ward, 2016). As shown in Figure 6(c), Kaolinite in the No. 6 coal seam occurs as fracture fillings. A similar cell-filling occurrence is common in many coals and closely associated strata, indicating it is an authigenic mineral (Ward, 2016). Illite in No. 6 coal seam occurs as fracture fillings, indicating a hydrothermal origin (Figure 6(d)).
The mode of occurrence of quartz is distributed in collodetrinite showing its terrigenous origin (Ward, 2002). Quartz is commonly distributed in the macerals as irregular particles in this coal, suggesting a terrigenous detrital origin. As shown in Figure 6(d), quartz is distributed as detrital grains in macerals throughout the samples.
Apatite is the most common phosphate mineral in coal (Ward, 2016). Apatite has been frequently identified in coals, most of which were associated with tonsteins or may be influenced by volcanic ash (Dai et al., 2015b; Hower et al., 1999; Spears et al., 1988). However, during the plant decay and humification processes within the peat swamp, phosphorus in plants tend to be released into the swamp or peat waters and precipitated elsewhere with other available ions, under an appropriate peat-water chemical environment, to form phosphate mineral (Ward et al., 1996). Both geological and geochemical data show that the research area was unaffected by magma activity since the formation of the coal seam (Bai, 2008). As shown in Figure 6(e), apatite occurs as cleat and fracture fillings, indicating it is an authigenic mineral. It is speculated that the apatite may either be formed by migration of hydrothermal solutions or from local remobilization of phosphate, formed earlier within the coal seam or associated strata (Kang and Li, 2016).
Meanwhile, goethite (Figure 6(i)) has also been found in the No. 6 coal seam and occurs as fracture fillings. Hematite and poorly crystallized Fe oxides and hydroxides may also be developed by the interaction of pyrite and other sulfides with the atmosphere in exposed coals (Silva et al., 2011; Ward, 2016). The spontaneous combustion of coal seams is widely observed throughout the Fukang mining area (Lu et al., 2008). The Xiaohuangshan open coal mine is just on the burned rock belt, and this would provide favorable conditions for the formation of goethite.
Occurrence of alkali metals in coal
The main occurrence of K in coal is as silicate minerals (Ward, 2002). For the Xiaohuangshan No. 6 coal seam samples, the determination coefficient between K2O and Al2O3 is 0.89 (Figure 7(a)), showing a significant positive correlation. Additionally, the determination coefficient between K2O and SiO2 is 0.59 (Figure 7(b)), which is also a positive correlation. The results indicate that the aluminosilicate minerals may be the main K carriers, rather than the main source of SiO2 in coal. It is speculated that aluminosilicate minerals from the Tianshan Mountain may be the main carrier of K.
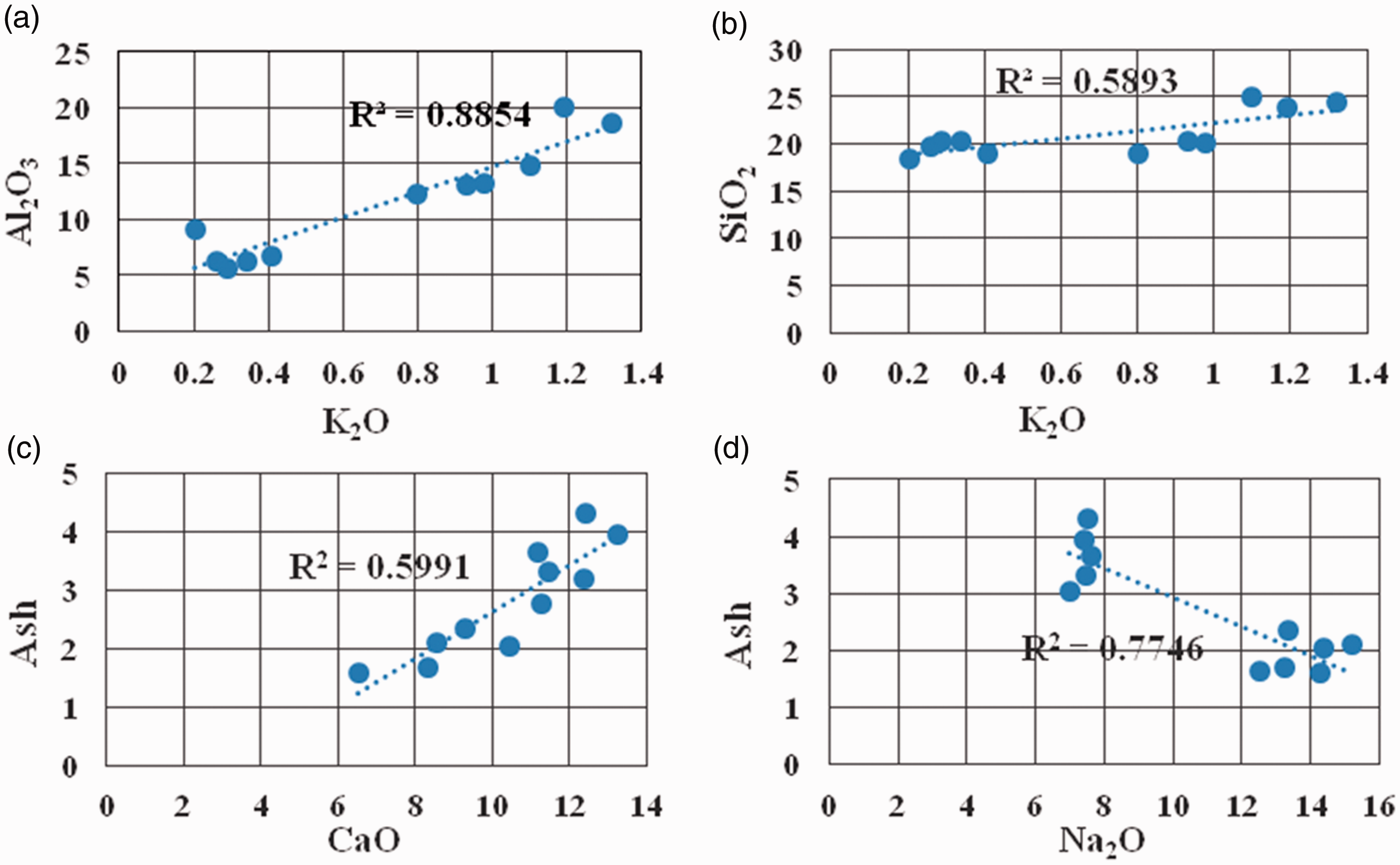
Relationship of Al2O3, K2O, SiO2, CaO, Na2O, and ash.
The main Ca-rich phases include carbonates (mainly calcite), sulfates (mainly anhydrite), phosphates, silicates, and organic matter (such as asphalt) (Dai et al., 2005; Ward, 2002, 2016). In coal ash from the Xiaohuangshan coal mine, the determination coefficient between CaO and ash is 0.60 (Figure 7(c)), indicating that Ca in coal is mainly present in the minerals. The LAT-XRD (Figure 5) patterns indicate that Ca is present in dolomite and calcite.
The correlation coefficient and determination coefficient between Na2O and ash are –0.88 and 0.77 in the No. 6 coal seam, respectively (Figure 7(d)), showing an obvious negative correlation; this indicates that the minerals are not the Na carriers, in spite of a few parts occurring as dawsonite and albite, which were detected by LAT-XRD (Figure 5). However, Na+ could be released while feldspar minerals are weathered, then combined with the organic structure of the coal matrix to form organic sodium. According to the sequential chemical extraction method, sodium species in coals mainly exist in four chemical forms, including those that are water-soluble, ammonium-acetate-soluble, acid-soluble, and acid-insoluble, which correspond to soluble salts, carboxylates, silicates and aluminates, and silico-aluminates, respectively (Li et al., 2018). According to the data from analyzing more than 600 drilling coal samples and more than 100 commercial coal samples collected in the Zhundong coalfield, water-soluble Na is widely recognized to be the most abundant species in the Zhundong coalfield (Bai et al., 2015; Li et al., 2018). As shown in Figure 1, the Xiaohuangshan coal mine is located in the northeast part of the Zhunnan coalfield, which is close to Zhundong coalfield. Na in some coal mines, which are adjacent to Xiaohuangshan coal mine (Wang, 2016), also occurs as water-soluble. The correlation coefficient between Na2O and coal ash in the No. 6 coal seam also shows a significant negative correlation. Therefore, it can be speculated that Na in the No. 6 coal seam exists as a water-soluble hydrated ion in coal organic structures.
Sources of alkali metals in the No. 6 coal seam
The contents and ratios of various elements in the sediments were linked to the surrounding physical and chemical conditions; this, in turn, reflects the variations in the sedimentary environment and the climate background (Algeo and Maynard, 2004; Chu et al., 2015; Tribovillard et al., 2006). The contents of Sr, Ba, and Rb, and the ratios of Sr/Ba, Sr/Cu, and Rb/Sr can qualitatively differentiate between the paleo-salinity and paleo-climate types of the media. The concentrations of Sr in saltwater and freshwater are generally 800–1000 × 10−6 and 100–3000 × 10−6, respectively. The Sr/Ba ratios lower and higher than 1 indicate that the sedimentary environments were freshwater and saltwater environments (sea phase or salty lake phase), respectively (Wang, 1996; Zhang et al., 2016). The Sr/Cu ratios ranging from 1 to 10 indicate a warm and humid climate, while Sr/Cu ratios greater than 10 indicate a dry and hot climate (Zhang et al., 2016; Lerman et al., 1995). Under the weathering action, Rb is relatively stable, whereas Sr is susceptible to leaching. Therefore, low Rb/Sr values indicate humid climates, while high Rb/Sr values indicate arid climates (Lei et al., 2017). The contents and ratios of trace elements in the samples are shown in Table 3. All the indicators of the trace elements demonstrate that the No. 6 coal seam had formed in a freshwater environment with a predominantly humid climate. The results are inconsistent with what was proposed by Chen (2014), who suggested that the higher Na contents in the Xinjiang coal come from the remnant salts following the evaporation of seawater during the coal-forming period (Han et al., 1999; Zhang et al., 2013). The paleo-geographic data of lithofacies indicate that the Jurassic coal seam of the Zhunnan coalfield constitute an inland fluvial and lacustrine facies (Figure 2). Additionally, the sodium concentrations of the paleo-water present during the coal-forming age were speculated to be very low since they were not affected by seawater, neither during the coal-forming stage nor during the later stages (Bai et al., 2015). Therefore, the high Na contents of the No. 6 coal seam samples indicate that they were not formed in the depositional period of the coal seam.
Some researchers (Bai et al., 2015; Wang et al., 2015) also found that the contents of Na, in an open coal mine in the Zhundong coalfield, decreased as the distance from the top of the coal seam increased. However, the contents of Na2O in the No. 6 coal seam are relatively high in the upper and bottom parts of the coal seam, while they are relatively low in the middle (Table 2). It is speculated that this may be related to the movement of water.
The main replenishment source of surface water and groundwater is snowmelt that runs off of the Tianshan Mountain in the southeast. There are several rivers that come from the snowmelt of the Tianshan. Due to topographical features, the flow of most rivers is from south to north. Rivers that run through the mountains traverse the coal-bearing stratum and flow through coal mining areas (Figure 3). Geological data show that quartz albitophyre, located in the recharge area of the groundwater, is distributed in the south and southeast of the coal field (Wang, 2016). Due to weathering, Na+ can be released from quartz albitophyre and carried to the coal-bearing stratum by the water. Therefore, this may be the main source of Na in No. 6 coal seam.
Moreover, this area exhibits the typical northern temperate continental arid climate, which is known for a long daylight duration. The annual average precipitation and evaporation values are 251.59 mm and 1543.81 mm, respectively (Gong et al., 2015). For the area of low precipitation and comparatively high evaporation capacity, annually, the topsoil layers have undergone salinization, and many saltwater lakes are distributed in Xinjiang province (Gong et al., 2017; Xu et al., 2007). In the Fukang area, the main chemical type of phreatic water is SO4·HCO3–Na·Ca (Li, 2014). Therefore, during the infiltration process of surface water, sodium in the surface soils is gradually brought into the coal seam, resulting in Na enrichment near the top region of the coal seam.
In contrast to the findings of Bai et al. (2015) on Na contents in the open-pit mine of the Zhundong coal mine, the Na contents in the coal seam near the bottom of the Xiaohuangshan coal mine were relatively high. The underlying Mesozoic formation water is dominated by SO4·HCO3–Na with ultrahigh or normal pressures in the Fukang area (Gong, 2017; Li et al., 2013; Wen et al., 2012). In addition, the Na2SO4-type Mesozoic formation water, with ultrahigh, pressure is often associated with the generation of hydrocarbons via the hydrocarbon resource rock in the coal systems (Voast and Montana, 2003). The connectivity between fractures, from the low metamorphism degree of the No. 6 coal seam (vitrinite reflectance of 0.55), is poor and the dominant pore types are micropore (Li et al., 2017; Wang et al., 2018b). Therefore, the Na2SO4 and NaHCO3 type waters in the underlying strata with ultrahigh or normal pressures have a relatively low flow rate and are rather stagnant (Gong et al., 2017; Li et al., 2013). The Na+ is enriched in those pores and fractures, indicating a higher Na content near the bottom of coal seam in comparison to the middle of the coal seam.
Conclusions
The No. 6 coal seam of the Fukang mining area has high volatility and ultralow ash and sulfur contents. The average content of Na2O in coal ash is 10.91%, which is much higher than the Na2O content (1%) in the ash of China’s steam coal; this indicates a great impact on the combustion and utilization of coal.
The minerals identified in the coal are dawsonite, dolomite, calcite, albite, kaolinite, quartz, and trace of apatite, goethite, illite. Alkalescent water and carbon dioxide from coalification offered favorable conditions for the formation of the carbonate minerals. The dawsonite in the No. 6 coals was probably derived mainly from the reaction between albite and CO2.
The minerals present in the coal are not Na carriers, and Na mainly combines with the organic structure of the coal matrix; K in coal mainly occurs in the form of silicate aluminosilicate minerals; Ca in coal mainly occurs in minerals, such as dolomite and calcite.
During the sedimentation period of the coal seam, the environment was a freshwater environment with warm and humid conditions. Therefore, the higher Na contents in the coal were not formed during the sedimentary period of the coal seam and may have leached from fluids during later stages.
The release of Na from albitophyre during their weathering may be the major source of Na in the No. 6 coal seam, and then Na+ was carried to the No. 6 coal seam by the surface water, phreatic, and high-pressure groundwater. Consequently, the Na contents are higher in the upper and bottom parts of the No. 6 coal seam in comparison to the middle part.
Footnotes
Acknowledgements
Special thanks are given to the Editor and reviewers for their careful reviews.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the program of investigation and assessment of the special and rare coal categories in China (No. DD20160187).
